Abstract
Background:
Severe hemophilia traditionally requires lifelong prophylactic treatment, which cannot fully prevent joint damage and significantly impacts patients’ quality of life. In recent years, gene therapy has been extensively researched as a potential cure with a single infusion, but it has not yet gained widespread acceptance.
Objectives:
This study examines the knowledge and attitudes of people with severe hemophilia regarding gene therapy to provide guidance to practitioners in the context of shared decision-making.
Design:
Cross-sectional observational study based on a standardized questionnaire.
Methods:
A questionnaire developed by Cutica, Ilaria et al. was evaluated in a self-reported, anonymized survey for a German cohort of 59 people with severe hemophilia. It covered sociodemographic and clinical characteristics, knowledge, attitudes, risk tolerance, and decision-making preferences.
Results:
Among 59 people with severe hemophilia A and 10 with severe hemophilia B, 59% expressed a predominantly rejective attitude toward gene therapy. Most had only general knowledge of gene therapy. Concerns about long-term side effects (e.g., cancer development) significantly contribute to a negative attitude toward gene therapy (p = 0.005). People with hemophilia and an annual bleeding rate ⩾1 show a significantly higher willingness to consider or accept gene therapy (p = 0.001). Rejection of gene therapy is significantly associated with high expectations regarding its duration of efficacy and the minimum clotting factor level.
Conclusion:
Further studies are needed to refine educational concepts for people with hemophilia and clear out misconceptions about gene therapy.
Plain language summary
We asked 69 German men with severe haemophilia A or B what they know and feel about gene therapy, a new one-time treatment that could help their bodies make the missing clotting factor. Most take regular infusions or the bispecific antibody emicizumab to prevent bleeds. Although 100% said they are satisfied with today’s care, three out of four still live with chronic joint pain and almost half avoid sports because of bleeding risk. Two-thirds had heard of gene therapy, mainly from their doctors or patient groups. Only about half could answer more than half of the knowledge questions correctly, showing many gaps and misconceptions. 59% said they would reject gene therapy right now. The biggest worry is long-term safety, especially fear of cancer or liver problems from the treatment and the steroid medicines used to manage side effects. People who expect the treatment to last “for life” or raise clotting factor to normal levels are more likely to say no when those guarantees are not yet proven. Now who is interested? Men who still have at least one bleed a year are much more willing to consider gene therapy, suggesting that living with frequent bleeds outweighs safety worries for some. Conclusion: Clear, easy-to-understand information is needed about how gene therapy works, its real benefits, limits, and side effects can help patients make informed choices together with their doctors. Addressing safety concerns directly and setting realistic expectations may increase acceptance over time
Introduction
Nowhere is the fragility of hemostasis more evident than in hemophilia. In hemophilia A, it is the cofactor of a serine protease, also known as clotting factor VIII; in hemophilia B, it is the serine protease itself, factor IX, that is deficient. 1 It is therefore dramatic to see the danger that this deficiency poses to the approximately 4000 registered individuals affected in Germany. 2 In severe forms of hemophilia, that is, with a factor activity of <1%, patients can suffer massive bleeding after minor trauma, resulting in painful joint damage. 3
Treatment of this X-linked inherited disorder with coagulation factors from pooled plasma concentrates has been available since the 1970s.3,4 This therapy required frequent intravenous infusions, which were both time-consuming and stressful. In recent decades, however, treatment options have improved considerably.5,6 Today, both plasmatic and recombinant factor concentrates and the bispecific monoclonal antibody emicizumab 7 are available to patients. These options are recommended for long-term bleeding prophylaxis in people with hemophilia (PWH) and a high bleeding tendency. 8 The development of factor concentrates with an extended half-life allows for longer intervals between infusions. 9 It has been proven that the prophylactic approach can delay the onset of hemophilic arthropathy by decades. 6
Unfortunately, the therapies mentioned so far offer only a lifelong prophylactic approach and continue to place a great burden on those affected. 10 Factor substitution cannot completely prevent bleeding,11,12 especially joint bleeding, which can progress to arthropathy and requires PWH to refrain from physically demanding activities. This can lead to social isolation and a significant loss of quality of life.13,14 They require knee or hip replacements at a higher rate than the general population, 15 which, together with regular substitution and inhibitor treatment, 16 leads to frequent hospitalizations and non-negligible costs to the healthcare system.17–19
In recent years, significant progress has been made in gene therapy (GT) for hemophilia.20–26 In this therapy, a functional copy of the deficient factor gene is introduced into the hepatocytes using a non-replicating, non-pathogenic adeno-associated viral (AAV) vector. After just one infusion, the hepatocytes begin to produce and secrete the coagulation factor. 9 This innovative treatment option offers the prospect of curing hemophilia with a single infusion.27–31 Etranacogene dezaparvovec and fidanacogene elaparvovec are already approved for the treatment of hemophilia B, and valoctocogene roxaparvovec for the treatment of hemophilia A in the United States and the European Union.24,32–35 In case of valoctocogene roxaparvovec, the efficacy of GT is particularly evident in the significant reduction of the annual bleeding rate (ABR) to 0.3 bleeds/year over 7 years, 36 leading to a significant improvement in quality of life. 37 In terms of safety, transaminase elevations are documented, which can usually be controlled by glucocorticoids.25,38
Despite these revolutionary advances, GT has not yet gained widespread acceptance. It is currently unclear whether this is due to a lack of information or other factors.
