Abstract
Patients with type 2 diabetes mellitus require insulin as disease progresses to attain or maintain glycaemic targets. Basal insulin is commonly prescribed initially, alone or with one or more rapid-acting prandial insulin doses, to limit mealtime glucose excursions (a basal–bolus regimen). Both patients and physicians must balance the advantages of improved glycaemic control with the risk of hypoglycaemia and increasing regimen complexity. The rapid-acting insulin analogues (insulin aspart, insulin lispro and insulin glulisine) all have similar pharmacokinetic and pharmacodynamic characteristics and clinical efficacy/safety profiles. However, there are important differences in the pharmacokinetic and pharmacodynamic profiles of basal insulins (insulin glargine, insulin detemir and insulin degludec). Insulin degludec is an ultra-long-acting insulin analogue with a flat and stable glucose-lowering profile, a duration of action exceeding 30 h and less inter-patient variation in glucose-lowering effect than insulin glargine. In particular, the chemical properties of insulin degludec have allowed the development of a soluble co-formulation with prandial insulin aspart (insulin degludec/insulin aspart) that provides basal insulin coverage for at least 24 h with additional mealtime insulin for one or two meals depending on dose frequency. Pharmacokinetic and pharmacodynamic studies have shown that the distinct, long basal glucose-lowering action of insulin degludec and the prandial glucose-lowering effect of insulin aspart are maintained in the co-formulation. Evidence from pivotal phase III clinical trials indicates that insulin degludec/insulin aspart translate into sustained glycaemic control with less hypoglycaemia and the potential for a simpler insulin regimen with fewer daily injections.
Keywords
Introduction
International treatment guidelines strongly recommend that patients with type 2 diabetes mellitus (T2DM) who present with significant symptoms of hyperglycaemia and/or dramatically elevated plasma-glucose levels (in the range 16.7–19.4 mmol/l [300–350 mg/dl]) and glycated haemoglobin (HbA1c) levels in the range 10–12% at diagnosis initiate treatment on insulin, as such patients are unlikely to achieve glycaemic control with diet or oral antidiabetic drug (OAD) monotherapy alone [Inzucchi et al. 2012]. In particular, patients exhibiting catabolic features or ketonuria, which reflects profound insulin deficiency, must be started on insulin rather than on oral therapy [Inzucchi et al. 2012]. Some reluctance to initiate insulin therapy has been noted among physicians and patients because of perceived problems with regimen complexity, weight gain and the increased risk of hypoglycaemia relative to OADs [Brod et al. 2014; Leiter et al. 2005].
Therapy with modern insulins has also, more rarely, been associated with the development of antibodies to the insulin analogues, which could reduce treatment efficacy. It has also been hypothesized that insulin analogues may be associated with an increased risk of cancer, possibly mediated by increased insulin-like growth factor 1-receptor activation or by sustained signalling by the insulin receptor, but to date there is no firm proof for this hypothesis [FDA, 2012].
Insulin therapy is generally associated with a moderate increase in body weight due to its anabolic effects and the reduction in glycosuria that results from treatment [Heller, 2004]. Modern insulins are associated with lower weight gain than their predecessors, however [Zinman et al. 2012].
