Abstract
Aim:
The correlation between soluble Klotho (sKlotho) levels and clinical outcomes remains inconclusive for patients undergoing maintenance haemodialysis (MHD). We aimed to evaluate the potential predictive significance of sKlotho in this population by conducting a meta-analysis.
Methods:
PubMed, Embase, Web of Science and Cochrane Library were comprehensively searched for studies concerning the association between sKlotho level and clinical outcomes including cardiovascular (CV) events and all-cause mortality. The pooled hazard ratios (HR) and 95% confidence intervals (CI) were generated using either random or fixed effects models. Sensitivity and subgroup analyses were used to explore heterogeneity sources.
Results:
Eight prospective studies with 992 MHD participants were included and reduced sKlotho levels predicted more adverse outcomes in this meta-analysis. The pooled HRs and 95% CIs related to CV events, mortality, or composite outcomes were 1.73 (95% CI 1.08–2.76,
Conclusion:
Lower sKlotho levels were associated with more CV events and all-cause mortality, suggesting that sKlotho may have predictive value in CKD patients receiving haemodialysis.
Introduction
Klotho is an identified gene in 1997 and exerted anti-aging effect. 1 It encodes a single chain transmembrane protein (mKlotho), which functions as a co-receptor for fibroblast growth factor-23. 2 There is another form of Klotho, secreted Klotho or soluble (sKlotho), which is generated from alternative splicing of the Klotho gene or ectodomain cleavage of mKlotho. 2 sKlotho can be released into the circulation and functions as a circulating factor, with anti-inflammatory, anti-oxidative, anti-apoptotic and anti-fibrotic effects.3,4 sKlotho is suggested to be a novel kidney protective protein because it has various biological activities. 5 Although Klotho has also been found in multiple organs, the kidney has the highest level of Klotho and Klotho is significantly decreased when kidney is diseased, demonstrating that the kidney is an important organ that is associated with the production of Klotho.6–8 Because Klotho is predominantly expressed in the kidney, there is a close association between sKlotho levels and kidney disease. 9 The sKlotho level decreased significantly in patients in early and intermediate chronic kidney disease (CKD) stages, and it decreased further in advanced CKD stages.10–12 For pre-dialysis patients, decreased sKlotho levels are associated with more adverse kidney outcomes including creatinine doubling, renal replacement therapy and death.11,13,14 These observations suggested that sKlotho may be a potential biomarker for the timely diagnosis and prognosis of CKD patients without dialysis.
Patients undergoing maintenance haemodialysis (MHD) had lower sKlotho levels and a higher risk of cardiovascular (CV) events and mortality than pre-dialysis CKD patients.14–17 Furthermore, sKlotho deficiency is accompanied by abnormal calcium and phosphorus metabolism or prominent vascular calcification (VC) in this population.17–20 It is well established that disturbed mineral metabolism and VC are common complications in patients on MHD, and these independently predict poor prognosis in this patient population. 21 This suggests that reduced sKlotho levels may also be correlated with increased adverse outcomes in haemodialysis patients. An increasing number of studies have suggested that reduced sKlotho levels predict a poor prognosis such as death or CV morbidity in MHD patients,15,22,23 indicating that sKlotho is a potential predictor for MHD patients. However, other studies have shown conflicting results in which sKlotho level was not associated with adverse outcomes in MHD patients.24–26 Thus, the prognostic role of sKlotho in MHD patients remains uncertain and it has not been systematically reviewed. We conducted this meta-analysis and systematic review in MHD patients to investigate the relationship between the sKlotho level and adverse outcomes including CV events and all-cause mortality.