The main objective of this study is to assess the knowledge and attitudes of people with severe hemophilia in Germany toward GT, with a particular focus on their informational needs, priorities, and concerns. Additionally, the study aimed to identify specific factors influencing patients’ willingness to choose GT, considering all the aforementioned aspects as well as their clinical condition. To date, no such investigation has been conducted for Germany.
Ultimately, the results should provide physicians and therapists with a better basis for informing and advising patients in the context of shared decision-making.
Methods
To assess patients’ knowledge and attitude, a questionnaire developed by Cutica et al. at the University of Milan and previously used on a cohort of Italian hemophilia patients 39 was used in this study for German patients. To achieve equivalence between the original questionnaire and the German version, the translation was done according to the guidelines of Beaton et al. 40 The questionnaire was then made available on the SurveyMonkey platform, ensuring anonymous data collection. The access was provided to the patients via a QR code in the newsletters of the patient organizations Deutsche Hämophiliegesellschaft e.V. and Interessengemeinschaft Hämophiler e.V. from June to October 2023.
The questionnaire consisted of four sections:
Patient information: Collected sociodemographic (age, education) and clinical data, including hemophilia type, severity, treatment details, bleeding symptoms, and presence of chronic arthropathy.
GT knowledge: Assessed understanding of GT, including its mechanism, risks, and benefits, through multiple-choice questions of varying difficulty.
Attitudes toward GT: Evaluated willingness to undergo GT, information needs for decision-making, perceived benefits, and concerns about potential consequences.
Psychological traits: Included standardized questionnaires on decision-making style (assessing habitual decision patterns) and risk-taking attitude (measuring likelihood of engaging in risky behaviors).
Participants were included in the study if they were adults (⩾18 years) with mild, moderate, or severe hemophilia A or B, and were able to read and understand the study materials, including the consent form and questionnaires. Patients with a pre-existing psychiatric diagnosis were excluded. A total of 101 patients accessed the link, 85 of whom completed the questionnaire. Within this group, the proportion of women was 10%. As the disease almost exclusively affects men, we attributed the relatively high proportion of women to the fact that they probably suffered from other coagulation disorders. However, due to the anonymity of the survey, it was not possible to track this and make a differentiated classification. Therefore, all female participants had to be excluded from the study. Finally, 69 male people with severe hemophilia A and B participated in the study. The survey took approximately 20 min to complete.
Statistical analysis of raw data was conducted using Microsoft Excel, BiAS for Windows 11.12, and GraphPad Prism (version 10.2.3). For ordinally scaled data, the Mann–Whitney U test was applied, while the chi-squared test was used for categorical variables. A p value of <0.05 was considered statistically significant. The analysis of individual decision-making style was conducted based on the work of Scott and Bruce. 41 This study was performed according to the principles laid out in the ICH guideline for good clinical practice E6(R2). 42
Results
Of the 69 patients included in the study, 100% were male with a median age of 50 years (SD 13.3, range 19–85). More than half of the respondents had an education beyond compulsory schooling. The cohort consisted exclusively of male people with severe hemophilia (residual factor activity <1% of normal). Of the patients, 86% had hemophilia A (n = 59) and 14% had hemophilia B (n = 10); 7% had formed inhibitors in history. At the time of the survey, 96% of the patients required regular bleeding prophylaxis. Seventy-one percent were receiving replacement therapy with recombinant coagulation factor, 13% with emicizumab, 10% with plasmatic coagulation factor, and 6% with other therapies. A detailed overview of the patient characteristics is provided in Tables A1 and A2.
