Abstract
Patients with functional dyspepsia, defined in the 2016 Rome IV criteria as bothersome clinical dyspepsia symptoms, experience markedly reduced quality of life. Several etiologies have been associated with the disorder. In the Rome IV criteria, the brain–gut axis was acknowledged as an important factor in the etiology of functional gastrointestinal (GI) disorders. The distinct subgroups of functional dyspepsia, epigastric pain syndrome (EPS) and postprandial distress syndrome (PDS), are treated differently: acid secretion inhibitors are recommended with patients with EPS, whereas prokinetic drugs as mosapride and acotiamide are recommended for patients with PDS. A previous study has reported that proton pump inhibitors (PPIs) and H2-blockers were equally effective in functional dyspepsia. A new drug, acotiamide, a muscarinic antagonist and cholinesterase inhibitor, has been shown to improve gastric motility in rodents and dogs, and to reduce PDS symptoms in patients in double-blind multicenter studies. The pharmacological mechanisms of acotiamide remain unknown; whether acotiamide alters gastric emptying and gastric accommodation in patients with functional dyspepsia remains an open question. Other emerging treatment options include Rikkunshito, a herbal medicine that improves gastric emptying through 5-hydroxytryptamine (5-HT)2B-mediated pharmacological action, and tricyclic antidepressants (TCAs). Different drugs are needed to accommodate the clinical symptoms and etiology in individual patients.
Introduction
Functional dyspepsia is a common disorder and can markedly impair the patients’ quality of life. Based on the Rome III classification criteria, the main symptoms of functional dyspepsia consist of bothersome postprandial fullness, early satiety, epigastralgia, and epigastric burning. 1 In 2014, the guideline for functional dyspepsia patients was also provided in Japan. 2 Functional dyspepsia is treated by two major categories of drugs: acid inhibitors such as H2-receptor antagonists and proton pump inhibitors (PPIs), and prokinetic drugs that accelerate disturbed gastrointestinal (GI) motility by modifying altered visceral sensitivity. In 2016, Rome IV criteria defined that the diagnosis of functional dyspepsia required bothersome clinical symptoms, and the brain–gut axis was acknowledged as an important factor in the etiology of functional GI disorders.3,4
Pathophysiology
Visceral hypersensitivity, 5 impaired gastric accommodation 6 and impaired gastric emptying are commonly reported by patients with functional dyspepsia.7–9 Involvement of several other mechanisms has also been suggested, including duodenal hypersensitivity to the luminal contents, small bowel dysmotility, psychological disturbances, 10 central nervous system disorders and Helicobacter pylori infection.11,12
H. pylori
Recently, the H. pylori eradication therapy was proposed as first-line treatment for H. pylori-infected dyspeptic patients, 13 however, the main justification for H. pylori eradication in patients with functional dyspepsia may be related to other potential beneficial effects such as prevention of gastric cancer and recurrence of gastric ulcer rather than symptomatic improvement. 14
Although H. pylori infection may affect gastroduodenal motility and viscerosensory function, the precise mechanism of its benefits in functional dyspepsia is not clear.15–18 Therefore, although H. pylori eradication does not have a direct beneficial effect on of the symptoms of functional dyspepsia, recent studies have reported that long-term H. pylori eradication therapy improved the symptoms of functional dyspepsia 19 and according to the Kyoto consensus meeting documents, symptoms were cured from 6 months to 1 year after eradication therapy in functional dyspepsia patients with H. pylori. 13 Further studies will be needed to confirm the long-term effect of eradication therapy. 20
Delayed gastric emptying
Delayed gastric emptying has been reported by gastric scintigraphy in a large proportion (up to 45%) of dyspeptic patients, 21 especially those with postprandial distress syndrome (PDS). Other strategies to evaluate the speed of gastric emptying include the paracetamol absorption test which can measure the gastric emptying of liquids, and the 13 C breath test, which can measure the gastric emptying of solids or liquids and can achieve an accuracy comparable with gastric scintigraphy. 22 We have previously reported that the Tmax value as the point of maximum speed of gastric emptying for a marker of gastric emptying in PDS patients was significantly greater than in healthy volunteers. 9 In addition, we have also reported that nizatidine significantly improved both gastric emptying and clinical symptoms in functional dyspepsia patients with impaired gastric emptying. 23
Subtypes and targeted therapy
The functional dyspepsia subtypes, epigastric pain syndrome (EPS) and PDS, require different treatment; patients with EPS benefit from acid secretion inhibitors, whereas patients with PDS benefit from prokinetic drugs such as mosapride and acotiamide. 2 Other treatment options in EPS include H2-blockers and PPIs.
