Abstract
Background:
Acquired aplastic anemia (AA) is rare bone marrow failure syndrome characterized by pancytopenia due to immune-mediated destruction of hematopoietic stem and progenitor cells, and leads to an increased risk of bleeding and infectious complications. The first-line treatment option for individuals with severe AA who are ineligible for allogeneic hematopoietic stem cell transplantation is triple therapy, comprised equine anti-thymocyte globulin, calcineurin inhibitor (CNI), and eltrombopag (EPAG). However, this approach is associated with considerable treatment-related complications, requires close inpatient monitoring as well as specialized care and expertise, limiting its feasibility in less-experienced centers. Emerging evidence suggests that double therapy with CNI and EPAG combination reduces the transfusion burden in severe AA.
Objective:
Evaluate the real-world effectiveness of CNI and EPAG combination therapy in reducing transfusion burden among adults with AA.
Methods:
We conducted a retrospective cohort study using Blue Cross Blue Shield Axis Database to examine the change in transfusion requirements in adult patients with newly diagnosed AA who have received combination therapy with CNI and EPAG. Individuals were stratified into baseline transfusion-independent, low, and high transfusion subgroups. Transfusion requirements were reassessed after 6 months from initiation of double therapy.
Results:
The majority of the 153 identified patients with AA started therapy shortly after diagnosis and were adherent to treatment and monitoring during the initial 6 months. Among patients with baseline transfusion requirements (97; 63.4%), 66% achieved TI status. Additionally, 75.5% of patients with a high baseline transfusion burden demonstrated a ⩾50% reduction in transfusion requirements.
Conclusion:
These findings underscore the efficacy and feasibility of double therapy with CNI and EPAG for adults with AA in the real-world setting, offering an alternative for patients when triple therapy is not possible.
Plain Language Summary
Acquired aplastic anemia is an rare condition where the bone marrow stops producing enough blood cells which can cause bleeding and infectious complications. The standard treatment for patients with severe aplastic anemia and who cannot have a bone marrow transplant is the combination of three medications: anti-thymocyte globulin (ATG), a cyclosporine (a calcineurin inhibitor or CNI), and eltrombopag (EPAG). Treatment with ATG can be difficult to administer since it requires close monitoring for complications, making it less practical in certain healthcare settings. In this study, we looked at how a two-drug regimen (CNI and EPAG) worked in real-world settings. Recent studies showed that this two-drug combination was effective in situations where ATG could not be given or was not available. We focused on the number of blood transfusions patients needed before and after starting treatment. We found that most patients started treatment soon after diagnosis and stayed on therapy consistently. Among patients who needed transfusions at the time of diagnosis, two-thirds no longer needed them after 6 months. These results suggest that the two-drug combination may be a practical and effective option for patients with aplastic anemia who cannot receive the standard three-drug regimen.
Introduction
Acquired aplastic anemia (AA) is a rare immune-mediated bone marrow failure disorder caused by cytokine-mediated cytotoxic destruction of hematopoietic and progenitor cells. 1 Patients with AA develop varying degrees of cytopenias which can range from a mild phenotype not requiring transfusions or supportive care (moderate AA), to very severe forms associated with significant risks of bleeding, infections, and mortality.
Allogeneic hematopoietic stem cell transplantation (allo-HCT) remains the only curative therapy for AA, but it is historically limited to younger patients (<40 years of age) with severe disease who have a matched sibling donor. 2 For transplant-ineligible patients with severe or very severe AA, 3 the current standard of care combines horse anti-thymocyte globulin (ATG), calcineurin inhibitor (CNI, such as cyclosporine A or tacrolimus), and eltrombopag (EPAG). 4 This regimen builds on evidence that immune suppression with ATG and cyclosporine A restores hematopoiesis and improves overall survival.5,6 The more recent addition of EPAG, a thrombopoietin receptor agonist, demonstrated better hematopoietic recovery across all cell lineages, resulting in superior complete hematological response rates at 3 and 6 months after treatment initiation, as well as a shorter time to response when compared to immunosuppressive therapy alone.4,7,8 Triple therapy has been shown to induce durable hematologic responses with some patients achieving long-term remission. 9 However, the administration of ATG requires specialized institutional and physician expertise, central venous access, and inpatient monitoring given its risk for serious adverse effects including anaphylaxis and serum sickness. These logistical challenges, along with cost and accessibility barriers, have prompted interest in an ATG-free alternatives, such as the combination of CNI and EPAG as front-line therapy for patients with severe AA. 10
The SOAR trial, an international, single-arm, multicenter, phase II study, evaluated 54 patients with severe AA who were ineligible for or did not have access to ATG and were treated with cyclosporine A and EPAG. 11 The combination of CNI and EPAG yielded hematologic improvements with an overall response rate of 46% by 6 months (25 of 54 patients; 95% confidence interval (CI): 33–60) and was successful in reducing the number of transfusions from baseline, suggesting that this oral, ATG-free regimen can be utilized in settings where use of ATG is not feasible.
Real-world data on CNI plus EPAG use for AA is limited to observational studies from specialized institutions.10,12–14 We examined patterns of care and outcomes of adult patients with newly diagnosed AA using the Blue Cross Blue Shield (BCBS) Axis administrative database, which captures commercial healthcare claims of approximately one-third of the United States’ population, providing a robust platform to assess real-world treatment practices. 15 In our previous publication using the same dataset, we described the initial management of patients with AA and identified that the combination of CNI and EPAG was the most frequently used first-line AA-directed therapy. 16 The purpose of the current study was to further characterize this group of patients and evaluate the efficacy of double therapy approach after 6 months of treatment.
