Abstract
Background:
Conditioning regimen selection significantly impacts the outcomes of allogeneic hematopoietic stem cell transplantation (HSCT) in patients with hematologic malignancies. However, comparative evidence between Busulfan–Fludarabine and Busulfan–Cyclophosphamide remains inconclusive.
Objectives:
To compare the efficacy and safety of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide as conditioning regimens prior to HSCT.
Design:
Systematic review and meta-analysis conducted in accordance with PRISMA 2020 guidelines.
Data sources and methods:
MEDLINE, CENTRAL, and Embase were searched through October 2024. Eligible studies included randomized controlled trials and cohort studies comparing Busulfan–Fludarabine and Busulfan–Cyclophosphamide in HSCT recipients. Primary outcomes included overall survival and acute graft-versus-host disease (GVHD). Risk ratios (RRs) with 95% confidence intervals (CIs) were pooled using random-effects models. Risk of bias was assessed using RoB 2.0 and the Newcastle–Ottawa Scale.
Results:
Eighteen studies (6 randomized controlled trials, 12 cohorts) comprising 2888 patients (1539 received Busulfan–Fludarabine, 1349 received Busulfan–Cyclophosphamide) were included. Busulfan–Fludarabine showed higher 1-year overall survival (RR 1.13, 95% CI: 1.01–1.26), but no significant difference at 2 or 5 years. Grade III–IV acute GVHD was significantly lower with Busulfan–Cyclophosphamide (RR 0.45, 95% CI: 0.21–0.98). Busulfan–Fludarabine resulted in lower 5-year non-relapse mortality (RR 0.63, 95% CI: 0.48–0.83), and significantly reduced pulmonary and gastrointestinal toxicities. Event-free survival favored Busulfan–Fludarabine at 2 and 5 years. No significant differences were found for relapse-related mortality, chronic GVHD, cytomegalovirus infection, or total mortality. Meta-regression identified conditioning regimen and graft source as contributors to 1-year survival variability.
Conclusion:
Busulfan–Fludarabine offers early survival and toxicity advantages, while Busulfan–Cyclophosphamide may reduce severe acute GVHD. Conditioning regimen selection should consider patient-specific factors. Further prospective trials are needed to guide clinical decisions.
Trial registration:
PROSPERO ID: CRD42025630836.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (HSCT) is a potentially curative treatment for hematologic malignancies. However, it is also associated with many complications, such as aGVHD (acute graft-versus-host-disease), 1 infection, sinusoidal obstructive syndrome (SOS), 2 neurologic complications, 3 and damage to cardiovascular, gastrointestinal, and respiratory systems. 4
Patients set to undergo stem cell transplantation are first treated with a conditioning regimen of certain drugs, which greatly influences the effects of HSCT. Two well-known conditioning regimens primarily used in myeloid malignancies (e.g., Acute Myeloid Leukemia (AML), Myelodysplastic Syndromes (MDS), Chronic Myeloid Leukemia (CML) are Busulfan–Fludarabine and Busulfan–Cyclophosphamide5,6; it may play a role in attenuating the adverse events or might even decrease their intensity depending on the drug regimen. An optimized conditioning regimen critically influences transplant preparedness and ultimately determines clinical success, defined as sustained engraftment without major complications.
Busulfan–Cyclophosphamide is reported to have higher incidences of regimen-related toxicity, aGVHD, and chronic graft-versus-host-disease (cGVHD), and a higher duration of hospital stay, while Busulfan–Fludarabine was reported to have higher cumulative incidence of graft failure and second HSCT.7,8 A study reported Busulfan–Fludarabine and Busulfan–Cyclophosphamide to have similar non-relapse mortality (NRM) rates, while overall survival was less in the patients conditioned with Busulfan–Fludarabine. 9 On the other hand, another study reported Busulfan–Fludarabine and Busulfan–Cyclophosphamide to demonstrate comparable outcomes like overall survival (OS), GVHD, and relapse rate (RR). 10
Given the conflicting evidence regarding conditioning regimen outcomes, we conducted this meta-analysis to quantitatively compare Busulfan–Fludarabine and Busulfan–Cyclophosphamide. We hypothesized that Busulfan–Fludarabine would demonstrate lower regimen-related toxicity and NRM, while Busulfan–Cyclophosphamide would be associated with superior disease control through reduced relapse incidence. Our primary objective was to statistically synthesize evidence across endpoints, including OS, relapse incidence, NRM, GVHD, and treatment-related complications. This comparative effectiveness analysis aims to inform evidence-based regimen selection for allogeneic HSCT candidates, optimizing survival while minimizing adverse events.
Methods
By the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, this comprehensive review and statistical synthesis was performed. The research protocol was officially registered with the International Prospective Register of Systematic Reviews (PROSPERO) with the ID number CRD42025630836. 11
Search strategy
A comprehensive digital search was conducted in MEDLINE, Central, and Embase from their inception until October 2024. The Medical Subject Headings (MeSH) keywords employed included “Busulfan” AND “Fludarabine” OR “BuFlu,” combined with “Busulfan” AND “Cyclophosphamide” OR “BuCy”. In addition, terms related to transplantation were incorporated, such as “Stem Cell Transplantation, Hematopoietic,” “Transplantation, Hematopoietic Stem Cell,” and “Allogenic Hematopoietic Stem Cell Transplantation.” Furthermore, the bibliographies of all identified articles were examined to find additional relevant studies. The details of the search strategy are summarized in the Supplemental File.
Study selection and eligibility criteria
All articles were imported into EndNote 20 (Clarivate, Philadelphia, PA, USA), and duplicates were removed manually and through algorithmic deduplication. Two authors (M.A.A. and A.F.) independently reviewed the titles and abstracts of the identified studies, excluding articles that did not address the research question or did not meet the predefined selection criteria. Any conflicts or disagreements were resolved through discussions with a third author (A.A.). The full text of the remaining articles was then reviewed to determine their relevance. A third investigator was consulted to resolve any discrepancies during the full-text assessment.
Eligibility criteria
The inclusion criteria for this research were as follows:
Patients with hematologic malignancies undergoing HSCT.
Treatment protocol involving Busulfan–Fludarabine.
Comparison group receiving Busulfan–Cyclophosphamide.
