Abstract
Background:
Anti-thymocyte globulin (ATG) is used as prophylaxis for graft-versus-host disease (GvHD) and graft failure (GF) in allogeneic hematopoietic stem cell transplantation (allo-HSCT) for patients with transfusion-dependent thalassemia (TDT). However, the optimal dose of ATG remains unknown.
Objectives:
To compare the 3-year overall survival (OS) of 8 mg/kg and 10 mg/kg ATG in allo-HSCT for patients with TDT.
Methods:
We compared two different doses of ATG for patients with TDT undergoing allo-HSCT from HLA-matched sibling donors (MSDs). Between 2015 and 2023, 147 patients were randomized to the 8 mg/kg ATG group and 148 patients to the 10 mg/kg ATG group. Primary endpoints were 3-year OS and thalassemia-free survival (TFS). Survival analysis for OS and TFS was performed using the Kaplan–Meier method.
Results:
The 8 and 10 mg/kg ATG groups had similar 3-year OS and TFS (both 98.6% (95% confidence interval [CI]: 96.8–100) vs 95.3% (95% CI: 91.9–98.7), p = 0.066). There were no significant differences in graft-versus-host disease and GF between groups. In the subgroup with co-transplantation of cord blood (CB) and bone marrow (BM) as stem cell sources, the 8 mg/kg ATG group had higher 3-year OS and TFS than the 10 mg/kg ATG group (both 100% (95% CI: not applicable) vs 86.5% (95% CI: 75.5–98.2), p = 0.012).
Conclusion:
This study provides evidence that an 8 mg/kg ATG is an effective treatment for TDT transplanted from MSDs. A notable finding was the superior survival associated with the 8 mg/kg ATG in the CB and BM co-transplantation. These results provide critical evidence for guiding ATG dosing strategies in allo-HSCT for TDT, particularly in the context of stem cell source selection.
Design:
Open-label, randomized clinical trial.
Trial registration:
ChiCTR-IPR-15005779 (Chinese Clinical Trial Registry; https://www.chictr.org.cn/showproj.html?proj=10208; date of registration: December 29, 2014).
Plain language summary
Anti-thymocyte globulin (ATG) is used to prevent graft failure and graft-versus-host disease in allogeneic hematopoietic stem cell transplantation. However, the optimal ATG dose for patients with thalassemia remains unclear. This study compared outcomes between a control dose (10 mg/kg) and an experimental lower dose (8 mg/kg) of ATG. The results indicated that both doses led to similarly high survival rates and the lower dose was associated with better outcomes in patients who received a co-transplantation of cord blood and bone marrow.

Keywords
Introduction
Transfusion-dependent thalassemia (TDT), an inherited hemolytic disorder resulting from defects in the globin gene, necessitates lifelong red blood cell transfusion and regular iron chelation. Allogeneic hematopoietic stem cell transplantation (allo-HSCT) with human leukocyte antigen (HLA)-matched sibling donors (MSDs) has been the gold standard treatment for TDT for decades, with thalassemia-free survival (TFS) reaching 80%–90%.1–3
Anti-thymocyte globulin (ATG) has been used as prophylaxis for graft-versus-host disease (GvHD) in allo-HSCT. It is associated with a reduced risk of graft failure (GF) and is accepted as part of standard conditioning regimens in allo-HSCT for patients with TDT. 4 However, the clinical outcomes associated with ATG dosage are variable, primarily due to differences in the formulation, dosing, and timing, as well as variations in donor types, stem cell sources, and intensity of conditioning regimens.5–9 Furthermore, ATG is associated with delayed immune reconstitution, which increases the risk of viral reactivation, particularly cytomegalovirus (CMV) or Epstein–Barr virus (EBV). 10
The optimal dosage of ATG in malignant hematological diseases has been extensively studied, with efforts aimed at balancing the prevention of GvHD, maintaining graft-versus-leukemia (GVL) effects, and reducing infection risks.10–14 However, in the context of allo-HSCT for TDT, the ideal dose of ATG remains unclear. ATG dosing varies across centers, with doses ranging from 4.5 to 12.5 mg/kg (rabbit ATG), despite different conditioning regimens being used across various centers.5–9 In a previous study, we reported a 3-year TFS of 97.8% in TDT patients who received MSDs transplanted with a 10 mg/kg dose of rabbit ATG. 15 Despite these findings, the optimal dose of ATG for preventing GF and GvHD in TDT patients remains unresolved. Therefore, we performed an open-label, randomized clinical trial to compare the outcomes of 3-year OS of 8 mg/kg ATG with those of 10 mg/kg ATG (Supplemental Material).
