Abstract
Several clinical trials with anti-CD20 antibodies have successfully treated Acute Lymphoblastic Leukemia. Nevertheless, systematic comparisons between different anti-CD20 antibody trials are rare, and a comprehensive evaluation of their efficacy and safety has yet to be performed. The purpose of this systematic review and meta-analysis was to assess the efficacy and safety of anti-CD20 antibodies in the treatment of acute lymphoblastic leukemia and to guide clinical decision-making regarding the use of anti-CD20 antibody therapy. According to the PRISMA guidelines, Embase, Cochrane Library, PubMed, Web of Science, and ClinicalTrials.gov were searched for clinical trials conducted up to November 1, 2024, for the evaluation of anti-CD20 antibodies (rituximab, obinutuzumab, and ofatumumab) and corresponding controls. After screening the literature and extracting data, study quality was assessed using the Cochrane ROB 2 tool (RCTs) and the Newcastle-Ottawa Scale (cohorts). Heterogeneity was assessed using the I² test. Based on the results of the heterogeneity test, meta-analysis was performed in RevMan 5.4 software with either a random-effects model or a fixed-effects model. We combined data from eight studies (n = 1330 patients, including two RCTs and six cohorts). Meta-analysis showed that anti-CD20 monoclonal antibodies significantly improved overall survival (OR = 1.89, 95% CI: 1.21–2.95, p = 0.005) and event-free survival [OR = 1.68, 95% CI: 1.32–2.14, p < 0.0001] after >1-year follow-up, and increased complete remission rates (p < 0.05). No significant differences were observed in common adverse events between groups. Subgroup analyses by study type did not alter these conclusions. Overall, anti-CD20 antibody therapy was more efficacious than the corresponding control and did not increase the incidence of grade 3–4 adverse events. Ofatumumab may be a more effective anti-CD20 antibody for the treatment of ALL.
Plain language summary
We combined data from 8 studies (n=1330 patients, including 2 RCTs and 6 cohorts). Meta-analysis showed that anti-CD20 monoclonal antibodies significantly improved overall survival [OR=1.89, 95% CI: 1.21–2.95, P=0.005] and event-free survival [OR=1.68, 95% CI: 1.32–2.14, P<0.0001] after >1-year follow-up, and increased complete remission rates (P<0.05). No significant differences were observed in common adverse events between groups. Subgroup analyses by study type did not alter these conclusions. Overall, anti-CD20 antibody therapy was more efficacious than corresponding control and did not increase the incidence of grade 3-4 adverse events. Ofatumumab may be a more effective anti-CD20 antibody for the treatment of ALL.
Introduction
Acute lymphoblastic leukemia (ALL) is a malignant disease of the hematopoietic system that originates from lymphoid progenitor cells and is characterized by abnormal proliferation and accumulation of immature cell precursors in the bone marrow, peripheral blood, and other tissues, followed by failure of normal hematopoietic production and destruction of organs. 1 The onset of ALL is bimodal, with the first peak occurring around age 5 and the second around age 50. 2 Approximately 60% of ALL patients are diagnosed before the age of 20. 3
Immunophenotypic analysis is essential for the diagnosis and classification of ALL, as specific morphology and cytochemistry are often lacking. Cytoplasmic CD19, cytoplasmic CD22, cytoplasmic CD79a, and B-cell surface immunoglobulin CD20 are common immunophenotypic markers indicating B lymphocyte lineage ALL.4,5 From pre-B cells to mature B cells, CD20, a leukocyte differentiation antigen, is expressed at all stages, with its expression progressively increasing as cell maturity. Nevertheless, lymphoid stem cells, plasma cells, and other organs do not express it and have a regulatory role in the proliferation and differentiation of B lymphocytes. Notably, CD20 expression occurs in approximately 30%–50% of cases, and CD20 positivity is defined as CD20 expression in the blast cell population being greater than or equal to 20%.6,7
There is debate regarding the association between CD20 expression and prognosis in ALL patients. The significance of CD20 expression in precursor B-ALL (BCP ALL) was initially used to evaluate the efficacy in children, and Borowitz et al. 8 found that CD20 expression was an independent risk factor for poor prognosis in children, while in the study by Jeha et al., 9 CD20-expressing patients had a better prognosis, with a 5-year event-free survival (EFS) rate of 84% (78% in the negative group) and a 5-year overall survival (OS) rate of 88% (83% in the negative group). Thomas et al. 10 conducted the first trial on adults and found that CD20+ patients had a worse overall survival (OS; 27% vs 40%, p = .03) and 3-year complete remission duration (CRD; 20% vs 55%, p < 0.001). The similar negative predictive significance of CD20 expression in adult B-cell precursor ALL was noted in the study conducted by Maury et al. 11 on 143 adult BCP-ALL patients. Mannelli et al. 12 reached a different conclusion from a molecular perspective. In this study, the minimal residual disease (MRD) remission rates at 10, 16, and 22 weeks were similar between 52 CD20+ BCP-ALL patients and 120 CD20- BCP-ALL patients (55% vs 45.3%, 53.6% vs 61.7%, 60% vs 59%), and positive expression had no significant impact on disease-free survival (DFS) and OS. These controversies have driven the research and evaluation of anti-CD20 monoclonal antibodies’ (mAbs’) efficacy and safety.
