Abstract
Background:
Efanesoctocog alfa is a first-in-class high-sustained factor VIII (FVIII) replacement therapy. In the phase III XTEND-1 (NCT04161495) study, once-weekly efanesoctocog alfa prophylaxis (50 IU/kg) was well-tolerated and achieved high-sustained factor levels in the normal to near-normal range (>40%) for most of the week.
Objective:
To report outcomes in previously treated participants with severe haemophilia A aged ⩾12 years from an observational study who switched to efanesoctocog alfa prophylaxis during XTEND-1.
Design:
Paired assessment of participants from an observational study who enrolled in the phase III XTEND-1 study.
Methods:
Seventy-eight participants switched from marketed standard half-life (SHL) or extended half-life (EHL) FVIII prophylaxis to once-weekly efanesoctocog alfa prophylaxis (50 IU/kg). Endpoints included annualized bleed rates (ABRs), treatment of bleeding episodes, injection frequency and FVIII consumption.
Results:
Pre-study, 44 (56%) and 34 (44%) participants received SHL FVIII or EHL FVIII prophylaxis, respectively. In the overall population, a significant reduction in ABR from 2.96 to 0.69 (p < 0.0001) was observed following the switch to efanesoctocog alfa prophylaxis as well as reductions in spontaneous, traumatic, joint and spontaneous joint ABRs (p < 0.0001). Significant reductions in mean weekly injection frequency were observed, from 2.8 to 1.0 in the SHL FVIII cohort (p < 0.0001) and from 1.8 to 1.0 in the EHL FVIII cohort (p < 0.0001). Mean annualized factor consumption reduced by 47% in the SHL FVIII cohort and 30% in the EHL FVIII cohort.
Conclusion:
Collectively, the results of this post hoc analysis demonstrate the benefits of once-weekly efanesoctocog alfa prophylaxis over SHL or EHL FVIII prophylaxis on bleed rates, injection frequency and consumption.
Trial registration:
Observational study: 242HA201/OBS16221; XTEND-1: NCT04161495 (https://clinicaltrials.gov/study/NCT04161495).

Introduction
People with severe haemophilia A (endogenous factor VIII (FVIII) level <1 IU/dL) are at risk of life-threatening bleeding episodes in addition to recurrent joint and muscle bleeds that may lead to joint damage and haemophilic arthropathy. 1 Prophylaxis, with standard half-life (SHL) or extended half-life (EHL) factor replacement therapies, or emicizumab, is standard of care for severe haemophilia A, with the aim of preventing spontaneous bleeds and preserving joint health. 1
Factor trough levels of 1%–3% have been found to be insufficient to prevent all bleeds in people with haemophilia. 1 Therefore, recently, clinicians have been targeting FVIII trough levels of 3%–5% or higher, 1 with evidence suggesting that targeting higher FVIII activity levels provides improved protection from bleeds, leading to better outcomes.2–6 However, despite recent treatment advances, joint bleeds still occur with current SHL and EHL FVIII prophylaxis, leading to joint damage, chronic pain and impaired mobility.6–11 Although prophylaxis with emicizumab, a non-factor therapy, has reduced the burden of treatment, some patients receiving emicizumab experience breakthrough bleeding episodes.4,12,13
Currently available SHL and EHL FVIII replacement therapies bind to von Willebrand factor (VWF) in the circulation. 14 Although VWF stabilizes and protects FVIII from degradation, the high affinity interaction prevents prolongation of FVIII half-life beyond the VWF half-life of ~15 h, thus imposing a ceiling on the achievable half-life with current FVIII replacement therapies. 14 Therefore, prophylaxis with SHL or EHL FVIII replacement typically requires injections 3–4 or 2–3 times per week, respectively, dependent on patient and factor replacement pharmacokinetics (PK) and target trough levels. 1 Sustaining higher FVIII levels with these therapies is possible with increased dosing frequency and/or higher doses. 1 However, frequent dosing increases treatment burden and reduces health-related quality of life, and can lead to reduced adherence and the requirement for central venous access devices, especially in children.1,15,16
Efanesoctocog alfa is a first-in-class high-sustained FVIII replacement therapy (Figure 1) uniquely designed to decouple recombinant FVIII (rFVIII) from endogenous VWF and overcome the VWF-imposed half-life ceiling.17–19 Efanesoctocog alfa consists of a single rFVIII fused to the Fc domain of human immunoglobulin G1, the D′D3 domain of VWF (FVIII-binding domain) and two XTEN polypeptides.17–19 The D′D3 domain prevents binding to endogenous VWF, while the Fc domain facilitates recycling through the neonatal Fc receptor-mediated pathway and the XTEN polypeptides shield from proteolytic degradation and reduce binding to clearance receptors.17–19 These components allow for a half-life that is three- to fourfold longer than EHL and SHL rFVIII therapies, as shown in a sequential PK study. 20 Efanesoctocog alfa was approved as ALTUVIIIO® by the United States Food and Drug Administration in early 2023, for use in adults and children with haemophilia A for routine prophylaxis to reduce the frequency of bleeding episodes, on-demand treatment and control of bleeding episodes, and perioperative management. 21 It was approved as ALTUVOCT® by the European Medicines Agency in 2024, for the treatment and prophylaxis of bleeding in patients with haemophilia A. 22

Current FVIII replacement therapy classification.