Methods
Study design
This single-center, open-label, randomized clinical trial was conducted at the First Affiliated Hospital of Guangxi Medical University in Nanning, Guangxi, China. Data analysis and outcome evaluation were conducted in a masked manner.
The primary endpoints were 3-year OS and TFS. The secondary endpoints were rates of engraftment, GF, GvHD-free and relapse-free survival (GRFS), grades 2–4, 3–4 acute GvHD (aGvHD), chronic GvHD (cGvHD), transplantation-related mortality (TRM), infection-related mortality (IRM), and post-transplantation lymphoproliferative disorder (PTLD). Inclusion criteria for the study were as follows: (1) diagnosis of beta TDT, using hemoglobin electrophoresis and genetic analysis; (2) blood transfusion dependence; (3) aged between 1 and 18 years; (4) having MSDs and the MSDs were recommended to donate bone marrow (BM) and peripheral blood stem cells, or cord blood (CB) and BM as stem cell source; (5) an ejection fraction greater than 50% of the cardiac tissue; and (6) normal results for pulmonary examinations, and normal kidney function. Exclusion criteria were as follows: (1) Human Immunodeficiency Virus positive result; (2) uncontrolled fungal, viral, or bacterial infections; (3) CMV or EBV levels exceeding 200 copies/mL, as determined by quantitative polymerase chain reaction (PCR); or (4) liver dysfunction (alanine aminotransferase or aspartate aminotransferase >4-fold the upper limit of the normal range based on our institution’s laboratory criteria).
The choice of two ATG dosages (8 and 10 mg/kg) was based on our center’s experience, where 10 mg/kg was the standard of care. 15 The 8 mg/kg dose was selected as a rational intermediate dose for initial de-escalation, balancing the need for effective immunosuppression against the goal of reducing drug-related toxicity. Once evaluated as eligible, patients were assigned to either the control group (10 mg/kg ATG) or the experimental group (8 mg/kg ATG) according to a randomization principle, after obtaining written consent. This trial used a simple randomization method using the random number table. The allocation ratio was 1:1. The study duration spanned from July 2015 to July 2024, covering patient recruitment and follow-up. Patients were enrolled between July 2015 and December 2023.
Conditioning regimen and GvHD prophylaxis
The conditioning regimen consisted of busulfan (Bu, 1 mg/kg i.v. 4× daily on days -9 to -6), fludarabine (Flu, 50 mg/m2 i.v. once daily from days -12 to -10), cyclophosphamide (Cy, 50 mg/kg i.v. once daily, days -5 to -2), and ATG (Genzyme Polyclonals S.A.S., Lyon, France) (2.5 mg/kg i.v. once daily, days -4 to -1 in the 10 mg/kg ATG group or 2.0 mg/kg i.v. once daily, days -4 to -1 in the 8 mg/kg ATG group). 15 Before transplantation, all patients received 20–30 mg/kg hydroxyurea orally once daily for 2–3 months. The GvHD prophylaxis included cyclosporine (CsA, 1.5–3 mg/kg/d), methotrexate (MTX, 15 mg/m2 × 1 d + 10 mg/m2 × 3 d), and mycophenolate mofetil (MMF, 250 mg × 30 d). 15
Stem cell collection
Patients with MSDs received G-CSF-mobilized BM and peripheral blood stem cells (G-BM and PBSCs), 15 or CB and BM stem cells. 16 Patients were assigned to two subgroups: (1) CB and BM: If a related HLA-matched CB was available, co-transplantation of CB and BM from the same sibling donor was preferred; and (2) G-BM and PBSCs: In the absence of a related HLA-matched CB, co-transplantation of mobilizing BM and PBSCs from a single sibling donor was considered. For the CB and BM subgroup, the median total mononuclear cell count was 2.1 × 108/kg (interquartile range (IQR) 1.4–3.2 × 108/kg), consisting of 2.8 × 107/kg (IQR 1.6–5.0 × 107/kg) from CB and 1.9 × 108/kg (IQR 1.1–2.9 × 108/kg) from BM. For the G-BM and PBSCs subgroup, G-CSF (5 mg/kg of body weight per day for 5 days) was used to mobilize the G-BM and PBSCs. 11 The median total mononuclear cell was 14.7 × 108/kg (IQR 11.3–18.0 × 108/kg), consisting of 6.5 × 108/kg (IQR 4.5–8.5 × 108/kg) from G-BM and 7.9 × 108/kg (IQR 5.5–11.2 × 108/kg) from PBSCs. The recommended ratio of mononuclear cells of BM to PBSCs was 1:1–2, as previously described. 15
