Abstract
Objective
This study aimed to provide an updated assessment of the clinical efficacy and safety of arthroscopic debridement combined with platelet-rich plasma injection for the treatment of knee osteoarthritis.
Methods
On 16 August 2025, we systematically searched multiple databases, including the Cochrane Library, Embase, Ovid Medline, PubMed, Web of Science, China Biology Medicine Disc, China National Knowledge Infrastructure, Wanfang, and VIP Chinese Science and Technology Journal Database (VIPC), for randomized controlled trials evaluating the efficacy of arthroscopic debridement combined with platelet-rich plasma injection in the management of knee osteoarthritis. Outcome measures included the following: (a) clinical response rate; (b) visual analog scale score; (c) Lysholm score; (d) Western Ontario and McMaster Universities Osteoarthritis Index score; and (e) adverse reactions. Data were recorded and analyzed using RevMan 5.4 software. The systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, and the research protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO) platform (registration number: CRD420251127571).
Results
A total of 17 studies involving 1587 patients were included. Meta-analysis results showed that compared with the control group, the treatment group demonstrated a statistically significant difference in the overall clinical response rate: odds ratio = 5.38; 95% confidence interval: 3.45, 8.38; p < 0.00001. Subgroup analyses based on follow-up duration (3, 6, and 12 months) demonstrated a p value of 1.00 and I² value of 0%. Meta-analysis results for each subgroup were as follows: odds ratio = 5.66; 95% confidence interval: 2.14, 14.94; p = 0.0005 for 3 months; odds ratio = 5.68; 95% confidence interval: 2.87, 11.27; p < 0.00001 for 6 months; and odds ratio = 5.40; 95% confidence interval: 2.44, 11.96; p < 0.0001 for 12 months. The treatment group also exhibited a statistically significant reduction in visual analog scale scores: mean difference = −0.62; 95% confidence interval: −1.03, −0.20; p = 0.004. Lysholm scores increased significantly in the treatment group: mean difference = 5.07; 95% confidence interval: 2.99, 7.14; p < 0.00001. The Western Ontario and McMaster Universities Osteoarthritis Index score indicated statistically significant difference: mean difference = −8.65; 95% confidence interval: −11.13, −6.18; p < 0.00001. Adverse reaction rate demonstrated no statistically significant difference: odds ratio = 0.73; 95% confidence interval: 0.28, 1.89; P = 0.52.
Conclusion
The overall clinical response rate of arthroscopic debridement combined with platelet-rich plasma injection for knee osteoarthritis was significantly higher than that of the control group. The visual analog scale scores were lower, indicating effective pain relief in patients with knee osteoarthritis. The Lysholm scores were higher, demonstrating a significant improvement in knee function compared to the control group. The Western Ontario and McMaster Universities Osteoarthritis Index scores were significantly lower than that of the control group, reflecting substantial efficacy of arthroscopic debridement combined with platelet-rich plasma injection in treating knee osteoarthritis by improving pain, stiffness, and functional impairment. The incidence of adverse reactions in the arthroscopic debridement combined with platelet-rich plasma injection group did not differ significantly from that in the control group, indicating that platelet-rich plasma injection does not increase the risk of adverse events.
