Abstract
Background:
Connective tissue diseases (CTDs) are a group of autoimmune disorders in which immune thrombocytopenia (ITP) represents a common and important manifestation. Current CTD-ITP management mainly relies on glucocorticoids and immunosuppressive agents.
Objectives:
This systematic review and meta-analysis aims to evaluate the efficacy and safety of immunosuppressive agents in patients with CTD-ITP.
Design:
Systematic review and meta-analysis.
Methods:
This study was conducted using eight databases up to July 22, 2024. Observational studies and experimental trials with relevant efficacy and safety data were included. Methodological quality were evaluated using the Newcastle-Ottawa Scale and the ROBINS-I tool. The Mantel–Haenszel formula with a random effect model was employed to estimate the overall effect size. Subgroup analyses were performed based on the study characteristics, clinical features, and treatment regimen.
Results:
Through integrating 24 studies (1 single-arm clinical trial, 1 case–control, and 22 cohort studies) involving 775 CTD-ITP patients, the pooled estimates of the optimal overall and complete response rate for immunosuppressive therapy were 82% (95% CI: 75–88) and 64% (95% CI: 56–72), with stable results after sensitivity analysis. The combined side-effect incidence was 31% (95% CI: 24–39). The pooled relapse rate was 30% (95% CI: 18–43) among 403 immunosuppressive therapy responders.
Conclusion:
Immunosuppressive treatments exhibited favorable efficacy and safety in CTD-ITP patients. Future larger-scale multicenter studies are needed.
Introduction
Connective tissue diseases (CTDs) are a spectrum of autoimmune conditions, including systemic lupus erythematosus (SLE), primary Sjögren’s syndrome (pSS), rheumatoid arthritis, and others. 1 Immune thrombocytopenic purpura (ITP) is a blood disorder characterized by a decrease in platelet (PLT) count lower than 100 × 109/L, and represents a significant hematological manifestation in various CTDs.2,3 The estimated prevalence of ITP was 12%–20% in patients with SLE4–6 and 12% in patients with pSS. 7 Severe thrombocytopenia is an indicative prognostic risk factor and is associated with a high mortality rate.8–10
Current treatment for CTD-associated immune thrombocytopenia (CTD-ITP) relies heavily on glucocorticoids (GCs) and immunosuppressive agents (ISAs), but repeating treatment courses of these medications may lead to a refractory condition and increased risk of infection.3,8 However, the efficacy and safety of immunosuppressive therapy for CTD-ITP patients have not been systematically evaluated. Furthermore, studies have shown that patients who achieve complete remission at an early stage have a better long-term prognosis. 11 Therefore, a comprehensive evaluation with meta-analysis is essential to consolidate and appraise the existing evidence, providing higher-quality treatment references for patients with CTD-ITP.
This review aims to evaluate the efficacy and safety of immunosuppressive therapy in patients with CTD-ITP. Through systematic review and meta-analysis, data were synthesized from diverse studies to provide clearer guidance on clinical management and further research.
Methods
The research protocol was registered on PROSPERO (CRD42023490991). This study was scheduled, conducted, and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Table S1). 12
Data sources and search strategy
An exhaustive search of the literature was conducted by a medical research librarian (A.A.G.) in the China National Knowledge Infrastructure, Cochrane Library, Google Scholar, Ovid Embase, Ovid Medline, PubMed, Scopus, and Web of Science Core Collection databases to find relevant articles published from the inception of each database to July 22, 2024. Databases were searched using a combination of keywords and controlled vocabulary for thrombocytopenia, CTD, and ISAs. The search was not limited by language, publication type, or year (full search strategies available in Table S2). The search was peer-reviewed by a second medical librarian using the Peer Review of Electronic Search Strategies (PRESS). 13 Forward and backward citation chasing was performed using CitationChaser to identify additional relevant studies not retrieved by the database search. 14
Definitions and eligibility
This study focused on the efficacy and safety of immunosuppressive therapies in CTD-ITP patients with PLT count <100 × 109/L. The exposures of interest included treatment with traditional ISAs, biologic agents, and/or GCs. A detailed list of therapeutic agents included can be found in Table S3.
The outcomes of interest included therapeutic response, ITP relapse rates, and side effects. Overall response (OR) included complete response (CR, PLT >100 × 109/L) and partial response (PR, PLT >30 × 109/L and at least twice of baseline PLT with absence of bleeding). 2 The American Society of Hematology 2 has emphasized three key time points for ITP assessment during follow-up: (1) initial response, defined as meeting response criteria within 1 month after the therapy initiation; (2) durable response, defined as therapy responses in 6 months; and (3) remission, defined as maintaining PLT >100 × 109/L after 12 months.