Anti-CD20 mAbs were first studied in non-Hodgkin’s lymphoma (NHL) and Burkitt lymphoma/leukemia, and used as part of the standard of care for FL, 13 DLBCL, 14 and CLL. 15 According to recent clinical studies, anti-CD20 mAbs also demonstrate strong antitumor effects in other hematologic cancers of the B lineage. Currently, three anti-CD20 mAbs, including rituximab, obinutuzumab, and ofatumumab, have been used for the treatment of hematologic diseases. To date, the efficacy and safety of these anti-CD20 mAbs have been fully validated in several large randomized controlled trials (RCTs) and cohort studies in ALL. To our knowledge, there have been few systematic comparisons between these trials. In this study, we pooled data from previous clinical trials and performed a systematic evaluation and meta-analysis to assess the efficacy and safety of the available anti-CD20 antibodies for the treatment of ALL and to assist clinicians in drug selection.
Methods
Protocol and registration
This study was performed according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 16 Before the formal start of this paper, we developed a study protocol following the Cochrane Collaboration model, which is registered with PROSPERO (CRD42024614896).
Inclusion and exclusion criteria
Inclusion Criteria: (1) The selected studies involved patients with a definitive Precursor Cell Lymphoblastic Leukemia diagnosis. (2) Studies were eligible if they incorporated obinutuzumab, ofatumumab, or rituximab. (3)We limited inclusion to articles written in English. (4) The studies had to have reported our primary outcomes: OS and EFS, together with the patient number for each. The included articles did not need to include all of the above results. The secondary outcomes of interest were complete remission (CR), EOI MRD negativity, and adverse event rate. Details were gathered when available. Three investigators (ZXM, YJX, and FJ) independently assessed study eligibility; disputes were handled through consensus.
Exclusion Criteria: (1)Study type: Conference abstracts, reviews, case reports, and non-randomized trials; (2)Single-arm studies; (3)Animal studies or research in vitro.
Search strategy
Embase, the Cochrane Library, PubMed, Web of Science, and Clinical Trials.gov were all systematically searched for clinical trials conducted up to November 1, 2024, for the evaluation of anti-CD20 antibodies (rituximab, obinutuzumab, and ofatumumab) and corresponding controls. The following headings and free words to combine search strategy were employed: “Precursor Cell Lymphoblastic Leukemia-Lymphoma”[MeSH Terms] AND “anti-CD20“ [Supplementary Concept] for Pubmed; (Precursor Cell Lymphoblastic Leukemia-Lymphoma): ti, ab,kw AND (anti-CD20): ti, ab,kw for the Cochrane Library; (acute lymphoblastic leukemia): ab, ti AND (anti-CD20) for Embase; (TS=(acute lymphoblastic leukemia)) AND (TS= (anti-CD20)) for Web of Science; “Acute Lymphoblastic Leukemia | anti-CD20” for ClinicalTrials.gov.
The detailed search strategy can be found in Table S1. Furthermore, we independently and manually screened reference lists of included literature, relevant systematic reviews, and meta-analyses to ensure a more comprehensive search.