Some patients with EPS have been reported to have an intractable disease; these are likely to relate to the various etiological factors contributing to functional dyspepsia, which overlap with other diseases such as irritable bowel syndrome (IBS), nonerosive reflux disease (NERD) and pancreatic dysfunction.
Dietary and lifestyle modification
Dietary recommendations in functional dyspepsia include eating smaller meals and avoiding high-fat meals which have been reported to aggravate clinical symptoms such as nausea and abdominal pain more than isocaloric high-carbohydrate meals. 24 In our recent study, 25 intake of dietary fat aggravated clinical symptoms of dyspepsia in patients attending the clinic.
Although Talley and colleagues reported that smoking, alcohol, aspirin and the use of nonsteroidal anti-inflammatory drugs (NSAIDs) was not associated with an increased risk of functional dyspepsia in outpatients presenting for endoscopy. 26 However, in view of the Rome IV criteria Stanghellini and colleagues recently recommended that besides more frequent, smaller meals and avoiding a high-fat diet, patients with functional dyspepsia should avoid NSAID use, coffee, alcohol, and smoking. 4 Randomized controlled trials (RCTs) and systematic studies are warranted to confirm the efficiency of adjusting of eating habits.
Standard drug treatments: anti-acid drugs
Several studies have reported that anti-acid therapy and prokinetic agents are effective for certain populations with functional dyspepsia.
H2-blocker therapy
H2-blockers (histamine H2 receptor antagonists) have been used as a first-line therapeutic drug for functional dyspepsia. In a 2012 review, Lacy and colleagues reported that in 12 RCTs that compared H2-blockers with placebo in a total of 2183 participants, 54% had a statistically significant improvement in dyspeptic symptoms with H2-blocker therapy compared with 40% in the placebo arm. 27
PPI therapy
Although PPIs also have been widely used for the treatment of dyspeptic symptoms in functional dyspepsia, evidence from RCTs suggests that the efficacy of PPIs in functional dyspepsia is limited28–30 and may be confined to those patients who have co-existing reflux symptoms. The efficacy of PPIs fails against that of placebo for nonacid-related symptoms.31–33
Comparison of PPI and H2-blocker therapy and other treatments
Generally, PPI treatment has been reported to reduce basal gastric acid secretion and gastrin-stimulated gastric acid secretion levels compared with patients treated with the H2-blocker H2RA. A comparison of PPI therapy with H2-blocker therapy in nonulcer dyspepsia showed a trend towards a better outcome based on global dyspepsia cure with a PPI, however, the difference was not statistically significant. 28 By contrast, Moayyedi and colleagues have reported that there were no differences in the effects of PPI and H2-blocker in functional dyspepsia.2,34 In the CADET-HN study, PPI treatment significantly improved clinical symptoms compared with patients with placebo and prokinetics. 35
Standard drug treatments: prokinetic drugs
Previous studies have reported that metoclopramide is not effective in functional dyspepsia.36,37 By contrast, a placebo-controlled trial reported a beneficial effect of domperidone (10–20 mg three times daily) with placebo. 38
A recent meta-analysis concluded that itopride, a D2 antagonist and acetylcholinesterase inhibitor, improves the symptoms of early satiety and postprandial fullness. 39 Moreover, itopride is more effective than domperidone in improving postprandial fullness and early satiety.40–43
Standard drug treatments: antidepressants
Since refractory functional dyspepsia involves psychiatric manifestations such as depression and anxiety, many physicians prescribe antidepressants for their patients. In Japan, according to the guidelines for the treatment of functional dyspepsia, the first-line therapy includes PPIs or prokinetics. For the treatment of refractory functional dyspepsia, 2 antidepressants such as tandospirone are used as second-line drugs in clinical practice in Japan. Koloski and colleagues have reported that antidepressant treatment significantly improves functional dyspepsia compared with placebo.44,45
Tricyclic antidepressants
Lu and colleagues reported in a systematic review that TCAs are effective in the treatment of functional dyspepsia symptoms. 46 Other studies have reported that TCAs are effective particularly for certain functional dyspepsia patients with chronic pain.44,47
However, improvement of clinical symptoms of functional dyspepsia treated with antidepressants has been reported to be limited by Talley and colleagues. 48 The disease subtype might have a role in treatment efficacy. Talley and colleagues later reported that patients with ulcer-like functional dyspepsia who are on TCAs or amitriptyline are more likely to report adequate symptom control than are patients with dysmotility-like functional dyspepsia. 47
Serotonin-specific reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs)
In contrast with TCAs, no improvement of clinical symptoms over placebo was seen in patients taking escitalopram (an SSRI).46,49 Moreover, Van and colleagues reported no significant improvements in symptom severity, anxiety score, depression score and health-related quality of life in patients receiving venlafaxine (an SNRI) compared with placebo. 49
Interestingly, according to a double-blind, placebo-controlled, multicenter study by Miwa and colleagues, the 5-HT1A agonist, tandospirone, improved abdominal symptoms such as upper abdominal pain and abdominal discomfort at 4 weeks. 50
To conclude, given the limited evidence for the efficiency of antidepressants, further studies will be needed to perform double-blind, multicenter studies to determine the usefulness of antidepressants for functional dyspepsia patients.