Experimental and observational studies reporting relevant data on therapy response were eligible for inclusion, regardless of whether safety information was reported. Publications were excluded if they involved secondary thrombocytopenia due to drug, pregnancy, infection, or malignancy; did not include human subjects; presented duplicate or retracted data; case reports or series; or if the full-text manuscript was not accessible. Studies with a sample size of fewer than 10 were also excluded to ensure statistical validity. There are no restrictions on the study time frame or language.
Study selection and data extraction
Search results from all databases were imported into an Endnote 21 library (Clarivate Analytics, London, United Kingdom). Duplicates were removed using the Yale Reference Deduplicator. 15 The deduplicated results were then imported into Covidence (Veritas Health Innovation Ltd, Melbourne, Australia), a systematic review software for screening and data extraction. The title-abstract selection and then the included full-text selection were both implemented by two independent reviewers (Y.Z. and X.Y.) through each phase of the review including screening, eligibility, and inclusion. Data was extracted by independent researchers in parallel and checked consensus. All the selection conflicts were resolved, and the extracted data form was assessed by a third independent reviewer (N.J.). Extracted data included study characteristics (first author, publication year, study design, study location, sample size, follow-up time), baseline clinical features (age, sex, ITP duration, PLT before treatment, CTD type, CTD duration, disease activity, organ involvements, and autoantibody profiles), treatment regimen (immunosuppressive regimen, previous, and other concurrent therapies), efficacy outcomes (OR, CR, PR, and evaluated endpoints if available), relapses, safety events (death, bleeding, infection, allergy, cytopenia, liver or kidney toxicity, gastrointestinal discomfort, central nervous system discomfort, and other side effects reported).
Study quality assessment
The risk of bias was assessed by two independent reviewers (Y.Z. and X.Y.) using the Newcastle-Ottawa Scale (NOS) applicable to case–control and cohort studies. The ROBINS-I (Risk Of Bias In Nonrandomized Studies of Interventions) tool was used to assess the risk of nonrandomized clinical trials. 16 Discrepancies were resolved by a third reviewer. The NOS comparability domain was applied to distinguish and determine the global risk of bias, categorized on a scale from 0 to 2, with 0 = high risk of bias, 1 = moderate risk of bias, and 2 = low risk of bias).
Data synthesis and analysis
The effect sizes were subjected to the Freeman–Tukey double arcsine transformation using “PFT” method in the “metaprop” R package, which is a widely accepted and commonly used method without requiring continuity correction for variables including 0 or 1 values. 17 Subsequently, the pooled response rate across studies was calculated using the inverse weighting method. The Mantel–Haenszel formula with a random effect model was employed to estimate the overall effect size, with 95% confidence intervals (95% CI) reported. Heterogeneity among studies was assessed using the Cochrane Q test and Ι2 statistic. Subgroup analyses were performed based on the following study characteristics: geographic region (North America, Europe, or Asia), study design (prospective, retrospective), language (Chinese, English), study population (refractory or broadly defined CTD-ITP), CTD subtype (SLE, pSS), ITP severity (PLT <100 × 109/L, PLT <50 × 109/L, PLT <30 × 109/L), immunosuppressive therapy regimen (rituximab (RTX), ISAs), and other combination therapy (with, without). Regarding refractory CTD-ITP, no unified definition has yet been established, and there are currently no relevant guidelines or expert consensus. All definitions reported in the included studies were reviewed and summarized in Table S4, after which we adopted the definition that was most commonly used and considered most appropriate. Refractory ITP was defined as having failed at least one course of GC pulse therapy (1 g/d for 3–5 days) and/or intravenous immunoglobulin (20 g/d for 3–5 days) and high-dose GCs in combination with at least one standard ISA, while the broadly defined one was CTD-ITP patients without additional restrictions. Differences between subgroups were tested using the chi-square test. Sensitivity analyses were conducted using the leave-one-out method to evaluate the influence on the overall results. All analyses were performed using R Project (Version 4.3.2, www.r-project.org), with the “meta” package for meta-analysis. P Values lower than 0.05 were considered statistically significant.