Satisfaction with the current treatment was high, with 64% of patients very satisfied and 36% satisfied. Despite the high level of patient satisfaction, 77% of patients experienced chronic arthralgia, and 13% experienced severe acute arthralgia. Regarding activities of daily living, Figure 1 illustrates that 48% of respondents felt impaired in sports, while 17% reported no impairment due to hemophilia. Sixty-eight percent of patients had heard about GT, mainly from physicians and patient organizations, 8% had heard about it through online seminars/conferences, and 6% used newspapers and the Internet as a source of information. Forty-four percent did not seek further information.

To objectify knowledge, patients were asked to rate statements about the availability, indications, mechanisms, and potential risks of GT for hemophilia, including its use in clinical trials, the role of viral vectors, eligibility criteria, and possible side effects such as liver inflammation and long-term risks. The list of statements is provided in Table A3. The majority of patients had only general knowledge about GT as illustrated in Figures 2 and 3, while specific questions, such as pre-existing liver disease and AAV vector immunity, were answered correctly by only a few patients. No patient was able to answer all the questions about GT correctly. 48% achieved a rate of over 50% correct answers, and 7% answered over 80% of the questions correctly.
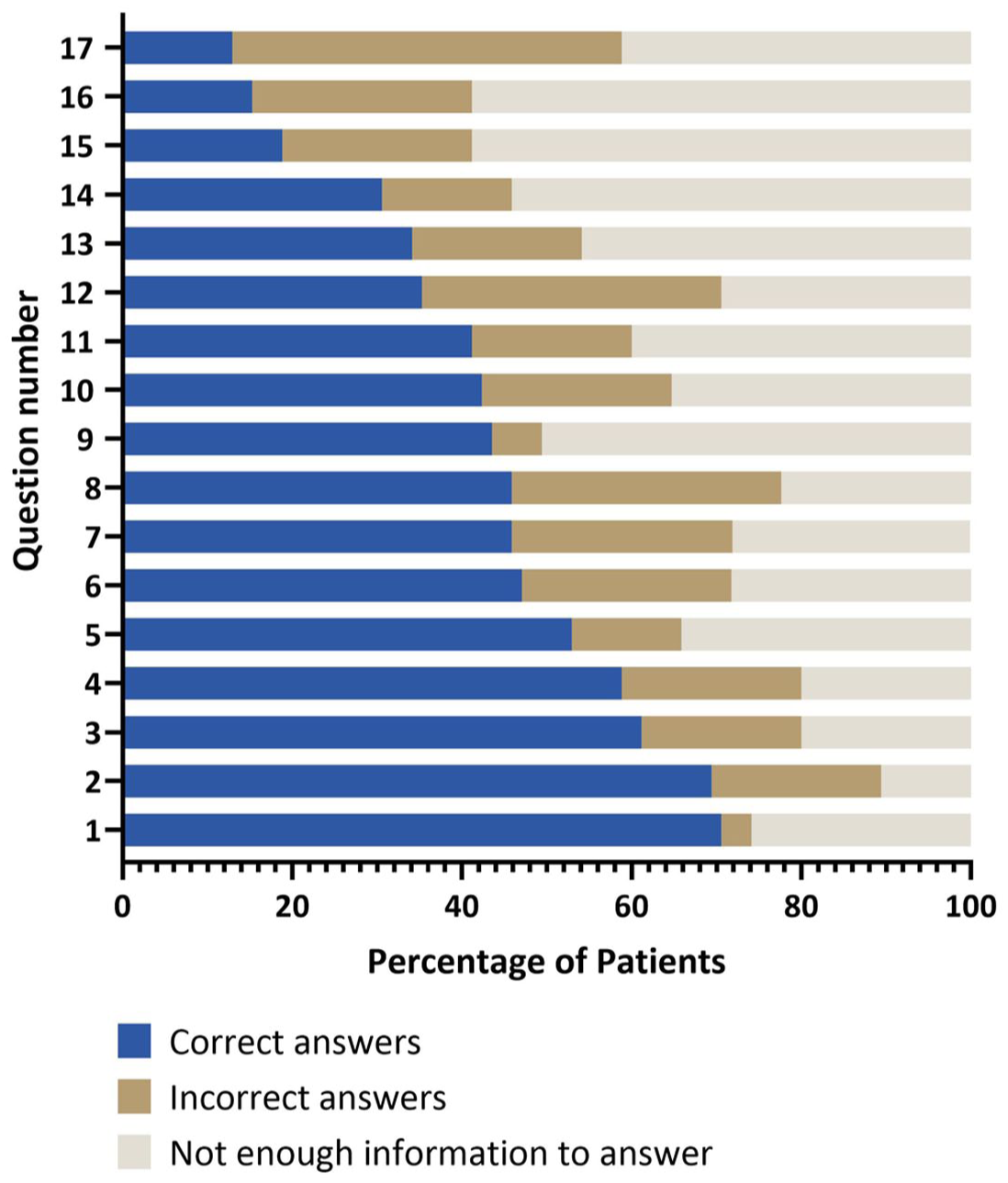

Based on their level of knowledge, 59% of patients said they would refuse GT. Patients’ attitudes toward GT were reflected in their hopes and concerns: they saw the greatest potential benefits in reducing bleeding episodes, avoiding infusions, and reducing chronic arthralgias. However, patients were concerned about potential long-term side effects such as cancer, short-term side effects such as possible liver inflammation, and the consequences of steroid therapy (Table 1).