Main outcome evaluated: OS and aGVHD.
Acceptable research methodologies: randomized controlled trials (RCTs) and cohorts.
The exclusion criteria were as follows:
Case reports, editorials, reviews, or studies without original data.
Studies without a control group or those not designed as RCTs or cohort studies.
Insufficient data for outcomes such as OS, aGVHD, and event-free survival (EFS).
Non-English publications or inaccessible full texts.
Conditioning regimens including additional drugs or protocols beyond Busulfan–Fludarabine and Busulfan–Cyclophospha-mide.
Data extraction and outcomes
Data extraction was conducted independently by two authors (T.A. and M.U.A.) using a predetermined Microsoft Excel spreadsheet, with any conflicts resolved by a third author (A.A.). The extracted data encompassed the following study characteristics: study ID, study design, number of male patients in each conditioning regimen, median age, conditioning regimens, diseases included in the study, GVHD prophylaxis, graft source, donor type, and median follow-up duration. The study’s primary outcomes were OS at 1, 2, and 5 years, and grade I, II–IV, and III–IV aGVHD. The secondary outcomes included EFS at 1, 2, and 5 years; limited and extensive cGVHD; relapse-related mortality (RRM); NRM at 1, 2, and 5 years; cytomegalovirus (CMV) infection; relapse-free survival (RFS) at 1, 2, and 5 years; relapse incidence (RI) at 1, 2, and 5 years; total mortality (TM); and regimen-related toxicity (RRT).
Risk of bias
Two reviewers (F.K. and H.K.) independently assessed the risk of bias in the selected studies using the Cochrane “Risk of Bias” tool (RoB 2.0) 12 for RCTs and the Newcastle–Ottawa Scale (NOS) for observational studies. 13 The RoB 2.0 tool evaluated bias based on randomization methods, deviations from intended interventions, missing outcome data, measurement of outcomes, and reporting biases. Studies were classified as having low risk, some concerns, or high risk of bias. For cohort studies, the NOS assessed quality across three domains: selection, comparability, and outcome. We applied thresholds to convert the Newcastle–Ottawa scores to Agency for Healthcare Research and Quality (AHRQ) standards (good, fair, and poor). The assessment was independently performed by two reviewers (T.A. and M.A.M.), with disagreements resolved by discussion or consultation with a third reviewer (A.A.). The selection domain was rated with a maximum of four stars, the comparability domain with a maximum of two stars, and the outcome domain with a maximum of three stars. Studies scoring 7–9 stars were rated as “low risk of bias,” studies scoring 5–6 stars were rated as “some concerns,” and studies scoring less than 5 stars were rated as “high risk of bias,” ensuring a comprehensive evaluation.
Data analysis
Review Manager (Version 5.4, The Cochrane Collaboration, 2020) was employed for statistical analyses. The effect measure of interest for categorical variables was the risk ratio (RR) with a 95% confidence interval (CI), while for continuous variables, the mean and standard deviation were used. All results were pooled using random-effects models. A p-value <0.05 was considered statistically significant. The I2 statistic 14 was used to assess heterogeneity across the studies, with values of 0%, 25%, 50%, and 75% indicating no, low, moderate, and high levels of heterogeneity, respectively. We performed a meta-regression using a mixed-effects model with treatment type, proportion of peripheral blood grafts, and their interaction as moderators. Log-odds of survival were the dependent variable, and heterogeneity was assessed using (Cochran’s Q statistic for residual heterogeneity) (QE) and τ2. Analyses were conducted in R using the metafor package, with significance set at p < 0.05.
Results
Search results
The original literature search yielded 3546 articles. Four hundred twenty-one studies were duplicates, which were removed. A total of 3127 unique articles remained. The articles were screened based on the titles and abstract; screening excluded 3046 articles, leaving 81 for full-text screening against eligibility criteria. Finally, 18 studies were included, with 6 RCTs and 12 cohorts. The selection is summarized in Figure 1.
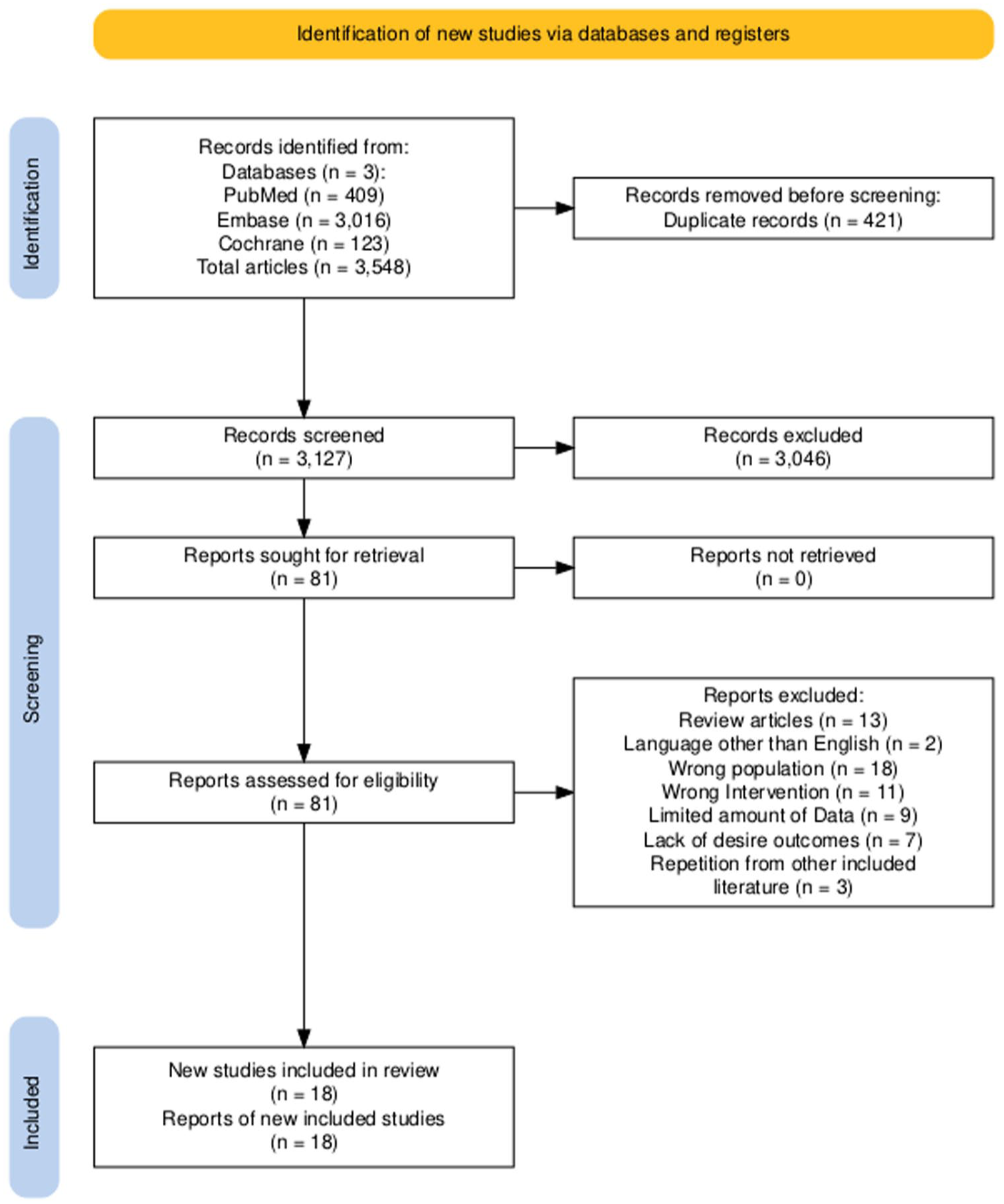
PRISMA flow chart.