Both the implementation and intensification of insulin therapy need to be balanced against the increase in the risk of hypoglycaemia that is associated with more intensive glucose-lowering treatment regimens [Leiter et al. 2005]. Non-severe, patient-managed hypoglycaemic events are commonly reported by patients with T2DM on insulin [Leiter et al. 2005]. Such events affect daily activities such as exercise, work and sleep with resulting psychological and emotional impact on the lives of both the patient and their family [Brod et al. 2012, 2013]. In addition, patients face the risk of hospitalization due to severe hypoglycaemia [Budnitz et al. 2011]. The occurrence of both severe and non-severe hypoglycaemia and the attendant fear of future such events can affect the clinical success of diabetes treatment and has been cited by physicians as a reason for selecting less ambitious treatment targets [Peyrot et al. 2012], and by patients, as a reason for reducing their insulin dose [Leiter et al. 2005]. Beyond these effects, hypoglycaemia increases both the direct and indirect treatment costs of diabetes management for healthcare systems [Lundkvist et al. 2005]. Aside from the potential for hypoglycaemia, basal–bolus insulin regimens are perceived as complex and challenging, with up to one-third of patients reporting regular nonadherence with their regimen [Peyrot et al. 2012]. While many patients with T2DM may start treatment on basal insulin, most patients will require additional prandial insulin to limit mealtime glycaemic excursions [Inzucchi et al. 2012]. Prandial glycaemic insulin coverage has been shown to play an increasingly important role in attaining tight overall glycaemic control in patients whose HbA1c is close to target [Monnier et al. 2003], especially for patients who have achieved target fasting blood-glucose levels but whose HbA1c levels remain above target despite 3–6 months of basal-insulin treatment [Inzucchi et al. 2012]. When prandial insulin is required, this can either be through the addition of mealtime short-acting insulin or by switching to an analogue premixed insulin formulation, which combines a rapid-acting insulin analogue and a protaminated, intermediate-acting version of the same insulin in a single injection [Inzucchi et al. 2012]. Although premixed insulins offer a simple injection regimen, and provide comparable glycaemic control to basal–bolus insulin regimens, they are relatively inflexible in terms of titration and dose timing, and are associated with higher rates of hypoglycaemia compared with basal-insulin regimens [Vaag et al. 2012; Liebl et al. 2012].
In order to reduce the burden of insulin regimens, research efforts in recent years have focused on modifying the properties of insulin to simplify therapeutic regimens, reduce the risk of hypoglycaemia, improve adherence with treatment and better match the physiological release of endogenous insulin [Tibaldi, 2014]. This article reviews the current evidence for the basal insulin degludec (IDeg) and the prandial insulin aspart (IAsp) in the context of other currently available insulin treatments, and how as a novel co-formulation (IDegAsp) they may lead to improvements in adherence and safety for patients with T2DM.
IDeg
Structure/pharmacokinetics and pharmacodynamics
To date, three basal insulin analogues have been approved for clinical use, i.e. insulin glargine (IGlar), insulin detemir (IDet) and IDeg, each of which has a different mode of protraction to extend the glucose-lowering potential of the insulin [Tibaldi, 2014].
IGlar is soluble at pH 4.0; when injected, the neutral pH of the subcutaneous environment causes IGlar to microprecipitate, thereby slowing its absorption into the circulation and prolonging the duration of action [Sanofi-Aventis, 2010]. By contrast, IDet forms weakly associated dihexamers in solution [Tibaldi, 2014], and upon subcutaneous injection IDet strongly self-associates, slowing absorption into the circulation [Novo Nordisk, 2009]. Furthermore, IDet binds to plasma albumin, which also slows dissemination to peripheral tissues [Novo Nordisk, 2009]. In IDeg, the addition of a fatty-diacid side chain (Figure 1a) to human insulin favours the formation of an insulin analogue that forms stable dihexamers while in solution at neutral pH (7.4) in the presence of phenol (Figure 1b) [Jonassen et al. 2012]. When injected subcutaneously these dihexamers assemble into long multihexamers (Figure 1c), and the relatively large molecular weight of these multihexamers slows their absorption, creating a subcutaneous depot of IDeg, from which monomers slowly disassociate into the circulation (Figure 1c) [Jonassen et al. 2012; Kurtzhals et al. 2011].