Importance of benefits, concerns, and information on gene therapy for hemophilia.

In order to understand patients’ expectations of the new treatment method, we asked respondents to define the minimum duration of efficacy of GT and the minimum guaranteed factor activity that would be acceptable for them to opt for this form of therapy. First, a clear tendency emerged that the willingness to undergo GT increases with a longer minimum duration of efficacy. Correspondingly, there is also a higher willingness to undergo GT with a higher guaranteed minimum factor activity. To determine the relevance of these aspects for the decision for or against GT, we performed Mann–Whitney U tests for non-normally distributed samples and compared the expectations of the group of PWH who would accept GT (n = 19) with those who would reject it (n = 50): There were significantly higher expectations in the group of refusers in terms of duration of efficacy (p = 0.003) and minimum factor level (p = 0.04; see also Figures 4 and 5).


We also performed comparative analyses between the two groups of respondents using chi-square tests to compare their sociodemographic and clinical characteristics. No significant correlations were found between respondents’ level of education, age, presence of chronic or acute arthralgia (Table A2), or knowledge of GT and their negative attitude. However, a highly significant difference was observed when comparing the two groups in terms of ABR: PWH with an ABR ⩾1 were significantly more likely to agree or accept GT (p = 0.001).
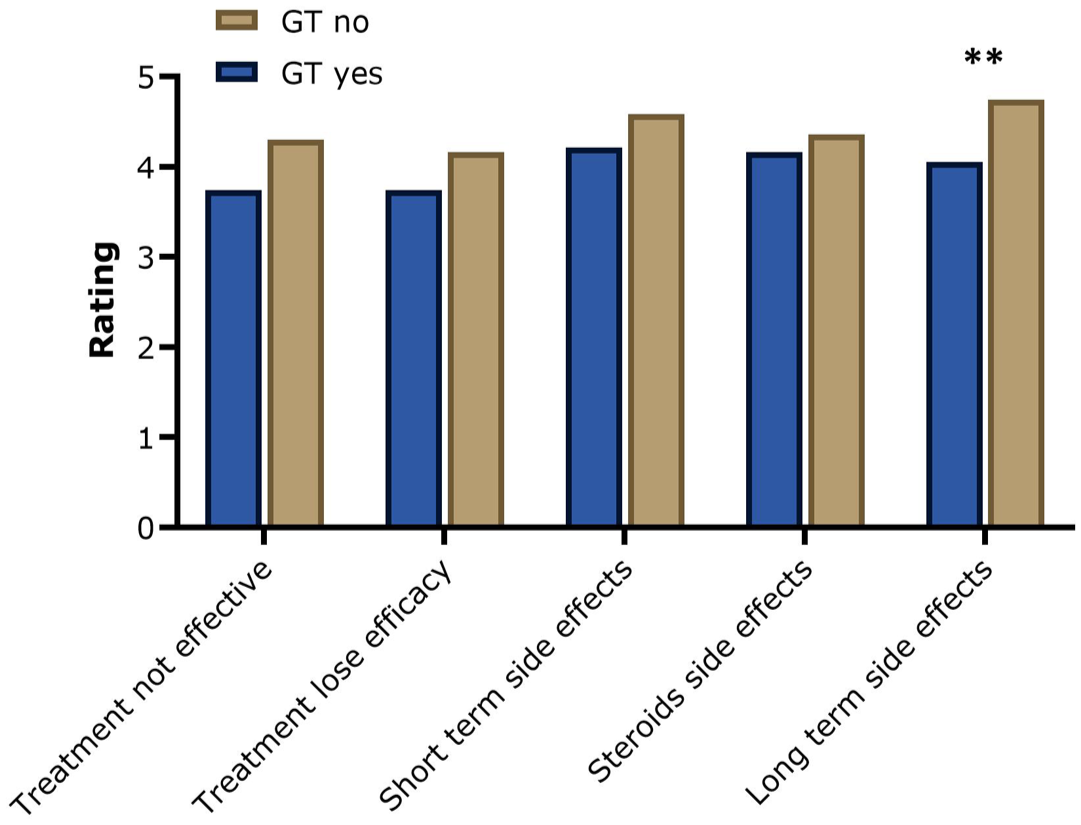
Regarding their assessment of the potential advantages and disadvantages of GT, our analysis showed a trend that people who would reject GT at the current time tended to rate the potential disadvantages higher and the benefits lower than those who would consider GT (Figures 6 and 7). However, a high significance was reached in the assessment of long-term side effects, such as the development of cancer: GT rejecter group rated this potential disadvantage significantly higher than GT acceptor group (p = 0.005).