In the phase III XTEND-1 (NCT04161495) study, once-weekly efanesoctocog alfa (50 IU/kg) prophylaxis achieved high-sustained factor levels in the normal to near-normal range (>40%) for the majority of the week and 15% at the end of the weekly dosing interval. 19 Once-weekly efanesoctocog alfa prophylaxis provided superior bleed protection versus prior factor prophylaxis with clinically meaningful improvements in physical health, pain, and joint health. 19 Here, we report a post hoc analysis comparing outcomes in participants receiving SHL and EHL FVIII prophylaxis in a prospective, observational study 23 and who switched to once-weekly efanesoctocog alfa prophylaxis in XTEND-1.
Materials and methods
Study design and study population
This post hoc analysis includes data on the subset of participants who received prophylaxis for ⩾6 months in the observational pre-study and had an efficacy period of ⩾6 months in Arm A of XTEND-1 (NCT04161495), an open-label, multicentre, phase III study of efanesoctocog alfa (Supplemental Figure 1). The study was performed in accordance with the Declaration of Helsinki and local regulations. The protocol was approved by institutional review boards and/or ethics committees at participating institutes (Supplemental Table 1).
The prospective, observational study (242HA201/OBS16221) enrolled participants ⩾12 years of age with severe haemophilia A (<1 IU/dL (<1%) endogenous FVIII) with ⩾150 exposure days to a FVIII product and/or cryoprecipitate. 23 Participants were required to be currently receiving a prophylactic or on-demand regimen with a marketed FVIII product. If receiving on-demand treatment, participants had to have had ⩾12 bleeding episodes in the 12 months prior to study enrollment. Participants with any concurrent clinically significant major disease, coagulation disorders other than haemophilia A, a history of a positive inhibitor test (⩾0.6 BU/mL) or clinical signs of decreased response to FVIII, or receiving non-factor therapies (fitusiran or emicizumab) were excluded. Marketed FVIII therapies were administered per standard local prescribing procedures or package insert. Participants were followed for ⩽12 months from January 25, 2019, until November 30, 2020, with study visits at baseline, and months 3, 6 and 12. Participants also attended their routine standard-of-care and as-needed clinic visits. Prospective data on bleeding episodes and FVIII treatment administration were recorded via electronic participant diaries and electronic case report forms.
After ⩽12 months in the observational pre-study, participants could enroll in XTEND-1 (NCT04161495), an open-label, multicentre, phase III study of efanesoctocog alfa. Study design and results of XTEND-1 have been previously reported (a summary of key exclusion and inclusion criteria can be found in Supplemental Table 2). 19 Participants on prior FVIII prophylaxis could enroll in Arm A of XTEND-1 to receive once-weekly efanesoctocog alfa (50 IU/kg) prophylaxis for 52 weeks. Participants on prior on-demand therapy regimen could enroll in Arm B to receive on-demand efanesoctocog alfa for 26 weeks and then once-weekly efanesoctocog alfa (50 IU/kg) prophylaxis for 26 weeks.