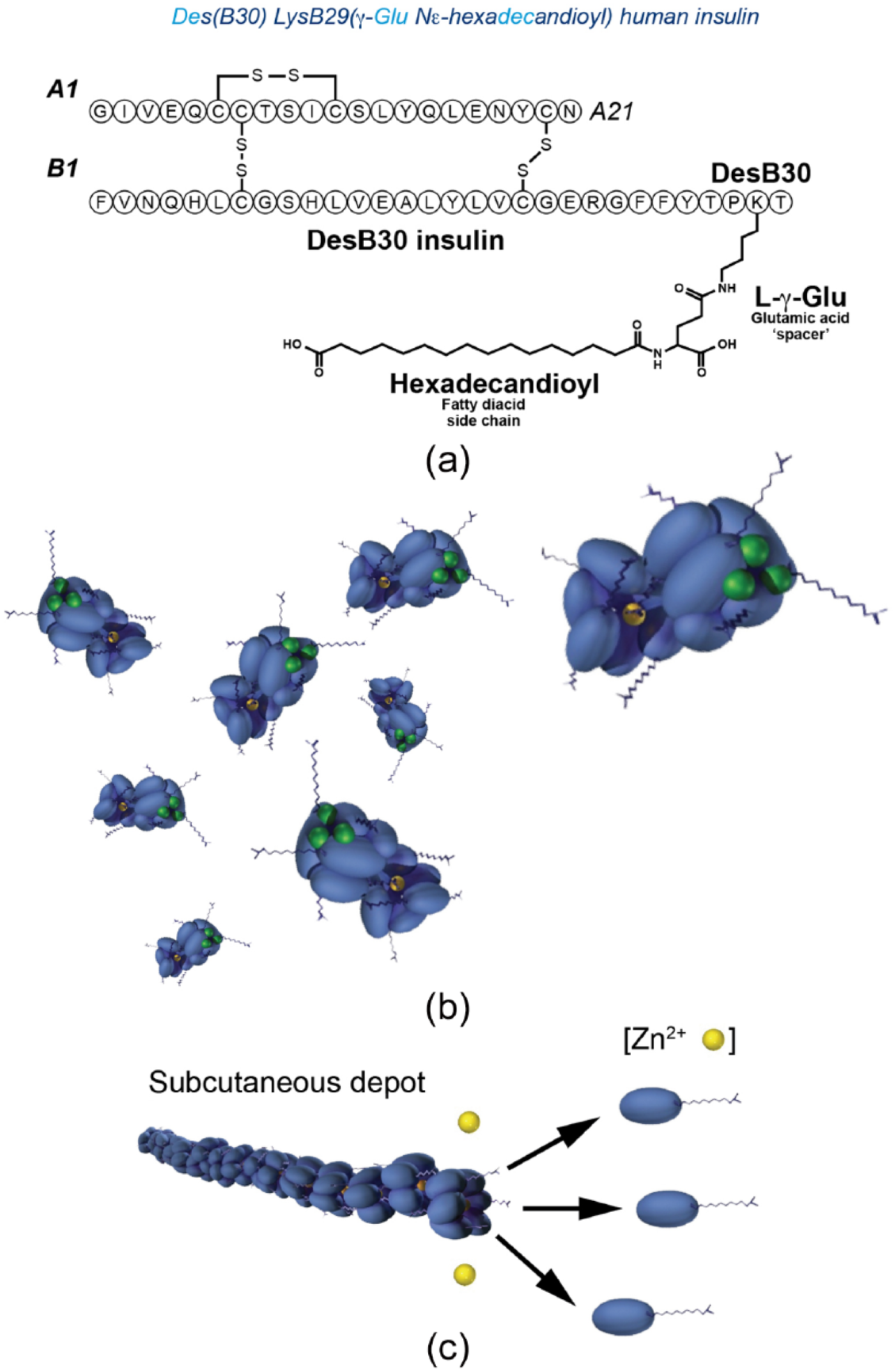
Schematic representations of (a) insulin degludec composition, (b) formation into dihexamers in solution, and (c) behaviour in the subcutaneous environment.
The resultant pharmacokinetic and pharmacodynamic properties of these three basal insulins have been investigated using blood sampling and euglycaemic-clamp experiments. These properties of IGlar, IDet and IDeg are summarized in Table 1. Studies have demonstrated that, due to its mode of protraction, IDeg has an ultra-long half-life, a duration of action that exceeds 42 h [Heise et al. 2012a], and reduced within-patient variability compared with IGlar [Heise et al. 2012b].