Study characteristics
This meta-analysis incorporated 18 studies, including 6 RCTs5,9,15–18 and 12 cohort studies,19–30 all meeting the eligibility criteria. Among the cohort studies, one was a prospective cohort, 30 while the remaining 11 were retrospective cohorts.19–29 Notably, three studies were presented as abstracts,15,23,28 with their full texts yet to be published. A total of 2888 patients were included in this analysis, with 1539 receiving Busulfan–Fludarabine and 1349 receiving Busulfan–Cyclophosphamide as treatments. Baseline characteristics of the included studies are summarized in Table 1.
Baseline characteristics.

Median follow-up in months for both arms.
Number without percentage.
Median follow-up in months without range.
Days in both arms.
ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ATG, anti-thymocyte globulin; BM, bone marrow; Bu, Busulfan; CD, cord blood; CML, chronic myeloid leukemia; CR, complement receptor; CS, cyclosporine; Cy, Cyclophosphamide; Flu, Fludarabine; MDS, myelodysplastic syndromes; MMF, mycophenolate mofetil; MP, mycophenolate; MPN, myeloproliferative neoplasms; MTX, methotrexate; NHL, non-Hodgkin lymphoma; PBSC, peripheral blood stem cells; TAC, tacrolimus.
Bias assessment
A total of 6 RCTs and 12 cohort studies were included in the quality assessment. One RCT was judged to have a low risk of bias while 5 RCTs have some concerns two of which due to domains of Deviations from the intended interventions and one of which due to domain of measurement of the outcome and two of which due to domains of Deviations from the intended interventions and Measurement of the outcome. Of the cohort studies, 11 were rated as good quality, whereas one was considered fair quality due to concerns in the selection domain. The detailed quality evaluation of the included studies is presented in Figure S1 and Table S2.
Meta-analysis of primary outcomes
Overall survival
This outcome was reported by 9 studies (1-year follow-up), 13 studies (2-year follow-up), and 8 studies (5-year follow-up). OS was significantly better in Busulfan–Fludarabine at 1 year (RR 1.13, 95% CI: 1.01–1.26: p = 0.03), but no significant differences were observed at 2 years (RR 1.01, 95% CI: 0.88–1.16; p = 0.86) or 5 years (RR 1.08, 95% CI: 0.99–1.17: p = 0.10) (Figures 2–4).
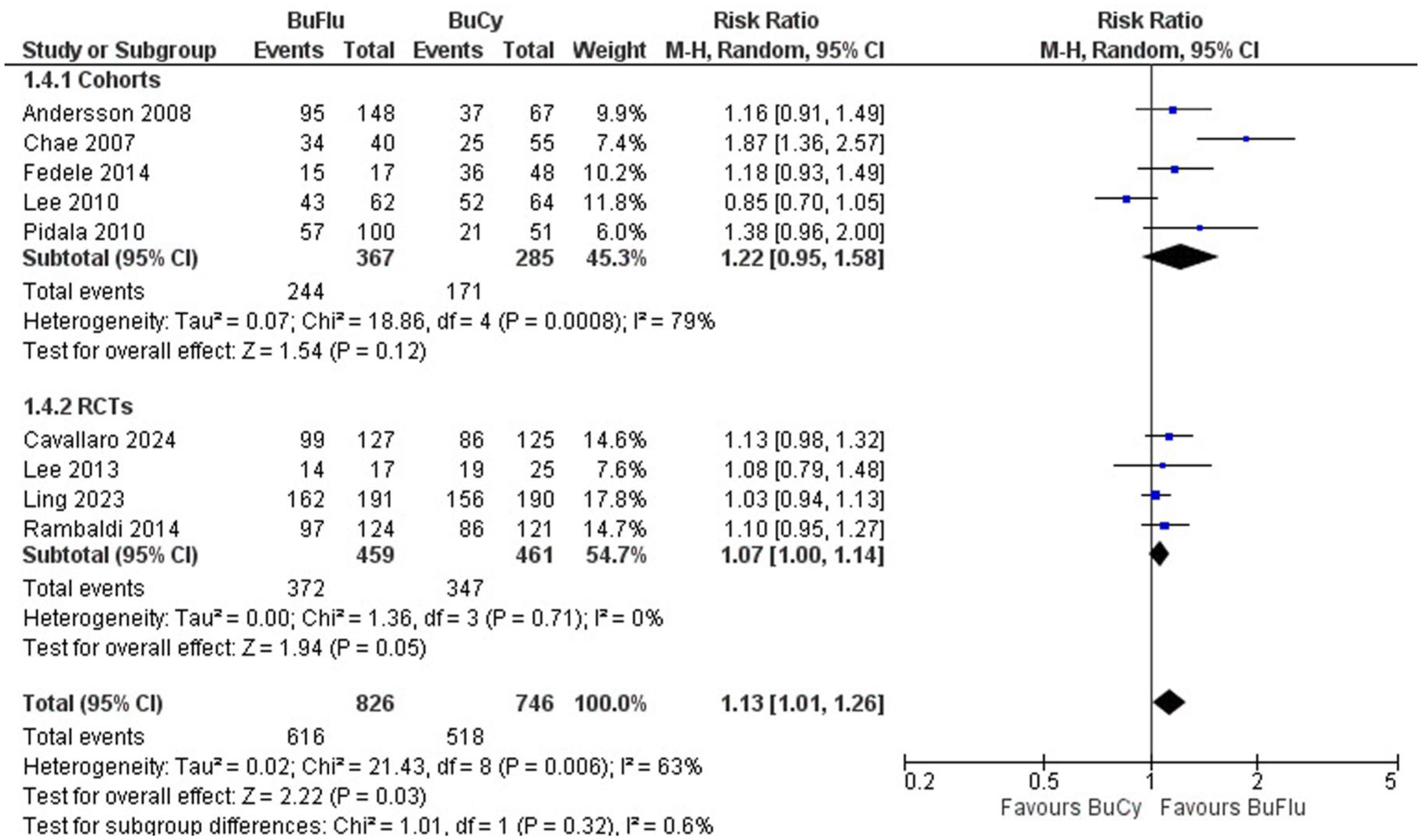
Forest plot of 1-year OS.