Outcomes and assessments
Outcomes included annualized bleed rates (ABRs) for treated bleeding episodes (overall, spontaneous, traumatic, joint and spontaneous joint), treatment of bleeding episodes, FVIII consumption and injection frequency. Annual factor consumption included routine prophylaxis doses and doses used to treat bleeding episodes. Weekly factor consumption was the total dose per participant divided by the total number of weeks on study. Bleeding episodes were defined as starting from the first sign of bleeding and ending ⩽72 h after the last FVIII injection to treat a bleed. 24 Symptoms of bleeding at the same location and injections administered ⩽72 h from the first injection were considered the same bleed, and any injection >72 h after the preceding one was considered the first injection to treat a new bleed in the same location. 24
Statistical analysis
To execute intra-patient comparisons of ABR during the efanesoctocog alfa treatment period and historical prophylaxis period, a non-inferiority test was performed among individuals who had ⩾6 months of historical data and ⩾6-month efficacy period during XTEND-1. The non-inferiority margin was estimated based on the known treatment effect between on-demand and prophylactic treatment. A meta-analysis of phase III registrational studies for rFVIII products that included both on-demand and prophylactic treatment regimens estimated a reduction of 31 bleeds per year between on-demand and prophylactic treatment, with a lower bound of 27 bleeds per year (i.e., there is a non-inferiority margin of 4). For a non-inferiority test of the null hypothesis (median difference in ABR exceeds or is equal to the non-inferiority margin) versus the alternative hypothesis (median difference in ABR is less than the non-inferiority margin), a sample size of 63 achieves 90% power to detect non-inferiority using a one-sided paired Wilcoxon Signed-Rank test at a 0.025 significance level when the actual mean of paired differences is 0 and the non-inferiority margin is 4.
Outcomes were reported for the overall observational study population and by FVIII replacement therapy, either SHL rFVIII and plasma-derived FVIII (herein referred to as SHL FVIII cohort), or EHL FVIII. Mean (95% confidence interval (CI)), mean difference (95% CI) and rate ratio for ABRs were estimated using a negative binomial regression model with treatment (on-study prophylaxis vs pre-study prophylaxis) as covariate. P Values relate to the null hypothesis: rate ratio (efanesoctocog alfa prophylaxis/pre-study prophylaxis) = 1. Mean difference (95% CI) and p values for weekly injection frequency, and weekly and annual consumption were estimated using paired t test. Data on treatment of bleeding episodes were presented descriptively. McNemar’s test was used to determine the significance of the difference in the number of participants with zero bleeding episodes. A two-sample t test was used to determine the statistical significance of the difference in the total dose and number of injections to treat a bleeding episode.
Results
Participant demographics and baseline characteristics
Seventy-eight participants were included in this post hoc analysis (Table 1). In the pre-study, 44 (56%) participants were receiving either SHL rFVIII (n = 36) or plasma-derived FVIII prophylaxis (n = 8; SHL FVIII cohort). Thirty-four (44%) participants were receiving EHL FVIII prophylaxis. All participants were male. At entry into the observational pre-study, the mean (standard deviation (SD)) age in the overall cohort was 33.4 (15.6) years and 78% (n = 61) of participants were 18–64 years of age. Participant demographics were similar in the SHL FVIII and EHL cohorts. In the SHL FVIII cohort most were receiving pre-study prophylaxis every other day (20%; 9/44), twice per week (27%; 12/44) or three times per week (41%; 18/44). Participants in the EHL cohort were receiving pre-study prophylaxis less frequently, most twice per week (32%; 11/34) or once per week (21%; 7/34).
Participant demographics and characteristics at entry into the observational pre-study.