Pharmacokinetic/pharmacodynamic properties of (a) basal and basal–bolus co-formulation; (b) currently available rapid-acting insulins in patients with type 2 diabetes.

NPH, neutral protamine Hagedorn insulin.
Efficacy and safety
Table 2 summarizes the glycaemic efficacy and hypoglycaemia risk for each of the three currently available basal insulins when used as part of a basal–bolus regimen for patients with T2DM. Studies have shown that, as part of basal–bolus therapy in patients with type 1 diabetes mellitus (T1DM) [Russell-Jones et al. 2004] or T2DM [Haak et al. 2005], IDet provides the same level of glycaemic control as neutral protamine Hagedorn (NPH, human insulin), with a similar overall risk of hypoglycaemia but significantly less within-patient variability in blood-glucose levels. When compared in treat-to-target trials in patients with T2DM, IGlar and IDet demonstrated similar levels of glycaemic control and similar risk of hypoglycaemia, but weight gain was lower with IDet [Hollander et al. 2008; Raskin et al. 2009].
Key phase III efficacy and safety data from treat-to-target trials of IAsp, IGlar, IDet, IDeg and IDegAsp co-formulation in basal–bolus therapy for patients with T2DM.

BIAsp 30, biphasic IAsp 30; BID, twice daily; CI, confidence interval; ETD, estimated treatment difference; Flex, flexible dose timing; FPG, fasting plasma glucose; HbA1c, glycated hemoglobin; HI, human soluble insulin; IAsp, insulin aspart; IDeg, insulin degludec; IDegAsp, insulin degludec/insulin aspart co-formulation; IDet, insulin detemir; IGlar, insulin glargine; MIX, human premix insulin; NPH, neutral protamine Hagedorn; NS, not significant; NR, not reported; OD, once daily.
The clinical efficacy and safety of IDeg recently has been compared with IGlar as part of the BEGIN® phase III clinical programme, which involved more than 5000 patients with T1DM or T2DM. In patients with T2DM, IDeg was associated with comparable glucose-lowering efficacy to IGlar (as expected due to the treat-to-target nature of the trials) [Zinman et al. 2012; Garber et al. 2012]. In insulin-naïve T2DM patients, similar rates of overall confirmed hypoglycaemia and lower rates of nocturnal hypoglycaemia were reported compared with IGlar when both insulin agents were administered once daily [Zinman et al. 2012]. In insulin-experienced T2DM patients, IDeg was associated with lower overall rates of hypoglycaemia and lower nocturnal hypoglycaemia rates than IGlar [Garber et al. 2012].
A preplanned meta-analysis of seven phase III trials in the BEGIN programme (five in T2DM, two in T1DM) reported significantly lower risk of hypoglycaemia with IDeg compared with IGlar in the overall pooled population (T1DM and T2DM), particularly after 16 weeks of treatment when the insulin dose had been stabilized [Ratner et al. 2013]. Furthermore, a
Glucose-lowering effect of insulin degludec (IDeg)
In comparisons of IDeg with IGlar in patients with T1DM and T2DM as part of the BEGIN programme, there were no differences in weight gain in any of the groups [Heller et al. 2012; Zinman et al. 2012]. Similarly, equal proportions (1% in both groups) of patients with T2DM treated with either IDeg or IGlar developed malignant neoplasms, and none of these were considered to be treatment related by investigators [Zinman et al. 2012].
In a pooled analysis of six phase III studies comparing IDeg and insulin IGlar in patients with T1DM and T2DM, the immunogenic responses to long-term treatment with the two preparations were similar and low, with no clinically relevant impact on HbA1c or total daily insulin dose [Vora et al. 2012].
In summary, the mode of protraction of IDeg and associated ultra-long pharmacokinetic and pharmacodynamic basal insulin properties are associated with effective glycaemic control and clinically meaningful differences in fasting blood-glucose levels and a lower risk of adverse events, including hypoglycaemia and, particularly, nocturnal hypoglycaemia [Garber et al. 2012; Zinman et al. 2012; Ratner et al. 2013; Vora et al. 2014].