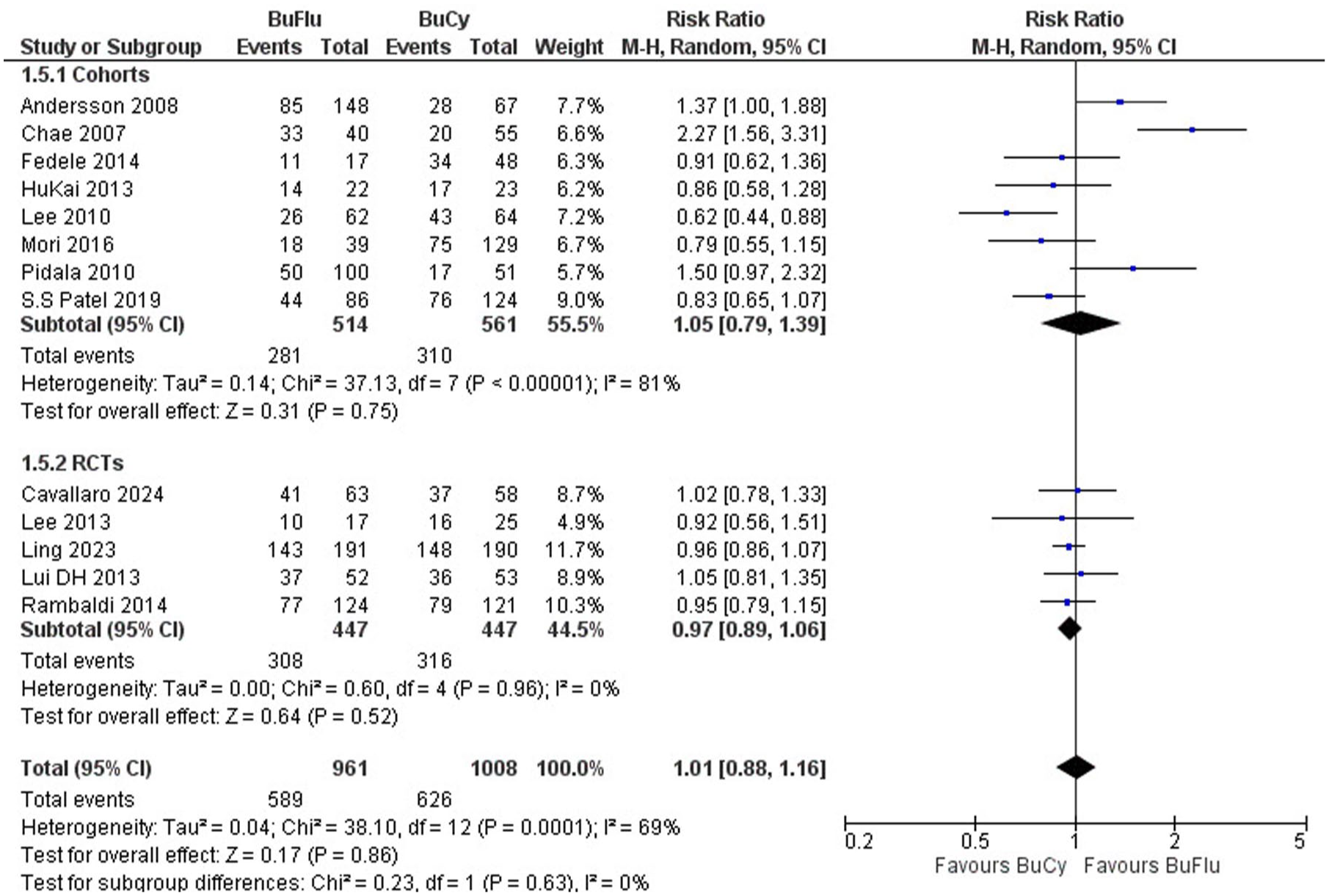
Forest plot of 2-year OS.

Forest plot of 5-year OS.
Acute graft-versus-host disease
This outcome was reported by 4 studies for grade I aGVHD, 9 for grade II–IV aGVHD, and 10 for grade III–IV aGVHD. aGVHD was significantly lower in Busulfan–Cyclophosphamide in grade III–IV (RR 0.45, 95% CI: 0.21–0.98; p = 0.04, I2 = 86%), while no significant difference was noted for grade II–IV aGVHD (RR 0.86, 95% CI: 0.55–1.34; p = 0.51, I2 = 78%) and grade I aGVHD (RR 1.50, 95%CI: 0.80–2.79; p = 0.20, I2 = 64%) (Figures 5–7).