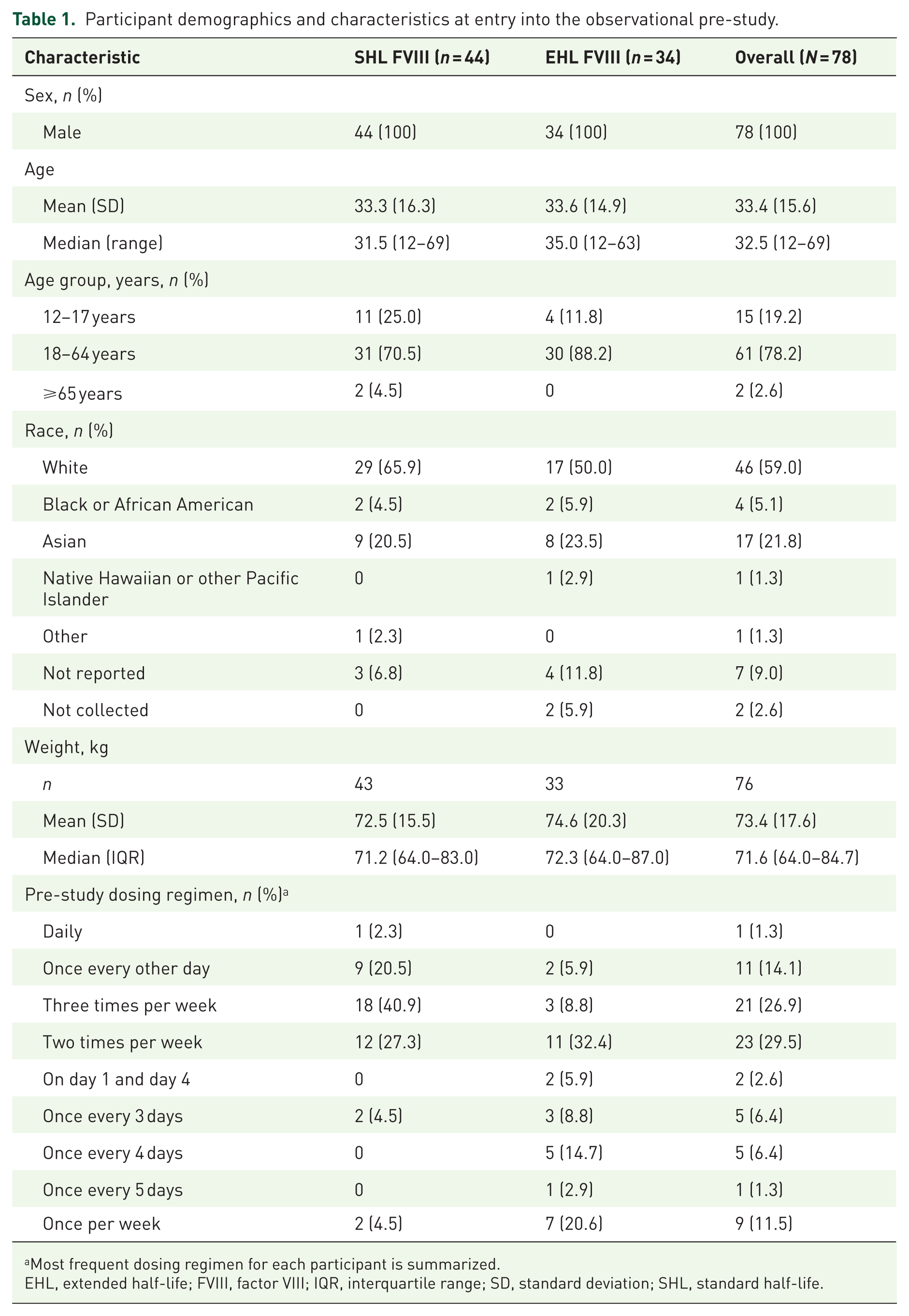
Most frequent dosing regimen for each participant is summarized.
EHL, extended half-life; FVIII, factor VIII; IQR, interquartile range; SD, standard deviation; SHL, standard half-life.
Annualized bleed rates
Once-weekly efanesoctocog alfa prophylaxis provided superior bleed protection to prior FVIII prophylaxis, evidenced by a significant reduction in overall ABR in the overall population following the switch (2.96–0.69, p < 0.0001; Table 2; Figure 2(a)). The reduction in overall ABR was slightly higher in participants who switched from SHL FVIII prophylaxis to efanesoctocog alfa prophylaxis (3.23–0.79), compared with those who switched from EHL FVIII prophylaxis (2.61–0.55; Table 2; Figure 2(b) and (c)). In the overall population, a significant reduction in mean joint ABR from 1.99 to 0.44 was observed following switch; corresponding reductions observed in the SHL FVIII and EHL FVIII cohorts were 2.30 to 0.46, and 1.59 to 0.43, respectively (Table 2; Figure 2). Significant reductions in spontaneous, traumatic and spontaneous joint ABRs were observed in the overall population, and in both FVIII cohorts (Table 2; Figure 2).