Keywords
Introduction
Knee osteoarthritis (KOA), one of the most common degenerative joint diseases, is pathologically characterized by cartilage and extracellular matrix deterioration, synovial inflammation, and subchondral bone remodeling. Clinically, it primarily presents as localized pain, deformity, and joint dysfunction, significantly affecting patients’ quality of life. 1 Studies have indicated that the global burden of KOA continues to increase, affecting approximately 7.6% of the world’s population. This burden is projected to rise by 60% to approximately 100% by 2050. 2 Currently, primary clinical management include medication (analgesics, topical, and oral nonsteroidal anti-inflammatory drugs), functional exercises, and reconstructive therapies. 3 In vitro and in vivo studies highlight the role of platelets in angiogenesis and tissue repair. 4 Platelet-rich plasma (PRP) is a concentrated preparation of platelets and growth factors obtained by centrifuging autologous whole blood. Injection of PRP at injury sites may promote tissue repair by releasing bioactive factors (growth factors, cytokines, and lysosomes) and adhesion molecules that trigger hemostatic cascade, new connective tissue synthesis, and vascular remodeling. 5 As an autologous biological agent, PRP shows promise in the management of musculoskeletal disorders. Although some controversy exists, current evidence supports its use in conditions such as mild-to-moderate osteoarthritis. 6
Arthroscopic joint debridement is an effective option for short-term symptom relief in patients with KOA. This procedure typically involves synovectomy, chondroplasty, loose body removal, meniscectomy, and ligament release. 7 Arthroscopy is a highly effective surgical tool, and careful patient selection enables clinicians to deliver effective treatment that aligns with evidence-based medical practice. 8 Research indicates that combining arthroscopic debridement and PRP injections effectively alleviate pain and improve joint function in patients with KOA, 9 and clinical studies investigating this combined approach have been increasing rapidly. Although several systematic reviews and meta-analyses have investigated this approach, the therapeutic efficacy of this combination remains controversial.10–12 Therefore, this study aimed to analyze the efficacy and safety of arthroscopic debridement combined with PRP injection in managing KOA through an updated systematic review, thereby providing evidence-based medical guidance for its clinical application.
Data retrieval and methods
Literature search strategy
Computer-assisted searches were conducted in the Cochrane Library, Embase, Ovid Medline, PubMed, Web of Science, China Biology Medicine Disc (CBM), China National Knowledge Infrastructure (CNKI), Wanfang, VIP Chinese Science and Technology Journal Database (VIPC), and other databases to identify randomized controlled trials (RCTs) evaluating arthroscopic debridement combined with PRP injection for KOA. The search covered literature from the inception of each database to 16 August 2025. Search terms included “Osteoarthritis,” “Platelet-Rich Plasma,” “Arthroscopy,” and “randomized controlled trial,” using a combination of subject headings and free-text terms. This systematic review was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and checklist, 13 and the research protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO) platform (Registration Number: CRD420251127571).
Inclusion criteria for literature
The inclusion criteria were as follows: (a) Study type. Published RCTs evaluating arthroscopic debridement combined with PRP injection for KOA, with no language restrictions; (b) Study design. No restrictions on blinding and allocation concealment; (c) Study population. Participants of any age who met the diagnostic criteria for KOA; (d) Interventions. Control group treated with arthroscopic debridement and the treatment group with a combination of anthroscopic debridement and PRP injection; and (e) Outcome measures. Studies must have included at least one of the following outcomes, including clinical efficacy rate, adverse reactions, visual analog scale (VAS) score, Lysholm score, or Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score.
Literature exclusion criteria
The exclusion criteria were as follows: (a) duplicate publications; (b) irrelevant literature, including lectures, systematic reviews, reviews, animal studies, case reports, and nonrandomized controlled trials; (c) studies with noncompliant intervention measures; (d) studies with ≤30 cases per group; (e) studies with noncompliant outcome measures; (f) studies with duplicate publications reporting identical results, in which case only the highest-quality paper was selected; (g) low-quality literature, defined as studies with modified Jadad scores of ≤1; and (h) studies for which full-text documents were unavailable.
Literature screening and data extraction
All retrieved literature was independently reviewed by two researchers. First, duplicate records were removed using NoteExpress software. Second, preliminary exclusion was conducted by examining titles and abstracts according to the inclusion and exclusion criteria. Finally, full-text articles were critically appraised to determine final inclusion strictly based on the criteria. In cases of disagreement regarding inclusion, the researchers initially discussed and analyzed the issues; if consensus was not reached, a third researcher made the final decision. Data were extracted by the two researchers from the included studies, including information on first author, publication year, baseline comparability (sex, age, disease duration, and pretreatment efficacy indicators), sample size, intervention measures, adverse reactions, and outcome measures. Extracted data were cross-checked by two evaluators, and, once confirmed for accuracy, statistical analysis was performed.