Methods and materials
The present meta-analysis was conducted in agreement with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria 27 and A Guide to Systematic Review and Meta-Analysis of Prognostic Factor studies. 28
Search strategy
Medical databases such as PubMed, Embase, Web of Science and the Cochrane Library (from their inception to 27 October 2019) were systematically searched to identify studies that assessed the prognostic value of sKlotho in patients with MHD. Two independent investigators (QFL and LXY) performed the search. Reference lists from each identified article were also examined. The following search terms were used: End stage renal disease or ESRD or end stage kidney disease or ESKD or uremic or dialysis or haemodialysis or haemodialysis or hemofiltration or HD or renal dialysis or renal dialysis; and Klotho or KL or alpha-Klotho or α-Klotho or α-KL or sKlotho or sKL; and death or mortality or all-cause mortality or total mortality or morbidity or cardiovascular event or cardiovascular disease or stroke or coronary artery disease or ischaemic heart disease or myocardial ischaemia or myocardial infarction or prediction or predictive or endpoint or outcome or survival or prognostic or prognosis.
Study selection and inclusion criteria
Titles and abstracts were screened by two reviewers (JHF and LLX) for potentially relevant studies and the full text of the relevant studies was evaluated for eligible studies based on the inclusion and exclusion criteria. The inclusion criteria were set as the following: (a) Patients with MHD history >3 months and age >18 years; (b) Cohort or longitudinal studies; and (c) Studies reporting the relationship between sKlotho levels and adverse clinical outcomes (such as mortality and CV events). The exclusion criteria were set as the following: (a) Patients with history of organ transplantation or immunosuppressants; (b) Retrospective cohort studies or observational studies, or publications other than English; (c) Non clinical studies including
Data extraction and quality assessment
Two reviewers (SS Li and QF Liu) extracted relevant data from the full text of the included studies using a standardised data extraction form. The obtained information was expressed as follows: first author, publication year, study characteristics (age, gender, location, study design and sample), assay utilisation of sKlotho, duration of follow-up and outcomes assessment including relative risk (RR), hazard ratio (HR) and odds ratio (OR). Any disagreements concerning date extraction were resolved by a third reviewer after re-examining the original data. The Newcastle–Ottawa Scale (NOS) was used to evaluate the risk of bias in the eligible publications. 29 The NOS assigned three key points including the selection of study participants, comparability of study groups and assessment of outcome. Each study was awarded several stars depending on the quality of the items. Studies that scored ⩾7 stars were considered to have a low bias risk and were rated as high-quality studies.
Data synthesis and statistical analysis
The HR and the 95% confidence interval (CI) that were reported in the study were directly extracted and converted to the pooled HR and 95% CI. If the HR was not available directly in the study, the indirect HR was calculated based on Kaplan–Meier curves, as previously described.
30
Otherwise, OR was estimated based on the provided data and combined with HRs to generate a pooled effect size. Heterogeneity among the studies was examined by the
Results
Literature search
There were 1301 studies that were retrieved from medical databases and 228 studies were removed after duplication. Among the remaining 1073 studies, 964 were excluded after screening the titles and abstracts, and 87 studies were excluded after examining the full texts in the remaining 109 studies. Among the remaining 22 full articles, 11 articles were included for qualitative synthesis and 8 eligible articles were selected for quantitative synthesis.15,22–26,31,32 The literature screening flow chart is summarised in Figure 1.