Pre-study and on-study annualized bleed rates. a

Mean (95% CI), mean difference (95% CI), and p values were calculated using negative binomial regression model with treatment (on-study prophylaxis vs pre-study prophylaxis) as a covariate. p Value relates to the null hypothesis: rate ratio (efanesoctocog alfa prophylaxis/prior prophylaxis) = 1.
ABR, annualized bleed rate; CI, confidence interval; EHL, extended half-life; FVIII, factor VIII; IQR, interquartile range; SHL standard half-life.
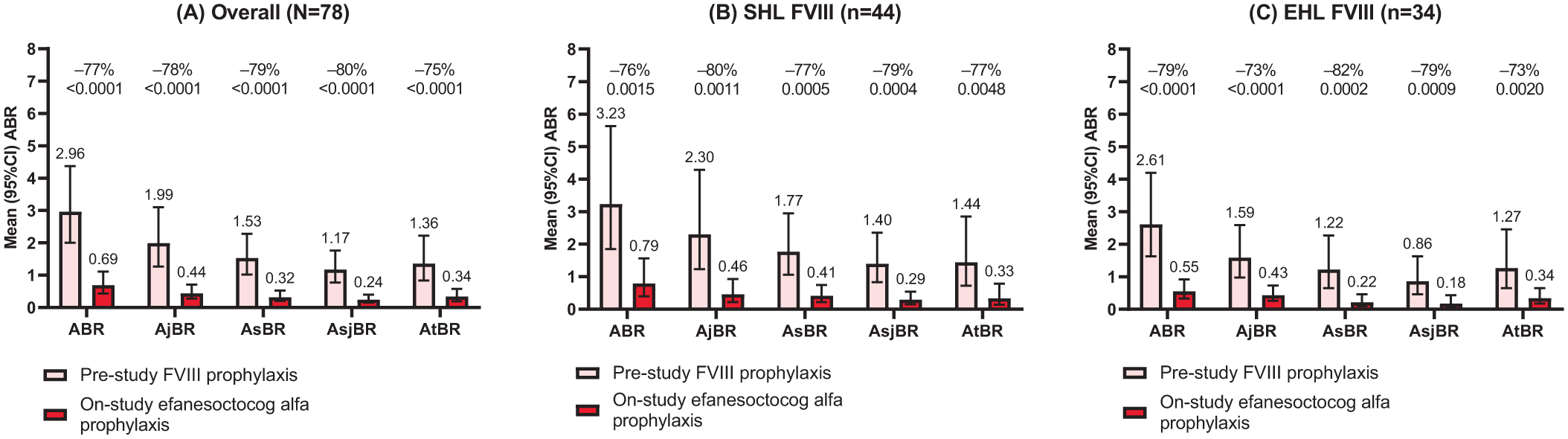
Pre-study and on-study annualized bleed rates with FVIII prophylaxis. A: Overall; B: SHL FVIII; C: EHL FVIII.
Bleeding episodes
The mean (SD) duration of observation period in the pre-study was 47.2 (6.7) weeks, and the efficacy period in XTEND-1 (on-study) was 49.6 (3.1) weeks. There were 132 bleeding episodes in the SHL FVIII cohort (n = 44) during the pre-study, and 33 during XTEND-1 (Table 3). In the EHL FVIII cohort (n = 34), there were 80 and 18 bleeding episodes, respectively. Following switch to efanesoctocog alfa prophylaxis, the proportion of participants with zero bleeding episodes increased from 43% (19/44) to 64% (28/44) in the SHL FVIII cohort (p = 0.0495), and from 41% (14/34) to 65% (22/34) in the EHL FVIII cohort (p = 0.0455; Table 3). In the overall cohort, 36 (46%) participants’ bleeding episodes reduced following switch to efanesoctocog alfa prophylaxis, including 15 (19%) participants who had ⩾3 bleeding episodes on their prior prophylaxis therapy during the observational study and had zero bleeding episodes on efanesoctocog alfa prophylaxis (Figure 3). Furthermore, 29 (37%) participants had no change in the number of bleeding episodes, and 13 (17%) participants experienced an increase in the number of bleeding episodes (Figure 3). Twenty-eight (36%) participants had a reduction in spontaneous bleeding episodes following a switch to efanesoctocog alfa prophylaxis, 39 (50%) participants had no change, and 11 (14%) had an increase (Supplemental Figure 2). Corresponding values for traumatic bleeding episodes were 27 (35%), 43 (55%) and 8 (10%) participants (Supplemental Figure 3).