Study selection data collection
All titles, abstracts, and full-text articles retrieved from the five databases and the corresponding reference lists were independently evaluated by two evaluators (ZXM and yjx) according to the inclusion criteria described above. Disagreements between the two writers were settled through discussion or, if necessary, by a third author (fj) who did not participate in the data collection process. Research publications with no full text available and duplicate articles were not included. A standardized data collection form was used to extract the following data: First author’s last name; year of publication; country in which the study was conducted; number of participants in each group; ratio of female participants to male participants; median age and range of participants; disease status of included participants; chemotherapy regimen used; type and dose of immunotherapy used during induction; median duration of follow-up (Months); and number of participants in each group with the outcome of interest.
Literature quality assessment
The risk of bias maps was assessed using Review Manager 5.4 software (The Cochrane Collaboration, 2020). The Cochrane Collaboration harmonized criteria (Cochrane ROB 2) were used to assess the risk of bias in RCTs, including bias resulting from the randomization procedure, bias due to deviations from the intended intervention, bias due to missing outcome data, bias in outcome assessment, and bias in reported outcome selection. Each bias criterion was categorized as “low,” “high,” or “unclear.” 17 The Newcastle-Ottawa Scale (NOS) was used to evaluate the quality of the included cohort studies, including study subject selection (0–4 points), between-group comparability (0–2 points), and outcome measures (0–3 points). A total score of 0–3 is classified as low quality, a total score of 4–6 as moderate quality, and a total score of 7–9 as high quality. 18 Assessments were conducted independently by ZXM and YJX, and disagreements were resolved through consultation with a third author (FJ).
Statistical analysis
RevMan 5.4 software was used for statistical analysis. Count data were expressed as odds ratios (OR) and corresponding 95% confidence intervals (CI). Statistical heterogeneity was assessed using Cochran’s Q test with the heterogeneity (I2) statistic. The I2 statistic quantifies the proportion of total variation among the studies, where an I2 value of 0%–25% indicates insignificant heterogeneity, 26%-50% indicates low heterogeneity, 51%–75% indicates moderate heterogeneity, and >75% indicates high heterogeneity. The statistical heterogeneity of the fixed-effects model was limited to I2 < 50% and p > 0.1. Otherwise, it indicates that there is statistical heterogeneity among studies, which needs to be further analyzed for the source of heterogeneity, and a random-effects model was used for the analysis after significant heterogeneity was excluded. Subgroup analysis was stratified by study design type. For publication bias assessment: when ⩾10 studies were available per outcome, we employed Egger’s linear regression test combined with visual inspection of funnel plots; when fewer than 10 studies were included, Doi plots and the LFK index were applied. 19 For all analyses, a two-tailed test was performed with a test level of α = 0.05.
Quality of evidence
Evidence certainty for key outcomes was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system to evaluate the quality of evidence. 20 RCTs are rated as high-quality evidence, while cohort studies are rated as low-quality evidence. Five factors that may downgrade the quality of evidence include: risk of bias, inconsistency, indirectness, imprecision, and publication bias. Three factors that may upgrade the quality of evidence include: large-effect, dose-response gradient, and plausible confounding that would change the effect. ZXM and YJX independently classified the overall quality of evidence as high, moderate, low, or very low. Disagreements were also resolved through consultation with a third author (FJ).
Results
Literature screening process and characteristics of the patients at baseline
We combined 1330 patients from eight clinical investigations (two RCTS21,22 and six cohort studies).23–28 PubMed, Cochrane Library, Embase, Web of Science, and ClinicalTrials.gov all provided 5678 titles and abstracts. A total of 972 articles were excluded due to duplication. Also, a total of 1944 articles were excluded due to irrelevance, 2151 articles were excluded due to inapplicable publication type, 394 articles were excluded due to incorrect population, 158 articles were excluded due to in vitro studies/animal testing, and 59 articles were further assessed for eligibility after rapid review. Of these: 39 cases where the full text could not be found, 9 cases where there was no control group, and 3 cases where the outcome of interest was not reported. The age of the participants varied greatly (range 7–83 years). This was because some studies only included instances from children and adolescents, while others included older individuals. Three recruited only Ph-negative ALL patients,21,23,26 two only de novo patients,23,26 and one only relapsed/refractory patients. 28 Moreover, five of the eligible studies recruited onlyCD20+ (CD20 expression ⩾ 20%) patients.21,25,27,28 The entire literature review and selection procedure is shown in Figure 1. Table 1 presents the main characteristics of the eight included studies.
