Abstract
Platelet-rich plasma (PRP) is defined as a volume of plasma with a platelet concentration higher than the average in peripheral blood. Many basic, preclinical and even clinical case studies and trials report PRP’s ability to improve musculoskeletal conditions including osteoarthritis, but paradoxically, just as many conclude it has no effect. The purpose of this narrative review is to discuss the available relevant evidence that supports the clinical use of PRP in osteoarthritis, highlighting those variables we perceive as critical. Here, recent systematic reviews and meta-analyses were used to identify the latest randomized controlled trials (RCTs) testing a PRP product as an intra-articular treatment for knee osteoarthritis, compared with an intra-articular control (mostly hyaluronic acid). Conclusions in the identified RCTs are examined and compared. In total, five recent meta-analyses and systematic reviews were found meeting the above criteria. A total of 19 individual trials were identified in the five reviews but only 9 were level of evidence I RCTs, and many had moderate or high risks of bias. At present, results from these RCTs seem to favor PRP use over other intra-articular treatments to improve pain scales in the short and medium term (6–12 months), but the overall level of evidence is low. As a result, clinical effectiveness of PRP for knee osteoarthritis treatment is still under debate. This is, prominently, the result of a lack of standardization of PRP products, scarceness of high quality RCTs not showing high risks of bias, and poor patient stratification for inclusion in the RCTs.
Keywords
Introduction
Platelets, also known as thrombocytes, are small cytoplasmic fragments derived from bone marrow megakaryocytes. Most platelet functions are directly connected with platelet activation, a process that occurs naturally after an injury in the wall of a blood vessel. Platelets are then exposed to collagen and other extracellular matrix proteins that stimulate their activation, resulting in the release of the content of their cytoplasmic granules. 1 Overall, platelets contain over 800 proteins and molecules, comprising cytokines, chemokines, membrane proteins, metabolites, messenger molecules, growth factors (GFs) and numerous soluble proteins. 2 As a result, besides their role in coagulation and hemostasis, platelets are also involved in vasoconstriction, inflammation, immune response, angiogenesis and tissue regeneration and consequently, they participate in numerous physiologic signaling mechanisms and are related to multiple pathologies.3–5
The therapeutic use of platelet concentrates was first described by Whitman in 1997, 6 although blood-derived fibrin glues were already used 30 years earlier to seal wounds and stimulate their healing. 7 In 1998, platelet concentrates started to be known as platelet-rich plasma (PRP), generally defined as a volume of autologous plasma containing a higher platelet count than peripheral blood (150,000–350,000 platelets/μl). 8 Thereafter multiple systems have been developed to concentrate platelets and remove erythrocytes (red blood cells; RBCs) and, in some cases, also leukocytes (white blood cells; WBCs), as reviewed elsewhere. 9 For the purpose of this review, PRP or PRP products refer to any product derived from a platelet concentrate (PC), optionally containing WBCs, from whole blood.
The rationale for its use is quite strong: it is considered well tolerated, it very rarely leads to complications, it is easy to prepare and administer and it is less aggressive than other therapeutic options that might be indicated for some patients, such as corticoid intra-articular injection or even surgery.10,11
The number of studies on PRP preparation methods and applications has grown exponentially since the late 1990s; however, there are still concerns regarding its clinical efficacy, mainly due to the heterogeneity of preparation methods and resulting products, the scarceness of high quality randomized controlled trials (RCTs), and the contradictory results that have been found so far.
Custom and commercial preparation methods for human use
Methodologies to prepare PRP and derived products vary widely. Briefly, they can rely on single centrifugation, double centrifugation, or blood selective filtration procedures, and on manual or automatic systems operated in open or closed circuits. Ex vivo, platelet activation can be triggered mechanically with freeze–thawing cycles, chemically with thrombin or calcium chloride, or endogenously. This last option implies the direct application of nonactivated PRP, to allow local tissue factors to elicit the process.9,12
The high number of variables involved has led to innumerable custom protocols developed in-house in the different research labs and also to many (proprietary) commercial systems for producing PRP products (see Table 1).
Examples of commercial PRP preparation systems.