Definitions
Engraftment was considered when the neutrophil count increased to >0.5 × 109/L, and the platelet count increased to >20 × 109/L over three consecutive days. GF was defined as the absence of donor-derived DNA on at least two consecutive PCR tests, no less than 1 week apart. CMV infection was characterized by CMV-DNA ⩾ 1000 copies/mL detected using two consecutive PCRs. GvHD was classified according to the Glucksberg and NIH classifications.17,18 OS was defined as the time from transplantation until death from any cause. TFS was defined as the time from transplantation to TDT recurrence and death or the last follow-up. GRFS was defined as the absence of relapse, death from any cause, grades 3–4 aGvHD, and cGvHD that requires systemic treatment. 19 TRM was defined as death resulting from transplantation rather than thalassemia recurrence. IRM was defined as the death of patients due to an infection and was the primary cause of death.20,21 We reported the incidence of hemorrhagic cystitis (HC) for all grades, which was judged according to the Droller criterion. 22
Statistical analysis
Sample size calculation was performed using PASS software (version 21.0.3, NCSS, LLC, Kaysville, UT, USA), based on the 3-year OS of 97.8% with 10 mg/kg ATG in the conditioning regimen at our center. 15 We hypothesized that, when the OS in the 8 mg/kg ATG group was below 90%, there was a significant difference compared to the 10 mg/kg ATG group.1–3 Assuming an alpha of 0.05 for a two-sided test and a statistical power of 0.80, the sample size was calculated to be 288 patients (144 in each group).
All statistical analyses were performed using IBM SPSS Statistics 27 (IBM Corp., Armonk, NY, USA) and R version 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables are summarized as medians and IQR, and categorical variables as proportions. Continuous variables were compared using the Mann–Whitney U test, whereas categorical variables were analyzed using the chi-square and Fisher’s exact tests. The survival analyses for OS, TFS, GRFS, and TRM were performed by the Kaplan–Meier method. Hazard ratios were estimated with the Cox proportional hazards model, which included hepatomegaly and stem cell source as covariates. The Fine and Gray model was applied to assess the cumulative incidence of outcomes in the presence of competing risks. For aGvHD within 100 days after transplantation, GF, death, and relapse without corresponding grades of aGvHD were considered competing risks. For cGvHD and moderate-severe cGvHD, GF, death, relapse, and loss to follow-up without corresponding grades of cGvHD were treated as competing risks.
Results
Study population
Between July 2015 and December 2023, 301 patients from the First Affiliated Hospital of Guangxi Medical University were evaluated. Two patients were excluded due to age criteria, and four due to uncontrolled bacterial infections. A total of 295 patients were eligible for the final analysis, with a median age of 7 years (IQR, 5–9 years, and range 2–17 years). The study flow diagram is shown in Figure 1. The median follow-up period was 59 months (IQR 31.3–81.8). In addition to liver size, other baseline characteristics, including age, sex (female donor-male recipient), splenectomy status, serum ferritin levels, ABO incompatibility, liver size, alanine aminotransferase levels, infused mononuclear cells, and infused CD34+ cells, were similar between the 8 mg/kg ATG and 10 mg/kg ATG groups (Table 1).

Patient flowchart.
Baseline characteristics.

BM, bone marrow; CB, umbilical cord blood; G, granulocyte colony-stimulating factor; HLA, human leukocyte antigen; LIC, liver iron content tested by magnetic resonance imaging; MNC, mononuclear cell; PBSCs, peripheral blood stem cells; T2*, spin-spin relaxation time.