Bleeding episodes and treatment of bleeding episodes.

Total number of spontaneous and traumatic treated bleeding episodes does not sum to the overall number of treated bleeding episodes as some bleeding episodes were ‘unknown’.
Total dose provided per bleeding episode.
Bleeding episodes with non-missing data.
Calculated by taking the sum of total dose for treated bleeding episodes divided by the sum of follow-up time in years among all participants in the cohort.
EHL, extended half-life; FVIII, factor VIII; IQR, interquartile range; SD, standard deviation; SHL, standard half-life.

Summary of pre-study and on-study treated bleeding episodes per participant.
Treatment of bleeding episodes
For the SHL FVIII cohort, the mean (SD) total dose to treat a bleeding episode pre-study was 35.1 (9.4) IU/kg and on-study was 48.9 (7.1) IU/kg (p = 0.0085; Table 3). Corresponding values for the EHL FVIII cohort were 38.5 (10.1) IU/kg and 46.0 (9.5) IU/kg, respectively (p = 0.2712). The annualized total dose for treated bleeding episodes reduced following switch to efanesoctocog alfa prophylaxis (Table 3). In the SHL FVIII cohort, annualized total dose for treated bleeding episodes reduced from 108.9 to 40.2 IU/kg/year, and in the EHL FVIII cohort, from 97.9 to 26.9 IU/kg/year. The number of injections required to treat a bleeding episode in the SHL FVIII cohort reduced from a mean (SD) of 1.9 (6.1) pre-study to 1.1 (0.3) on-study (p = 0.1388; Table 3). A similar reduction in the number of injections required to treat a bleeding episode was observed in the EHL FVIII cohort, from a mean (SD) of 1.6 (2.2) to 1.1 (0.2; p = 0.3559).
Injection frequency
Switching from prior FVIII prophylaxis to efanesoctocog alfa prophylaxis led to a significant reduction in weekly injection frequency for both SHL FVIII and EHL FVIII cohorts (Table 4; Supplemental Figure 4(A)). Participants in the SHL FVIII cohort had a reduction in their mean (SD) weekly injection frequency from 2.8 (0.9) to 1.0 (0.0), a mean (95% CI) change of −1.8 (−2.1, −1.5) (p < 0.0001). The reduction in mean injection frequency observed in the EHL FVIII cohort was also significant, 1.8 (0.7) to 1.0 (0.0), a mean (95% CI) change of −0.8 (−1.1, −0.6; p < 0.0001). Overall mean (SD) annual injection frequency, including both routine prophylaxis and doses for bleeding episodes, for participants in the SHL FVIII cohort was significantly reduced, from 151.2 (50.6) injections per year to 56.0 (3.1; p < 0.0001), with a reduction from 142.9 (52.8) to 54.8 (1.7) injections per year for routine prophylaxis (p < 0.0001; Table 4; Supplemental Figure 4(B) and (C)). The reduction in mean (SD) annual injection frequency in the EHL FVIII cohort was also significant, from 104.2 (35.0) to 55.4 (1.8) injections per year (p < 0.0001), and a decrease in mean (SD) routine prophylaxis injection frequency from 98.0 (34.3) to 54.6 (1.5) per year (p < 0.0001; Table 4; Supplemental Figure 4(B) and (C)).
Injection frequency and factor consumption.

Weekly injection frequency defined as the total number of injections given to a participant divided by the number of weeks a participant was in the study. Includes both prophylaxis and doses to treat bleeding episodes.
p Values calculated using paired t test.
Includes both prophylaxis and doses to treat bleeding episodes.
EHL, extended half-life; FVIII, factor VIII; SD, standard deviation; SHL, standard half-life.