Results
Study characteristics
A total of 21,864 citations were identified through systematic literature searching. After removing duplicates, 11,718 remained for title-abstract screening. Full-text review was conducted for 237 articles, with 24 meeting the inclusion criteria (Figure 1). No other studies were added after citation chasing covering the bibliography of all included articles. There were 213 studies excluded due to inappropriate study design, incorrect study population, irrelevant interventions or outcomes, insufficient sample size, missing detailed original data, or conference abstracts (Table S5). A majority of the included studies originated from Asia (16 studies, 66.7%), 5 from Europe, and 3 from North America. Five studies were prospectively designed, while the rest were retrospective. There was one single-arm clinical trial, one case–control study, and the remaining were cohort studies. All the included articles reported a minimum follow-up duration of 6 months. Among the studies, 19 focused on immunosuppressive therapy for refractory CTD-ITP, with 14 addressing refractory SLE-ITP and 8 discussing refractory pSS-ITP. Regarding ITP severity, 8 studies included patients with severe ITP with PLT <30 × 109/L, 6 recruited patients with PLT <50 × 109/L, and 10 defined ITP as PLT <100 × 109/L. In terms of treatment regimens, ISAs included cyclosporin A (CsA), tacrolimus, sirolimus, and others; whereas the most commonly prescribed biological agent was RTX. Hydroxychloroquine (HCQ) was regarded as an immune modulator. GCs were also of concern, which were generally maintained and gradually reduced to a minimum maintenance dose after the addition of immunosuppressive therapies. Fifteen studies reported on RTX, with six utilizing standard doses (375 mg/m2 per week for 4 weeks, or 1000 mg/m2 on day 1 and day 15),11,18–22 eight exploring low-dose regimens (100–200 mg/m2 per week for 2–4 weeks),23–29 and the other two30,31 did not specify dosage. There were 12 studies11,31–40 evaluating the efficacy of traditional immunosuppressants, including 2 on CsA (5 mg/kg/d),27,33 3 on tacrolimus (2–3 mg/d),34–36 and other 2 on sirolimus (0.5–1 mg/d initially, with plasma concentrations maintaining at 6–15 ng/mL for 6 months).37,38 Details on specific regimens, preintervention, and concurrent therapies can be found in Table 1. The demographic, clinical, and immunological characteristics of participants are detailed in Table S6.