IAsp
Structure/pharmacokinetics and pharmacodynamics
Rapid-acting insulin analogues have been developed using amino-acid substitutions of human insulin to reduce the propensity for insulin to aggregate into dimers and hexamers. Thus, insulin analogues that promote the formation of monomers provide faster absorption of insulin into the bloodstream. Three rapid-acting insulin analogues are currently available, i.e. IAsp, insulin lispro and insulin glulisine, all of which have been developed based on this principle. Insulin lispro utilizes a proline to lysine substitution at B28 and a lysine to proline substitution at B29 (Figure 3a), insulin glulisine has an asparagine to lysine substitution at B3 and a lysine to glutamic acid substitution at B29 (Figure 3b) and IAsp has a single proline to aspartic acid switch at position B28 (Figure 3c). [Tibaldi, 2014].

Amino-acid sequence changes used in the rapid-acting insulin analogues (a) insulin lispro, (b) insulin glulisine and (c) IAsp.
As a result of these modifications, each of these insulin analogues rapidly achieves maximum plasma concentration (and consequently glucose-lowering effect) (Figure 4). IAsp and insulin glulisine reach maximum plasma concentrations at approximately 40 min and 55 min, respectively, after subcutaneous injection (Figure 4c for IAsp) [Tibaldi, 2014], and insulin lispro reaches peak plasma concentration at approximately 68 min after injection [Tibaldi, 2014]. These pharmacokinetic/pharmacodynamic characteristics allow patients who require prandial insulin to inject closer to mealtime, instead of the 30 min interval recommended for human insulin [Tibaldi, 2014]. All three rapid-acting insulin analogues have similar published pharmacokinetic and pharmacodynamic profiles (Table 1) [Home, 2012].

Glucose-lowering effect of (a) IDeg and (b) IDegAsp co-formulation observed in euglycaemic-clamp studies as well as (c) IAsp in healthy adult subjects.
Efficacy and safety
Rapid-acting insulins represent important therapeutic options for patients who are unable to achieve glycaemic targets on regimens with basal insulin alone [Inzucchi et al. 2012]. IAsp and the other rapid-acting insulin analogues offer equivalent or improved reductions in HbA1c and greater reductions in postprandial blood-glucose levels when compared with human insulin, as shown in several clinical trials [Bretzel et al. 2004; Dailey et al. 2004; Ross et al. 2001]. In clinical trials, the overall hypoglycaemia rates reported with the different rapid-acting insulin analogues are also similar to, or lower than, those associated with short-acting human insulin [Bretzel et al. 2004; Dailey et al. 2004; Ross et al. 2001].
Basal and rapid-acting insulin co-formulation
The goal of insulin management for patients with both T1DM and T2DM is to provide physiological insulin replacement that closely mimics the endogenous secretion of insulin, providing peak insulin coverage after main meals with a rapid-acting insulin combined with sustained 24-h basal insulin coverage from a basal insulin [Abrahamson, 2010]. Although premixed insulins and basal–bolus insulin regimens have provided additional treatment options for patients requiring better glycaemic control, both approaches have limitations. These include the burden of multiple injections, complex titration schedules associated with basal–bolus regimens, and incomplete 24-h basal coverage with premixed insulins. Therefore, there is a clinical need for a combination insulin formulation that can provide sustained and stable basal insulin coverage with an effective prandial insulin bolus from a single injection [Abrahamson, 2010].
Premixed insulin formulations consist of intermediate-acting insulin combined with prandial insulin coverage for a main meal within a single injection. Protamination of the short-acting insulin analogue delays its absorption, resulting in an intermediate-acting insulin that can be combined with the short-acting form of the same insulin analogue resulting in a biphasic insulin preparation, e.g. biphasic insulin aspart 30 (BIAsp 30) [Garber, 2009; Tibaldi, 2014]. Premixed insulins offer equivalent glycaemic control to basal–bolus insulin regimens but may increase the overall risk of confirmed hypoglycaemia and tend to be associated with greater weight gain compared with basal insulin regimens [Ilag et al. 2007]. In addition, premixed insulins are relatively inflexible in dose timing and the dose of the basal and prandial components cannot be titrated separately, making dosing complicated [Inzucchi et al. 2012]. Also a ‘shoulder’ effect occurs in the hours following mealtime due to interaction between the protaminated and nonprotaminated insulins that prolongs the action of the short-acting prandial component, potentially increasing the risk of hypoglycaemia [Cengiz et al. 2012].