Acotiamide, a novel pharmacological treatment
The new drug, acotiamide, a muscarinic antagonist and cholinesterase inhibitor has been shown to improve gastric motility and gastric emptying in rodents and dogs (Table 1).51,52
Clinical symptoms and experiments for acotiamide usage.

ACh, acetylcholine; AChE, acetylcholine esterase; EPS, epigastric pain syndrome; FD, functional dyspepsia; PDS, postprandial distress syndrome.
Acotiamide monotherapy
Double-blind multicenter studies have shown a significant improvement in PDS symptoms such as postprandial fullness, early satiety, upper abdominal bloating in patients treated with acotiamide, as reported by Matsueda and colleagues. 53
In Japan, acotiamide for functional dyspepsia is covered by insurance. Therefore, several Japanese studies have reported acotiamide to be associated with improvement of clinical symptoms in patients with functional dyspepsia (Table 1),54–56 one of these was a long-term study. 57 According to our data, acotiamide treatment relatively improved lower abdominal symptoms, although this finding was not statistically significantly (Table 1). 58
Mechanisms of action
Although, as shown in Table 1, many studies have reported the relationship between gastric motility and treatment with acotiamide,59–61 there are no human data to clarify the pharmacological mechanisms of acotiamide. Thus, it is still an unsolved issue whether acotiamide alters gastric emptying and gastric accommodation in patients. Recently, in a guinea pig model, Ikeo and colleagues demonstrated that acotiamide improved stress-induced impaired gastric accommodation, 62 and Nagahama and colleagues reported oral administration of acotiamide to stimulate postprandial gastroduodenal and colonic motor activities in conscious dogs. 63
Using the 13C-acetate breath test, we have observed that acotiamide can improve functional dyspepsia symptoms and ameliorate impaired gastric emptying. 55 In our study, we tried to clarify whether acotiamide can improve gastric emptying through upregulation of acylated ghrelin and leptin levels in patients with functional dyspepsia. Interestingly, we addressed that the percentage of improvement of the acylated ghrelin/total ghrelin levels in patients treated with acotiamide was significantly higher than in patients treated with rabeprazole alone. 55 Considering previous reports that acylated ghrelin is associated with appetite and gastric emptying, 64 acylated ghrelin levels associated with acotiamide use may underlie the symptom amelioration and restore gastric emptying. Seto and colleagues have reported that acotiamide exerted an impact on the expression of genes related to the expression of neuromedin U, known as a stress-related neuropeptide. 60 Acotiamide may thus act directly on the gut and also indirectly through the brain–gut axis, potentially via as the effects of ghrelin in the central nervous system. 60 Further research in animal models and in humans is still needed to clarify whether acotiamide can affect both gastric accommodation and gastric emptying and to elucidate the pharmacological mechanisms of its action.
Acotiamide as an add-on therapy
We compared the severity of PDS and EPS symptoms in patients treated with acotiamide and rabeprazole combination therapy compared with rabeprazole monotherapy (Table 1). In our results, acotiamide–rabeprazole combination therapy significantly improved both PDS- and EPS-like symptoms compared with rabeprazole monotherapy (Table 2). These results suggest that combination therapy can stabilize both of EPS and PDS symptoms.
Efficiency of acotiamide and rabeprazole combination therapy for clinical symptoms for 4 weeks.

Clinical symptoms assessed by visual analogue scale (VAS scale: 0–10; 0 = absent, and 10 = maximal) acotiamide and rabeprazole combination therapy versus rabeprazole monotherapy.
