Abstract
Background:
Chemotherapy-induced thrombocytopenia (CIT) commonly exacerbates the difficulty of cancer treatment, increasing bleeding risks and potentially reducing chemotherapy dosage, ultimately impacting its efficacy. However, there are limited studies about avatrombopag application in acute lymphoblastic leukemia (ALL) CIT.
Objectives:
We aimed to evaluate the efficacy and safety of avatrombopag in treating CIT patients diagnosed with ALL.
Design:
This retrospective study, using propensity score matching, included 42 pairs of cases treated with and without avatrombopag (CAT: 54 cases, CAT+: 30 cases).
Methods:
Data of CIT-ALL children were retrospectively collected. The primary endpoint was platelet count (PC) response rate on day 10 ± 2 (defined as an increase of PC to ⩾75 × 109/L with the exclusion of platelet transfusion). Secondary efficacy endpoints, safety endpoints, and factors that predict PC response were also analyzed.
Results:
In the avatrombopag group, the PC response rate was prominently higher on day 10 ± 2 (89.1%) versus the control group (56.4%, p = 0.005). On day 10 ± 2, the difference in median PC change from baseline was predominantly distinct in the avatrombopag group compared to the control group (p = 0.001). In the avatrombopag group, platelet recovery to ⩾25 and ⩾50 × 109/L was faster (p = 0.001, p = 0.002), and quicker platelet reaching ⩾75 × 109/L and ⩾100 × 109/L was achieved (p = 0.023, p = 0.011). The avatrombopag group not only increased the nadir PC (p = 0.009) but also reduced the total platelet transfusion compared to the control group (p = 0.047). Only one case (2.4%) experienced bleeding events after medication. Nine cases of secondary thrombocythemia were noted without other adverse events. There was no difference in event-free survival between the two groups (p = 0.648). Drug administration was prediction factor for PC response.
Conclusion:
Avatrombopag is a potentially safe and effective treatment option for CIT in pediatric ALL.
Plain language summary
Chemotherapy-induced thrombocytopenia, a common hematologic toxicity in chemotherapy, increases the bleeding risk. Thrombopoietin receptor agonists are second-generation drugs designed to mimic the action of thrombopoietin, which promote platelet production. However, there is currently no related research in CIT acute lymphoblastic leukemia pediatrics. In our study, 42 pairs of cases treated with and without avatrombopag were included. We found that avatrombopag can improve the platelet response rate, and reduce the platelet transfusion. Treatment related adverse events occurred at a low rate and only nine cases of secondary thrombocythemia were noted. Avatrombopag may be effective and safe in CIT ALL patients.
Introduction
As a prevalent childhood cancer, acute lymphoblastic leukemia (ALL) often requires chemotherapy, a crucial component in its treatment. Chemotherapy-induced thrombocytopenia (CIT) occurs when anticancer chemotherapy drugs suppress bone marrow megakaryocytes, leading to the platelet count (PC) below 100 × 109/L. CIT is a common hematologic toxicity in chemotherapy, 1 resulting in a reduction in relative dose intensity (RDI), delays in treatment, and even treatment termination, thereby impacting the antitumor efficacy and adversely affecting the long-term survival of patients. 2 Children with hematological malignancies whose incidence and severity of CIT are higher than those with solid tumors, require more platelet transfusions. 3 Although platelet transfusions are frequently administered for managing thrombocytopenia, they only provide short-term benefits while carrying transfusion-related adverse events and potentially resulting in refractory platelet transfusions. 4 Research has demonstrated that managing or preventing CIT with thrombopoietic drugs helps maintain RDI and can extend overall survival, notably in curable malignant tumors. 5
Thrombopoietin receptor agonists (TPO-RAs), the second-generation drugs, are designed to mimic the action of thrombopoietin that activates the TPO receptor (TPO-R). The activation of TPO-R further activates the JAK/STAT and MAP kinase pathways, which promote the maturation and proliferation of megakaryocytes, subsequently stimulating platelet production and increasing platelet counts. 6 However, previous TPO-RAs had some limitations. Romiplostim carries certain potential adverse events, such as the generation of neutralizing antibodies, 7 and eltrombopag’s efficacy is constrained by dietary considerations and specific requirements regarding the timing of administration. 8 Contrastingly, avatrombopag, a novel oral nonpeptide TPO-RA, demonstrates immunoneutrality and lacks dietary intake restrictions. 9 In the United States, avatrombopag has been approved for use in treating thrombocytopenia in adults with chronic liver disease. 10 However, avatrombopag’s application in pediatric CIT is considered off-label, as there is currently no related research in this specific population. Hence, we retrospectively assessed the efficacy, safety, and predictive factors in PC response rate of pediatric ALL.