Flowchart of methods used for study identification. The flowchart is designed following the PRISMA guidelines (http://www.prisma-statement.org).
Basic characteristics of included studies.
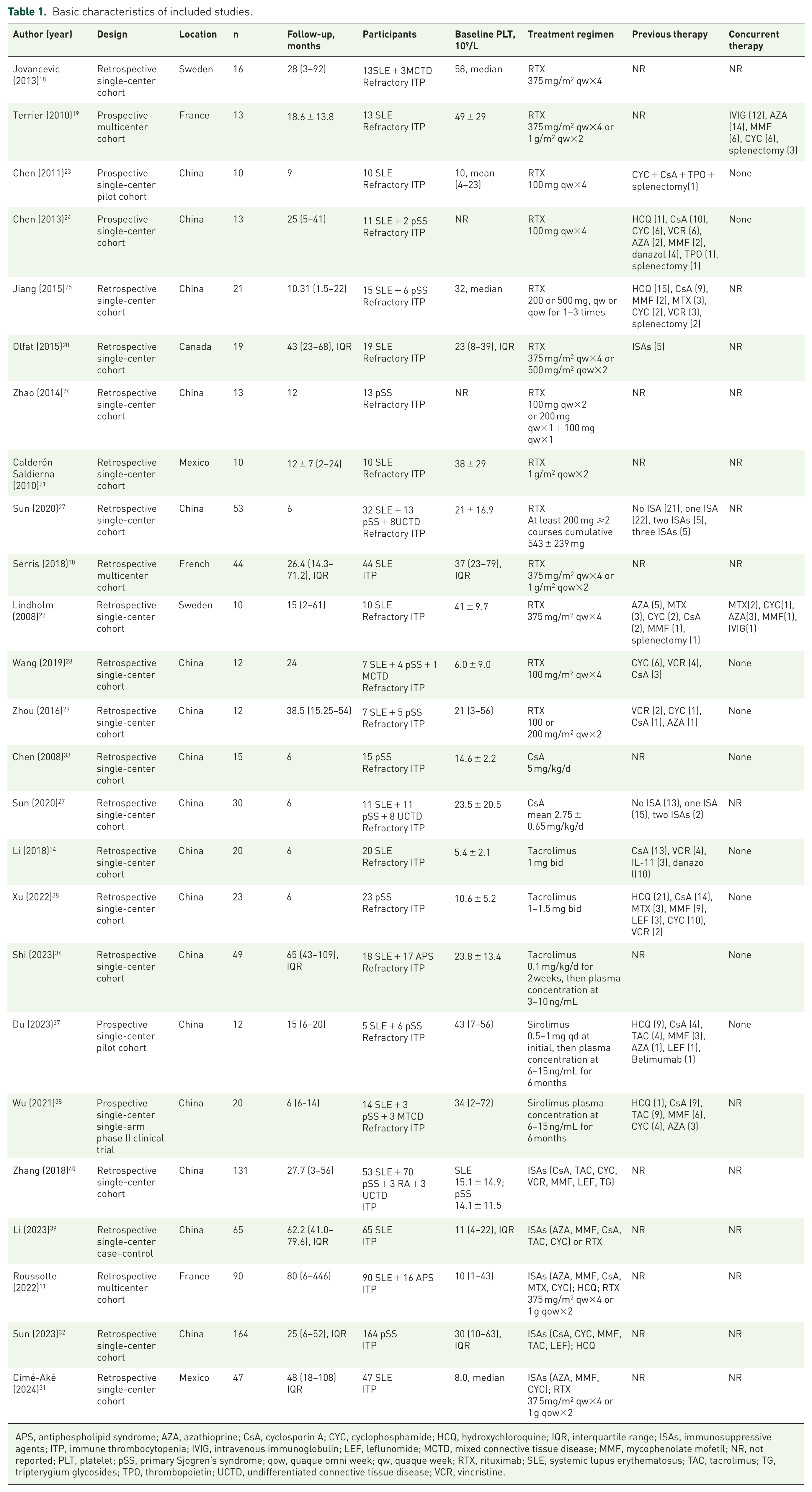
APS, antiphospholipid syndrome; AZA, azathioprine; CsA, cyclosporin A; CYC, cyclophosphamide; HCQ, hydroxychloroquine; IQR, interquartile range; ISAs, immunosuppressive agents; ITP, immune thrombocytopenia; IVIG, intravenous immunoglobulin; LEF, leflunomide; MCTD, mixed connective tissue disease; MMF, mycophenolate mofetil; NR, not reported; PLT, platelet; pSS, primary Sjogren’s syndrome; qow, quaque omni week; qw, quaque week; RTX, rituximab; SLE, systemic lupus erythematosus; TAC, tacrolimus; TG, tripterygium glycosides; TPO, thrombopoietin; UCTD, undifferentiated connective tissue disease; VCR, vincristine.
Risk of bias assessment
The risk of bias in the included observational studies and one interventional research were assessed using the NOS comparability domain and ROBIN-I tool, respectively. Nine studies had a low risk of bias, 10 showed a moderate risk, and 5 exhibited a high risk of bias (Tables S7 and S8).
Efficacy
In Figure 2, the pooled optimal overall response rate (ORR) was 82% (95% CI: 75–88) across 24 studies involving 775 patients, while the pooled optimal complete response rate (CRR) was 64% (95% CI: 56–72) across 21 studies involving 610 patients. High heterogeneity was observed, with I2 values of 80% and 68%, respectively, and Q test p values were both less than 0.01.

Forest plot of the pooled optimal overall response and complete response of immunosuppressive therapies in CTD-ITP patients.
Subgroup analyses of the optimal response rates were conducted based on the study location, types of study design, publication language, and risk of bias, as detailed in Table 2. It was observed that studies from North America reported a higher optimal CRR of 90% (72%–100%) than the other regions (p = 0.01), with a similar trend in ORR (p = 0.09). No significant differences were found among the subgroups in study design, language, or bias risk.
Subgroup analysis of immunosuppressive therapy responses in CTD-ITP patients.