Factor consumption
Participants in the SHL FVIII cohort had a 47% reduction in their weekly FVIII consumption (including both prophylaxis and doses to treat bleeding episodes) following switch to efanesoctocog alfa prophylaxis; mean (SD) weekly FVIII consumption reduced from 96.3 (68.4) IU/kg to 51.3 (3.0) IU/kg, a mean (95% CI) change of −45.0 (−66.0, −24.0; p < 0.0001; Table 4; Supplemental Figure 4(D)). Similarly, in the EHL FVIII cohort, weekly FVIII consumption was reduced by 30%, from 72.9 (31.0) IU/kg to 50.9 (2.0) IU/kg, a mean (95% CI) change of −22.0 (−32.8, −11.1; p = 0.0002). Switching to efanesoctocog alfa resulted in a reduction in routine prophylaxis dose factor consumption for both the SHL FVIII and EHL FVIII cohorts (Table 4; Supplemental Figure 4(E)). Overall, mean (SD) annual consumption for the SHL FVIII cohort reduced from 5021 (3541) IU/kg to 2676 (155) IU/kg, and for the EHL FVIII cohort from 3808 (1612) IU/kg to 2654 (106.9) IU/kg after switching to efanesoctocog alfa (Table 4; Supplemental Figure 4(F)).
Discussion
The results of this post hoc analysis, comparing outcomes in participants on SHL FVIII and EHL FVIII replacement prophylaxis prior to switching to efanesoctocog alfa prophylaxis, builds upon the results of the phase III XTEND-1 study that reported superiority of efanesoctocog alfa prophylaxis over pre-study FVIII prophylaxis as a key secondary endpoint. 19 In this analysis, we present additional details of the improved bleed protection provided by once-weekly efanesoctocog alfa prophylaxis compared with prior SHL FVIII and EHL FVIII prophylaxis. Importantly, significant reductions in spontaneous and spontaneous joint ABRs were observed across cohorts. In addition, across all three cohorts, there were significant reductions in traumatic and joint ABRs. Therefore, the percentage of participants with zero bleeds increased from 43% to 64% in the SHL FVIII cohort and from 41% to 65% in the EHL cohort following switch to efanesoctocog alfa prophylaxis. Similar results were observed in matching indirect treatment comparison analyses, which found that once-weekly efanesoctocog alfa prophylaxis is associated with a significantly lower ABR compared with both SHL and EHL FVIII replacement therapy prophylaxis.25,26
Increasing evidence suggests higher sustained FVIII activity levels are needed in people with haemophilia to reduce bleeds, with factor activity levels of between 15% and 50% required to achieve a near-zero joint bleed rate.2–6 In XTEND-1, participants receiving once-weekly efanesoctocog alfa (50 IU/kg) prophylaxis had high-sustained FVIII activity levels in the normal to near-normal range (>40%) for 4 days and 15% at the end of the weekly dosing interval. 19 Furthermore, results from a phase I sequential PK study have demonstrated that efanesoctocog alfa has a three- to fourfold longer half-life, and three- to sixfold greater area under the FVIII activity-time curve than EHL and SHL FVIII comparators.20,27 Here, we confirm that once-weekly efanesoctocog alfa prophylaxis provides greater bleed protection than current SHL and EHL prophylaxis.
Although approximately two-thirds of participants receiving efanesoctocog alfa prophylaxis did not experience a bleeding episode on study, bleeding episodes did occur. A post hoc analysis of bleeding episodes during XTEND-1 indicated that the bleeding event rate for treated bleeds was low and stable over the 52 weeks on study for individuals using efanesoctocog alfa prophylactically, with most treated bleeds occurring during Days 5–7 of the weekly dosing interval. 28 Participants receiving efanesoctocog alfa for bleed treatment had fewer injections compared with their prior therapy, with most bleeding episodes resolving with a single injection. In this post hoc analysis, the dose of efanesoctocog alfa used to treat a bleeding episode was higher than prior SHL and EHL therapies; however, this is because the dose of efanesoctocog alfa given for bleed treatment was predefined in the study protocol as a single dose of 50 IU/kg, with the possibility to use a 30 IU/kg dose to treat mild to moderate bleeding episodes within 2–3 days of prophylaxis.