Methods
Patients and study design
A retrospective analysis was conducted on pediatric ALL patients undergoing CCCG-ALL-2020 chemotherapy protocol at Southern Hospital’s pediatric ward from January 2020 to July 2023. We de-identified all patient details in our manuscript. The main inclusion criteria were (1) Age ranging from 1 month to <18 years; (2) Diagnosis of ALL meeting the World Health Organization (WHO) criteria 11 ; (3) Pediatric cases experienced CIT who underwent the CCCG-ALL-2020 chemotherapy protocol; (4) Predicted survival time ⩾3 months. The major exclusion criteria were (1) Patients with cardiovascular diseases, chronic active hepatitis, liver disease, or cirrhosis; (2) Combined with other blood diseases; (3) Having inherited thrombophilia and severe thrombosis; (4) Severe infection leading to disseminated intravascular coagulation; (5) Bone marrow morphology abnormalities during CIT.
The avatrombopag group comprised 62 pediatric ALL-CIT cases from 36 patients who received avatrombopag treatment. The control group included 250 pediatric ALL-CIT cases from 94 patients who did not receive avatrombopag treatment. Due to the majority of patients in the Vincristine, Doxorubicin, L-asparaginase, Prednisone (VDLP) chemotherapy stage not achieving bone marrow morphological remission, 85 cases from this stage were excluded (19 from the avatrombopag group and 66 from the control group). One case was excluded due to concomitant aplastic anemia. Based on different chemotherapy stages, 226 cases were stratified into subgroups: CAT group and CAT+ group, subjected to propensity score matching using a 1:1 nearest-neighbor approach. Covariates utilized included gender, age, body mass index (BMI), bone marrow megakaryocyte status, disease classification, risk level, baseline platelet count, and infection status. The avatrombopag group was used as the reference standard for matching. Clinical data of CIT cases included demographics, ALL risk stratification, infection status, hematological parameters (changes in platelet count, neutrophil, and hemoglobin), bleeding-related clinical manifestations, avatrombopag usage, complications, bone marrow examinations at various time points (megakaryocyte count), and prognosis. Megakaryocyte inadequate was defined as the number of megakaryocytes smear fewer than 7 within a 1.5 cm × 3.5 cm area in bone marrow.
Chemotherapy regimen
The CCCG-ALL-2020 protocol (Supplemental Table S1) was administered to all enrolled pediatric patients. This protocol involves various chemotherapy phases, including VDLP, CAT, CAT+, etc., with patients stratified based on their risk levels. Detailed risk stratification criteria can be found in Supplemental Table S2.
Avatrombopag treatment
In the avatrombopag group, pediatric patients with ALL are administered avatrombopag treatment when experiencing CIT with platelet count <75 × 109/L, and the first day of medication was the starting point. Correspondingly, the first day of platelet count <75 × 109/L was the starting point in the control group. Patients weighing less than 30 kg received 10 mg daily, while those ⩾30 kg received 20 mg daily. Discontinuation criteria is a platelet count ⩾100 × 109/L. During the treatment period, blood routine tests were conducted at least three times per week, along with coagulation function tests and liver and kidney function tests conducted once a week. Platelet transfusion was allowed when platelet count <30 × 109/L or having clinical bleeding risks. Platelet-raising drugs, including rhIL-11, rhTPO, TPO-RAS except avatrobopag, and caffeic acid tablets, were not allowed.
Endpoint assessment
Platelet count response rate was defined as the proportion of cases with platelet recovery, whether through avatrombopag treatment or spontaneous correction. Platelet recovery was defined as a platelet count ⩾75 × 109/L, excluding the influence of platelet transfusion. Based on the 10- to 14-day timeline for hematopoietic progenitor cells to differentiate into megakaryocytes 12 and avatrombopag’s pharmacokinetics, 13 which peak between days 10 and 13, the PC response rate on day 10 ± 2 was used as the primary efficacy endpoint. In addition to the PC response rate on day 3 ± 2, day 7 ± 2, and day 14 ± 2, the following endpoints were also secondary efficacy endpoints: the time of recovery to PLT ≥25 × 109/L, ≥50 × 109/L, ≥75 × 109/L, and ≥100 × 109/L, the time of platelet beginning to rise, the nadir and peak platelet count. Additional secondary efficacy endpoints included total platelet transfusion and CIT-related bleeding events conforming to the WHO bleeding grading criteria. 14 To compare medication characteristics in different chemotherapy stages, cases were subgrouped into two groups (CAT group and CAT+ group) for subgroup analysis.