Bias risk assessment and quality evaluation
Multivariate assessments were conducted using the Cochrane Risk of Bias Tool, specifically evaluating the following: (a) random sequence generation (selection bias); (b) allocation concealment (selection bias); (c) blinding of participants and personnel (performance bias); (d) blinding of outcome assessors (detection bias); (e) completeness of outcome data (attrition bias); (f) selective reporting of results (reporting bias); (g) other potential biases. Two researchers independently assessed each identified study according to the Cochrane Risk of Bias Assessment Manual, assigning the risk of bias as low, high, or unclear. Any discrepancies were resolved through discussion or consultation with a third researcher. Findings were recorded and visualized using RevMan 5.4 software (Review Manager 5.4, The Cochrane Collaboration, London, UK).
The quality of the included literature was assessed using the modified Jadad scoring system, focusing on four key criteria: random sequence generation, allocation concealment, use of blinding, and withdrawal or dropout rates. A composite score was calculated based on these criteria, with scores of 1–3 indicating low quality and 4–7 indicating high quality. The final assessments were cross-verified by two reviewers, and any discrepancies were resolved by a third researcher.
Statistical analysis
Statistical analyses were performed using RevMan 5.4 software. Heterogeneity among the included studies was assessed. A p value of >0.1 and an I² value of <50% were considered indicative of acceptable heterogeneity, allowing the use of a fixed-effects model. When the p value was ≤0.1 or I² value was ≥50%, heterogeneity was considered significant, and further analyses were conducted to identify potential sources of variation. For categorical data, the odds ratio (OR) was used as the effect measure, whereas the weighted mean difference (WMD) was used for continuous data. All effect measures were calculated with 95% confidence intervals (CI). A p value <0.05 was considered indicative of statistical significance. Sensitivity analyses were conducted by sequentially excluding individual studies or reestimating the pooled results to evaluate the robustness of the findings. For outcomes including more than 10 studies, funnel plots were used to assess potential publication bias.
Results
Literature search results
Searching the databases yielded 477 articles: Cochrane Library (18), Embase (63), Ovid (79), PubMed (16), Web of Science (69), CBM (31), CNKI (107), Wanfang (42), and VIPC (52). Using NoteExpress document management software, 76 duplicate publications were removed. Initial screening based on titles and abstracts eliminated 358 publications, including lecture materials, systematic reviews or review articles, animal studies, case reports, and irrelevant papers. After full-text review, 26 additional publications were excluded. Ultimately, 17 publications were included in this study.14–30 The literature search and screening process is illustrated in Figure 1.

Flowchart of the literature search and screening process.
Basic characteristics and quality assessment of included literature
This study included 17 published articles involving a total of 1587 patients. Of these, 833 patients were assigned to the treatment group and 754 to the control group. The basic characteristics of each included study are presented in Table 1. The publication years of the included studies ranged from 2015 to 2025. All studies described the use of “random” methods; however, only 10 studies14,16,17,19,20,23–26,28 reported complete random allocation procedures. The remaining 7 studies15,18,21,22,27,29,30 mentioned “random” but did not specify the procedures. Nine studies14,16,17,20,23–26,28 used random number tables for allocation concealment, one study 22 used sealed opaque envelopes, and the remaining studies did not specify the concealment method. Among all studies, two22,30 implemented double-blind methods, whereas the others did not report blinding. The quality assessment results of each study are presented in Figures 2 and 3.
Basic characteristics of the included studies.

PRP: platelet-rich plasma.
① Clinical effective rate; ② VAS score; ③ Lysholm score; ④ WOMAC score; ⑤ Adverse reaction.

Summary of risk of bias in the included trials.

Risk of bias analysis of the included trials.