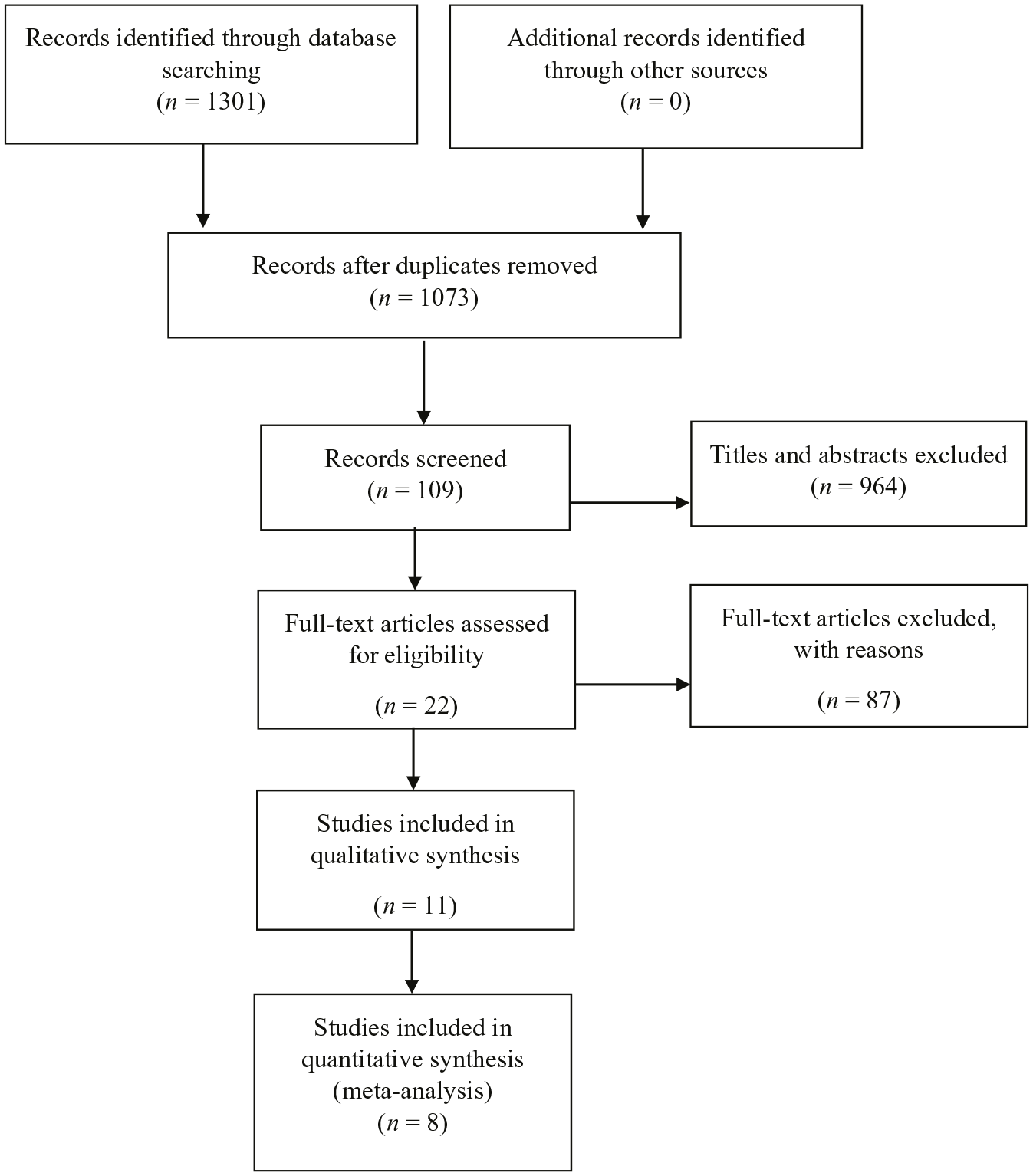
Study screening flow.
Study characteristics
The current meta-analysis included eight eligible prospective studies with 992 MHD participants. The sample size ranged from 30–239 and follow-up period ranged from 18–66 months. The effect indicators, including four HRs,15,22,23,25 one RR 32 and one OR, 24 and 95% CIs were retrieved directly from six of the eight studies.26,31 Based on the median sKlotho or sKlotho tertiles levels, two studies reported CV events,24,31 four studies reported all-cause mortality25,26,32,22 and two studies reported composite CV events and all-cause mortality.15,23 The study characteristics are summarized in Table 1. The NOS scores ranged from 6–8 stars and the average score was 7.3 stars. Four studies were of good quality. The detailed NOS information is displayed in Table 2.
Characteristics of the included studies.