The first safety endpoint was the occurrence of hepatorenal dysfunction during avatrombopag treatment. Renal dysfunction was defined as creatinine >2× upper limit of normal (ULN). Abnormal liver function was defined as abnormal aspartate aminotransferase (AST) and/or alanine aminotransferase (ALT) levels combined with abnormal total bilirubin levels and excluding other causes: ALT or AST > 3 × ULN, TBIL > 2 × ULN after 7 days of treatment when the baseline was normal. When baseline was abnormal, ALT or AST > 2 × baseline level and value >3 × ULN, or value >8 × ULN, TBIL increased >1 × ULN or its value >3 × ULN. The second safety endpoint involved assessing adverse events based on the National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0 (NCI-CTCAE 5.0). 15 Lastly, the safety analysis focused on assessing avatrombopag’s potential effects on tumor cell proliferation and leukemia progression, by comparing the minimal residual disease (MRD) differences between the two groups before and after treatment, recording the prognosis of patients with follow-up information and computing the event-free survival (EFS). EFS is defined as the time from the date of diagnosis to the occurrence of the first event or the date of the last follow-up. Events include leukemia relapse, death from any cause, and secondary neoplasms.
Factors predicting PC response rate after CIT in children with ALL
Predicting factors of PC response rate were evaluated using a logistic model. The specific factors are shown in Table 3.
Statistical analysis
The mean ± standard deviation representation was used to compare continuous variables that followed a normal distribution between groups, employing t tests. Median (interquartile range, IQR) were used by continuous variables with skewed distribution, and group comparisons were performed with the Mann–Whitney U test. Count variables were presented as frequency (percentage), and it used the χ2 test or Fisher’s exact test to compare two groups. Multifactorial analysis was conducted using logistic regression. The EFS rate was estimated using the Kaplan–Meier method. To identify protective factors for EFS, Cox regression models were utilized, with results expressed as hazard ratios. A statistically significant difference was set at a level of p < 0.05. Data analysis was used by SPSS (SPSS, Chicago, IL, USA) and Prism5 (GraphPad Software, La Jolla, CA, USA).
Results
Propensity score matching
A total of 226 medical records were included in two groups. Differences were significantly observed between the subgroups in terms of CAT group infection (p = 0.002), phenotype (p = 0.045), and CAT+ group infection (p = 0.029), D46 megakaryocyte (p = 0.022), indicating no comparability. Employing a 1:1 nearest-neighbor matching method resulted in 42 pairs (27 pairs for CAT, and 15 pairs for CAT+). In baseline data between the two groups, no statistically significance existed after matching, demonstrating favorable baseline comparability (Supplemental Tables S3 and S4 and Supplemental Figure S2).
Patient characteristics and treatment exposure
Altogether, 84 cases with a median age of 4 years were enrolled. The initial demographics and disease characteristics of the two group were largely comparable. Among them, there were 75 cases classified as B-cell ALL, and 9 cases classified as T-cell ALL. Of the total, 59.5% (50 cases) were categorized as low risk, and the remaining 40.5% (34 cases) as intermediate risk (Table 1). The median time of medication initiation was day 11 (IQR 10–13 days) after chemotherapy in the avatrombopag group, with a median cumulative dose of 80 mg (IQR 50–160 mg).
Patient characteristics.

CAT and CAT+ are different stages of chemotherapy in CCCG-ALL-2020 regimen. D46 were defined as the 46th day of remission induction therapy. MRD <0.01% was considered negative. The hemoglobin value collected was the value within 7 days before taking the medicine.
BMI, body mass index; MRD, minimal residual disease.