Engraftment
All patients achieved primary engraftment, and none experienced primary GF. The median time to neutrophil engraftment was 11 days (IQR 10–13) in both the 8 and 10 mg/kg ATG groups. The median time to platelet engraftment was 14 days (IQR, 12–20) in both the 8 and 10 mg/kg ATG groups (Table 2). Chimerism evaluation and therapeutic interventions were based on methods outlined in our previous study. 15 The overall incidence of mixed chimerism (MC) was 7.8%. No patients progressed to secondary GF.
Transplantation outcomes.

aGvHD, acute graft-versus-host disease; ATG-8, 8 mg/kg ATG group; ATG-10, 10 mg/kg ATG group; cGvHD, chronic graft-versus-host disease; CI, confidence interval; CMV, cytomegalovirus; EBV, Epstein–Barr virus; GRFS, graft-versus-host disease and relapse-free survival; HC, hemorrhagic cystitis; HR, hazard ratio; IQR, Interquartile range; OS, overall survival; PRES, posterior reversible encephalopathy syndrome; TFS, thalassemia-free survival; TRM, transplant related mortality; VOD/SOS, hepatic veno-occlusive disease/sinusoidal obstruction syndrome.
Outcomes
The 3-year OS, TFS, and GRFS of all patients were 96.9% (95% CI: 95.0–98.9), 96.9% (95% CI: 95.0–98.9), and 88.9% (95% CI: 84.7–92.0), respectively. The 3-year OS of the 8 mg/kg ATG group was similar to that of the 10 mg/kg ATG group (98.6% (95% CI: 96.8–100) vs 95.3% (91.9–98.7), p = 0.066; Figure 2). The 3-year TFS in both groups was consistent with the OS. The 3-year GRFS of the 8 mg/kg ATG group was similar to that of the 10 mg/kg ATG group (88.9% (95% CI: 82.4–93.0) vs 88.9% (95% CI: 82.4–93.1), p = 0.949; Figure 2). Nine patients died (six with severe infections, two with PTLD, and one with hemorrhage; two in the 8 mg/kg ATG group and seven in the 10 mg/kg ATG group). No significant difference in TRM was observed between the 8 mg/kg (1.4%, 95% CI: 0–3.2) and 10 mg/kg (4.7%, 95% CI: 1.9–9.5) ATG groups (p = 0.066).

Kaplan–Meier curves for (a) overall survival, (b) thalassemia-free survival, and (c) GvHD-free and relapse-free survival.
Graft-versus-host disease
The cumulative incidences of grades 2–4, 3–4 aGvHD, and moderate-severe cGvHD for all patients were 15.0% (95% CI: 11.1–19.4), 5.7% (95% CI: 3.4–8.9), and 4.6% (95% CI: 2.6–7.5), respectively. The cumulative incidences of grades 2–4, 3–4 aGvHD, and moderate-severe cGvHD were similar between the 8 mg/kg ATG and 10 mg/kg ATG groups (17.6% (95% CI: 11.8–24.3) vs 12.4% (95% CI: 7.6–18.3), p = 0.200; 7.2% (95% CI: 3.7–12.4) vs 4.2% (95% CI: 1.7–8.5), p = 0.270; and 5.0% (95% CI: 2.2–9.6) vs 4.2% (95% CI: 1.7–8.4), p = 0.750; Table 2 and Figure 3).

Nelson-Aalen curves for grade 2–4 aGvHD (a), grade 3–4 aGvHD (b), overall cGvHD (c), and moderate-severe cGvHD (d). For grades 2–4 aGvHD and grades 3–4 aGvHD, GF, death, and relapse without corresponding grade aGvHD were considered as competing risks. For cGvHD and moderate-severe cGvHD, GF, death, relapse, and loss to follow-up without corresponding grade cGvHD were considered as competing risks.
Other complications
The rates of CMV reactivation and EBV reactivation were similar between the 8 mg/kg ATG and 10 mg/kg ATG groups (33.3% vs 35.8%, p = 0.655, and 12.9% vs 14.9%, p = 0.630; Table 2). However, the incidence of HC in the 8 mg/kg ATG group was significantly lower than that in the 10 mg/kg ATG group (15.0% vs 25.7%, p = 0.022; Table 2).