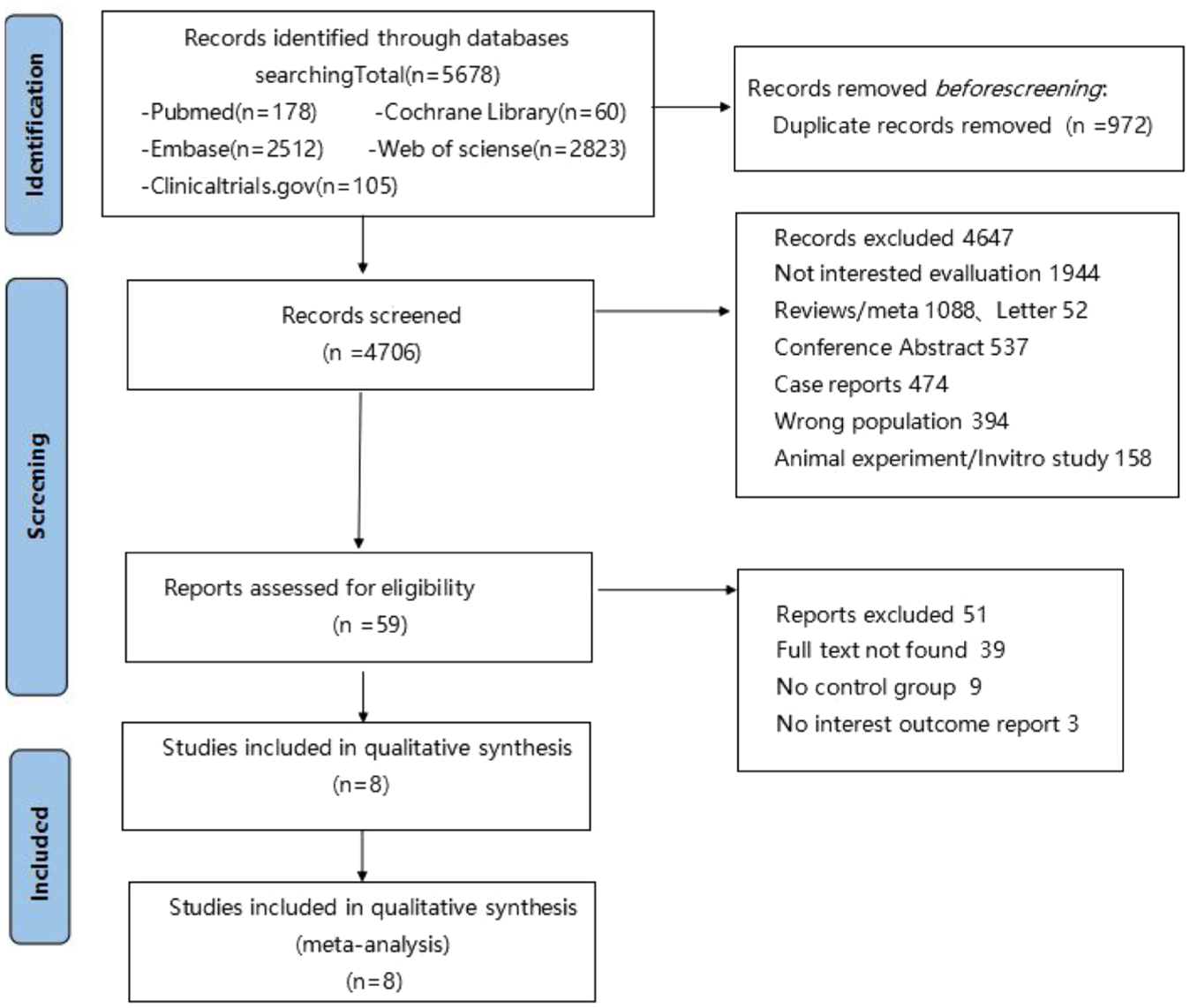
Study search, selection, and inclusion process.
Characteristics of the included clinical trials for patients with ALL and outcome events.

Outcomes Reported: ①OSR; ②EFSR; ③EOI CRR; ④EOI MRD-negative rate; ⑤AE.
One study reported mean age ± standard deviation (range). 27
B-ALL, B-cell Acute Lymphoblastic Leukemia; BCP, B-cell precursor; CAR-T, chimeric antigen receptor T-cell therapy; CD20+, ⩾20% CD20 expression; CD20−, <20% CD20 expression; CVT, Conventional Therapy; F/M, Female/Male; GRAALLR, Group for Research on Adult Acute Lymphoblastic Leukemia Regimen; Mab, Monoclonal antibody therapy; NA, Not Available; O, Ofatumumab; Ph-, Philadelphia chromosome negative; R, Rituximab; r/r, relapsed/refractory; UKALLR, United Kingdom Acute Lymphoblastic Leukemia Regimen.