Efficacy analysis of avatrombopag
The response to avatrombopag was achieved rapidly, with a greater proportion of cases in the avatrombopag group reaching a platelet response on day 7 ± 2 compared to the control group: 59.5% versus 39.1% (p = 0.049). In the avatrombopag group, the PC response rate on day 10 ± 2 showed a notable increase compared to the control group (p = 0.005), and the treatment difference was the greatest: 32.7% (Figure 1). The median platelet counts in the avatrombopag group were higher than that in the control group on day 7 ± 2 (78.5 × 109/L vs 60 × 109/L, p = 0.015). In addition, the treatment difference was still significant on day 10 ± 2 (p < 0.001) and 14 ± 2 (p = 0.002; Figure 2(a)). Since day 10 ± 2, the difference value of median platelet counts in the avatrombopag group exceeded the control group significantly (p = 0.001). The change from baseline in median platelet count was predominantly different from that of the control group on day 14 ± 2 (p = 0.005; Figure 2(d)). The median time for platelet recovery to ⩾50 × 109/L in the avatrombopag group was 2 days (IQR 1.75–4.25 days), while it was 4 days (IQR 2–7 days) in the control group (p = 0.002). Avatrombopag group causes grade 4 thrombocytopenia for a shorter period of time (p = 0.001). The median time for platelet recovery to ⩾100 × 109/L in the avatrombopag group was 8 days (IQR 6–10 days), compared to 9 days (IQR 7–12 days) in the control group (p = 0.011). Avatrombopag group increases the nadir platelet count (p = 0.009), and it can also reduce platelet transfusions (p = 0.047; Table 2). Once platelet counts in all pediatric patients exceeded 100 × 109/L, no cases of grade 1 thrombocytopenia were reported. All patients required pharmacological intervention when CIT recurred.

Platelet count response rate for all children at different time points. The percentages of patients who responded on day 3 ± 2, day 7 ± 2, day 10 ± 2, and day 14 ± 2 both in the AVA group and the control group. Difference was found between the two groups on day 7 ± 2 (p = 0.049) and day 10 ± 2 (p = 0.005).

(a–c) Median platelet counts over time in all cases, CAT phase and CAT+ phase. (d–f) Median change from baseline in platelet count over time in all cases, CAT phase and CAT+ phase. Error bars indicate 25th percentile and 75th percentile. CAT and CAT+ are different stages of chemotherapy in CCCG-ALL-2020 regimen.
Comparison of efficacy endpoints between the two groups in all children, CAT and CAT+ phase.
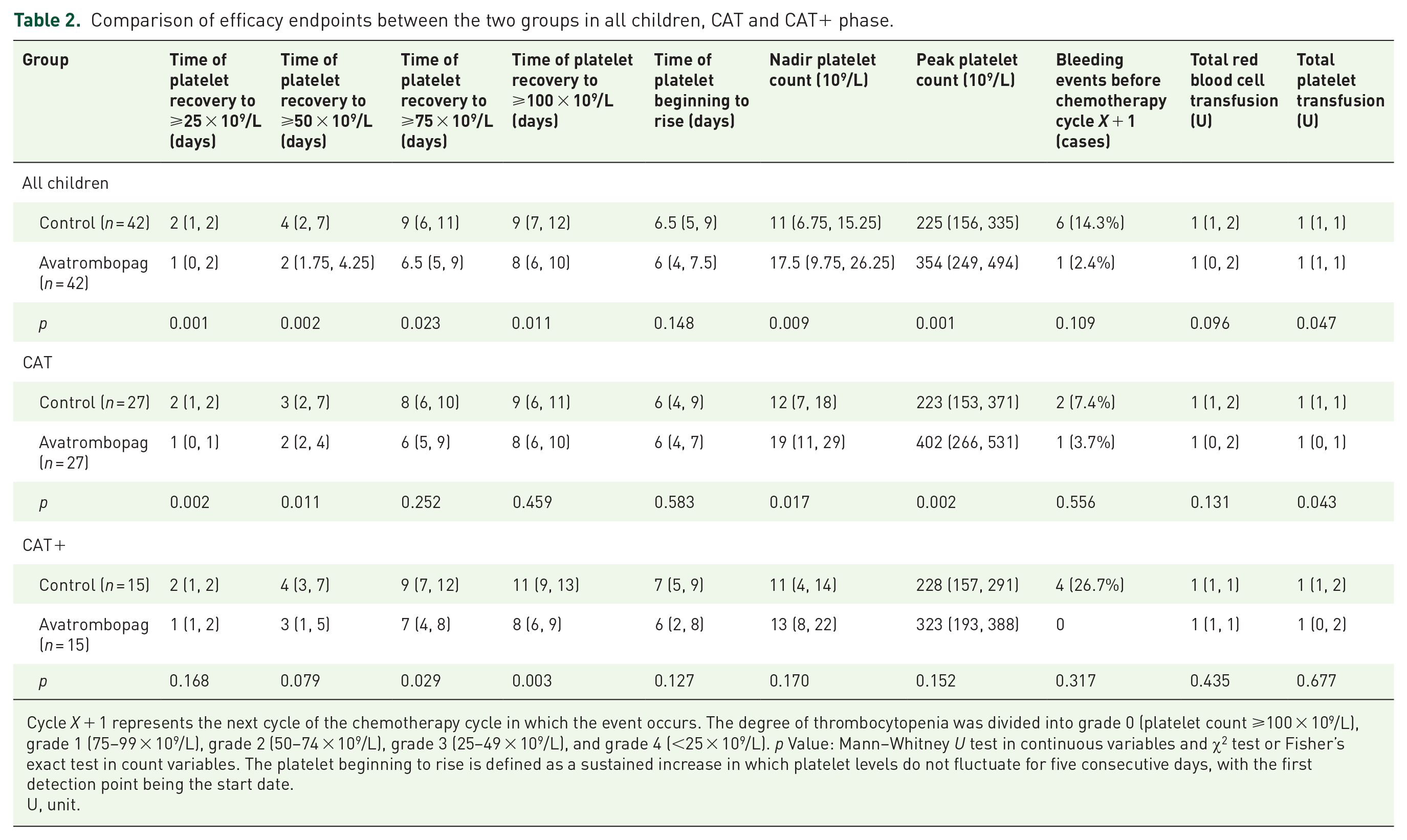
Cycle X + 1 represents the next cycle of the chemotherapy cycle in which the event occurs. The degree of thrombocytopenia was divided into grade 0 (platelet count ⩾100 × 109/L), grade 1 (75–99 × 109/L), grade 2 (50–74 × 109/L), grade 3 (25–49 × 109/L), and grade 4 (<25 × 109/L). p Value: Mann–Whitney U test in continuous variables and χ2 test or Fisher’s exact test in count variables. The platelet beginning to rise is defined as a sustained increase in which platelet levels do not fluctuate for five consecutive days, with the first detection point being the start date.