Outcomes of subgroups
Baseline characteristics were comparable between the two ATG doses in the subgroups (Table 3). In the CB and BM subgroup, the 3-year OS, TFS, and GRFS were 94.1% (95% CI: 89.3–99.3), 94.1% (95% CI: 89.3–99.3), and 89.4% (95% CI: 80.7–94.4), respectively. The 3-year OS in the 8 mg/kg ATG group was higher than that in the 10 mg/kg ATG group (100% (95% CI: not applicable) vs 86.5% (95% CI: 75.5–98.2); p = 0.012; Table 4). The 3-year TFS followed a similar pattern, with the 8 mg/kg ATG group exhibiting higher rates than the 10 mg/kg ATG group. Five patients died in the 10 mg/kg ATG group (three with severe infections and two with PTLD). The 3-year GRFS was also higher in the 8 mg/kg ATG group than in the 10 mg/kg ATG group (95.8% (95% CI: 90.4–100) vs 80.6% (95% CI: 68.7–94.6), p = 0.021). The cumulative incidence of aGvHD and cGvHD was similar between the two groups (Table 4).
Characteristics of the patients and donors.

IQR, interquartile range; LIC, liver iron content, tested using magnetic resonance imaging; MNCs, mononuclear cells; T2*, spin-spin relaxation time.
Transplantation outcomes of the subgroups.
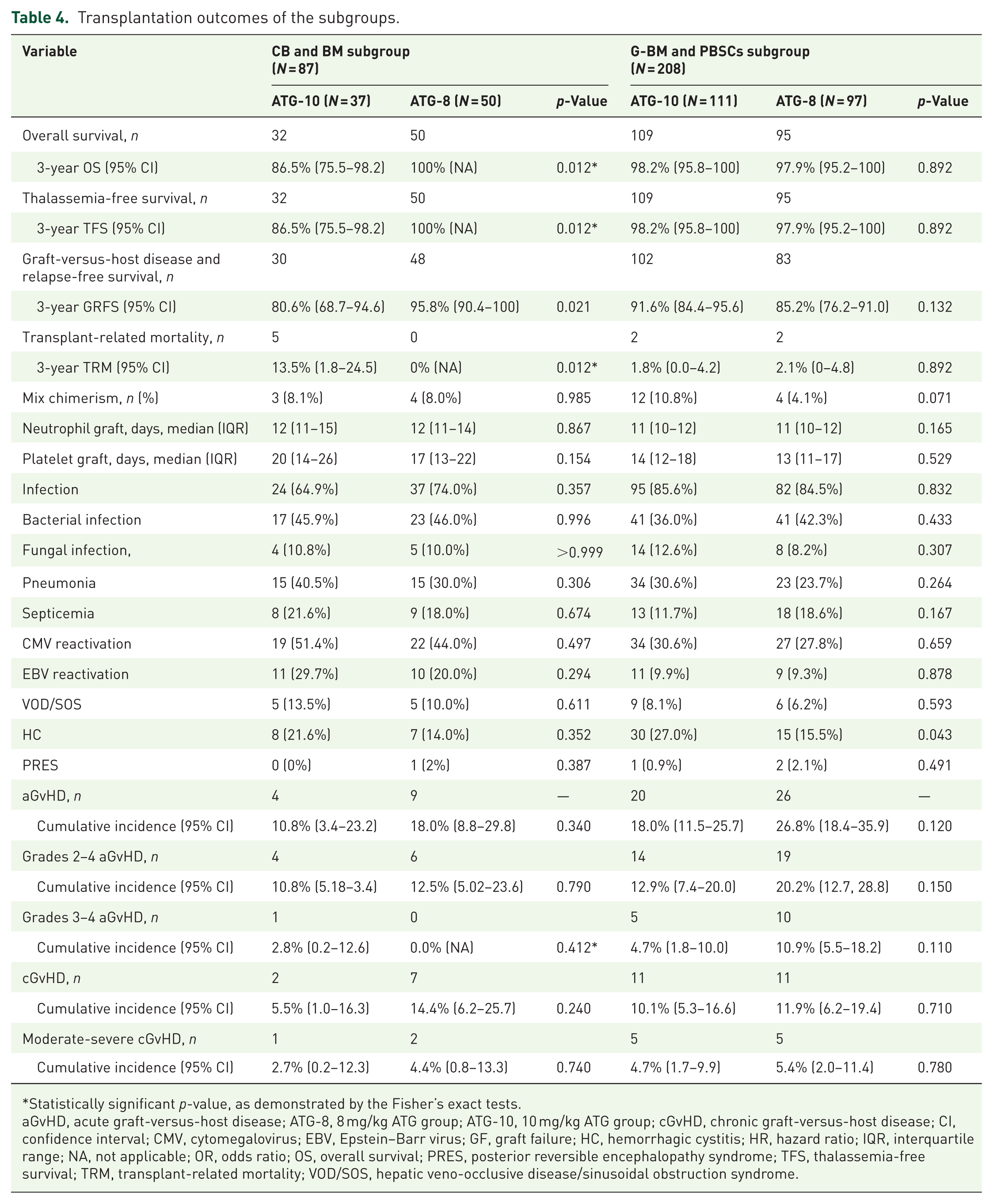
Statistically significant p-value, as demonstrated by the Fisher’s exact tests.