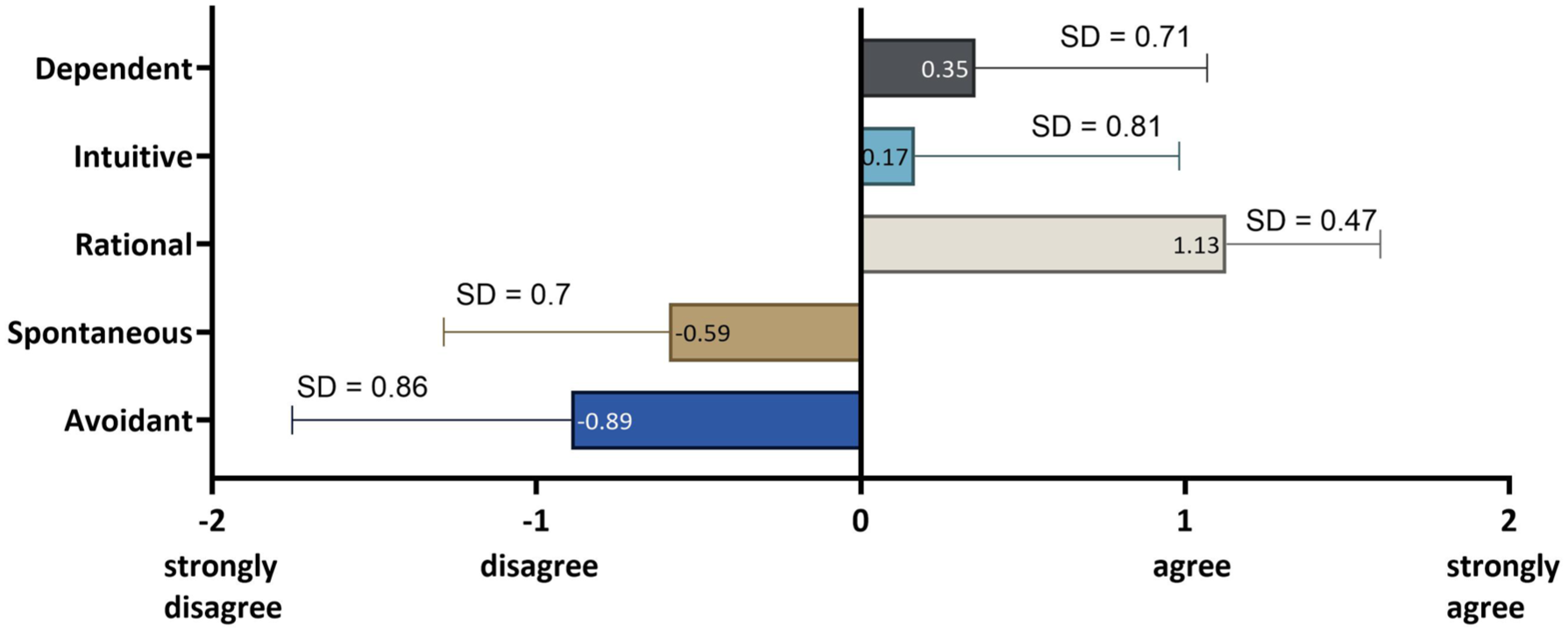
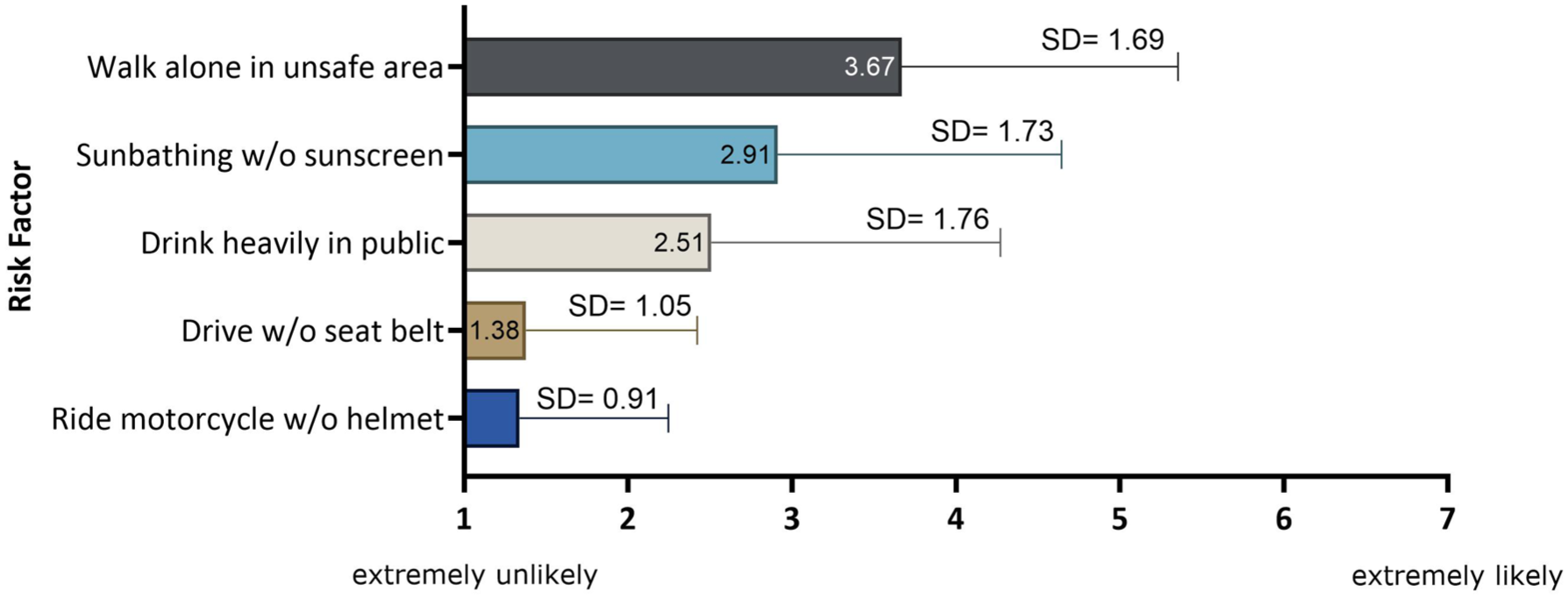
Finally, we examined the entire cohort of patients with respect to their individual decision-making style. The methodology was based on the work of Scott and Bruce 41 on the collection and analysis of decision-making preferences. The results showed that respondents had a predominantly rational decision-making style (n = 51), that is, based on a thorough consideration of alternatives and facts (Figure 8). The low prevalence of spontaneous and intuitive styles underscores the group’s rational, controlled decision-making process. We performed a Craddock–Flood chi-square test to assess whether decision-making style influences the decision to undergo GT. There was no significant difference between the group which would choose GT and the group which would reject it. As shown in Figure 9, the group of respondents exhibited low risk-taking behavior overall, particularly in areas associated with clear safety rules. This is reflected in the low mean scores for activities such as “drinking in public” or “driving without a seat belt,” while slightly higher scores are seen for less directly safety-related behaviors such as “sunbathing without sun protection.”
Discussion
This study is the first in Germany to investigate the knowledge of individuals with hemophilia about GT. In view of the rapid progress in research in this field, particularly with regard to gene therapies for hemophilia A and B, which show impressive long-term results36,43–46 and have been approved in the European Union for a few years,23,35 an analysis of the knowledge of affected persons seems urgently necessary. Using a questionnaire, we investigated patients’ knowledge, concerns, and hopes regarding GT, as well as the sociodemographic and disease-related characteristics of the study participants.
Compared to the study conducted by Cutica et al. 39 from the University of Milan, both investigations included a similarly sized patient cohort in which the majority of participants had severe hemophilia. The average age of the patients was also comparable, with a similarly broad age range. Furthermore, a high proportion of participants in both studies had an educational background beyond compulsory schooling, indicating a well-informed patient population.