Moreover, Mayanagi and colleagues have reported a trend towards improved symptoms with acotiamide–esomeprazole combination therapy, although the finding was not statistically significant (Table 1). 65 In patients with epigastric pain, improvement of other symptoms such as postprandial abdominal fullness may be gradually abrogated by EPS symptoms and, the patients’ quality of life may worsen. Since the majority of patients with functional dyspepsia have comorbid gastroesophageal reflux disease (GERD) and IBS, combination therapy with acotiamide and PPIs such as rabeprazole may be considered for the treatment of functional dyspepsia patients.
Vonoprazan, a drug with potential for repurposing to functional dyspepsia
In Japan, a new drug, vonoprazan, a potassium-competitive acid blocker (P-CAB) is used for the treatment of acid-related disease such as reflux esophagitis and gastric ulcers. Further studies will be needed as to whether vonoprazan can efficiently manage functional dyspepsia symptoms.
Rikkunshito: a Japanese herbal medicine treatment
In Japan, herbal medicine is very popular and covered by national health insurance. Herbal medicine is an empirical therapeutic drug with a long history. Although it is used widely, scientific evidence showing clinical benefits is scarce.
Rikkunshito is a well-known medicine in herbal agents and is prepared from eight crude herbs: Rhizoma Atractylodis lanceae, ginseng root, Pinellia tuber, Poria sclerotium, jujube, satsuma mandarin peel, licorice root, and ginger. In Japan, Rikkunshito is commonly used for dyspeptic symptoms (Table 3). 66
Efficiency of Rikkunshito in improvement for clinical symptoms and animal models.
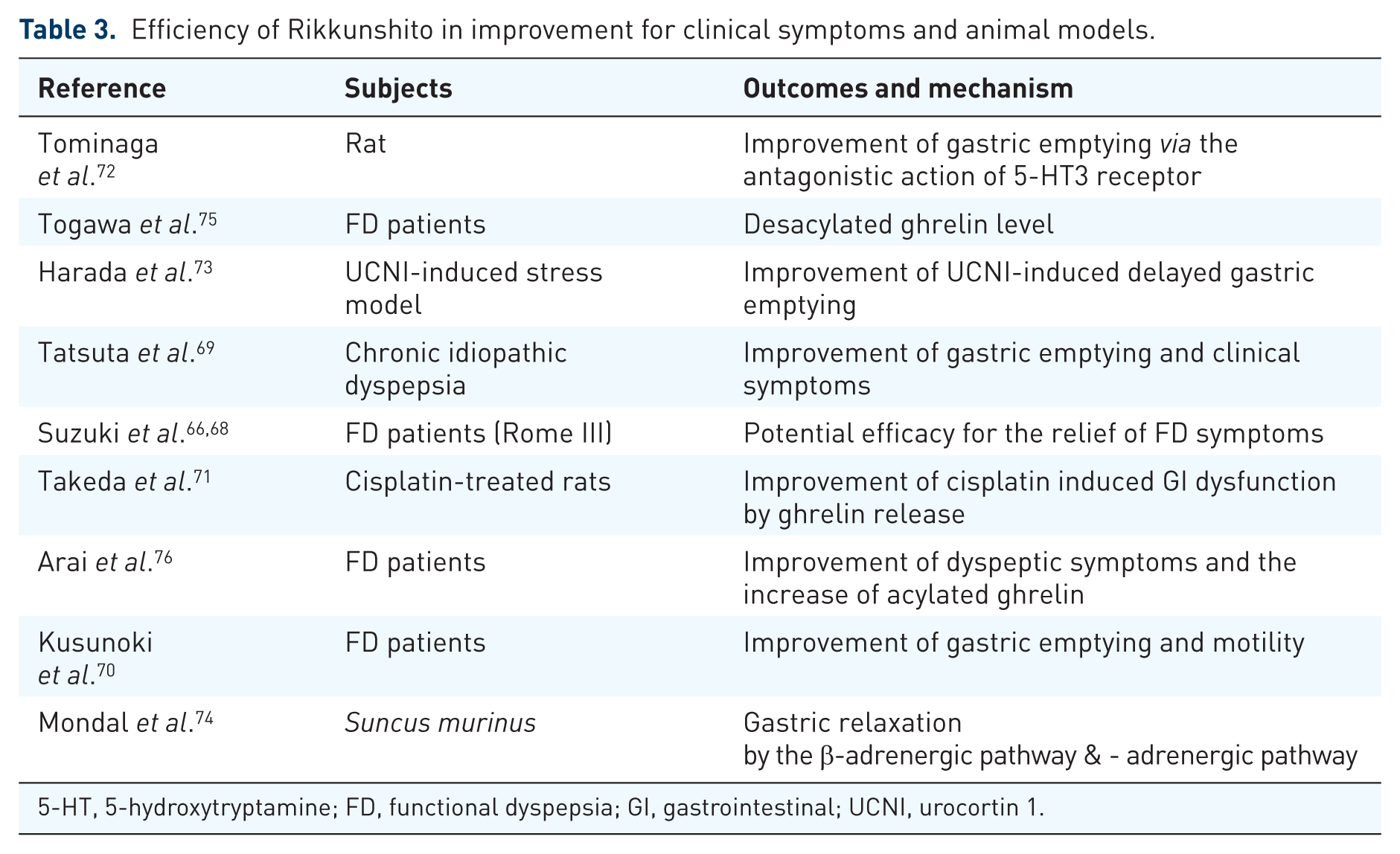
5-HT, 5-hydroxytryptamine; FD, functional dyspepsia; GI, gastrointestinal; UCNI, urocortin 1.