Forest plot of grade 3–4 aGHVD.

Forest plot of grade 2–4 aGHVD.

Forest plot of grade 1 aGHVD.
Meta-analysis of secondary outcomes
Event-free survival
Eight studies at 1-year follow-up reported this outcome, 10 studies at 2-year follow-up, and 4 studies at 5-year follow-up. EFS at 2 years showed borderline significance (RR 1.18, 95% CI: 1.00–1.38; p = 0.05) and 5 years follow-up (RR 1.11, 95% CI: 1.01–1.24; p = 0.04) were significantly higher in Busulfan–Fludarabine, though 1-year EFS was not significantly different between the two groups (RR 1.10, 95% CI: 0.97–1.25; p = 0.15) (Table S3).
Chronic graft-versus-host-disease (cGVHD)
Three studies reported this outcome for limited cGHVD and nine studies for extensive cGHVD. The results showed that there were no significant differences between the limited (RR 0.76, 95% CI: 0.44–1.32, p = 0.33, I2 = 52%) and extensive cGVHD (RR 0.92, 95% CI: 0.66–1.29; p = 0.63, I2 = 36%) (Table S4).
Relapse-related mortality
This outcome was reported by 9 studies (695 patients in Busulfan–Fludarabine and 645 in Busulfan–Cyclophosphamide). The results showed no significant difference between Busulfan–Fludarabine and Busulfan–Cyclophosphamide considering relapse-related mortality (RR 0.93, 95% CI: 0.76–1.14, p = 0.48, I2 = 36%) (Table S5).
Non-relapse-related mortality
Eight studies reported the outcome in 1-year follow-up, 8 studies at 2-year follow-up, and 5 studies at 5-year follow-up. The results showed that NRM was significantly lower in the Busulfan–Fludarabine at 5-year follow-up (RR 0.63, 95% CI: 0.48–0.83, p = 0.001, I2 = 12%). However, there was no significant difference between the two groups at 1-year (RR 0.70, 95% CI: 0.41–1.17, p = 0.17, I2 = 62%) and 2-year follow-up (RR 0.76, 95% CI: 0.47–1.23, p = 0.26, I2 = 74%) considering the NRM (Table S6).
Cytomegalovirus infection CMV infection
This outcome was reported by 8 studies (338 in Busulfan–Fludarabine vs 433 in Busulfan–Cyclophosphamide). Results showed no significant differences between the two regimens for CMV infection (RR 0.93, 95% CI: 0.62–1.37, p = 0.70, I2 = 64%) (Table S5).
Relapse-free survival
This outcome was reported by 7 studies in the 1-year follow-up group, 8 in the 2-year follow-up period, and 4 in the 5-year follow-up period. The results showed that there was no significant difference between the two regimens considering the RFS at all follow-up durations: 1-year (RR 1.00, 95% CI: 0.80–1.25, p = 0.99, I2 = 9%), 2-year (RR 1.04, 95% CI: 0.85–1.27, p = 0.70, I2 = 10%), and 5-year (RR 1.17, 95% CI: 0.90–1.52, p = 0.24, I2 = 0%) (Table S7).
Relapse or progression incidence
Seven studies reported this outcome at 1-year follow-up, 8 studies at 2-year follow-up, and 4 studies at 5-year follow-up. The results showed that there was no significant difference between two regimens at all follow-ups: 1-year (RR 1.00, 95% CI: 0.80–1.25, p = 0.99, I2 = 9%), 2-year (RR 1.04, 95% CI: 0.85–1.27, p = 0.70, I2 = 10%), and 5-year (RR 1.17, 95% CI: 0.90–1.52, p = 0.24, I2 = 0%) (Table S8).
Total mortality
Eight studies reported this outcome. The results showed no significant difference between the two regimens for TM (RR 0.80, 95% CI: 0.60–1.06, p = 0.12, I2 = 74%) (Table S5).
Regimen-related toxicity
(a) Cardiac toxicity: The toxicity was reported by four studies, with results showing no significant difference between the two regimens (RR 0.63, 95% CI: 0.39–1.03, p = 0.07, I2 = 0%) (Table S9).
(b) Liver toxicity: This toxicity was reported by 9 studies (785 patients in Busulfan–Fludarabine vs 562 in Busulfan–Cyclophosphamide). The results showed no significant difference between the two groups (RR 0.73, 95% CI: 0.45–1.18, p = 0.20, I2 = 52%) (Table S9).
(c) Neurological toxicity: This outcome was reported by three studies. The results showed no significant difference between the two groups (RR 0.56, 95% CI: 0.17–1.91, p = 0.36, I2 = 0%) (Table S9).
(d) Pulmonary toxicity: This toxicity was reported by five studies. The results showed that pulmonary toxicity was significantly lower in the Busulfan–Fludarabine regimen (RR 0.42, 95% CI: 0.18–0.97, p = 0.04, I2 = 0%) (Table S9).
(e) Renal toxicity: The toxicity was reported by three studies. The results showed no significant differences between the two regimens (RR 0.45, 95% CI: 0.17–1.19, p = 0.11, I2 = 0%) (Table S9).
(f) Gastrointestinal toxicity: The toxicity was reported by two studies with results showing that the Busulfan–Fludarabine has significantly lower GI toxicities (RR 0.63, 95% CI: 0.49–0.81, p = 0.0002, I2 = 0%) (Table S9).
Sensitivity analysis for heterogeneity
The sensitivity analysis revealed significant reductions in heterogeneity across various outcomes upon removing specific studies (see Figures S2–S5).
Meta-regression analysis
The meta-regression revealed significant residual heterogeneity among studies (QE(12) = 51.24, p < 0.0001), indicating that the included moderators did not fully explain variability in survival outcomes. However, the overall test of moderators was statistically significant (QM(3) = 11.60, p = 0.0089), suggesting that treatment, peripheral blood graft proportion, or their interaction contributed meaningfully to explaining heterogeneity.
Individually, none of the moderator coefficients reached statistical significance, including the interaction term between treatment and graft source (β = 0.8888, CI: −0.8248 to 2.6023, p = 0.3093). Visual inspection suggested a trend toward improved survival with higher PB graft proportion in the Busulfan–Cyclophosphamide group, although this effect was not statistically confirmed (Figure S6).