Observation indicators
Clinical response rate
Eleven studies14,18–21,23–25,28–30 were included for comparison of the overall clinical response rate, involving a total of 1039 patients: 545 in the treatment group and 548 in the control group. Heterogeneity testing demonstrated a p value of 1.00 and I² value of 0%, indicating no statistically significant heterogeneity among the studies. Therefore, a fixed-effects model was applied. The pooled results were as follows: OR = 5.38; 95% CI: 3.45, 8.38; p < 0.00001. The difference was statistically significant, indicating that the overall clinical response rate of arthroscopic debridement combined with PRP injection for KOA was significantly higher than that of the control group, as shown in Figure 4.

Clinical efficacy rate.
Subgroup analyses were conducted according to follow-up duration (3 months, 6 months, and 12 months), yielding a p value of 1.00 and I² value of 0%. These results indicated minimal heterogeneity among the three follow-up subgroups, with no statistically significant heterogeneity. The results were as follows: 3 months: OR = 5.66, 95% CI: 2.14, 14.94, p = 0.0005; 6 months: OR = 5.68, 95% CI: 2.87, 11.27, p <0.00001; and 12 months: OR = 5.40, 95% CI: 2.44, 11.96, p < 0.0001. These findings indicated statistically significant differences, demonstrating that arthroscopic debridement combined with PRP injection for KOA achieved a clinically significant overall response rate as early as 3 months posttreatment in this study. Moreover, the therapeutic effect persisted through 12 months without diminishing over the follow-up period. The results are shown in Figure 5.

Forest plot of subgroup analysis of clinical efficacy.
VAS score
Twelve studies14–24,28 compared VAS scores, involving a total of 1101 patients: 544 in the treatment group and 557 in the control group. Heterogeneity testing demonstrated a p value of <0.00001 and I² value of 98%, indicating statistically significant heterogeneity among the studies. Exclusion of individual studies did not substantially alter heterogeneity, and no clear source of heterogeneity was identified. Therefore, a random-effects model was applied. The pooled results were as follows: mean difference (MD) = −0.62, 95% CI: −1.03, −0.20), p = 0.004. The difference was statistically significant, indicating that the VAS scores for KOA were lower in the treatment group than in the control group. These results demonstrated that arthroscopic debridement combined with PRP injection effectively reduces pain in patients with KOA, as shown in Figure 6.

Forest plot of VAS scores. VAS: visual analog scale.
Lysholm score
Eleven studies14–20,22,23,25,27,28 compared Lysholm scores in 985 patients, with 486 in the treatment group and 499 in the control group. Heterogeneity testing demonstrated a p value of <0.00001 and I² value of 91%, indicating statistically significant heterogeneity among the studies. Exclusion of individual studies did not substantially alter heterogeneity, and no clear source of heterogeneity was identified. Therefore, a random-effects model was applied. The pooled results were as follows: MD = 5.07, 95% CI: 2.99, 7.14, p < 0.00001. The difference was statistically significant, indicating that arthroscopic debridement combined with PRP injection significantly improved knee function in patients with KOA compared to the control group, as shown in Figure 7.