An RCT conducted in Japan showed improvement of epigastric symptoms for dysmotility. 67 In addition, Suzuki and colleagues reported that Rikkunshito improved symptoms of epigastric pain and postprandial fullness in functional dyspepsia patients based on the Rome III criteria in multicenter trials. However, they did not acknowledge the significant improvement of clinical symptoms in patients on placebo (Table 3). 68
Rikkunshito has been reported to improve gastric emptying in patients with functional dyspepsia 69 and to decrease other GI symptoms such as abdominal pain, heartburn, and abdominal distension through the improvement impairment of expansion of proximal stomach (Table 3). 70
The effects of Rikkunshito are likely to be mediated via the effect on ghrelin levels. Rikkunshito has been shown to increases ghrelin levels in Sprague–Dawley rats treated with cisplatin, 67 and several studies have evaluated its effects on ghrelin levels in human studies. Moreover, Rikkunshito increased acylated ghrelin levels in the plasma and regulated signs of GI dysmotility such as the delay of gastric emptying in a Sprague–Dawley rat model. 71 In animal models, Rikkunshito accelerates gastric emptying through antagonism of 5-HT3 receptors (Table 3). 72 Harada and colleagues reported Rikkunshito improved acylated ghrelin levels and impaired gastric emptying in the urocortin 1-induced stress models. 73 They suggested supplementation of exogenous acylated ghrelin or enhancement of endogenous acylated ghrelin secretion by the treatment of Rikkunshito will be effective for amelioration of functional dyspepsia symptoms because acylated ghrelin has a strong efficiency of GI motility. In addition, Mondal and colleagues have also reported that Rikkunshito affected gastric relaxation in a Suncus murinus model. 74
In one study, Rikkunshito improved symptoms of functional dyspepsia through the elevation of deacylated ghrelin levels in H. pylori-negative patients, and the levels of deacylated ghrelin correlated with the efficacy of Rikkunshito. 75 In another study that did not control for H. pylori status, Rikkunshito also ameliorated dyspeptic symptoms and increased acylated ghrelin levels. 76 Rikkunshito has been suggested to improve gastric emptying through 5-HT2B-mediated pharmacological action. 67 Takeda and colleagues also speculated that the improved gastric motility in association with Rikkunshito use is mediated through ghrelin activity as a GI function controlling peptide.
Patient-tailored treatment
Functional dyspepsia is diagnosed on the basis of clinical symptoms and patient’s medical history; endoscopy can exclude organic diseases such as gastric ulcer and gastric cancer. The first-line therapy for patients with EPS-type functional dyspepsia includes anti-acid therapy such as PPIs, H2-blockers and vonoprazan. First-line treatment of PDS-type functional dyspepsia includes prokinetic drugs such as itopride, metoclopramide and acotiamide. If these treatments and combined therapies are ineffective, functional dyspepsia should be considered as treatment-resistant, and patients should be treated with second-line therapies such as antidepressants or a herbal medicine such as Rikkunshito (Figure 1).

Flowchart of the treatment of FD patients. FD, functional dyspepsia; H2RA, histamine type-2 receptor antagonist; Hp, Helicobacter pylori; PPI, proton pump inhibitor.
Conclusion
Choosing the correct treatment for functional dyspepsia is difficult, and even though the prevalence of functional dyspepsia is increasing, pharmacological therapeutic options remain limited. Development of new drugs to treat the disorder is, therefore, a priority.
Footnotes
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the Ministry of Education, Culture, and Science and from the Ministry of Health, Japan.