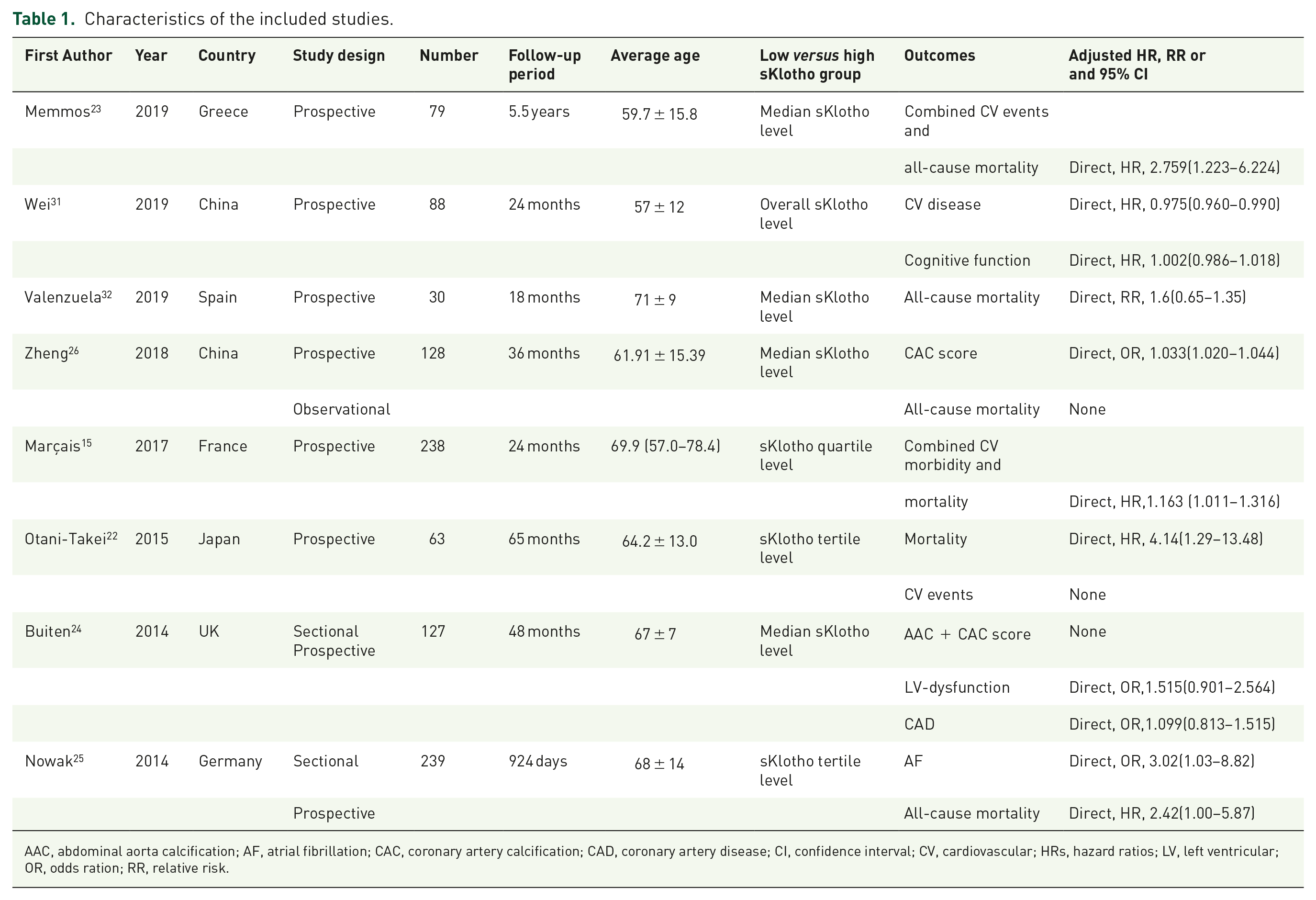
AAC, abdominal aorta calcification; AF, atrial fibrillation; CAC, coronary artery calcification; CAD, coronary artery disease; CI, confidence interval; CV, cardiovascular; HRs, hazard ratios; LV, left ventricular; OR, odds ration; RR, relative risk.
NOS scores of the cohort studies included.