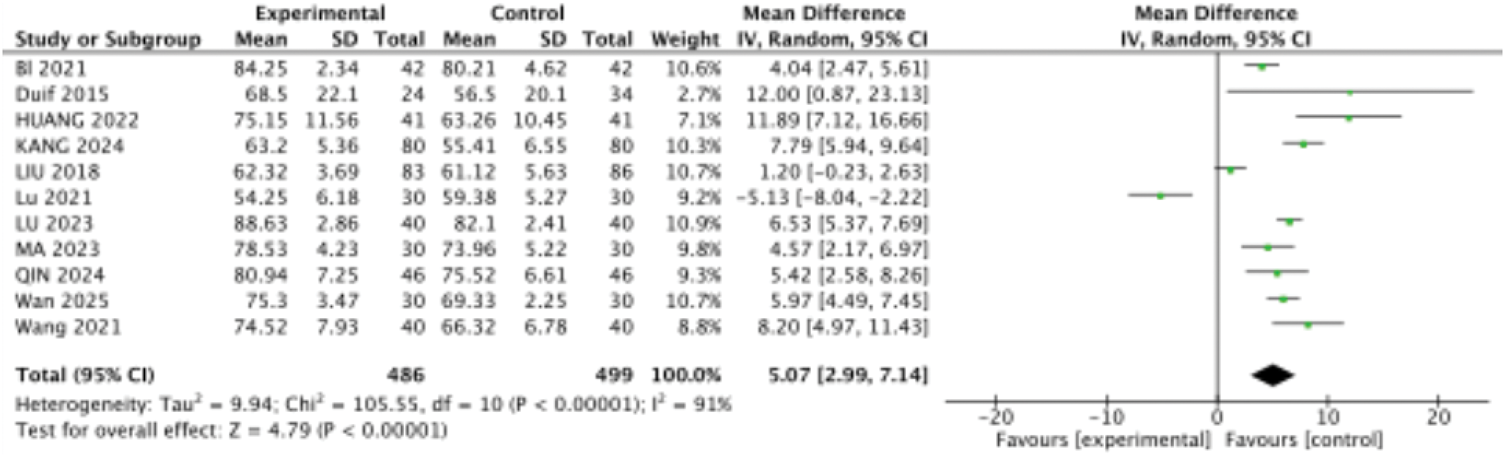
Forest plot of Lysholm scores.
WOMAC score
Three studies14,23,24 assessed WOMAC scores in 282 patients, including 141 in the treatment group and 141 in the control group. Heterogeneity analysis demonstrated a p value of 0.005 and I² value of 81%, indicating statistically significant heterogeneity. Sequential exclusion of individual studies revealed that removing the study by Li 23 (2025) resulted in a p value of 0.61 and I² value of 0%. The original studies suggested that heterogeneity likely arose from differences in follow-up duration: the study by Li 23 (2025) had a 6-month follow-up, that by Huang 14 (2022) had a 6-week follow-up, and that by Qin 24 (2024) had a 1-month follow-up. These findings indicate that variations in follow-up duration may be a primary source of heterogeneity.
After excluding the LI 2025 study, p = 0.61 and I² = 0%, a fixed-effects model was applied. The results demonstrated an MD of −8.65 (95% CI: −11.13, −6.18; p < 0.00001). This difference was statistically significant, indicating that arthroscopic debridement combined with PRP injection for KOA resulted in significantly lower WOMAC scores than in the control group. This combination therapy effectively improved patients’ pain, stiffness, and functional impairment (Figure 8).

Forest plot of WOMAC scores. WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
Adverse reactions
Four studies16–18,28 compared adverse reaction rates, including 199 patients in the treatment group and 202 patients in the control group, for a total of 401 cases. Heterogeneity analysis revealed a p value of 0.36 and I² value of 0%, indicating no statistically significant heterogeneity among the studies. Therefore, a fixed-effects model was applied. The results were as follows: OR = 0.73; 95% CI: 0.28, 1.89; p = 0.52, indicating that the incidence of adverse reactions in the arthroscopic debridement combined with PRP injection group did not differ significantly from that in the control group. These findings suggested that no significant difference in safety was observed between the two groups and that compared with the control group, PRP injection does not increase the risk of adverse events (Figure 9).

Forest plot of adverse reactions.
Publication bias
For the three outcome measures, including overall efficacy rate, VAS score, and Lysholm score, more than 10 studies were included and analyzed using funnel plots to assess publication bias (Figures 10–12). The results indicated that the distributions across the three funnel plots were not entirely symmetrical, suggesting the presence of potential publication bias. Sensitivity analyses performed using the one-by-one exclusion method for the three outcome measures yielded robust and stable results.