Discussion
The selection of a conditioning regimen is a critical factor in the success of HSCT, particularly for patients with hematologic malignancies. These regimens aim to achieve myeloablation or immunosuppression, enabling donor cell engraftment while eradicating residual malignant cells. 31 Busulfan, a myeloablative alkylating agent, is frequently used in these regimens due to its ability to induce DNA cross-linking, ultimately leading to cell apoptosis. 32 Busulfan therapy is effective; however, high doses pose challenges such as toxicity, a narrow therapeutic range, and variability in pharmacokinetics among patients. 33 Bu has traditionally been combined with Cyclophosphamide and has been effective in treating acute leukemias, 34 However, it is associated with significant toxicities and a high rate of transplantation-related mortality, particularly in older and frail patients. 35 Fludarabine, a purine analog, offers a potentially lower toxicity profile while preserving the immunosuppressive effectiveness of Cyclophosphamide. 36 Unlike Cyclophosphamide, it is associated with significantly lower NRM and fewer regimen-related toxicities, such as sinusoidal SOS, a severe liver complication caused by endothelial damage and inflammation. 37 The Busulfan–Fludarabine combination offers several advantages, including minimal hepatotoxicity, and when administered at the right time, it enhances Busulfan-induced cytotoxicity by interfering with the repair of DNA damage caused by radiation and alkylating agents. 27 An additional factor influencing HSCT outcomes is the graft-versus-leukemia (GVL) effect, where the donor’s immune system detects and eliminates cancerous cells, playing a crucial role in the therapeutic efficacy of HSCT for treating blood cancers. 38 Busulfan–Cyclophosphamide regimens are known to enhance the GVL effect. 39
In our meta-analysis comparing Busulfan–Fludarabine and Busulfan–Cyclophosphamide as conditioning regimens for HSCT in patients with hematologic malignancies, the primary outcomes revealed that Busulfan–Fludarabine showed a significant improvement in OS at 1 year, but no significant differences were found at 2 or 5 years. GVHD was significantly lower in the Busulfan–Cyclophosphamide group, particularly for grade III–IV aGVHD. Regarding secondary outcomes, Busulfan–Fludarabine demonstrated better EFS at both 2 and 5 years, but no difference was seen at 1 year. NRM was significantly lower in the Busulfan–Fludarabine group at 5 years, with no differences observed at 1 or 2 years. There were no significant differences between the regimens in terms of relapse-related mortality, CMV infection, RFS, or TM. Busulfan–Fludarabine was associated with significantly lower pulmonary and lower GI toxicity compared to Busulfan–Cyclophosphamide, while both regimens showed similar results in terms of cardiac, liver, neurological, renal, and upper GI toxicities. Studies have demonstrated that the Busulfan–Fludarabine regimen is a safe and effective option for allo-HSCT in patients with hematological malignancies. For instance, Slavin et al. highlighted the efficacy of flu (180 mg/m2) in a reduced-intensity conditioning regimen, achieving an OS rate of 85% and a Disease-Free Survival (DFS) rate of 81% after a median follow-up of 8 months in HLA-matched peripheral blood stem cell transplants. 40 Similarly, Chae et al. found that the Busulfan–Fludarabine regimen offers better survival outcomes than Busulfan–Cyclophosphamide, primarily due to its association with reduced rates of SOS, Treatment-Related Mortality (TRM), and GVHD. 20 In addition, Dai et al. 41 emphasized the effectiveness of Busulfan–Fludarabine as a conditioning regimen for patients with acute leukemia undergoing allo-HSCT, noting its strong antileukemic effects and low rate of transplant-related adverse events.
Busulfan–Fludarabine demonstrated a 13% relative improvement in 1-year OS compared to Busulfan–Cyclophosphamide. While this finding was statistically significant, the clinical benefit appears modest, especially given the lack of a corresponding OS advantage at 2 and 5 years. In absolute terms, assuming a baseline 1-year OS of 65% in the Busulfan–Cyclophosphamide group (based on pooled data), this translates into an approximate increase to 73.5% in the Busulfan–Fludarabine group, an absolute gain of 8.5 percentage points. Such improvement may hold value, particularly in older or frail patients, though it must be weighed against long-term survival equivalence.
A potential concern with the Busulfan–Fludarabine regimen is that its reduced organ toxicity could lead to a diminished antileukemic effect or a higher risk of relapse compared to the Busulfan–Cyclophosphamide regimen. 42 Nonetheless, this potential trade-off between toxicity and relapse risk underscores the importance of patient selection when choosing a conditioning regimen. For patients with advanced age or pre-existing organ dysfunction, the reduced intensity of Busulfan–Fludarabine may outweigh the slightly increased relapse risk. The heterogeneity observed in 1-year OS data highlights differences in study designs and populations. Studies involving younger and healthier cohorts might favor Busulfan–Cyclophosphamide due to its greater myeloablative intensity, whereas studies including older or high-risk patients might show a stronger preference for Busulfan–Fludarabine because of its improved safety profile.
By contrast, the incidence of severe (grade III–IV) aGVHD was 55% lower in the Busulfan–Cyclophosphamide group than in the Busulfan–Fludarabine group. Assuming a baseline risk of 20% for grade III–IV aGVHD with Busulfan–Fludarabine, this corresponds to an estimated reduction to 9% with Busulfan–Cyclophosphamide. This marked decrease is clinically significant, particularly for patients at elevated risk of immune-mediated complications, and may guide regimen selection in such cases.
The immunomodulatory effect observed with Busulfan–Cyclophosphamide may be explained by the active metabolites of Cyclophosphamide, which suppress T-cell proliferation and mitigate donor T-cell-mediated injury to critical target organs such as the skin, gastrointestinal tract, and liver—hallmark sites of aGVHD pathology. 43 Supporting this, Russell et al. 44 described a conditioning regimen comprising high-dose Fludarabine (250 mg/m2), Busulfan (12.8 mg/kg), and thymoglobulin, which achieved an exceptionally low grade III–IV aGVHD rate of just 3%, with chronic GVHD observed in 38% of patients over 2 years. While other studies, including Paina et al., 45 reported a numerically higher incidence of both acute and chronic GVHD with Busulfan–Cyclophosphamide compared to Busulfan–Fludarabine, these differences did not reach statistical significance, suggesting variability in patient populations and prophylaxis strategies may influence outcomes.