Two stars could be awarded for this item. Studies that controlled for age or eGFR were awarded one star, respectively.
eGFR, estimated glomerular filtration rate; NOS, Newcastle–Ottawa Scale.
Correlation between sKlotho and CV events
One study reported that overall sKlotho level was correlated with cerebrovascular disease [HR, 0.975 (0.960–0.990)]
31
and one study demonstrated that median sKlotho was not associated with CV events such as heart dysfunction (OR, 1.515, 95% CI 0.901–2.564) or CAD (OR, 1.099, 95% CI 0.813–1.515).
24
Another study demonstrated that the sKlotho level was associated with the development of AF (OR, 1.515, 95% CI 0.901–2.564).
25
The pooled result of the association between the sKlotho level and CV events was a pooled HR of 1.73 (95% CI, 1.08–2.76,

Forest plots of low sKlotho levels and cardiovascular events.
Correlation between sKlotho and all-cause mortality
Four studies reported the relationship of sKlotho level and all-cause mortality.22,25,26,32 Among the four studies, one study demonstrated that sKlotho level was strongly correlated with all-cause mortality,
22
but the remaining three studies did not show this association.25,26,32 Zheng

Forest plots of low sKlotho levels and all-cause mortality.
Correlation between sKlotho and composite adverse clinical outcomes
CV events and all-cause mortality were combined into the composite adverse clinical outcomes for further analysis. Five HRs and one OR were pooled in our meta-analysis. Overall, there was a remarkable difference in the rate of composite adverse clinical outcomes (pooled HR, 1.75, 95% CI 1.19–2.57,

Forest plots of low sKlotho levels and combined cardiovascular events and all-cause mortality.
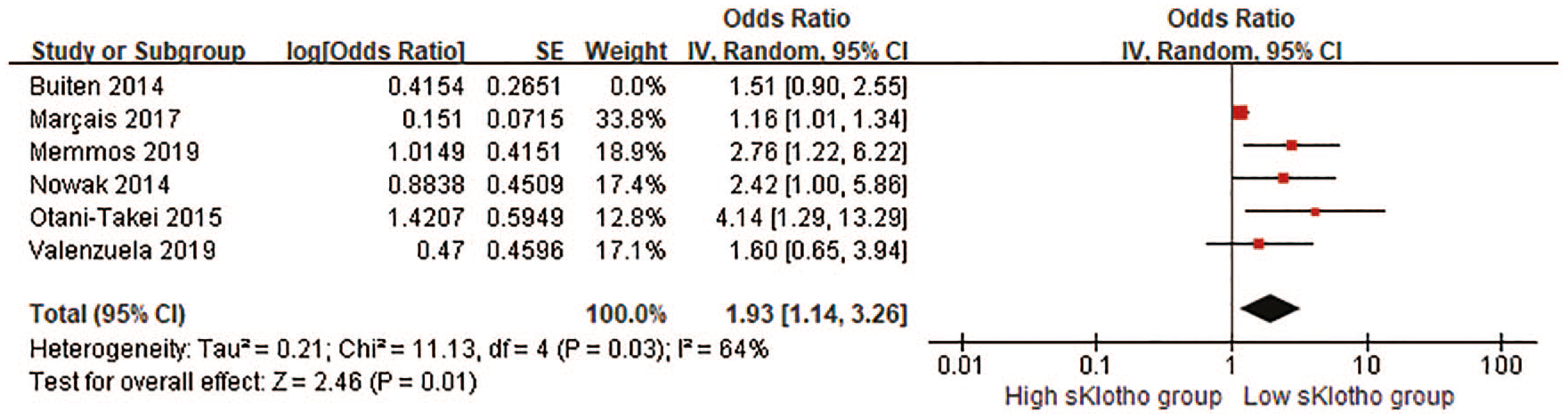
Forest plots of low sKlotho levels and combined cardiovascular events and all-cause mortality after excluding Buiten’s study.