In the pre-study, the weekly injection frequency was 2.8 and 1.8 for participants receiving SHL FVIII and EHL FVIII prophylaxis, respectively. Upon switching to efanesoctocog alfa prophylaxis, participants received once-weekly injections, leading to a reduction of 88 injections for prophylaxis per year in those receiving SHL FVIII, and a reduction of 43 injections per year for the EHL FVIII cohort. Reducing the burden of prophylaxis, while improving protection against bleeds, may contribute to a better quality of life for people with haemophilia, reducing the need to plan and consider treatment in their day-to-day lives. 29
The prospective, observational study presented here and also reported by Chowdary et al., 23 is representative of a global population of people with severe haemophilia A. The overall ABRs in this observational study were 3.23 and 2.61 for the SHL FVIII and EHL FVIII cohorts, respectively. Of note, adherence was not recorded in the observational pre-study; therefore, we are unable to determine whether compliance with prophylaxis influenced the ABRs observed. However, ABRs reported in the observational study were similar to those reported in a recent systemic review that assessed ABRs in observational studies of FVIII replacement therapies, with a mean (95% CI) pooled estimate ABR of 2.0 (1.3, 2.6) for EHL FVIII therapies, and 4.8 (3.6, 6.1) for SHL FVIII therapies. 30
Although the haemophilia treatment landscape has expanded to include alternatives to FVIII replacement-based approaches with the emergence of rFVIIIa mimetics, rebalancing agents and gene therapies, patients may require additional hemostatic support in case of breakthrough bleeds or surgery.12,13,31–36 Efanesoctocog alfa prophylaxis allows for maintenance of high-sustained FVIII levels in the normal to near-normal (non-haemophilia) range for the majority of the week; this level of protection is not achievable with currently available mimetics, rebalancing agents or gene therapy, which provide variable and non-durable levels of FVIII or FVIII-like activity.
Limitations
Limitations in this switching study include the fact that outcomes were assessed post hoc and should therefore be considered exploratory and hypothesis generating. The relatively small number of patients who were eligible for inclusion in this analysis (n = 78) may also limit the generalizability of this analysis.
Conclusion
Results presented here and for the overall population in the prospective observational study 23 highlight that outcomes with current SHL and EHL prophylaxis are suboptimal, with joint bleeds still occurring, which can damage joints, cause chronic pain and impair mobility.7–11 The changing treatment landscape in haemophilia indicates progress towards elevated treatment goals, and attainment of health equity is increasingly possible. 37 Collectively, the results of this post hoc analysis demonstrate the benefits of once-weekly efanesoctocog alfa prophylaxis over SHL and EHL prophylaxis on bleed rates, injection frequency and factor consumption. A phase III study evaluating the long-term safety and efficacy of prophylaxis with efanesoctocog alfa (XTEND-ed; NCT04644575) is ongoing.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251398986 – Supplemental material for Outcomes in participants switching from FVIII replacement therapy to efanesoctocog alfa prophylaxis in XTEND-1: a post hoc analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251398986 for Outcomes in participants switching from FVIII replacement therapy to efanesoctocog alfa prophylaxis in XTEND-1: a post hoc analysis by Sophie Susen, Roshni Kulkarni, Keiji Nogami, Flora Peyvandi, Barbara Konkle, Elena Santagostino, Umer Khan, Annemieke Willemze, Linda Bystrická, Jennifer Dumont and Pratima Chowdary in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors, Sanofi and Sobi gratefully thank the families and all investigators for participating in these studies. This study was funded by Sanofi (Cambridge, MA) and Sobi (Stockholm, Sweden). Biostatistical/programming support for this post hoc analysis was provided by Kirti Nagar, Rahul Gupta and Jagadish Annavarapu of Sanofi. Coordination of the development of this manuscript and facilitation of author discussion was provided by Alicia Mack, PharmD, of Sanofi and Nick Fulcher, PhD, of Sobi. The authors acknowledge medical writing support provided by Sarah Rupprechter, PhD, and Ashleigh Pulkoski-Gross, PhD, CMPP, of Avalere Health Global Limited, and funded by Sanofi and Sobi. The authors were responsible for all content and editorial decisions, and received no honoraria related to the development of this publication.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