Busulfan–Fludarabine also demonstrated favorable outcomes in EFS at 2 and 5 years, with 18% and 11% relative improvements, respectively, compared to Busulfan–Cyclophosphamide. These improvements, while statistically significant, reflect modest absolute benefits, roughly 6%–10% increases in patients remaining relapse-free and alive without complications over time. This suggests better long-term disease control and fewer regimen-related adverse events with the Busulfan–Fludarabine regimen, respectively, is likely attributable to its lower RRT, which reduces NRM and allows patients to remain event-free for a longer duration.45,46
Several studies corroborate our findings of better long-term EFS with Busulfan–Fludarabine. For instance, Russell et al. 44 found that Busulfan–Fludarabine regimens were associated with better long-term DFS in AML patients, especially those with intermediate-risk disease. Multiple clinical trials have indicated that Busulfan–Fludarabine, when used as a myeloablative conditioning regimen, results in fewer RRTs, reduced NRM, and higher DFS rates in comparison to Busulfan–Cyclophosphamide for allo-HSCT.28,47,48 The heterogeneity in 1-year EFS is consistent with the variability seen in the mentioned studies, where differences in patient characteristics, disease risk, and donor types significantly impact short-term outcomes.
While both Busulfan–Fludarabine and Busulfan–Cyclophosphamide effectively suppress host immunity to prevent graft rejection, the similar rates of cGVHD suggest that neither regimen confers a significant advantage in modulating the long-term alloimmune response. It is also worth noting that the heterogeneity observed in our analysis for limited cGVHD (I2 = 52%) was eliminated after removing the study by Chae et al. 20 This suggests that variability in patient populations, conditioning protocols, or reporting methods in specific studies may have influenced the overall results. For example, Chae et al. 20 included a cohort with a higher proportion of unrelated donors, a known risk factor for cGVHD, which may have biased the findings.
Our meta-analysis found no significant difference in RRM between Busulfan–Fludarabine and Busulfan–Cyclophosphamide (RR: 0.93, p = 0.48, I2 = 36%). Ben-Barouch et al. 34 reported no significant differences in relapse rates or RRM between Busulfan–Fludarabine and Busulfan–Cyclophosphamide in a large cohort of patients undergoing myeloablative conditioning, aligning with our findings. However, several concerns are raised that the slightly less myeloablative intensity of Busulfan–Fludarabine may be associated with a marginally higher risk of relapse in high-risk hematologic malignancies, particularly when minimal residual disease is present before transplantation. 48 In addition, Busulfan–Fludarabine was associated with a 37% lower risk of NRM at 5 years when compared to Busulfan–Cyclophosphamide. Clinically, this reduction may translate to a decrease in NRM from approximately 25% to 16%, primarily due to reduced toxicity-related complications. This benefit is particularly meaningful in patients with existing comorbidities or organ dysfunction, where minimizing treatment-related mortality is a key priority.
CMV infection remains a major post-transplant complication, often influenced by patient and donor CMV serostatus and immune reconstitution rather than conditioning regimen choice. The similar rates of CMV infection between Busulfan–Fludarabine and Busulfan–Cyclophosphamide suggest that neither regimen significantly impacts CMV reactivation risk. For example, Xuan et al. 49 found no significant differences in CMV reactivation between various conditioning regimens, suggesting that other factors, such as donor and recipient CMV serological status, aGVHD, T-cell depletion, and the use of anti-thymocyte globulin, are more critical determinants of CMV risk.
While the lower RR for cardiac toxicity in the Busulfan–Fludarabine group suggests a trend toward reduced risk, it did not reach statistical significance (RR: 0.63, 95% CI: 0.39–1.03, p = 0.07, I2 = 0%). Cyclophosphamide has well-documented cardiotoxic effects, particularly at high doses. The underlying mechanism of high-dose Cyclophosphamide-induced cardiac toxicity is thought to involve endothelial injury, which allows toxic metabolites to escape into surrounding tissues, causing myocyte damage, interstitial hemorrhage, and edema. Fludarabine, on the other hand, lacks significant cardiotoxicity, which may explain the trend favoring Busulfan–Fludarabine.50,51 Although our analysis did not find a statistically significant difference, Busulfan–Fludarabine’s lower cardiotoxicity may still be clinically relevant in patients with known cardiovascular comorbidities.
The Busulfan–Fludarabine regimen’s trend toward lower liver toxicity is consistent with studies, which indicate that Fludarabine is rarely associated with transient elevations in liver enzymes during treatment and is infrequently linked to clinically significant acute liver injury with jaundice. 5 Neurological toxicity was reported by three studies and showed no significant differences between the two regimens. Both regimens include Busulfan, which is associated with dose-dependent neurotoxicity, including seizures and encephalopathy. However, the addition of Fludarabine in Busulfan–Fludarabine versus Cyclophosphamide in Busulfan–Cyclophosphamide does not appear to significantly alter the risk of neurological complications. Existing studies reported comparable rates of neurotoxicity between the regimens. Importantly, prophylactic measures, such as the use of anti-seizure medications (e.g., levetiracetam), have significantly mitigated the neurotoxic effects of Busulfan in modern practice, reducing overall neurological toxicity in both regimens. 52
Cyclophosphamide is associated with renal toxicity, primarily due to its active metabolite acrolein, which causes direct tubular and glomerular injury. However, the lack of significant differences between the two regimens in our analysis suggests that renal toxicity is not a major differentiator between Busulfan–Fludarabine and Busulfan–Cyclophosphamide, possibly due to advancements in supportive care, such as hydration protocols and the use of mesna to mitigate Cyclophosphamide’s renal effects.53,54
The meta-regression analysis examining 1-year OS in our study provides crucial insights into the comparative effectiveness of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide conditioning regimens in HSCT. The significant omnibus test of moderators (QM(3) = 11.60, p = 0.0089) for 1-year OS indicates that conditioning regimen type, peripheral blood graft proportion, and their interaction collectively explain meaningful variation in early survival outcomes. This finding is particularly relevant given that 1-year survival rates in contemporary studies comparing Busulfan–Fludarabine and Busulfan–Cyclophosphamide typically range from 66% to 85%, making the detection of moderator effects clinically meaningful. 35 The persistent residual heterogeneity (QE(12) = 51.24, p < 0.0001) suggests that additional unmeasured factors continue to influence 1-year survival outcomes beyond the included moderators. The non-significant individual moderator coefficients, despite the significant omnibus test, may reflect the multifactorial nature of 1-year survival determinants. Early survival after transplantation depends on complex interactions between conditioning intensity, graft characteristics, patient factors, and supportive care measures. In addition, the visual trend suggesting improved 1-year survival with higher peripheral blood graft proportion in the Busulfan–Cyclophosphamide group, while not statistically confirmed, aligns with known biological mechanisms. Peripheral blood stem cells provide faster neutrophil and platelet recovery compared to bone marrow grafts, which may be particularly beneficial in the context of more intensive conditioning regimens like Busulfan–Cyclophosphamide. 55
The interaction term coefficient (β = 0.8888, CI: −0.8248 to 2.6023, p = 0.3093) suggests considerable uncertainty about the magnitude and direction of this interaction effect on 1-year survival. However, the upper confidence interval boundary indicates potential for a clinically meaningful benefit, particularly relevant for early survival outcomes where rapid engraftment can prevent infectious complications. The observed trend has important implications for treatment selection, as 1-year survival differences may inform risk-benefit calculations for individual patients.