Sensitivity and subgroup analysis of included studies
Although the positive association remained significant after omitting any a single study in the sensitivity analysis, moderate heterogeneity was no longer observed after removing Marçais
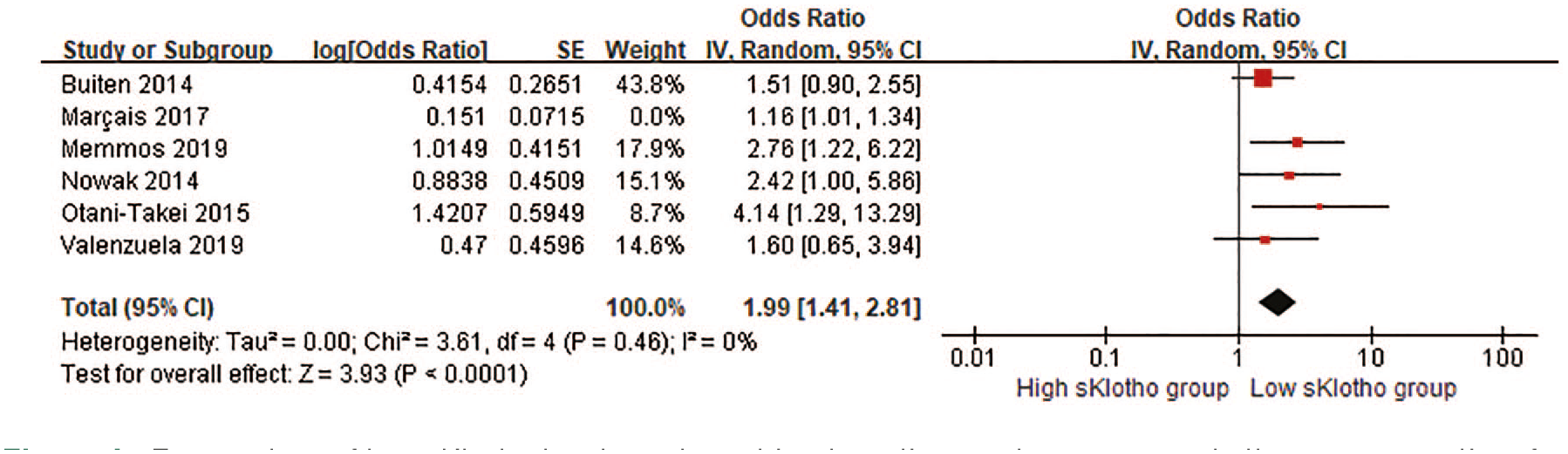
Forest plots of low sKlotho levels and combined cardiovascular events and all-cause mortality after excluding Marçais 2017’s study.
A subgroup meta-analysis was conducted to investigate the possible origins of heterogeneity in this study (Table 3). The eligible studies were divided into subgroups by age (⩾65 years or <65 years), sample size (⩾200 or <200), sKlotho level (median sKlotho and not median sKlotho), follow-up time (⩾36 months or <36 months) and study quality (⩾7 stars or <7 stars). The positive association was still observed in the subgroups by age and sKlotho level, but it was attenuated by a shorter follow-up time (<36 months, Figure 7), lower quality of the study (<7 stars) and a larger sample size (⩾100). No significant heterogeneity was detected in all subgroups except for age and sKlotho level, indicating that age and sKlotho level may be the main origins of heterogeneity.
Results of subgroup analysis by age, sample size, follow-up time and study quality.

CI, confidence interval; HR, hazard ratio.