U, unit.
Subgroup analyses was performed in two chemotherapy phases. In the CAT phase, the difference in median platelet counts between the avatrombopag and control group which occurred on day 10 ± 2 was still significant on day 14 ± 2 (Figure 2(b)). The median time for platelet recovery to ⩾25 × 109/L in the avatrombopag group was 1 day (IQR 0–1 days), while it was 2 days (IQR 1–2 days) in the control group (p = 0.002). The avatrombopag group had median time for platelet recovery to ⩾50 × 109/L of 2 days (IQR 2–4 days), while the control group was 3 days (IQR 2–7 days; p = 0.011). The nadir platelet can remain at a higher level after the administration of avatrobopag compared with the control group (19 × 109/L vs 12 × 109/L, p = 0.017; Table 2). In CAT+ phase, platelet counts remained at a low level in the control group, but was at a higher level in the avatrombopag group on day 7 ± 2 (60 × 109/L vs 78.5 × 109/L, p = 0.013; Figure 2(c)). The most predominant change from baseline in median platelet count between the two groups was observed on day 10 ± 2 (167 × 109/L vs 69.5 × 109/L, p = 0.020; Figure 2(f)). In the avatrombopag group, platelet recovery to ⩾75 × 109/L and 100 × 109/L occurs in a shorter time (p = 0.029, p = 0.003). No case experienced chemotherapy delays or dose reductions exceeding 15%.
Among 83 pediatric cases classified as CIT grades 3–4, a total of 7 cases experienced bleeding events: 3 cases during CAT phase, and 4 cases during CAT+ phase. All cases experienced grade 1 bleeding events due to CIT. Hemorrhagic events were more prevalent in the control group, with six cases (14.3%) compared to only one case (2.4%) in the avatrombopag group (p = 0.109).
Safety analysis of avatrombopag treatment
Nine cases of secondary thrombocythemia were noted, while this study did not observe any events related to hepatic and renal dysfunction or other adverse events. It is noteworthy that no case interrupted or suspended avatrombopag treatment for adverse effects. During the CAT phase, the difference in MRD change between day 19 and day 46 did not achieve statistical significance compared to the control group following avatrombopag administration (p > 0.05), and none of the cases experienced an increase in MRD after medication (Figure 3(b)). Subsequently, the comparison of EFS between the avatrombopag group and the control group indicated no statistically significant difference (p = 0.648; Figure 3(a)). No trend of deterioration in clinical outcomes of ALL was observed in avatrombopag group. However, given the relatively short duration of avatrombopag treatment with a median follow-up of only 13.4 months, further observation is warranted.