Quality evaluation of included studies
Both studies were randomized
One study did not use blinding of patients, clinical staff, or statistical analysts, and one did not report the assigned hiding schemes. Both studies reported complete data, none reported results selectively, and none were clear about the presence of other sources of bias.21,22 The results are shown in Figure 2.

Risk of bias graph.
All Cohort studies were high-quality literature, except for one that scored 6 points.23–28 The results are shown in Table 2.
Quality assessment results of included cohort studies (score).

Meta-analysis of efficacy outcome
OS Seven studies reported OS rates, including two RCTs and five cohort studies. Among them, two studies reported 2-year OS rates,24,28 four studies reported 3-year OS rates,22,23,25,27 and one study reported a 4-year OS rate. 26 There was statistical heterogeneity among the studies (p = 0.08, I² = 47%), and a random-effects model was used for the meta-analysis. The pooled meta-analysis found that the OS was significantly better in patients who received anti-CD20 antibodies, with pooled ORs of 1.89 (95% CI (1.21, 2.95), p = 0.005). Interestingly, when subgroups were analyzed by study type, the results showed that in the RCT study, the anti-CD20 antibody carried a higher OSR than conventional chemotherapy. However, there was no statistical significance (OR = 1.23, 95%CI (0.93, 1.64), p = 0.15). In the cohort study, patients receiving anti-CD20 antibodies had 2.82 times more OSR than those receiving conventional chemotherapy (OR = 2.82, 95% CI (1.58, 5.05), p = 0.0005). The results are shown in Figure 3.

Forest plot of OSR in two groups of patients.
EFS Seven studies reported EFS rates, including two RCTs and five cohort studies. Among them, three studies reported 2-year EFS rates,21,25,28 three studies reported 3-year EFS rates,22,23,27 and one study reported 4-year EFS rate. 26 There was no statistical heterogeneity among the studies (p = 0.41, I² = 2%), and a fixed-effect model was used for the meta-analysis. The pooled meta-analysis found that the EFS was significantly better in patients who received anti-CD20 antibodies, with pooled ORs of 1.68 (95% CI (1.32, 2.14), p < 0.0001). Subgroup analysis showed that the EFS rate in both RCT and cohort studies was higher in the mab group than in the control group (OR = 1.43, 95% CI (1.08, 1.90), p = 0.01) versus (OR = 2.65, 95% CI (1.64, 4.28), p < 0.0001). The results are shown in Figure 4.

Forest plot of EFSR in two groups of patients.
CRR Seven studies reported CR rates, including two RCTs21,22 and five cohort studies.23–26,28 There was no statistical heterogeneity among the studies (p = 0.82, I² = 0%), and a fixed-effect model was used for the meta-analysis. The pooled meta-analysis showed that patients in the mab group were more likely to achieve CR than those in the conventional chemotherapy group (OR = 1.76, 95% CI (1.10, 2.82), p = 0.02). When subgroups were analyzed by study type, although a higher CR rate was also observed in the mab group in the RCTs, it did not reach statistical significance (OR = 1.39, 95%CI (0.79, 2.43), p = 0.25). The results are shown in Figure 5.

Forest plot of CRR in two groups of patients.
EOI MRD Negativity Rates Four studies reported EOI MRD negativity rates, including two RCTs21,22 and two cohort studies.23,28 There was statistical heterogeneity among the studies (p = 0.05, I² = 62%), and a random-effects model was used for the meta-analysis. Pooled results suggest that, compared with the control group, ALL patients did not achieve a higher rate of EOI MRD negativity from rituximab-based chemoimmunotherapy (OR = 1.54, 95% CI (0.81, 2.96), p = 0.19). The results are shown in Figure 6.