Until now these compromises have been necessary because the available basal insulins could not be co-formulated with rapid-acting insulin. In the case of IDet, mixing with rapid-acting insulin analogues has been shown to result in the formation of mixed hexamers that adversely affects the glucose-lowering efficacy of the co-formulation [Cengiz et al. 2012]. IGlar is soluble in acidic solution at pH 4.0 and forms slowly dissolving microprecipitates on subcutaneous injection [Sanofi-Aventis, 2010], preventing co-formulation with rapid-acting insulins that are soluble at pH 7.4. However, in the presence of zinc and phenol, the basal insulin analogue IDeg forms stable dihexamers in solution at neutral pH, which do not interact with IAsp, therefore allowing for the first time the co-formulation of a 24-h basal insulin with a rapid-acting insulin analogue in the form of IDegAsp [Novo Nordisk, 2013].
IDegAsp: a novel insulin co-formulation
IDegAsp is a novel co-formulation of basal IDeg and rapid-acting IAsp (ratio 70% IDeg: / 30% IAsp) available as a single subcutaneous injection [Novo Nordisk, 2013]. In solution, IDeg forms soluble dihexamers at neutral pH [Jonassen et al. 2012], as described previously (Figure 1), whereas IAsp exists as distinct hexamers [Heise et al. 2014]. IDeg and IAsp remain separate entities in solution and size exclusion chromatography studies have shown that there is no evidence of physical or chemical interaction between the two insulins in co-formulation [Havelund, 2013]. Once injected into the subcutaneous environment, IDeg dihexamers immediately self-associate forming stable multihexamers [Kurtzhals et al. 2011], from which IDeg monomers slowly dissociate, providing stable basal insulin coverage (Figure 5) [Heise et al. 2014]. In contrast, the rapid-acting IAsp component consists of hexamers that quickly dissociate into rapidly absorbed insulin monomers, giving short-acting insulin coverage to control postprandial increases in blood glucose [Heinemann et al. 1993].

Distinct modes of protraction of the IDegAsp co-formulation components upon injection.
Pharmacokinetics/pharmacodynamics of IDegAsp
Pharmacodynamic studies have demonstrated that the glucose-lowering effect of once-daily IDegAsp is characterized by a distinct peak action (from IAsp), and a separate basal action that lasts for more than 30 h, at steady state (Figure 3c) [Heise et al. 2014]. Furthermore, at steady state IDegAsp has been shown to exhibit a dose-proportional glucose-lowering effect [Nosek et al. 2013]. Pharmacodynamic modelling of a twice-daily IDegAsp regimen has indicated that twice-daily dosing of IDegAsp could provide 24-h basal coverage and prandial control for two main meals [Heise et al. 2014]. Taken together, the pharmacokinetic and pharmacodynamic characteristics of IDegAsp [Table 1] indicate that IDegAsp may offer the advantages of true 24-h basal coverage with effective prandial glycaemic control for one or two meals achieved with a lower risk of hypoglycaemia and with fewer injections than previously available regimens.