Avatrombopag safety analysis. (a) EFS between two groups. The probability of EFS between two groups, and there is no statistically significant difference (p = 0.648), HR = 0.635 (95% CI: 0.088–4.563). (b) The D19 MRD and D46 MRD level in the avatrombopag group and the control group. D19 and D46 were defined as the 19th and 46th day of remission induction therapy.
Predictors of PC response rate
In univariate analysis, two variables were significantly likely to predict PC response: age (odds ratio (OR) 1.18, 95% confidence interval (CI): 0.98–1.43; p = 0.079) and with avatrombopag medication (OR 0.17, 95% CI: 0.05–0.66; p = 0.010). Further multivariate analysis indicated that drug administration was prediction factor for PC response (Table 3). Compared with the control group, avatrombopag had no significant effect on the levels of neutrophils and hemoglobin. But when comparing within groups, it can be found that avatrombopag significantly elevated hemoglobin levels (p = 0.002) after 7 days of medication (Figure 4).
Univariate and multivariate analysis of factors predicting the PC response rate in CIT patients.

The hemoglobin value collected was the value within 7 days before taking the medicine. D19 and D46 were defined as the 19th and 46th day of remission induction therapy. Logistic regression model was conducted.
BMI, body mass index; CIT, chemotherapy-induced thrombocytopenia; Hb, hemoglobin; PC, platelet count.

Avatrombopag affects hemoglobin and neutrophil level. Above pictures show the neutrophil and hemoglobin level before treatment and after treatment (Tx) in the avatrombopag group and control group. The data collected were the values of 1 day before treatment and the values of 7 days after treatment.
Discussion
As far as we know, the study assessing the efficacy and safety of avatrombopag in treating pediatric patients with CIT following ALL is the initial report. Although platelet counts can naturally recover, avatrombopag has been shown to accelerate this natural recovery process. We observed a superior PC response in the avatrombopag group. The avatrombopag group exhibited a rapid PC response, reaching 59.5% on day 7 ± 2. The response rate is much higher compared to a trial of solid tumor CIT, with only 37.8% effective rates after 1 week. 16 A study suggested that the time required for hematopoietic progenitor cells to differentiate into megakaryocytes, which produce platelets, is typically 10–14 days. 12 Researches about pharmacological characteristics of avatrombopag indicated that avatrombopag induces a dose and exposure-dependent elevations in platelet count. 17 The onset of the platelet count increase was observed within 3–5 days, with peak effects recorded between 10 and 13 days. Subsequently, platelet counts gradually declined, returning to near baseline values by day 35. 13 In our study, the observation time points for the efficacy endpoint were based on the above characteristics, and we found that the intergroup difference in median platelet counts started from day 7 ± 2, and the most prominent difference was noted on day 10 ± 2, which was consistent with the pharmacological characteristics. Avatrombopag was found to lack sustained efficacy, likely due to the mechanisms of CIT, which makes bone marrow suppression inevitable, 18 as well as its pharmacological characteristics that result in short duration of efficacy. The pattern of platelet changes of avatrombopag medication in this study may provide real-world evidence for further prophylactic medication research.
Subgroup analysis focusing on different chemotherapy stages revealed that there were differences in efficacy between different chemotherapy phases. The heterogeneous rates of CIT occurrence across different chemotherapy regimens may stem from the various impacts of different chemotherapy drugs on platelet and physiologic megakaryocyte generation.19,20 Additionally, different chemotherapy agents may influence platelet destruction and distribution.21,22 Therefore, further studies are required to investigate the efficacy of TPO-RAs in managing CIT across various chemotherapy phases. The differences in time of platelet recovery to ⩾75 and 100 × 109/L were not significant in the CAT chemotherapy phase. Lack of recovery difference may be due to the relatively short duration of CIT and a substantial incidence of spontaneous platelet recovery, rather than a lack of avatrombopag efficacy in increasing platelet counts. 23 Compared to the CAT phase, children in the CAT+ chemotherapy phase did not show a significant reduction in the duration of grade 3 or 4 thrombocytopenia after taking avatrombopag. This may be due to the impact of two previous rounds of chemotherapy, as well as the stronger chemotherapy intensity and longer duration of chemotherapy.