aGvHD, acute graft-versus-host disease; ATG-8, 8 mg/kg ATG group; ATG-10, 10 mg/kg ATG group; cGvHD, chronic graft-versus-host disease; CI, confidence interval; CMV, cytomegalovirus; EBV, Epstein–Barr virus; GF, graft failure; HC, hemorrhagic cystitis; HR, hazard ratio; IQR, interquartile range; NA, not applicable; OR, odds ratio; OS, overall survival; PRES, posterior reversible encephalopathy syndrome; TFS, thalassemia-free survival; TRM, transplant-related mortality; VOD/SOS, hepatic veno-occlusive disease/sinusoidal obstruction syndrome.
In the G-BM and PBSCs subgroup, the 3-year OS, TFS, and GRFS rates were 98.1% (95% CI: 96.2–100.0), 98.1% (95% CI: 96.2–100.0), and 88.4% (95% CI: 83.4–92.3), respectively. The 3-year OS, TFS, and GRFS, as well as the incidence of aGvHD and cGvHD, in the 8 mg/kg ATG group were comparable to those in the 10 mg/kg ATG group (Table 4). However, the incidence of HC in the 8 mg/kg ATG group was lower than that in the 10 mg/kg ATG group (15.5% vs 27.0%; p = 0.043; Table 4).
Among patients receiving 10 mg/kg ATG, the 3-year OS and TFS in the CB and BM subgroup were lower than those in the G-BM and PBSCs subgroup (86.5% vs 98.2%, p = 0.003; 86.5% vs 98.2%, p = 0.003, respectively). The 3-year GRFS was not significantly different between the CB and BM subgroups and the G-BM and PBSC subgroups (80.6% vs 91.6%, p = 0.063). The 3-year OS, TFS, and GRFS of the 8 mg/kg ATG group with co-transplantation of CB and BM subgroup were similar to those of the 10 mg/kg ATG group with co-transplantation of G-BM and PBSCs subgroup (100% vs 98.2%, p = 0.341; 100% vs 98.2%, p = 0.341; and 95.8% vs 91.6%, p = 0.338, respectively).
Discussion
To the best of our knowledge, this is the first randomized clinical trial comparing different doses of ATG in allo-HSCT for patients with TDT. The results showed the OS and TFS of 8 mg/kg ATG were similar to those of 10 mg/kg ATG (Rabbit). The incidence of GVHD and MC, as well as the cumulative incidences of grades 2–4, 3–4 aGvHD, and moderate-severe cGvHD, were similar between the two groups.
ATG has been used in allo-HSCT for preventing GvHD for decades. Previous studies have demonstrated that 4.5 mg/kg ATG (Rabbit) reduces both the incidence of grades 2–4 aGvHD and cGvHD in patients with malignant hematological disorders who have undergone MSDs.12,23 A series of randomized clinical trials have confirmed that 7.5–10 mg/kg ATG (Rabbit) effectively balances GvHD and infections in patients with these disorders who have been transplanted with haploidentical donors.20,23 The use of ATG in TDT patients undergoing allo-HSCT with MSDs has shown equivalent effectiveness in preventing aGvHD, cGvHD, and GF and improving event-free survival and OS. Claire Galambrun et al. reported that adding ATG to the conditioning regimen reduced the incidence of GF from 35% to 10% in allo-HSCT for patients with TDT.4 However, the optimal dose of ATG in allo-HSCT for patients with TDT remains unknown. The doses of ATG in allo-HSCT for patients with TDT have been inconsistent across centers, depending on baseline factors such as the type of donors, recipient’s age at transplantation, presence of splenomegaly, ABO incompatibility, and frequency of transfusions before transplantation.24–26
The 3-year outcomes with 10 mg/kg ATG in the G-BM/PBSCs group (OS: 98.2%, TFS: 98.2%, GRFS: 91.6%) were similar to our previous report (OS: 97.8%, TFS: 97.3%, GRFS: 89.5%), 15 indicating the reproducibility of our protocol. Within this established protocol, the outcomes of 8 mg/kg ATG were similar to those of 10 mg/kg ATG, suggesting that 8 mg/kg ATG should be considered a new standard option, as it provides comparable efficacy with a more favorable safety profile.