A key strength of this meta-analysis is the inclusion of RCTs and observational studies, providing a comprehensive evaluation of the efficacy and safety profiles of Busulfan–Fludarabine and Busulfan–Cyclophosphamide. The use of sensitivity analyses to address heterogeneity further strengthens the validity of the findings, particularly for outcomes with high initial variability, such as OS and NRM. In addition, our analysis covers a broad range of primary and secondary outcomes, offering insights into survival, relapse, and regimen-related toxicities.
However, the study has several limitations. First, the small number of included studies (n < 10) precluded a formal assessment of publication bias, as statistical tests (e.g., funnel plots, Egger’s regression) are underpowered and unreliable in such cases (Sterne et al., 2011)56,57. Heterogeneity in study populations, protocols, and follow-up durations remained a challenge despite sensitivity analyses. Differences in patient characteristics, such as age, disease risk, and comorbidities, may have influenced the pooled estimates. Furthermore, the reliance on retrospective studies for some outcomes introduces potential biases, including selection bias and unmeasured confounding. The inclusion of three studies available only as abstracts may also undermine the reliability of extracted data due to limited methodological details and a lack of peer review. In addition, the absence of reported data on long-term quality of life and late complications restricts our ability to assess the regimens’ enduring effects, which is an important consideration for clinical decision-making. Finally, variability in GVHD prophylaxis and supportive care practices across studies limits the generalizability of the findings.
In conclusion, Busulfan–Fludarabine and Busulfan–Cyclophosphamide regimens offer distinct advantages and drawbacks. Busulfan–Fludarabine provides improved early OS and better long-term EFS with lower toxicity, making it suitable for older or comorbid patients. Busulfan–Cyclophosphamide, while associated with higher toxicity, may offer a stronger anti-leukemic effect in high-risk populations. Optimal regimen selection should consider patient-specific factors, disease characteristics, and potential trade-offs between efficacy and toxicity.
Conclusion
This meta-analysis provides a robust comparison of Busulfan–Fludarabine and Busulfan–Cyclophosphamide as conditioning regimens for allogeneic HSCT in patients with hematologic malignancies. While Busulfan–Fludarabine demonstrated superior outcomes in terms of 1-year OS, long-term EFS, reduced NRM, and lower rates of pulmonary and gastrointestinal toxicities, Busulfan–Cyclophosphamide was associated with a significantly lower incidence of severe grade III–IV aGVHD, suggesting a potential advantage in immune-mediated disease control. Importantly, both regimens exhibited comparable efficacy regarding relapse-related mortality, TM, and chronic GVHD. These findings underscore the need for individualized conditioning regimen selection based on patient-specific factors such as age, comorbidities, disease risk, and tolerance to toxicity. Future prospective trials with standardized protocols and long-term follow-up are warranted to further refine regimen choice and optimize patient outcomes in HSCT.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251407534 – Supplemental material for Efficacy and safety of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide as a conditioning regimen prior to hematopoietic stem cell transplant in hematologic malignancy patients: a meta-analysis of randomized controlled trials and observational studies
Supplemental material, sj-docx-1-tah-10.1177_20406207251407534 for Efficacy and safety of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide as a conditioning regimen prior to hematopoietic stem cell transplant in hematologic malignancy patients: a meta-analysis of randomized controlled trials and observational studies by Aizaz Ali, Muhammad Abdullah Ali, Abdullah Afridi, Hammad Ali, Umair Ul Haq, Muhammad Osama, Muhammad Umar Afridi, Touba Azeem, Mian Aban Masaud, Fazia Khattak, Fiza Safeer, Areeba Khan, Muhammad Sufyan Darwesh, Nimra Ehsan, Wahab Zia, Gulmeena Riffat, Abdul Moeez and Kamil Ahmad Kamil in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251407534 – Supplemental material for Efficacy and safety of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide as a conditioning regimen prior to hematopoietic stem cell transplant in hematologic malignancy patients: a meta-analysis of randomized controlled trials and observational studies
Supplemental material, sj-docx-2-tah-10.1177_20406207251407534 for Efficacy and safety of Busulfan–Fludarabine versus Busulfan–Cyclophosphamide as a conditioning regimen prior to hematopoietic stem cell transplant in hematologic malignancy patients: a meta-analysis of randomized controlled trials and observational studies by Aizaz Ali, Muhammad Abdullah Ali, Abdullah Afridi, Hammad Ali, Umair Ul Haq, Muhammad Osama, Muhammad Umar Afridi, Touba Azeem, Mian Aban Masaud, Fazia Khattak, Fiza Safeer, Areeba Khan, Muhammad Sufyan Darwesh, Nimra Ehsan, Wahab Zia, Gulmeena Riffat, Abdul Moeez and Kamil Ahmad Kamil in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