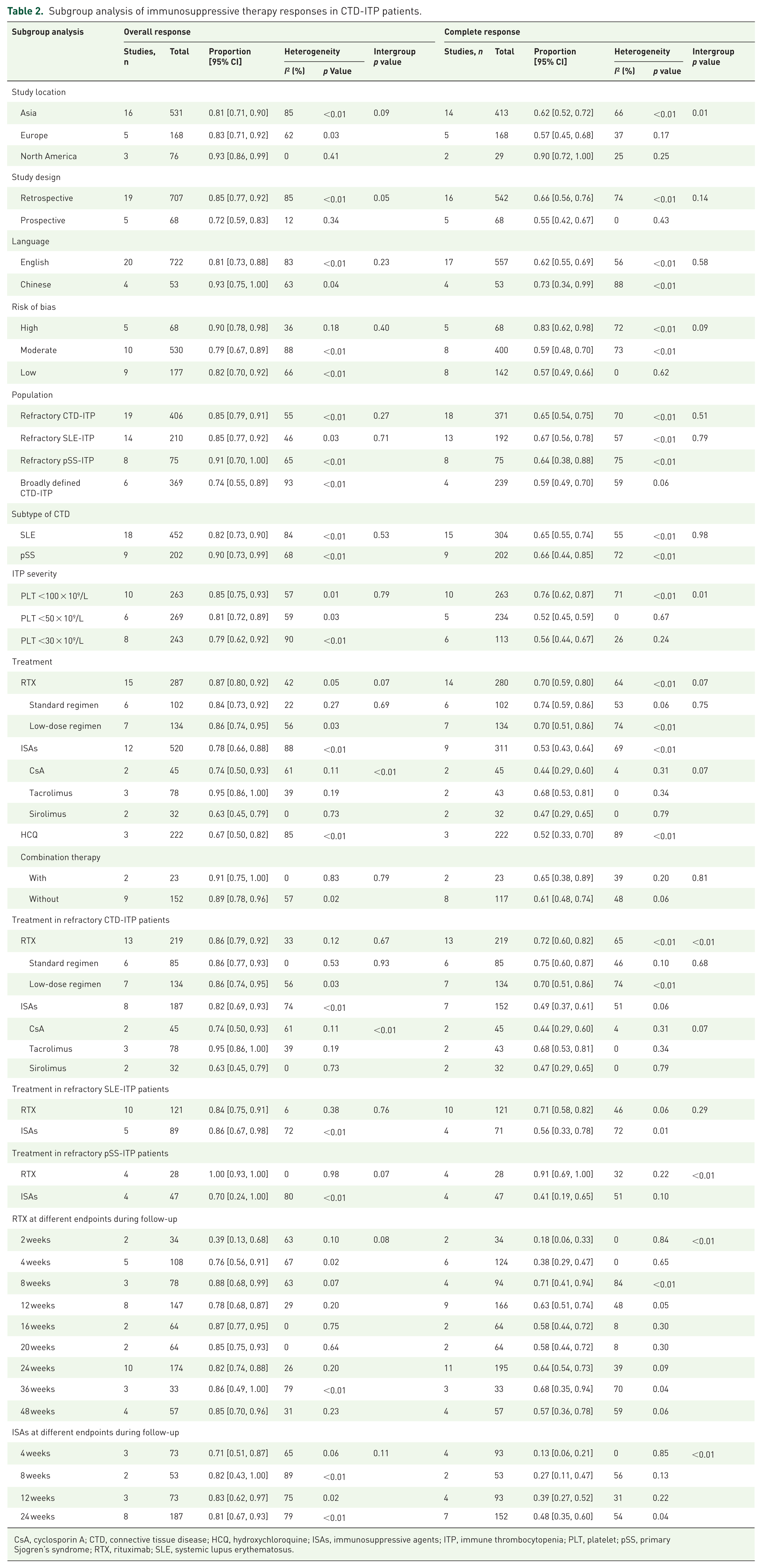
CsA, cyclosporin A; CTD, connective tissue disease; HCQ, hydroxychloroquine; ISAs, immunosuppressive agents; ITP, immune thrombocytopenia; PLT, platelet; pSS, primary Sjogren’s syndrome; RTX, rituximab; SLE, systemic lupus erythematosus.
For different CTD subtypes, the optimal ORR in SLE-ITP patients was 82% (73%–90%) with a CRR of 65% (55%–74%); while in pSS-ITP patients, 90% (73%–99%) achieved OR, and 66% (44%–85%) achieved CR. No statistically significant differences were discovered. Regarding ITP severity, patients with moderate to severe ITP (PLT <30–50 × 109/L) exhibited a lower optimal CRR, compared to those with a broader definition, while no difference was observed between the PLT lower than 30 or 50 × 109/L groups. Similarly, ORR was lower in the moderate to severe ITP group, although its p value did not reach significance.
We also conducted detailed subgroup analyses by medication class and dosages. Three studies11,32,39 reported the therapeutic efficacy of HCQ in CTD-ITP patients, with an ORR of 67% (50%–82%) and a CRR of 52% (33%–70%). Patients receiving RTX showed an optimal ORR of 87% (80%–92%) and CRR of 70% (59%–80%). There were no significant differences between the standard-dose and low-dose RTX groups. Additionally, the ORR for traditional ISAs was 78% (66%–88%) and the CRR was 53% (43%–64%). Specifically for tacrolimus, CsA, and sirolimus, the ORRs were 95% (86%–100%), 74% (50%–93%), and 63% (45%–79%), respectively, while the CRRs were 68% (53%–81%), 44% (29%–60%), and 47% (29%–65%), with tacrolimus displaying the best overall efficacy (p < 0.01).