Both studies clearly show that general information on GT is better known than specific knowledge on eligibility criteria, adverse events, and the mechanism of action, indicating a need for patient education in these particular areas. Short- and long-term side effects in general, and potential carcinogenesis in particular, are the most important concerns of those affected by hemophilia and, as the comparative analyses of both studies show, contribute significantly to GT rejection.
Altogether, the results of the present study indicate a notable increase in participants’ knowledge of GT compared to the findings of Cutica’s study. Indeed, the current study demonstrates a generally higher level of knowledge, with 48% achieving a score above 50% correct answers, compared to only 35% in the Italian sample. This difference may be attributed to the fact that this study was conducted 2 years after Cutica’s research, during which time GT has advanced further, with studies demonstrating long-term efficacy of treatments such as valoctocogene roxaparvovec over 7 years, 36 thereby contributing to its growing establishment and the increased availability of reliable information.
To ensure comparability with the study by Cutica et al., the original questionnaire was used without changes. Patients were not given any information about other new treatment options such as second-generation mimetics, as these were not yet approved at the time of the survey. 47 Therefore, it is unlikely that differences in preferences are related to new therapies.
In our evaluation, all study participants were satisfied with their current treatment regimen (64% very satisfied, 36% satisfied). Interestingly, 77% of respondents reported chronic arthralgia, and 21% experienced more than three bleeding episodes within 1 year. Individual cases of high bleeding frequency were attributed to occupational stress (e.g., construction work) or medical procedures. Overall, 83% of patients reported that they had experienced limitations in various aspects of their lives as a result of their disease. The described limitations are reflected in numerous publications48–51 and underline their relevance in daily clinical practice.
It is particularly noteworthy that, despite these considerable burdens, 59% of the study participants refused GT. Both the high level of satisfaction and the negative attitude are consistent with the findings of our colleagues at the University of Milan. 39
The striking discrepancy between the high level of satisfaction with current therapy and the reported burden of disease contributes to other findings of our study on the decision-making of hemophilia patients: For the first time, ABR is shown to be the only disease marker that highly significantly discriminates between GT refusers and GT acceptors. At the same time, the ABR serves as a key parameter to measure the success of GT in global studies.52–55 This dual function confirms the relevance of ABR from a clinical and patient-centered perspective. While other disease characteristics, such as chronic arthralgia or acute pain events, do not show comparable differences between the groups of those who would refuse GT or accept it, the ABR appears to accurately reflect the patient’s level of suffering and provides a clear basis for decision-making.
At the same time, the discrepancy between subjective satisfaction and objective disease severity suggests that the decision to undergo GT is not solely determined by clinical symptoms but is significantly influenced by patients’ expectations, subjective risk assessment, and individual decision-making style. Regarding the acceptance of GT, patients who would reject GT had significantly higher expectations regarding the duration of efficacy (p = 0.003) and the minimum factor level (p = 0.04) than those who would accept GT.
Overall, however, the majority of study participants rejected GT. On the one hand, this skepticism may be a reflection of the current lack of long-term evidence: While existing data confirm reductions in bleeding rates, stable factor expression for up to 7 years,36,46 and improved quality of life,22,37 the therapy’s effect on slowly evolving complications such as hemophilic arthropathy remains unknown.
On the other hand, the fact that the study population is characterized by a predominantly rational and risk-averse decision-making style might as well contribute to the rejection. 56 These findings are in line with the results of the Cutica study. In this context, the discrepancy between high disease burden and rejection of GT reflects the risk-averse thinking as the expectation of higher efficacy duration and guaranteed factor levels seems to represent a condition under which affected individuals would perceive GT as predictable and supported by clear evidence, and therefore as a viable option to consider.
Furthermore, our study participants were classified as rational decision-makers if they answered questions such as “I check my sources of information before making a decision” or “I make decisions in a logical and systematic manner” positively. For individuals who make decisions based on evidence and systematic thinking, access to trustworthy educational materials and clear medical advice is essential.
To address this, a three-step educational approach could be adopted: First, initial self-directed learning through online materials; second, a consultation with the treating physician to assess individual eligibility, discuss personal risks and benefits, and allow time for reflection; and third, a follow-up discussion to address any remaining questions before the final decision is made.
While several educational formats on GT for PWH already exist,57–60 they are predominantly available in English. It is therefore necessary to develop clear, independent resources in German to ensure equal access to information and enable informed decision-making.