Efficacy and safety of IDegAsp
Phase II and phase III studies have demonstrated comparable glycaemic efficacy and similar hypoglycaemia rates for IDegAsp compared with standard basal–bolus treatment (IDet plus mealtime IAsp) in T1DM [Hirsch et al. 2012], and compared with IGlar in insulin-naïve T2DM [Heise et al. 2011]. Similarly, a randomized open treat-to-target study, also in insulin-naïve patients with T2DM, showed that, compared with BIAsp 30, IDegAsp provided comparable overall glycaemic control with a significantly lower risk of hypoglycaemia [Niskanen et al. 2012]. In a 26-week, phase III, open-label, treat-to-target trial in a Japanese population of insulin-naïve T2DM subjects (
While this evidence is promising, patients who require additional mealtime glycaemic control require 1–3 injections of short-acting insulin per day (as is often the case with premixed insulins, which are dosed two or three times daily) [Inzucchi et al. 2012]. Clinical studies have therefore also evaluated the efficacy and safety of IDegAsp taken twice daily (Table 2). A 26-week, treat-to-target, open-label, phase III trial involving 394 insulin-naïve patients with T2DM reported that twice-daily IDegAsp provided effective reduction in HbA1c comparable with BIAsp 30, with superior reductions in FPG levels. Rates of confirmed hypoglycaemia were 54% lower with IDegAsp than BIAsp 30 (
In addition, two large phase III trials have investigated the efficacy and safety of twice-daily IDegAsp in insulin-experienced patients. In a 26-week, multinational, treat-to-target trial of 447 patients randomized to either IDegAsp or BIAsp 30 both dosed twice daily, IDegAsp was noninferior in reducing HbA1c and superior in lowering FPG levels (ETD −1.14 mmol/l; 95% CI −1.53, −0.76;
Subsequent meta-analysis of these two phase III trials have demonstrated that, whilst the improvements in HbA1c were similar in both treatment arms, overall rates of confirmed and severe hypoglycaemia were numerically lower, and nocturnal hypoglycaemia rates were significantly lower, with twice-daily IDegAsp compared with BIAsp 30 [Vaag et al. 2013]. Moreover, the differences between treatments became more pronounced when the analysis considered the ‘maintenance period’ after 16 weeks, by which time the insulin dose is expected to have stabilized, following initial dose titration [Vaag et al. 2013]. A further meta-analysis of these trials demonstrated that when patients who achieved HbA1c lower than 7% in the IDegAsp and BIAsp 30 arms were compared, both nocturnal- and overall-confirmed hypoglycaemia rates were significantly lower in the IDegAsp arm (
More recently, a 26-week, phase III trial in T2DM patients that compared twice-daily IDegAsp with once-daily IDeg plus additional mealtime IAsp in a basal–bolus regimen reported reductions in HbA1c in both arms with no significant difference between treatments (ETD 0.18%; 95% CI −0.04, 0.41;
Conclusion
As the development of insulin analogues continues, better options for basal, prandial and basal–bolus regimens are emerging for patients with T2DM who require insulin therapy. Novel insulin formulations provide better balances between glycaemic control, the risk of hypoglycaemia and convenience, allowing more dose flexibility and offering the potential for simpler regimens for patients. The development of the ultra-long-acting basal insulin, IDeg, has made possible the first co-formulation of basal and rapid-acting insulins, in which the basal and prandial glucose-lowering effects of the individual insulin components remain distinct. Evidence from the IDegAsp phase III clinical trial programme suggests that the pharmacokinetic and pharmacodynamic properties of IDeg in IDegAsp translate into effective glycaemic control with greater reductions in FPG levels and less hypoglycaemia compared with existing treatment options in insulin-naïve or insulin-experienced patients. IDegAsp therefore appears to offer a promising treatment option for patients who may benefit from the simpler regimen of one injection given once or twice daily.
Footnotes
Acknowledgements
SA contributed to the design, writing and review of the manuscript. ZJ contributed to the writing of the manuscript. GF assisted in the preparation of the manuscript, including content addition, review of layout, editing and involvement in the original study design and publication of the included studies.
Funding
Medical writing assistance was provided by apothecom scopemedical, London, UK, funded by Novo Nordisk.
Conflict of interest statement
SA has served on speakers’ bureau for Novo Nordisk. ZJ has no conflicts to disclose. GF has served on advisory panels for Novo Nordisk, Janssen, Boehringer Ingelheim, Merck Sharp & Dohme and Sanofi-Aventis.