Most of the included studies focused on therapies for refractory CTD-ITP. RTX had a higher CRR than traditional ISAs (72% vs 49%, p < 0.01), with no difference in ORRs. Furthermore, in refractory pSS-ITP patients, RTX showed a more pronounced CRR advantage over traditional ISAs (91% vs 41%, p < 0.01). A similar trend was also shown in refractory SLE-ITP; however, the p value for intergroup comparison was not significant.
We summarized the response rates at different time points, shown in Table 2. In refractory CTD-ITP patients, RTX infusion presented an initial ORR of 76% (56%–91%) and CRR of 38% (29%–47%). Continued follow-up revealed a durable ORR of 82% (74%–88%) and CRR of 64% (54%–73%), with 57% (36%–78%) ultimately achieving remission. For patients receiving traditional ISAs, 71% (51%–87%) met the OR criteria during the initial phase, while only 13% (6%–21%) were restored to normal PLT levels within the first month. However, the proportion of patients achieving durable OR increased to 81% (67%–93%) and CR to 48% (35%–60%) at 6 months posttreatment.
Sensitivity analysis
Sensitivity analyses were conducted by omitting one study each time to assess its impact on the overall results. As illustrated in Figure S1, the pooled effect sizes for optimal ORR and CRR remained robust, with no significant influence from any single study and only subtle changes in heterogeneity. It was suggested that the results of our meta-analysis were relatively reliable. Additionally, we conducted a sensitivity analysis to analyze the influence of pediatric population. The results showed that, before and after excluding all studies involving pediatric participants, no statistically significant differences were observed in the CRR (p = 0.74) or ORR (p = 0.66) of immunosuppressive therapy.
Relapse
Among all the included articles, 16 reported on relapse events during follow-up. The relapse rate was defined as the proportion of patients experiencing ITP recurrence among those therapy responders. In Figure S2, the pooled relapse rate was 30% (18%–43%) among 403 immunosuppressive therapy responders.
Subgroup analysis revealed a higher relapse rate of 41% (28%–54%) in patients who received RTX, compared with 14% (2%–34%) in those treated with traditional ISAs. Notably, the difference in relapse rates was not significant (p = 0.17) in the two dosage groups, with a relapse rate in studies evaluating standard-dose RTX of 54% (28%–79%) and those evaluating low-dose RTX of 32% (17%–49%). Furthermore, six studies19,21,22,25,29,31 reported relapse time, and demonstrated the first relapse mostly occurred 6–15 months after the initial RTX infusion.
Safety
Altogether, 19 studies with 476 CTD-ITP patients reported safety events (Table 3). A total of 15 severe adverse events (3.2%) were reviewed, including 11 deaths (2.3%), for which severe pulmonary infection was the main cause. The other four cases (0.8%) suffered active bleeding, with intracerebral, subarachnoid, airway, and gastrointestinal hemorrhages. Notably, infection (58 cases, 12.2%) was the most common adverse event of immunosuppressive therapy, primarily presenting as pneumonia or upper respiratory tract infections. Of these, 32 infections occurred in the RTX group and the pooled adverse event incidence was 14% (9%–20%). The second most common safety event was allergic reactions (16 cases, 3.4%). The vast majority occurred in participants receiving RTX, showing a combined side-effect incidence of 4% (1%–8%), while only one allergy case occurred in a patient treated with tacrolimus. Neither infection nor allergy incidence was statistically different between RTX dosage subgroups. Additionally, nine cases (1.9%) reported cytopenia, seven of whom were prescribed traditional ISAs.
Adverse events during follow-up of immunosuppressive therapies in CTD-ITP patients.

CNS, central nervous system; CsA, cyclosporin A; CTD, connective tissue disease; GI, gastrointestinal; HSV, herpes simplex virus; ISAs, immunosuppressive agents; ITP, immune thrombocytopenia; RTX, rituximab; VZV, varicella zoster virus.
Our meta-analysis indicated that the pooled adverse event incidence of immunosuppressive therapy was 31% (24%–39%; Figure S3). Subgroup analysis showed that the combined side-effect incidence for RTX was 30% (23%–37%) and 33% (19%–48%) for traditional ISAs. No significant differences were discovered between groups with different RTX dosages.