A, automatic; ACP, Autologous Conditioned Plasma; C, closed; CE, European Union CE mark; DS, double spin; F, filtration; FDA, United States Food and Drug Administration; GPS, Gravitational Platelet Separation System; M, manual; NR, not reported; O, open; OA, osteoarthritis; PEF, platelet enrichment factor [(platelets in PRP)/(platelets in whole blood)]; PRP, platelet-rich plasma; RBC, red blood cell; SA, semi-automatic; SC, semi-closed; SS, single spin; WBC, white blood cell.
Due to their complex composition and to inter- and intra-individual differences, 13 each PRP is unique and very difficult, if not impossible, to characterize. As a result, PRPs are usually defined in terms of a few critical variables and a detailed description of the preparation protocol. These variables usually include (1) the proportion of platelets in PRP to platelets in whole blood (platelet enrichment factor, PEF), (2) presence/absence of WBCs, and (3) method of activation. Yet, many authors believe this is not enough and different systems for a more complete and standardized description of platelet-derived product characteristics have been proposed (see Table 2).
Most commonly used PRP classification systems.

PRP, platelet-rich plasma; RBC, red blood cell; WBC, white blood cell.
Mishra and colleagues proposed to classify PRPs with two parameters: firstly, ‘type’ of PRP: (1) increased WBCs and no activation; (2) increased WBCs and activated; (3) minimal/no WBCs and no activation; (4) minimal/no WBCs and activated; and secondly, its platelet enrichment factor, A if the PRP contains a platelet concentration at or above five times the baseline, or B if platelet concentration is less than five times the baseline. 14 On the other hand, the PAW (Platelets, Activation, White cells) classification system includes at least three variables: (1) the absolute platelet concentration (P); (2) the method of activation (A); (3) the presence or absence of WBCs and neutrophils (W) relative to the baseline. Platelets are categorized as P1 (baseline) to P4 (>1.2 million platelets/µl), activation as either exogenous (X) or not, and WBCs and neutrophils as either above or below baseline. 15 Mautner and colleagues advocated for reporting at least platelet concentration (cells/µl, volume injected), leukocyte concentration, including the concentration of neutrophils (if >1%), RBC concentration and activation by exogenous agents. 16 And more recently still, the DEPA (Dose of injected platelets, Efficiency of production, Purity of the PRP, Activation of the PRP) classification system was proposed. 17 This system takes into account (1) the dose of injected platelets from A (>5 billion) to D (<1 billion); (2) platelet capture efficiency from blood from A (>90%) to D (<30%); (3) the % platelets compared with RBCs and leukocytes in the PRP from A (>90%, very pure) to D (<30%, whole blood PRP); and, (4) the activation process. Thus, an ‘AAA’ DEPA score refers to an injection of PRP with a very high dose of platelets (>5 billion) with little contamination from RBCs and with very high platelet recovery efficiency from blood. However, none of these methods has been formally adopted and PRP descriptions are still very heterogeneous.
Current regulatory framework
Across the European Union (EU) PRP is regulated under Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community code relating to medicinal products for human use. 18 The Directive is then assumed and adapted in each Member State according to their own national regulatory frame. For instance, in Spain, the AEMPS (Agencia Española de Medicamentos y Productos Sanitarios, Spanish Agency of Medicines and Health Care Products) published in May 2013 a ‘Report on the Use of Platelet-Rich Plasma’ (INFORME/V1/23052013). 19 As a result, in Spain, PRP can only be administered by a ‘medical doctor, a dentist or a podiatrist’ and under his/her sole responsibility, as long as all of the guarantees of quality, efficacy, traceability, and pharmacovigilance are met. The report was timely and relevant, since it elevated PRP to the category of a medicament for human use, and as a result, its regulation is much stricter than when it was considered a blood-derived product. It was well received, 20 but surprisingly, with the current European and national regulatory framework, only the preparation procedure, and not the product itself, is regulated, and it does not include any requirements about its composition or its effectiveness. In fact, the AEMPS itself points out that there exists some confusion in this type of autologous product between the medicament production procedures and the medicament itself. In commercial products, a CE marking is sought, to attest to the manufacturer’s declaration that the product complies with the essential requirements of the relevant European health, safety and environmental protection legislation, but this marking makes no reference to its clinical efficacy. 21 The above-mentioned EU Directive has not been translated into national law all across Europe, and therefore PRP products are still considered as blood derivatives in some EU countries.