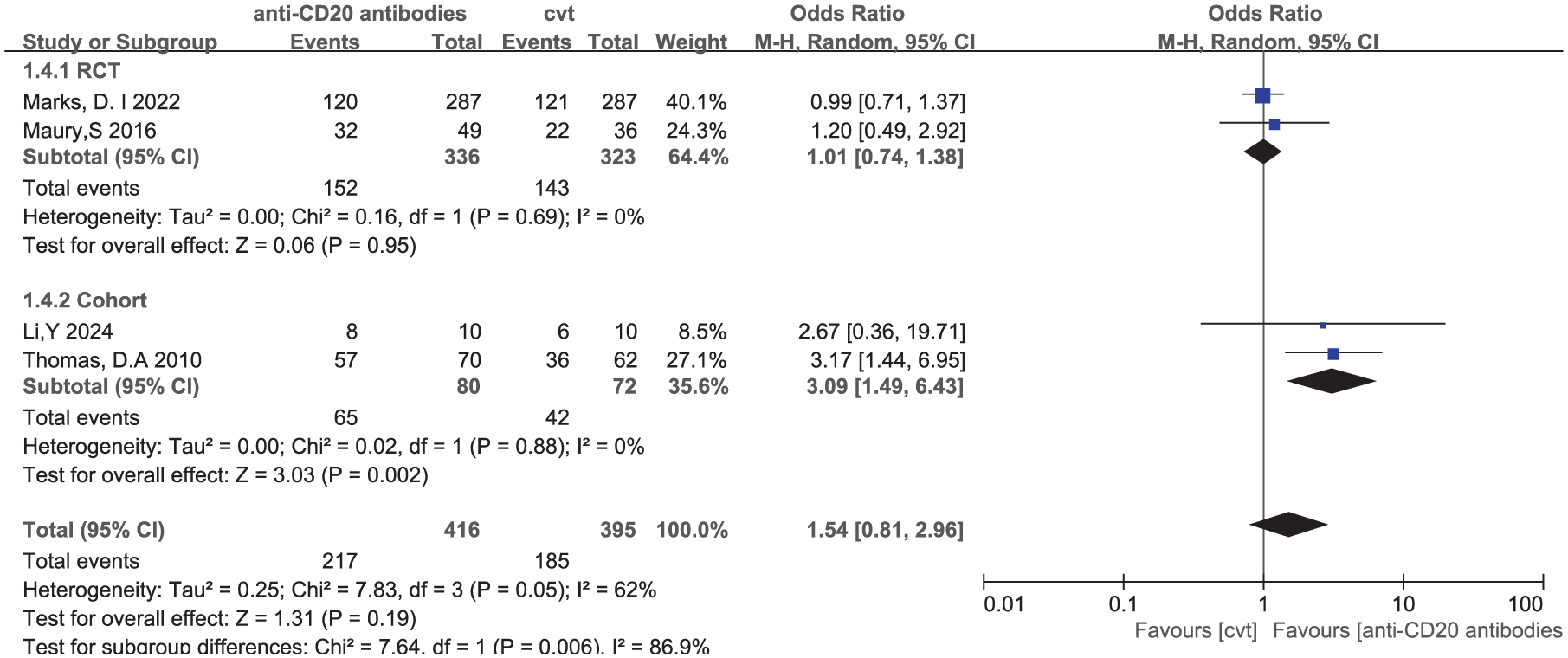
Forest plot of EOI MRD-negative rates in two groups of patients.
Meta-analysis of safety outcome
Among the included studies, only three articles reported adverse events, and both RCTs reported grade 3–4 adverse reactions during the induction phase.21,22 One cohort study reported cases of headache and infection. 28 There were no statistically significant differences in the incidence rates of headache, peripheral neuropathy, infection, fungal infection, bleeding, nausea, vomiting, diarrhea, constipation, pancreatitis, mucositis, pain, rash, elevated AST, elevated ALT, elevated bilirubin, elevated creatinine, and cardiac lesions between the anti-CD20 mab group and the conventional chemotherapy group (p > 0.05). However, the small number of studies and low event rates constrained the statistical power of these safety analyses.
The results are shown in Table 3.
The results of the meta-analysis of safety outcome indicators in two groups of patients.

GRADE evidence quality assessment results
Among the 20 outcome measures in RCTs, 3 (15%) were of very low quality, 17 (85%) were of low quality, and there was no high-quality evidence. All four outcome measures included in the cohort studies were upgraded due to a large effect, with all measures ultimately classified as moderate quality. Detailed results are shown in Tables S4 and S5.