Discussion
To the best of our knowledge, this is the first systematic review and meta-analysis focusing on the efficacy and safety of immunosuppressive therapy in patients with CTD-ITP. Across 24 studies involving 775 CTD-ITP patients, the pooled estimate of optimal ORR for immunosuppressive treatment was 82% (75%–88%), and the optimal CRR was 64% (56%–72%), with a combined side-effect incidence of 31% (24%–39%).
ITP is a common hematologic manifestation in CTD patients, 42 with a prevalence of 8%–20% in SLE3,4 and 5%–12% in pSS, 43 respectively. Decreased PLT production and increased PLT destruction play crucial roles in CTD-ITP pathophysiology, primarily mediated by autoantibodies. PLT antigens induce the differentiation from B-cells into plasma cells to secrete autoantibodies, together with CD8+ T-cells participating in PLT destruction. The mechanisms of ITP associated with SLE 3 and pSS 32 are fundamentally similar, which may support their parallel therapeutic responses in our review.
With highly heterogeneous clinical manifestations, CTD-ITP could range from asymptomatic cases to life-threatening bleeding. Currently, there are no guidelines or recommendations for ITP treatment in patients with CTD. However, an increased bleeding risk has been identified in lupus patients with PLT <30 × 109/L, which is regarded as the setpoint for initiation of therapy, 44 consistent with the threshold in primary ITP guidelines. 2 In our review, patients with PLT <30–50 × 109/L exhibited a much lower optimal CRR, compared to those with mild ITP. Our findings suggested the necessity of taking ITP severity into account when formulating the therapeutic strategy.
Corticosteroids, with or without intravenous immunoglobulin, are the first-line therapy regimen for CTD-ITP, inducing rapid responses in approximately 60%–80% of patients; however, only 30%–50% maintain sustainable responses or remissions after treatment discontinuation.3,45 Therefore, to achieve more durable responses and reduce long-term steroid toxicity, immunosuppressants or biologics are often necessary.
HCQ is considered an immunomodulator and cornerstone medicine. Our study summarized the therapeutic efficacy of HCQ in CTD-ITP, with an ORR of 67% (50%–82%) and a CRR of 52% (33%–70%). Consistent with the previous reviews, 46 HCQ serves as a steroid-sparing agent, contributing to overall remissions in over 60% of SLE-ITP patients.
Calcineurin inhibitors, including CsA and tacrolimus, can reduce intracellular calcineurin activity and release of interleukin-2, thereby inhibiting T lymphocyte activation. This literature review identified two studies examining the efficacy of GC + CsA in pSS-ITP. Sun et al. 27 reported an ORR of 76.6%, while Chen et al. 33 observed an ORR of 86.6%. Compared to CsA, tacrolimus was associated with higher efficacy in patients with refractory SLE-ITP34,36 and refractory pSS-ITP 35 (pooled ORR 95% vs 74%, pooled CRR 68% vs 44%), as well as lower incidence of adverse events (pooled side-effect incidence 22% vs 35%). Furthermore, Shi et al. found that patients on tacrolimus were more likely to be able to taper steroids to less than 15 mg/day of GCs within the first 3–6 months. 36 Taken together, tacrolimus may be a more advantageous treatment strategy for patients with CTD-ITP compared with CsA.
Sirolimus is another immunosuppressant inhibiting T-cell activation and proliferation through targeting the mammalian target of rapamycin. Our analysis indicated that sirolimus achieved comparable efficacy to CsA in refractory CTD-ITP (pooled OR 63% vs 74%, pooled CR 47% vs 44%), with no severe adverse events, consistent with findings from a randomized clinical trial (RCT) in patients with primary ITP. 47 As for long-term response, one study 48 discovered that GC + sirolimus was more effective than GC + CsA (68% vs 39%) in patients with chronic ITP. However, the follow-up periods of these CTD-ITP cohorts were all limited to 6 months, preventing the assessment of sustained remission. Further investigations with longer follow-up periods are warranted.
Mycophenolate mofetil (MMF) has been extensively studied in primary ITP, with good efficacy, although its potential bone marrow suppression effect and unknown long-term response remain concerns. 44 A multicenter RCT showed that the steroid-MMF combined regimen induced a higher efficacy than steroid monotherapy (91.5% vs 63.9%) and reduced the refractory or relapse risk. 49 However, relevant therapy data in CTD-ITP are scarce and limited to case reports.