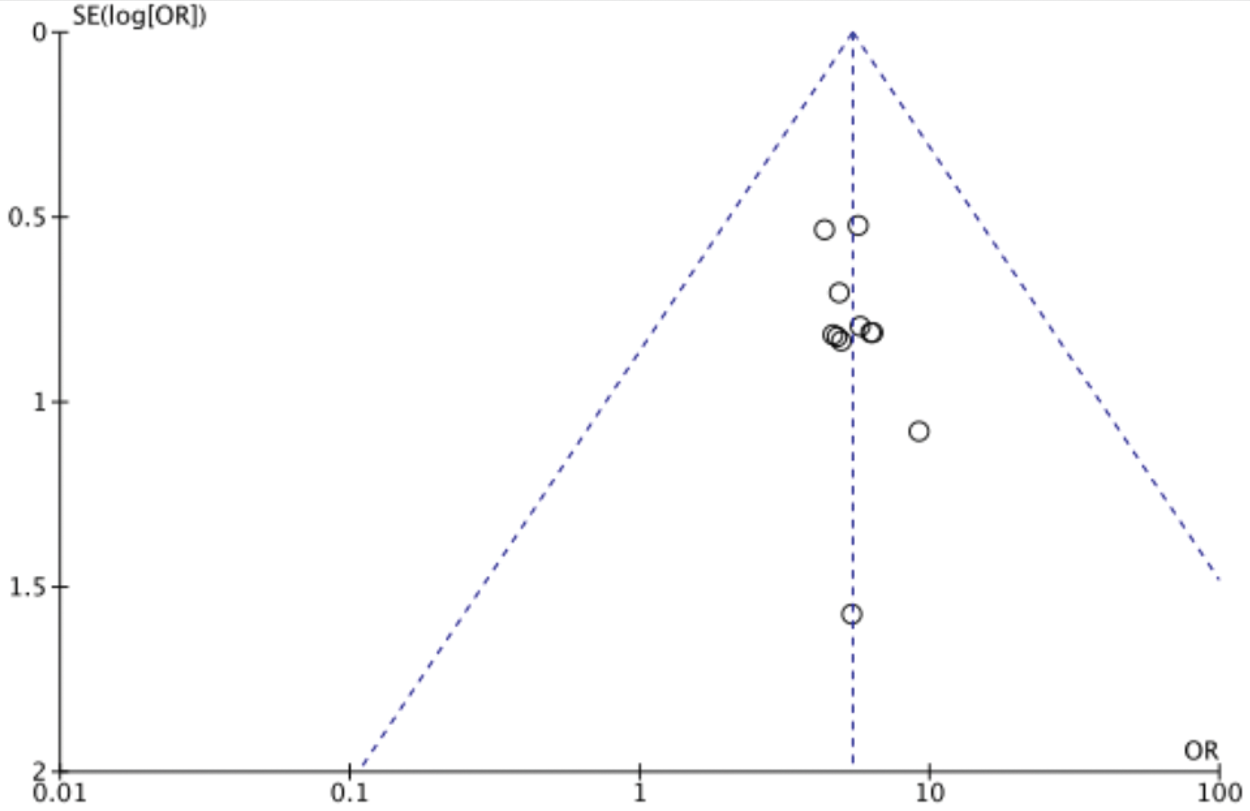
Funnel plot of total clinical efficacy.

Funnel plot of VAS scores. VAS: visual analog scale.