In the United States (US), on the other hand, PRP products are regulated according to the Food and Drug Administration (FDA)’s 21 CFR 1271 of the Code of Regulations, and do not follow the US FDA’s traditional regulatory pathway that includes animal studies and clinical trials. 22 PRP preparation systems are generally brought to the market through a 510(k) application. This route implies that the device is ‘substantially equivalent’ to another previously cleared and limited to the same indications. However, it should be noted that, as is the case in EU with the CE label, the clearance applies only to the device and its intended use in an operative setting and makes no claim about its effectiveness for a particular indication. 23 Evidently, all commercial systems included in Table 1 have achieved a EU CE mark or US FDA 501(k) approval.
Evidence of clinical efficacy of PRP-derived products in musculoskeletal injuries/diseases
The efficacy of PRP-derived products in its various forms (direct liquid/injection, gel, clot, release rate, etc.) either from commercial brands or in-house protocols, has been tested by means of in vitro studies, preclinical and clinical trials for oral and maxillofacial surgery, treatment of chronic ulcers, ophthalmology, dermatology, and injuries and pathologies associated to tendon, muscle, cartilage and bone, among other fields.24–31
Ligament and tendon repair focus the attention of most studies addressing soft tissue regeneration. However, even in this extensively investigated field there is no consensus on PRP efficacy and most authors highlight the need for more rigorous RCTs. A recent review by the Cochrane collaboration evaluated the evidence supporting clinical efficacy in soft tissue injuries. Of the identified trials (19 randomized and quasi-randomized trials), 16 were judged at high or unclear risk of bias. 32 It was also pointed out that PRP preparation methods lacked standardization and quantification of the PRP applied to the patient. The review concluded that there is currently insufficient evidence to support the use of platelet-rich therapy for treating musculoskeletal soft tissue injuries.