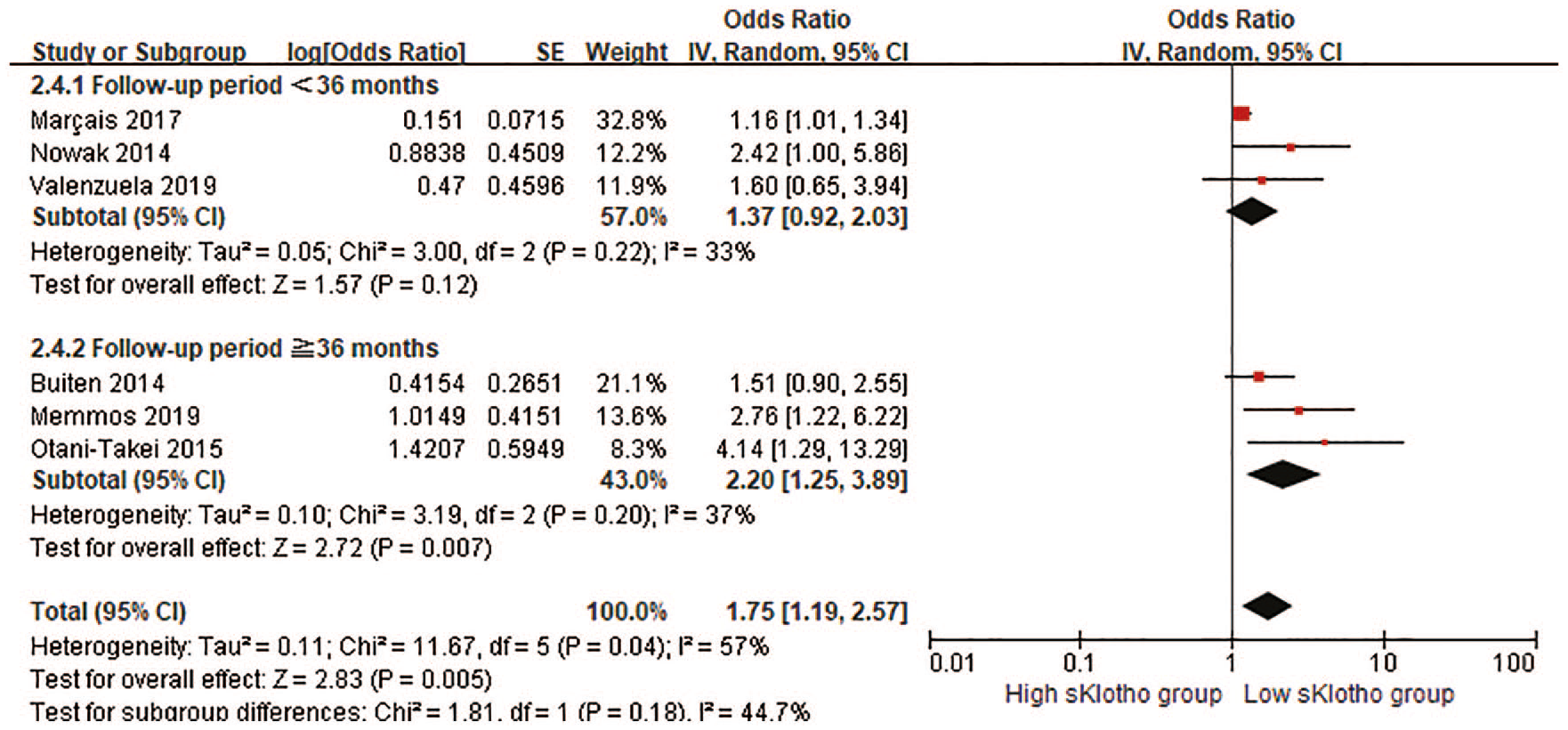
Forest plots of low sKlotho levels and combined cardiovascular events and all-cause mortality in the subgroup by follow-up time.