Publication bias
We used the LFK index (Doi plot asymmetry quantification index) to assess publication bias. The results showed that OSR (LFK = 5.15), EFSR (LFK = 4.07), CRR (LFK = 3.98), and MRD negativity rate (LFK = 4.22) all exhibited severe asymmetry. The Doi plot (Figures S1–S4) visually demonstrates that the scatter plot deviates from the unbiased triangle region, indicating a high risk of overpublication of positive results. This finding aligns with the downgrading of evidence quality by GRADE, suggesting that current results may overestimate the true efficacy of anti-CD20 monoclonal antibodies. Future studies should include unpublished data or conduct prospective trial registrations to correct for bias.
Discussion
Research evidence indicates that anti-CD20 mAbs act by inducing antibody-dependent cytotoxicity, cell death due to apoptosis, complement-dependent cytotoxicity, and antibody-dependent cell-mediated phagocytosis for B-cell depletion.29,30 This meta-analysis is the first to examine all available RCTs and cohort studies to compare the outcomes of patients receiving CD20-targeted immunotherapy to those receiving conventional chemotherapy. We discovered that in terms of pooled outcomes, B-ALL patients treated with anti-CD20 mAbs had higher OSR and EFSR than those receiving conventional chemotherapy. This conclusion is supported by the study of Baek et al., 31 which, compared with a historical control group receiving an identical chemotherapy backbone without rituximab, demonstrated superior outcomes for the rituximab-containing regimen in both relapse-free survival (2-year RFS: 55.3% vs 39.7%) and overall survival (2-year OS: 60.8% vs 45.1%). In addition, only one study 28 has evaluated the efficacy of anti-CD20 mAb as rescue therapy in patients with r/r B-ALL. It suggested that rituximab could improve the long-term prognosis of these patients with multiline therapy-failed B-ALL compared to CAR-T therapy alone. However, this conclusion is derived from a single study, and the efficacy of rituximab in the salvage therapy setting should be interpreted with caution and requires validation in future studies. Regarding induction therapy, our pooled analysis found that using anti-CD20 mAb as part of frontline treatment increased the chance of achieving CR at the end of induction. However, in a subgroup analysis based on the kind of study, we found that in RCTs, although better OSR, CRR, and EOI MRD-negative rates were observed in the anti-CD20 mAb group, the differences did not reach statistical significance. Hoshitsuki et al. 32 reported similar findings in pediatric and young adult cohorts, indicating no significant improvement in EOI MRD-negative rates following rituximab treatment in young patients.
All eight included studies, except one, 26 reported on rituximab. Rituximab, the first-generation mouse/human chimeric CD20 mAb, is a well-known example of a targeted therapy that was the first therapeutic mAb approved by the U.S. Food and Drug Administration (FDA) for cancer and has since been used extensively over the past ten years in the treatment of a range of B-cell-derived diseases. 33
Ofatumumab is a second-generation, fully human anti-CD20 IgG1 MoAb that has demonstrated activity in previously untreated or refractory inert NHL and refractory CLL. 34 It binds to a novel epitope of CD20 and has been found to elicit more potent complement-dependent cytotoxicity in vitro than rituximab.35,36 The results of Sasaki et al.’s trial 26 indicated that combining ofatumumab with the hyper-CVAD regimen was practicable and safe, and ofatumumab showed a moderate improvement in survival rate in the CD20 < 20% subgroup. In the CD20-positive subgroup, a trend of better 4-year OS rate and 4-year EFS rate was observed with ofatumumab compared to R (67% vs 51%; p = 0.097), (64% vs 43%; crude p = .054). In a single-arm phase II trial conducted at MD Anderson Cancer Center (NCT01363128), 37 69 B-ALL patients were enrolled. After one cycle of ofatumumab treatment, the CR rate was 94%, and the MRD-negative rate was 65%. After a median follow-up of 44 months, 64% of patients were alive, with 37 cases (54%) still in their first CR. The 4-year RFS and OS rates were 60% and 68%, respectively. It is critical to note that this suggestion of potential superiority is based solely on numerical trends from a propensity score-matched analysis and did not reach conventional levels of statistical significance. Therefore, the appropriate use of ofatumumab in ALL should be investigated in larger, prospective clinical trials.