Application of PRP for cartilage regeneration and OA treatment, our field of interest, has been getting more and more attention over the last decade. Comprehensive review of in vitro and preclinical validation studies is beyond the scope of this review but can be found elsewhere. 33
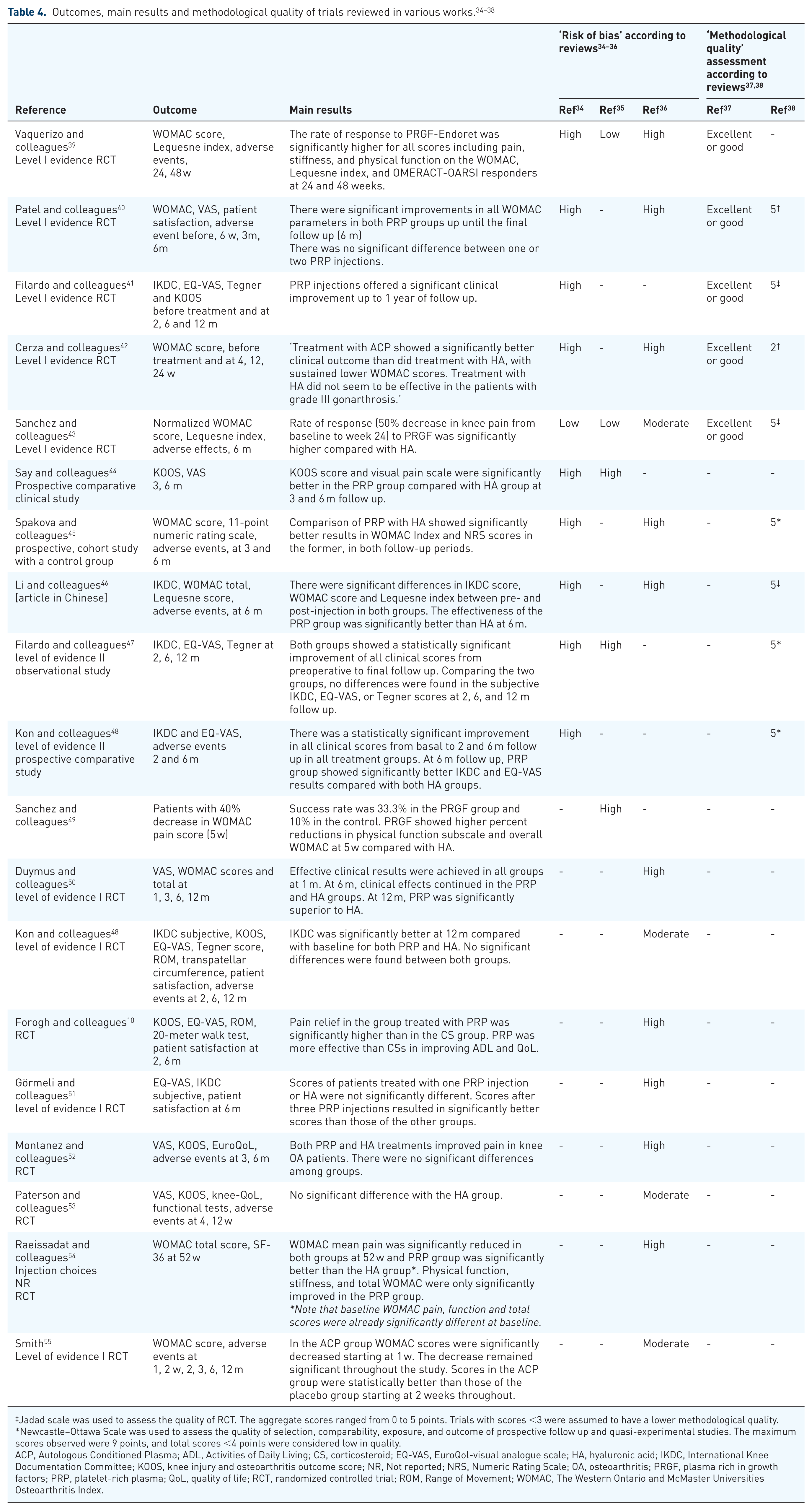
Here we comment on available evidence compiled from five recent meta-analyses and systematic reviews.34–38 These included randomized or quasi-randomized clinical trials that evaluated intra-articular (IA) injection(s) of PRP-derived products (commercial or custom protocols) with other IA interventions [hyaluronic acid (HA), corticosteroids (CSs), saline or other] for osteoarthritis (OA) treatment. A total of 19 individual trials were identified in the five reviews but only 9 were level of evidence I RCTs. Some were evaluated in more than one or even in all five reviews. Trial details are summarized in Tables 3 and 4, including the characteristics of the PRP used, the interventions, controls, outcome measures and the main results. The main conclusions of the five reviews consistently favored the use of PRP products over other IA treatments (in most cases HA), in particular in terms of pain improvement up to 12 months, but the global levels of evidence assigned in the reviews varied. Scales to evaluate methodological quality or risk of bias differed among the reviews which might account for these differences. Laudy and colleagues, Anitua and colleagues and Shen and colleagues used some version of the Cochrane collaboration risk of bias tool.34–36 Meheux and colleagues used the modified Coleman methodology score and Chang and coworkers used the Jadad scale for RCTs and the Newcastle–Ottawa Scale for quasi-experimental studies.37,38
i.e. 3/1/8 means three injections at a 1-week interval at 8 ml of PRP each.
CS, corticosteroid; DS, double spin; HA, hyaluronic acid; LP-PRP, leukocyte-poor PRP; LR-PRP, leukocyte-rich PRP; MW, molecular weight; NR, not reported; PRGF, plasma rich in growth factors; PRP, platelet-rich plasma; RCT, randomized clinical trial; SS, single spin.
is connected to the values immediately below, (5.2±1.5)X(4.8±1.8). According to Mishra classification, PRP is labelled “A” if platelet concentration is “at or above 5 times the baseline” and B otherwise, and in this case, concentration is in the cut-off number.