CB is an attractive alternative source of hematopoietic stem cells. Unrelated cord blood stem cell transplantation (CBT) has achieved the same outcomes in patients with hematological malignancies compared with BM transplantation or peripheral blood stem cells transplantation.27,28 The application of ATG in CBT is a matter of controversy. Previous studies have reported that lower dosages of ATG after CBT contributed to early immune reconstitution and improved outcomes in pediatric patients. 29 Data from the European Society for Blood and Marrow Transplantation reported that ATG was associated with lower OS and higher non-relapse mortality in adult patients with hematological malignancies receiving CBT. The use of ATG increased TRM, and IRM and PTLD were identified as the main causes of TRM. 30
The outcomes of unrelated CBT in patients with TDT have generally been unsatisfactory, with OS and TFS reported to be approximately 62–88.3% and 21–73.9%, respectively.30,31 However, the outcomes of related CBT in patients with TDT were comparable to those of BM transplantation, provided the CB unit has an adequate number of nucleated cells. 32 In a previous report, the OS and TFS of 70 patients transplanted with related CBT were 96% and 81%, respectively. 32 Promising results have also been observed after co-transplantation of CB and BM-derived hematopoietic stem cells harvested from the same MSDs. 16
In our study, the 3-year OS and TFS of 87 patients receiving CB and BM subgroups were higher than those stated in previous reports, which might be attributed to our intensive conditioning regimen and ATG use. However, the optimal dose of ATG in CBT or co-transplantation of CB and BM for patients with TDT remains unknown. In this study, patients receiving CB and BM as stem cell sources at 8 mg/kg ATG had a reduced risk of infection-related death and a lower risk of PTLD compared to the 10 mg/kg ATG group. For centers employing CB and BM co-transplantation, our data indicate that 8 mg/kg ATG is the preferred dose over 10 mg/kg, as it is associated with superior survival and a reduction in severe complications.
Limitations
This study has limitations that also highlight pathways for future research. Firstly, the single-center design necessitates external validation. Second, subgroup analyses are exploratory due to the modest sample sizes and require confirmation in larger, dedicated cohorts, particularly in the CB and BM subgroups (n = 87). Finally, data on long-term immune reconstitution dynamics and viral reactivation beyond the median follow-up are lacking, warranting future studies to validate the durable benefits of the reduced ATG dose.
Conclusion
This study demonstrated that 8 mg/kg and 10 mg/kg ATG had comparable 3-year OS, TFS, and GRFS in patients with TDT transplanted with MSDs. A dosage of 8 mg/kg ATG had similar cumulative incidences of grades 2–4 and 3–4 aGvHD and moderate-to-severe cGvHD as those of 10 mg/kg ATG. The results suggest that 8 mg/kg ATG can be considered when receiving co-transplantation of G-BM and PBSCs as stem cell sources, especially when receiving co-transplantation of CB and BM as stem cell sources.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261416816 – Supplemental material for Comparing two different doses of anti-thymocyte globulin in allogeneic hematopoietic stem cell transplantation for transfusion-dependent thalassemia: an open-label, randomized clinical trial
Supplemental material, sj-docx-1-tah-10.1177_20406207261416816 for Comparing two different doses of anti-thymocyte globulin in allogeneic hematopoietic stem cell transplantation for transfusion-dependent thalassemia: an open-label, randomized clinical trial by Qiulin Huang, Rongrong Liu, Baoshi Zheng, Lianjin Liu, Hongwen Xiao, Meiqing Wu, Lingling Shi, Gaohui Yang, Zhenbin Wei, Qi Zhou, Yinghua Chen, Yanye Liu, Jing Fan, Xuemei Zhou, Ruolin Li, Zhiyu Zeng, Zhongming Zhang and Yongrong Lai in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