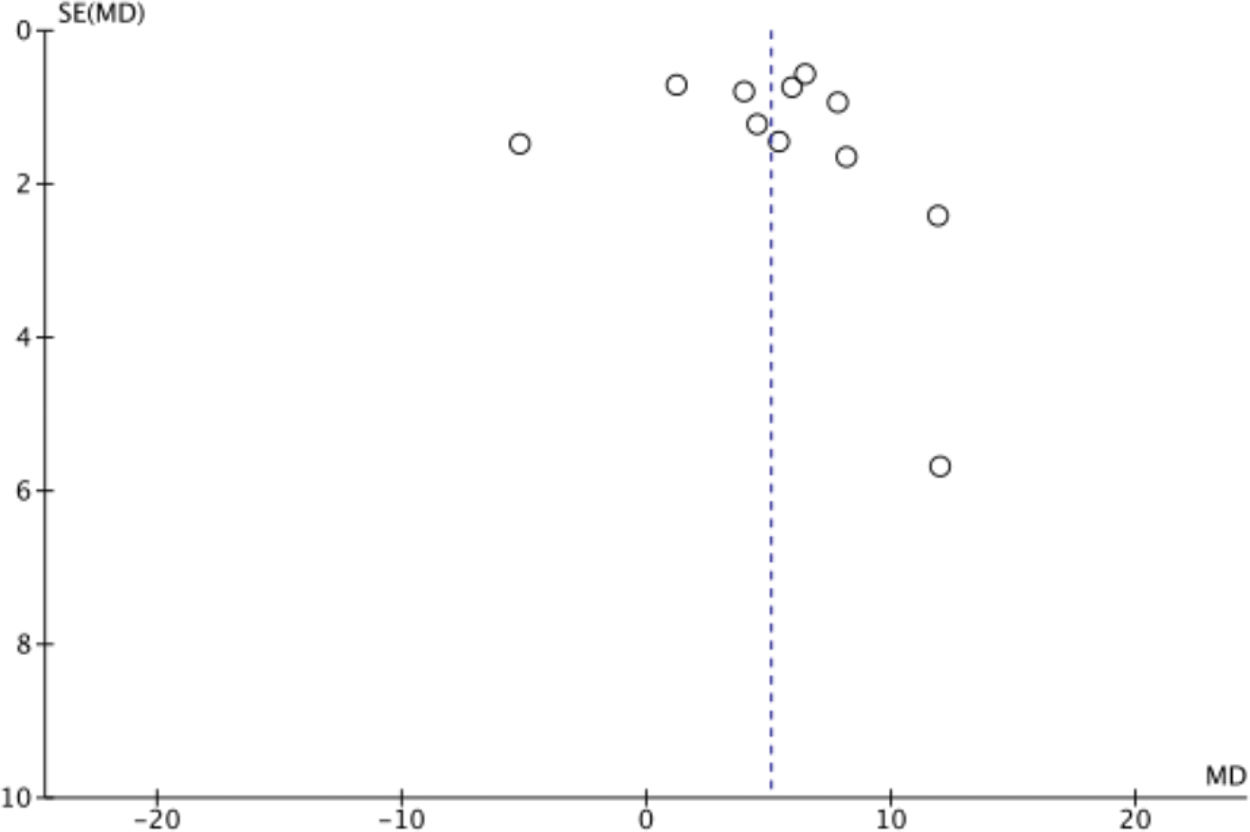
Funnel plot of Lysholm scores.
Discussion
Efficacy of arthroscopic debridement combined with PRP therapy for KOA
This study included 17 RCTs involving a total of 1587 patients. The findings of this study are consistent with those of the previous two meta-analyses,11,12 confirming that arthroscopic debridement combined with PRP injection is a reliable and safe treatment for KOA. The meta-analysis revealed that the total effective rate in the group treated with arthroscopic debridement combined with PRP therapy for KOA was significantly higher than that in the control group. The therapeutic effect manifested rapidly and remained stable for at least one year. In addition, the treatment group demonstrated superior VAS, Lysholm, and WOMAC scores compared with those in the control group, indicating significant therapeutic benefits. Future studies should focus on identifying key factors influencing treatment efficacy, such as follow-up duration, PRP dosage, and the severity of KOA.