Jadad scale was used to assess the quality of RCT. The aggregate scores ranged from 0 to 5 points. Trials with scores <3 were assumed to have a lower methodological quality.
Newcastle–Ottawa Scale was used to assess the quality of selection, comparability, exposure, and outcome of prospective follow up and quasi-experimental studies. The maximum scores observed were 9 points, and total scores <4 points were considered low in quality.
ACP, Autologous Conditioned Plasma; ADL, Activities of Daily Living; CS, corticosteroid; EQ-VAS, EuroQol-visual analogue scale; HA, hyaluronic acid; IKDC, International Knee Documentation Committee; KOOS, knee injury and osteoarthritis outcome score; NR, Not reported; NRS, Numeric Rating Scale; OA, osteoarthritis; PRGF, plasma rich in growth factors; PRP, platelet-rich plasma; QoL, quality of life; RCT, randomized controlled trial; ROM, Range of Movement; WOMAC, The Western Ontario and McMaster Universities Osteoarthritis Index.
As a result, for instance, the RCT by Cerza and colleagues, which was included in four out of the five reviews, received different assessments depending of the review selected. Risk of bias was considered high in two of them,34,36 while methodological quality was either considered low 38 or ‘excellent or good’. 37 In another example, the RCT by Patel and colleagues 40 was considered to have a high risk of bias in two reviews,34,36 while its methodological quality was graded as excellent in the other two.37,38
The clinical trial performed by Sanchez and colleagues was generally well considered by all reviews, with low34,35 or moderate 36 risk of bias, and with ‘excellent or good’ methodological quality 37 and with the best score for RCTs. 38
Additional relevant points consistently raised in the reviews were that more high quality RCTs are needed and that PRP formulations need to be standardized to allow comparison across studies.
At present, more clinical trials are underway. At least 60 clinical trials can be found in the EU (clinicaltrialsregister.eu) and US (clinicaltrials.gov) trial registries with the search terms ‘platelet-rich plasma’ and ‘osteoarthritis’ (access date: March 4, 2018) from which further results will hopefully soon be available.
Proposal for a new approach
As mentioned, in light of the above results, current evidence seems to favor PRP over other IA treatments (HA or CSs) for the treatment of OA. However, the clinical efficacy of PRP therapy remains an open discussion. Here we advocate for a change in the focus of the field to consider PRP (and derived products) as conventional drugs, in the sense that compositions (not only platelet concentrations or preparation methods), doses, and indications with demonstrated clinical efficacy, are established, and all regardless of the patient basal platelet count. We believe this approach would facilitate the demonstration of its clinical efficacy, and therefore is more likely to result in clinically effective products.
This would involve defining additional variables for each application, which could be classified into those related to product characterization and dosage, and those related to PRP administration procedure:
(A). Product characterization and dosage (what, how much)
Product description should include volume administered and platelet absolute concentration, concentration of WBCs (including neutrophils) and RBCs, and (optimally) GFs. The concentrations of platelets, WBCs and RBCs are fairly easy to determine, but routine quantification of GF concentration is more troublesome. Further, due to the highly complex nature of PRP, it is not fully understood which of the many factors/proteins in its composition are responsible for its effects. The most abundant GFs [platelet-derived growth factor (PDGF)-BB, transforming growth factor (TGF)-β1, vascular endothelial growth factor (VEGF), and epidermal growth factor (EGF)] are the most probable candidates, but there are not many mechanistic studies that have looked at this. As an example, Bendinelli and colleagues showed that PRP anti-inflammatory effects on human chondrocytes are the result of an inhibition of nuclear factor (NF)-κB transactivation mediated by hepatocyte growth factor (HGF) using a specific competitive inhibitor of HGF (an HGF-antagonist/angiogenesis inhibitor, NK4). 56 Some research supports that platelet concentration is correlated with the concentration of GFs. Positive correlations have been found between platelet concentration and PDGF-AB and TGF-β157–59; VEGF57,58; and EGF and PDGF-BB. 57 But some other studies found poor or no correlations.60–62 Therefore, standardizing the platelet dose administered might not warrant equivalent efficacy.