B-cell-targeted therapy is currently one of the most commonly used treatment for ITP. RTX, a chimeric monoclonal anti-CD20 antibody, targets autoreactive B-cells, thereby achieving therapeutic effects. In primary ITP patients, RTX has become the preferred second-line therapy with ORR and CRR ranging from 52%–73% to 20%–54%, respectively, and sustained remission rates of 40%–60% at 1 year, 30%–50% at 2 years, and 20%–30% at 5 years.50,51 Consistently, we found an optimal ORR of 87% (80%–92%) and CRR of 70% (59%–80%) among CTD-ITP patients treated with RTX. Additionally, in refractory CTD-ITP patients, it was shown that 76% achieved OR with 38% achieving CR in the first month; the durable response rate increased to 82% and 64% at 6 months, and the 1-year remission rate reached 57% (36%–78%). Furthermore, most participants discontinued previous ISAs before RTX infusion and gradually reduced GC doses to a minimum, while PLT responses were maintained for nearly 2 years.20,21,25
The standard regimen of RTX is intravenous administration of 375 mg/m2 per week for 4 weeks, while the regimen of 1000 mg/m2 on day 1 and day 15 is considered an alternative with similar efficacy. 52 Meanwhile, a low-dose regimen has also been explored, defined as 100–200 mg/week for 2–4 weeks. Subgroup analysis revealed no significant differences between the standard and low dosage groups in terms of efficacy outcomes, relapse, or adverse event incidence in CTD-ITP patients, consistent with results in primary ITP.51,53 Additionally, it was suggested that dosage and infusion interval adjustment were not related to response improvement, but repeated administration may be effective.25,30 Low-dose RTX may have promising for clinical application, with similar efficacy and safety compared to the standard regimen.
However, certain participants experience RTX treatment failure. One possible explanation is that RTX cannot deplete plasma cells, which may differentiate into long-lived plasma cells promoted by elevated B-cell activating factor (BAFF) levels. 3 Therefore, the BAFF inhibitor belimumab has emerged as an extra option for CD20-targeting nonresponders. A prospective persistent or chronic primary ITP cohort 54 reported a CRR of 66.7% and an ORR of 80% with RTX-belimumab combined therapy. Another study 55 discovered belimumab monotherapy with 90% ORR in refractory SLE-ITP patients. The therapeutic potential of belimumab in refractory ITP is promising and deserves further exploration.
Considering the CTD-ITP heterogeneity and the treatment accessibility, combined GC and traditional ISAs therapy appears to be the first-line option, but potential cytopenia and severe infections should be noted during medication. On the other hand, RTX therapy demonstrated excellent efficacy and well tolerability, especially for refractory CTD-ITP patients, although its long-term remission and recurrence remain unclear. In summary, it is essential to optimize treatment strategies individually, grounded in our understanding of the disease’s pathophysiology.
Several potential limitations warrant mention. First, the included studies exhibit relatively high heterogeneity. To address this, detailed subgroup analyses were conducted. Regrettably, only one study specifically focused on pediatric CTD-ITP, making it difficult to conduct intergroup analyses. To explore the potential influence of age on therapy efficacy, a sensitivity analysis was performed. Although no significant differences were observed, the limited evidence base indicates that our findings should be interpreted with caution, particularly when generalized to underage patients. Second, there was no highest-quality evidence derived from RCTs, and sample sizes were small. It was difficult to draw unbiased comparative conclusions due to the absence of head-to-head studies focusing on different immunosuppressive therapies. Third, the intergroup p values were calculated using chi-square tests without effect sizes accompanying, thus the observed statistical significance should be considered exploratory rather than confirmatory.
Conclusion
In conclusion, this study conducted a comprehensive literature review and meta-analysis on immunosuppressive therapy for patients with CTD-ITP, providing evidence of favorable efficacy and safety. However, the durability of therapy responses remains limited. These findings should therefore be interpreted with caution, and large-scale, high-quality prospective studies are needed to validate the efficacy and clarify the long-term sustainability.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261422963 – Supplemental material for Efficacy and safety of immunosuppressive therapy in connective tissue disease-related immune thrombocytopenia: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207261422963 for Efficacy and safety of immunosuppressive therapy in connective tissue disease-related immune thrombocytopenia: a systematic review and meta-analysis by Yupei Zhang, Xiaxiao Yan, Alyssa A. Grimshaw, Yanhong Wang, Mengtao Li, Xiaofeng Zeng, Evelyn Hsieh and Nan Jiang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