Safety of arthroscopic debridement combined with PRP therapy for KOA
Four studies16–18,28 reported adverse reactions. The meta-analysis indicated high consistency among the findings, with no significant differences observed in safety profiles. PRP, an autologous blood concentrate rich in platelets and other bioactive substances, demonstrates good safety. It effectively alleviates pain symptoms in patients with knee joint conditions and improves activities of daily living and quality of life in patients with KOA. 31 Arthroscopic debridement is a minimally invasive procedure widely used in clinical practice. It offers advantages such as minimal trauma, fewer adverse reactions, and the ability to bear full weight within one day postoperatively, resulting in high patient acceptance. 32 The safety of both treatments has also been demonstrated in previous studies.33,34
Possible mechanisms of action
The precise pathogenesis of osteoarthritis remains unclear. 35 However, multiple proinflammatory cytokines and growth factors have been identified in osteoarthritic joints and are known to play significant roles in disease progression. Research indicates that several key molecules serve as therapeutic targets for the management of osteoarthritis. 36 Inflammatory mediators such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-1 (IL-1) can activate multiple signaling pathways, including NF-κB, MAPK, JAK/STAT, and PI3K/AKT/mTOR. Through multilevel phosphorylation cascades, these mediators ultimately enter the nucleus to regulate the expression of key genes associated with osteoarthritis progression, such as matrix metalloproteinases (MMPs), A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) enzymes, TNF-α, IL-1β, and IL-6. This triggers cell proliferation, differentiation, inflammation, apoptosis, and cartilage degradation, thereby contributing to the pathogenesis of osteoarthritis. 37 The advantage of PRP lies in its ability to stimulate growth factor production and suppress inflammatory mediators. Upon activation, platelets rapidly release multiple growth factors from alpha granules, including transforming growth factor-beta (TGF-β) and insulin-like growth factor-1 (IGF-1). These growth factors act synergistically with coagulation factors, cytokines, and other platelet proteins to influence chondrocytes, promote chondrocyte-derived cartilage matrix synthesis, and mitigate the inflammatory effects of cytokines associated with osteoarthritis progression. 38 Arthroscopic debridement stimulates new cartilage formation by creating microfractures in the underlying bone tissue. These microfractures allow bone marrow to infiltrate the defect area, forming a “super blood clot” that delivers growth factors and stem cells to the injury site. This process facilitates healing by promoting the formation of a fibrocartilaginous layer within the damaged region. 39 Combining PRP therapy with arthroscopic debridement in KOA treatment can further suppress the expression of inflammatory factors and activate relevant growth factors, thereby enhancing the healing process.
Limitations and outlook
Although arthroscopic debridement combined with PRP injection for KOA is widely discusses in clinical practice, it remains controversial. This combined therapy has not yet been universally recommended by international guidelines; therefore, the clinical dosage of PRP, treatment duration, and optimal therapeutic concentration remain undetermined. Future research is required to explore these areas in greater depth, aiming to standardize PRP preparation protocols, including platelet concentration, white blood cell content, and red blood cell contamination, as well as establish the optimal treatment duration and injection frequency. Such advances will facilitate the development of internationally accepted clinical practice guidelines to guide the broader application of PRP.
This study provided further insights by sequentially excluding included studies to assess heterogeneity in WOMAC scores, suggesting that the efficacy of arthroscopic debridement combined with PRP injection for KOA may be related to treatment duration. However, the study has certain limitations. Some of the included studies were of lower quality, and the sample sizes were relatively small. Consequently, subgroup analyses examining KOA severity and the effects of PRP injection concentration or dosage on treatment efficacy could not be performed. Future research should focus on high-quality, large-sample, multicenter, and long-term RCTs to enable more robust systematic reviews and meta-analyses. This will provide stronger evidence-based support for the clinical application and development of these therapies.
Footnotes
Acknowledgments
Language refinement and grammar checking for this paper were assisted by DeepL. The authors assume full responsibility for all scientific content and have carefully reviewed and revised the generated text.
Author contributions
Li Fengzhen and Lu Jinyan drafted the initial manuscript. Wu Fengbiao performed the software analysis. Xu Zichun managed the data, and Chen Yulong provided methodological guidance. Zhou Honghai contributed to drafting sections of the text and completed the final review. All authors reviewed and approved the manuscript.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
None.