Discussion
In this study, we systematically investigated the association of the sKlotho level with adverse clinical outcomes in MHD patients. We showed that a reduction in the sKlotho level was strongly associated with an increase in CV events and all-cause mortality in this population. To the best of our knowledge, this is the first meta-analysis and systematic review providing insights into the association of sKlotho level with adverse outcomes in MHD patients. The results suggested that sKlotho may have prognostic role for MHD patients.
sKlotho is generated from mKlotho ectodomain cleavage or alternative splicing, so sKlotho can be released into the extracellular space and it functions as a circulating hormone. The kidney is the major organ that is responsible for the production and metabolism of Klotho (both mKlotho and sKlotho). 6 Thus, if the kidney is diseased, sKlotho loss immediately occurs and the sKlotho level reflects the state of kidney function. 35 A strong positive association between sKlotho and kidney function has been well demonstrated in a recent meta-analysis that involved nine publications with 1457 patients. 9 sKlotho deficiency also caused deterioration of kidney function and accelerated CKD progression. Consistent with this notion, we recently reviewed the predictive value of sKlotho in CKD patients who were not receiving dialysis. We found that patients with a low sKlotho level had a high risk of kidney function deterioration or death (pooled HR, 1.64, 95% CI 1.19–2.26). 30 The findings suggested that sKlotho is a feasible biomarker for CKD diagnosis and progression.
MHD patients had more CV complications and a higher mortality rate despite past improvements in prognosis.
36
Screening potential biomarkers is of importance to reduce CV events and mortality in MHD patients. Unfortunately, an ideal biomarker has not been available until now in clinical practice.
37
Available evidence for patients with MHD is characterised by progressive sKlotho deficiency that is caused by almost or complete loss of kidney function.11,15,17,22 As a potential cardio-renal protective factor, theoretically, progressive reduction in sKlotho in patients with MHD may affect clinical outcomes. Previous observational studies demonstrated that the reduced sKlotho level was correlated with cardiac parameters such as left ventricular hypertrophy, heart dysfunction, coronary artery calcification and AF, independent of traditional CV risk factors.19,20,38,39 This means that sKlotho may have a beneficial effect against CV complications and mortality. A previous prospective study by Otani-Takei
However, several studies contribute to these inconsistent results. In Nowak
There are several reasonable explanations for this association.
This study has several limitations. First, the meta-analysis enrolled a small number of studies with a relatively small sample size and thus the strength of our results may be reduced. Second, there is moderate heterogeneity in this study. To find the sources of heterogeneity, we conducted a subgroup analysis. We found that there was no heterogeneity among subgroups except for the subgroups by age and sKlotho level. Thus, heterogeneity may result from age and sKlotho level. Because sKlotho was originally identified as a life-span factor, it is not surprising that sKlotho level is decreased with aging. 58 This means that sKlotho expression is influenced by aging. Aging is also associated significantly with increased adverse outcomes in MHD patients in previous studies.26,59,60 In this context, the effect of sKlotho on clinical outcomes may be weakened by aging. There may be an interaction between sKlotho and aging. Thus, aging is an important confounder and the source of heterogeneity. In addition, sKlotho level (median sKlotho level or not median sKlotho level) was another source of heterogeneity among studies, although no heterogeneity exits within this subgroup. We observed that studies with median sKlotho levels had a shorter follow-up and smaller sample size, thus, differences in methodologies among studies may explain the heterogeneities. Third, we only included eight English articles and excluded non-English articles. Publication bias could not be avoided. Finally, although six adjusted effect measures (five HRs and one OR) were obtained directly from the full text of the manuscript, the other two adjusted HRs were not obtained directly. Therefore, our conclusion may be less convincing due to the limitations after omitting these two studies.
Thus, our findings demonstrated that a decreased the sKlotho level was associated with more detrimental clinical outcomes. The study provided evidence that sKlotho may have predictive performance in CKD patients who are receiving haemodialysis despite several limitations. Further larger scale prospective studies with high quality are needed to confirm our conclusions.
Footnotes
Acknowledgements
We thank Jodi Smith, PhD for editing the English text of a draft of this manuscript.
Author contributions
GYL conceived the study and revised the manuscript. QFL and LXY performed the literature search and analysed the original data. JHF and LLX screened the literature and identified the eligible studies. SSL and QFL extracted the data and wrote the manuscript. All authors had read, revised and approved this manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was granted partly from the Social Development Foundation of Kunshan (KS18058) and partly from the Technological Development Foundation of Clinical Medicine of Jiangsu University (JLY20180108), Jiangsu Province, China.