In this context, as with cellular therapy products and biologicals, the application of potency assays may play a key role in defining the quality of PRP-derived products and assuring its efficacy for the desired indication.63,64 Potency is the specific ability or capacity of a product, as indicated by appropriate laboratory tests, to effect a given result.65,66 In the case of PRP-derived products, given their high complexity, a combination of multiple biologic or analytical assays (i.e. an assay matrix) should be considered. 67 Ideally, these assays should be rapid, sensitive, reproducible and cost-effective. This approach has already been used, for example, to evaluate the potential benefit of PRP treatment in patients with long bone nonunion, 68 and to establish a relevant angiogenic potency assay for a commercial product. 69 Normalizing the use of potency assays in commercial PRP products may be helpful to fully characterize available PRP products and evaluate their different biological effects (anti-inflammatory, chondroprotective, proliferative effects, etc.) to select the most appropriate product for each indication. 67
(B). Procedure variables (when, where, how, how often)
Variables such as proper timing, treatment periodicity, location and technique for injection would need to be selected to establish efficacy in each indication. Clinical trials included in Tables 3 and 4 are very dissimilar with respect to these details.
Interventions vary from a single injection,10,40,44 two monthly injections, 54 three injections at 15-day intervals48,52 or 21-day intervals;41,46 however, the most frequent treatment strategy is to apply 3-weekly PRP injections.39,43,45,47,49,51,53,55 With respect to indications, inclusion criteria were also diverse across studies. It is relevant that some trials discriminated between patients with early and consolidated OA using the Kellgren-Lawrence (K–L) grading system,48,52 the Ahlbäck classification 40 or cartilage degeneration by magnetic resonance imaging. 41 These trials have consistently found that PRP, compared with HA, showed better performance in younger patients affected by cartilage lesions or early OA than in older patients or with more advanced pathology.40,41,48,52
But, in addition, it should be taken into account that OA is a very heterogeneous and multifactorial pathology. It is now known that in OA patients who present the same symptoms (disease), the underlying mechanisms causing them (illness) might be different. 70 Different OA phenotypes are emerging that will most likely require different treatment approaches. Notably, mechanical, inflammatory and metabolic phenotypes have been proposed. 71 Therefore, PRP might be effective for some patients but not others. In particular, treatment with PRP-derived products might only be effective in patients with ‘inflammatory’ or ‘mechanical’ OA due to PRP’s anti-inflammatory and regenerative potential suggested by existing preclinical and clinical trials. Clinical efficacy might be easier to demonstrate if OA patients are stratified.
Location and technique for the injection, as well as post-injection recommendations are, also very diverse. As for location and technique, various approaches have been used including lateral,41,45,48 supero-lateral,10,42 para-patellar, 55 and lateral mid-patellar, 54 among others. After-injection recommendations also vary, including rest (10 or 20 min of immobilization40,51,54); movement (flexion and extension, 5 min 39 ; passive flexion and extension 10 times, followed by 10 min rest in the supine position 53 ; bend and extend the knee a few times to allow the PRP to spread throughout the joint before becoming a gel41,48); or even a combination of both, independent of the order: passive flexion and extension 10 times, followed by 10 min rest in the supine position 10 or 15–20 min of rest, followed by active knee flexing and extending so PRP could spread evenly. 54 But this variability is not likely to affect efficacy.
Novel approaches: allogenic PRPs
At least three recent works have described the use of allogenic PRP clinically. Smrke and colleagues performed a case study in which a 50-year-old male with type 2 diabetes suffering from a comminuted fracture of the tibia and delayed union was treated with a graft composed of allogenic platelet gel mixed with autologous cancellous bone. 72 To avoid immunogenic reactions, the allogeneic PC was ABO- and RhD-matched, leukocyte-depleted, irradiated and activated by human thrombin. At week 14 after the procedure, no platelet or human leukocyte antigen (HLA) class I antibodies were detected. At 12 months after the procedure, the non-union was resolved, and full load-bearing was achieved.