Conclusion
Our study is the first to assess attitudes of PWH in Germany toward GT. While all respondents were satisfied with their current treatment, 59% rejected GT. Rejection was unrelated to age, education, or pain level, but PWH with an ABR ⩾1 were more willing to consider GT (p = 0.001). Concerns about long-term side effects (p = 0.005) and high expectations regarding duration of efficacy and minimum factor levels contributed to rejection. Overall, participants showed only general knowledge of GT. Compared with the Italian cohort studied by Cutica et al., knowledge levels appeared higher, suggesting growing awareness. Further studies should explore how education can improve understanding and acceptance of this therapy.
Limitations of the study
X-linked recessive hemophilia primarily affects men. In extremely rare cases, when both parents are carriers of a diseased gene or in monosomies such as Turner syndrome, women can also show clinical symptoms of the disease.61,62 An exclusively male study population may lead to gender bias that does not adequately reflect the perception of the burden of disease,62,63 the associated impairments, and the specific experiences of symptomatic carriers—and thus also their attitude toward GT, leaving the population of women with hemophilia understudied. 64
Although the questionnaire is based on the instrument developed by Cutica et al., it has not been pilot-tested or validated prior to data collection. Therefore, the validity and reliability of the instrument may be limited, as potential ambiguities were not identified in advance.
In addition, the focus on people with severe factor deficiency might contribute to the limited generalizability as only the responses from this group were submitted. Further studies are needed to explore attitudes toward GT across different severity levels.
The average age of our study participants was 50 years, with 7% under the age of 35. This may skew findings related to joint damage, needs, and risk tolerance, underscoring the need for studies including younger patients.65–67
About 50% of respondents had higher education, diverging from the general German population 68 contradicting the results of the eTHINK study by Mrakotsky et al., 69 which suggested deficits in intellectual and adaptive skills in children with hemophilia. Similar evidence has been found in individuals with other chronic diseases.70,71 Higher education likely facilitated understanding of GT, indicating a potential selection bias among study participants.
A trend emerged where those rejecting GT rated its disadvantages higher and benefits lower than those open to it, but differences were not statistically significant due to the small sample size (n = 69). Larger, more representative studies are needed to validate these trends.
No formal a priori power analysis was performed prior to data collection, as the study was exploratory in nature, and the total number of potential participants was limited due to the rarity of the disease, meaning that statistical significance for small effects may be limited.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261428774 – Supplemental material for An analysis of attitudes toward gene therapy in people with severe hemophilia in Germany, a survey-based cross-sectional study
Supplemental material, sj-docx-1-tah-10.1177_20406207261428774 for An analysis of attitudes toward gene therapy in people with severe hemophilia in Germany, a survey-based cross-sectional study by Svetlana Babayeva, Aida Peyvandi, Christian Schepperle, Günter Auerswald, Steffen Rauchfuss, Flora Peyvandi, Ilaria Cutica and Wolfgang Miesbach in Therapeutic Advances in Hematology
Footnotes
Appendix A
List of statements to rate as true or false on gene therapy.

| Number | Statement |
|---|---|
| 1 | In your country, no hemophilia patient has ever been treated with gene therapy |
| 2 | Gene therapy avoids constant infusions of the missing clotting factor |
| 3 | Several gene therapy clinical trials are currently ongoing worldwide in hemophilia patients |
| 4 | Gene therapy is possible for hemophilia A |
| 5 | Gene therapy is indicated in pediatric patients |
| 6 | Gene therapy is indicated for severe forms of hemophilia |
| 7 | One of the most frequent side effects of gene therapy is the increase of liver inflammation |
| 8 | Gene therapy aims to introduce into the body a functional copy of factor VIII or FIX gene |
| 9 | A pre-existing immunity to adeno-associated viral vectors allows to undergo gene therapy |
| 10 | Gene therapy is possible for hemophilia B |
| 11 | Gene therapy is assessed on the basis of the reduction in the annual bleeding rate |
| 12 | Gene therapy consists of a single intravenous infusion |
| 13 | Adeno-associated viral vectors are used for gene therapy for hemophilia |
| 14 | Gene therapy may have possible long-term side effects of cancer development |
| 15 | Gene therapy clinical trials are ongoing in patients with inhibitors |
| 16 | It might happen that patients in gene therapy may need steroids |
| 17 | Hemophilia patients with previous liver disease were eligible for gene therapy in clinical trials |
Acknowledgements
We thank the patient organizations and the patients for distributing and fulfilling the questionnaire.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