Elting et al. 24 indicated in cancer patients undergoing chemotherapy, the risk of thrombocytopenic bleeding increases threefold and bleeding grade rises once the platelet count drops below 75 × 109/L, and major bleeding events are relatively common in patients with grades 3–4 CIT. Our study confirms that avatrombopag can shorten the duration of grade 3 and grade 4 thrombocytopenia, accelerating the recovery of platelet counts to levels above 75 × 109/L, thereby lowering the risk of bleeding complications. Although no major bleeding events were recorded, there were fewer bleeding events due to thrombocytopenia in the avatrombopag group. The lack of significance could be due to the small sample size. We anticipate that larger trials may reveal more significant differences in future studies.
In the context of CIT, a crucial risk of exogenous platelet-generating drugs usage is the potential development of the underlying disease. The TPO-R gene (c-MPL) is expressed in megakaryocytes, CD34+ hematopoietic stem cells, and progenitor cells. It is present in most acute myeloid leukemia and myelodysplastic syndromes cells, but in contrast, c-MPL expression is low or nearly absent in ALL. 25 Currently, no events leading to myelofibrosis, myelodysplastic syndromes, or leukemia progression related to TPO-RA therapy have been discovered.26–28 In our study, the results did not indicate a trend toward promoting the proliferation of ALL cells in vivo or worsening clinical outcomes. However, due to the short exposure time to avatrombopag and early termination of follow-up, it is difficult to determine whether there is a causal relationship.
TPO-RAs are safe for CIT patients, with a pooled adverse event rate of only 3%. 29 In our study, avatrombopag demonstrated safety and good tolerability in pediatric cancer patients, with only nine cases of secondary thrombocytosis recorded, and no thromboembolic events. This aligns with the findings reported by Murphy, 30 who concluded that the thrombosis risk in immune thrombocytopenic purpura patients treated with TPO-RA treatment did not significantly increase. Khorana et al. 31 identified elevated platelet counts (>350 × 109/L) as an independent risk factor for increased thrombosis in cancer patients. Selecting 100 × 109/L as the threshold for discontinuing treatment can help prevent excessive platelet elevation, thus reducing the increased risk of thrombosis.
There are reports indicating that approximately 50% of patients experience anemia, and 50% exhibit neutropenia after chemotherapy, 32 which has a damaging effect on hematopoietic stem cells and megakaryocytic progenitor cells. TPO-R is present not only on the surface of platelets and megakaryocytes but also on CD34+ hematopoietic stem cells and progenitor cells. 33 Thus, TPO-RAs play an essential role in severe aplastic anemia. 34 In addition, an animal experiment had shown and exhibited a protective influence on hemoglobin but lacked the ability to shorten the duration of neutropenia. 35 Avatrombopag, in our study, can increase hemoglobin levels in intergroup. Given limited number of cases and the potential role of spontaneous patient recovery, further verification is needed to elucidate the effects of avatrombopag on hemoglobin and neutrophil.
Nonetheless, there were some limitations in our study. Being a retrospective study, it is a relatively small cohort with selection bias. The study lacks randomization, preventing the quantification of avatrombopag’s efficacy compared to the control group. Therefore, randomized controlled trials are needed to further validate the efficacy characteristics of avatrombopag in pediatric CIT.
Conclusion
Given that our study confirmed avatrombapag can raise the platelet nadir, shorten the duration of thrombocytopenia, reduce bleeding risks, and decrease the need for platelet transfusions. Although it was a retrospective cohort study, avatrombopag does show great promise as the innovative drug for treating pediatric patients with CIT.
Supplemental Material
sj-docx-1-tah-10.1177_20406207241304300 – Supplemental material for Efficacy and safety of avatrombopag in the treatment of chemotherapy-induced thrombocytopenia in children with acute lymphoblastic leukemia: a single-center retrospective study
Supplemental material, sj-docx-1-tah-10.1177_20406207241304300 for Efficacy and safety of avatrombopag in the treatment of chemotherapy-induced thrombocytopenia in children with acute lymphoblastic leukemia: a single-center retrospective study by Huiyan Yang, Jingyu Gao, Yongsheng Ruan, Zhaokun Chen, Ruihan Fang, Lei Zhang, Zhibiao Wang, Tiantian Yi, Qian Zhang, Yang Luo, Libai Chen and Xuedong Wu in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