GA101 (Obinutuzumab) is a type II humanized IgG1 anti-CD20 antibody with Fc engineering, after glycoengineering modification, can enhance its binding affinity to the FcγRIII receptor on immune effector cells, thereby enhancing ADCC and direct apoptosis.38–40 In the animal experiments conducted by Awasthi et al., 41 Obinutuzumab outperformed rituximab in the areas of inducing cell death and ADCC, as well as in rituximab-sensitive/resistant BL and pre-B-ALL xenograft mice. Studies have also reported that obinutuzumab monotherapy is effective in patients with relapsed/refractory indolent and aggressive NHL and patients with chronic lymphocytic leukemia (CLL) of B-cell origin.42–45 The clinical role of obinutuzumab in ALL is currently being defined. In the ongoing, prospective randomized PALG ALL7 ‘OVERALL’ trial (NCT04920968), obinutuzumab is being directly compared against rituximab in patients with newly diagnosed B-ALL. The results of this head-to-head comparison will delineate the therapeutic potential of obinutuzumab and are poised to improve future treatment strategies.
This study confirms that anti-CD20 mabs significantly improve survival outcomes in B-ALL patients (OSR: OR = 1.89, 95% CI: 1.21–2.95; EFSR: OR = 1.68, 95% CI: 1.32–2.14) without increasing severe adverse events. These findings directly support and validate current clinical guidance, such as the ELN 2024 recommendations and the NHS England Clinical Commissioning Policy, which endorses the addition of rituximab to frontline chemotherapy for adults with CD20-positive B-ALL. 46
However, GRADE evidence profiling reveals critical limitations: 85% of core outcomes (OSR, CRR, and MRD negativity rate) in RCTs were rated very low quality due to bias and imprecision, while EFSR evidence was downgraded to low quality owing to bias. Although cohort studies were upgraded to moderate quality based on large effect sizes, they remain susceptible to real-world bias risks. Within this evidence framework, rituximab may be considered for young patients with high CD20 expression—its relatively robust EFS benefit (low-quality evidence) potentially enables cost savings through reduced relapse rates. In contrast, novel agents like ofatumumab, despite demonstrating survival trends (4-year OS 67% vs 51%), require rigorous health economic validation of their price premiums.
Finally, our findings should be interpreted with caution due to some study limitations. First, the vast majority of qualifying research was retrospective cohort studies that used previous cohorts as control groups, which may have introduced bias. Second, there was statistical heterogeneity for some outcomes (e.g., OS and EOI MRD negativity), which was likely attributable to the variations in study design, population characteristics, and differences in anti-CD20 antibody dosing schedules. Importantly, specific limitations of key included studies warrant consideration. In the UKALL14 trial, 22 a lower rituximab dose (four doses) was used compared to the standard regimen in studies like GRAALL (16–18 doses), which may have limited its observed efficacy and explains the nonsignificant OS benefit in our RCT subgroup. In addition, the survival benefit in the Thomas et al. Study 23 was largely confined to younger adults, limiting its generalizability to older patients. Furthermore, some studies only included adults under 65, who often had better outcomes than older patients. However, owing to insufficient available data, further research is needed to evaluate outcomes in this patient population. Finally, due to a limited number of studies that reported adverse events, the difference in adverse events between the anti-CD20 mAb group and the control group was not significant. Therefore, more research is definitely needed to investigate the efficacy and safety outcomes of anti-CD20 mAbs therapy as part of induction or salvage therapy in ALL patients.
Conclusion
This systematic review and meta-analysis indicate that patients with ALL who received CD20-targeted immunotherapy as part of induction or salvage therapy had significantly higher OS and EFS rates than control patients and did not have an increased incidence of grade 3–4 adverse events. The use of anti-CD20 antibodies as part of induction or salvage therapy improved the chances of achieving a CR. Although these data indicate that anti-CD20 mAb is an appealing alternative for the treatment of B-ALL, more research is needed to evaluate the significance of the outcomes and toxicities of this medication.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251401313 – Supplemental material for The efficacy and safety of anti-CD20 antibody for the treatment of B-ALL: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251401313 for The efficacy and safety of anti-CD20 antibody for the treatment of B-ALL: a systematic review and meta-analysis by Xiaomei Zhou, Jiaxi Yang, Jing Fan, Xupai Zhang, Xiao Wang and Hai Yi in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
We acknowledge all original study authors for their contributions. Special thanks to Dr. Yihai for methodological guidance.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and publication of this article.
Competing interests
The authors declare that there is no conflict of interest.
Availability of data and materials
This study did not produce any new data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