As a follow up, a prospective clinical study was conducted by the same group to treat long bone nonunions using the same type of allogenic product (allogenic platelet gel and autologous cancellous bone) in nine patients. 73 They used random single-donor allogenic PCs [ABO and RhD-matched, serologically HIV, hepatitis B virus, hepatitis C virus and lues-negative, leukocyte-depleted, and irradiated] from standard blood bank stocks. As in the case study, screening for HLA antibodies class I, and human platelet antibodies was performed before implantation and after 3 months, also without detecting any sign of immunologic reactions. At 1 year after surgery, seven out of the nine patients treated achieved complete healing.
More recently, Bottegoni and colleagues performed a prospective open-label, uncontrolled, single-center, pilot study with 60 patients. 74 Participating patients (aged 65–86 years) suffered symptomatic early or moderate knee OA (Ahlbäck grade I–III) and were affected by hematologic disorders, preventing autologous PRP treatment. Effectiveness, as measured with the International Knee Documentation Committee (IKDC), knee injury and osteoarthritis outcome score (KOOS) and EuroQol-visual analogue scales (EQ-VAS), was varied. As noted in other trials, younger patients with lower degree of degeneration showed a better response. In addition, they did not report any severe complications related to the allogenic nature of the PRP.
Interestingly, a multicentric study was recently performed to standardize a clinical grade procedure for the preparation of allogeneic PCs from umbilical cord blood. 75 As mentioned by the authors, many umbilical cord blood units donated for hematopoietic transplant are deemed unsuitable due to low stem cell content. This prompted some banks from the Italian Cord Blood Network to perform a cooperation program among delivery rooms, cord blood banks and blood transfusion services to develop this standardized protocol, scalable to other national centers, or for translation to other countries.
Other experts have also highlighted that this avenue ought to be explored in future research as a way to deliver PRP therapy to the highest possible number of patients, including those with hematologic disorders or elderly patients.76,77
Still, important issues need to be resolved before the use of this type of product could be generalized. Most notably, safety issues, including what measures need to be taken to eliminate the allergenic potential and to inactivate all pathogens; and secondly, regulatory issues, such as the global consideration of PRP as a drug/medicament for human use.
Final remarks/conclusions
In this work, we have reviewed current available evidence supporting the use of PRP derivatives to treat OA. At present, results from randomized clinical trials seem to favor PRP used over other IA treatments such as HA injections, to improve pain scales in the short and medium term (6–12 months). However, poor methodological quality of available trials and variability in PRP preparations, confound definite demonstration of clinical efficacy. Here we have proposed an alternative approach in which PRPs are considered as conventional drugs for human use. That is, compositions (not platelet concentrations or preparation methods), doses and indications for which clinical efficacy can be demonstrated are identified, regardless of the individual patient basal platelet count.
Additionally, RCTs in which OA patients were stratified suggest that PRP is more effective in those with lower degree of cartilage degeneration or OA grade. Therefore, careful patient stratification, even with respect to OA phenotype, should be encouraged for all trials since it might facilitate proving clinical efficacy for each indication.
The use of allogenic PRP should also be explored, provided safety and regulatory issues can be resolved satisfactorily in order to make PRP available for the highest number of patients possible.
Concluding, at present the therapeutic potential of PRP products in OA remains unfulfilled and without further standardization its clinical efficacy will remain an open debate.
Footnotes
Acknowledgements
FJB and EFB conceived and designed the study. LGC, JM and CRR collected and assembled the data. LGC and EFB drafted the article. All authors performed review/editing of the manuscript and approved the final version before submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LGC is supported by contract FPU13/06041 (Ministerio de Educación, Cultura y Deporte). CRR is supported by the Miguel Servet program contract CPII15/00013 and EFB is supported by Grant PMP15/00032 both from Fondo Investigación Sanitaria-Spain. This work was supported by grants PI12/00329 and PI16/02124 (Fondo de Investigación Sanitaria), IN607A 2017/11 from Axencia Galega de Innovación (GAin) and project 0245_IBEROS_1_E, an Interreg (POCTEP)-EU Program with FEDER funds. CIBER-BBN is a national initiative of the Instituto de Salud Carlos III.
Conflict of interest statement
The authors declare that there is no conflict of interest.