Methods
We conducted an observational claims-based study focused on the utilization and efficacy in reducing the transfusion burden of double therapy, CNI plus EPAG, as first-line treatment of AA. We leveraged the BCBS Axis claims database and included patients with a diagnosis of AA who met the following criteria: (1) ⩾18 years of age; (2) first AA diagnosis (the International Classification of Diseases 10th Edition D61.3, D61.89, D61.9) occurred during July 1, 2016 to June 30, 2022, and had AA diagnosis from at least one inpatient or two outpatient visits ⩾30 days apart; (3) covered by BCBS continuously from 6 months before to 6 months after diagnosis of AA, including prescription drug coverage; and (4) started treatment within 6 months from AA diagnosis. Patients were excluded if they had: (1) unknown baseline characteristics, (2) received ATG and/or allo-HCT after AA diagnosis, or (3) had a diagnosis of acute myeloid leukemia or myelodysplastic syndrome/neoplasm within 6 months before or after AA diagnosis. We defined concurrent treatment as having prescriptions for a CNI (either cyclosporine A or tacrolimus) and EPAG within 3 months and with ⩾1 month overlap during the 6-month observation period.
We evaluated the starting dose of EPAG and testing frequency for CNI levels within 6 months of treatment initiation to ensure adequate dosing and monitoring. We used the median proportion of days covered (PDC) to asses adherence to treatment with CNI and EPAG. We recorded the number of red blood cell (RBC) and platelet (PLT) transfusions at baseline (8-week period before initiation of AA-directed treatment) and at the end of 6 months of treatment (response assessment period, 8-week period after 6 months of treatment). Patients were categorized based on their baseline RBC and/or PLT transfusion requirements into three groups: transfusion-independent (TI, zero transfusions of RBC and PLT), (2) low transfusion burden (LTB, 1–3 transfusions), and (3) high transfusion burden (HTB, ⩾4 transfusions). The outcomes of interest were transfusion independence for all subgroups, and a reduction by ⩾50% for patients with HTB during the response assessment period.
Descriptive statistics were generated for all variables and outcomes of interest, including frequency distributions for categorical variables, and mean, standard deviation, and median (and percentiles) for continuous variables. We used paired Student t test to assess transfusion burden differences between baseline and the response assessment period. We used multivariable logistic regression analysis to assess factors associated with transfusion independence among HTB subgroups. All statistical tests were two-sided with a type I error of 0.05 and conducted using R Version 4.2.2 (The R Project for Statistical Computing, Vienna, Austria). The Yale Human Investigation Committee determined that this study did not directly involve human subjects. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement. 17
Results
We identified 793 patients with a diagnosis of AA in the BCBS database. After implementing eligibility criteria for concurrent double therapy, a total of 153 patients were included in the final cohort (Figure 1). The median age of diagnosis was 51 (interquartile range (IQR): 34–59) years, with 34.6% (n = 53) patients <40 years of age. Most patients were female (n = 81, 52.4%) and were in the Southern (n = 48, 33.4%) or Midwestern (n = 42, 29.4%) US regions. Based on social deprivation index (SDI), the majority of patients (n = 68, 44.4%) were in the first tertile (corresponding to the highest socioeconomic status), whereas 25.5% were in the third (lowest) tertile. Sixty-seven patients (43.8%) had an Elixhauser Comorbidity Score of ⩾3, indicating higher comorbidity severity (Table 1).

Cohort selection.
Baseline characteristics of 153 patients with aplastic anemia.

Comorbidities measure.
IQR, interquartile range; SDI, social deprivation index; SES, socioeconomic status.
Treatment characteristics
The median time from AA diagnosis to initiation of CNI plus EPAG therapy was 17 (IQR: 8–29) days and patients remained on double therapy for nearly 6 months (median 171 days, IQR: 134–183). The PDC was 93.9% (IQR: 72.9–99.4), indicating high adherence to treatment. The majority of patients (n = 122, 79.5%) started both drugs within 30 days apart, with 19 (12.4%) starting on the same day.
A total of 123 patients (80.4%) had CNI levels measured at least once during the first month of treatment. Patients had a median of three CNI level measurements (IQR: 1–5) in the first month, and nine (IQR: 3–14) within the first 3 months. Cyclosporine A was the most commonly used CNI, prescribed in 136 patients (88.9%), while 9 patients (5.9%) received only tacrolimus, and 8 patients (5.2%) switched between CNI agents during the treatment period. The median starting dose for EPAG was 150 mg (n = 108, 70.6%) with 2 patients (1.3%) starting at higher doses and 43 (28.1%) at lower doses.
Transfusion assessments
At baseline, 56 patients (36.6%) were TI, while 97 patients (63.4%) were transfusion-dependent at baseline. Among transfusion-dependent patients, 48 (49.5%) had a LTB, and 49 (50.5%) had a HTB. Most TD patients (n = 70, 72.2%) received both RBC and PLT transfusions, whereas 14 (14.4%) received only RBC transfusions and 13 (13.4%) only PLT transfusions. The median number of baseline RBC and PLT transfusions was three (IQR: 2–5) and five (IQR: 4–6), respectively.
Transfusion requirements were reassessed 6 months after initiating double therapy during the response assessment period. Of the 97 patients who were transfusion-dependent at baseline, 64 (66.0%) became TI, including 34 (70.8%) with LTB and 30 (61.2%) with HTB. Additionally, 37 of 49 patients (75.5%) with baseline HTB achieved a ⩾50% reduction in transfusion burden while only 7 patients (14.3%) received a higher number of transfusions by the end of 6 months. Among the 56 patients who were TI at baseline, 44 (78.6%) remained TI by the end of the response assessment period (Figure 2). Across all 153 patients with AA, the mean reduction in transfusion requirements between baseline and end of the response assessment period was 1.2 (95% CI: 0.59–1.81; p < 0.001).
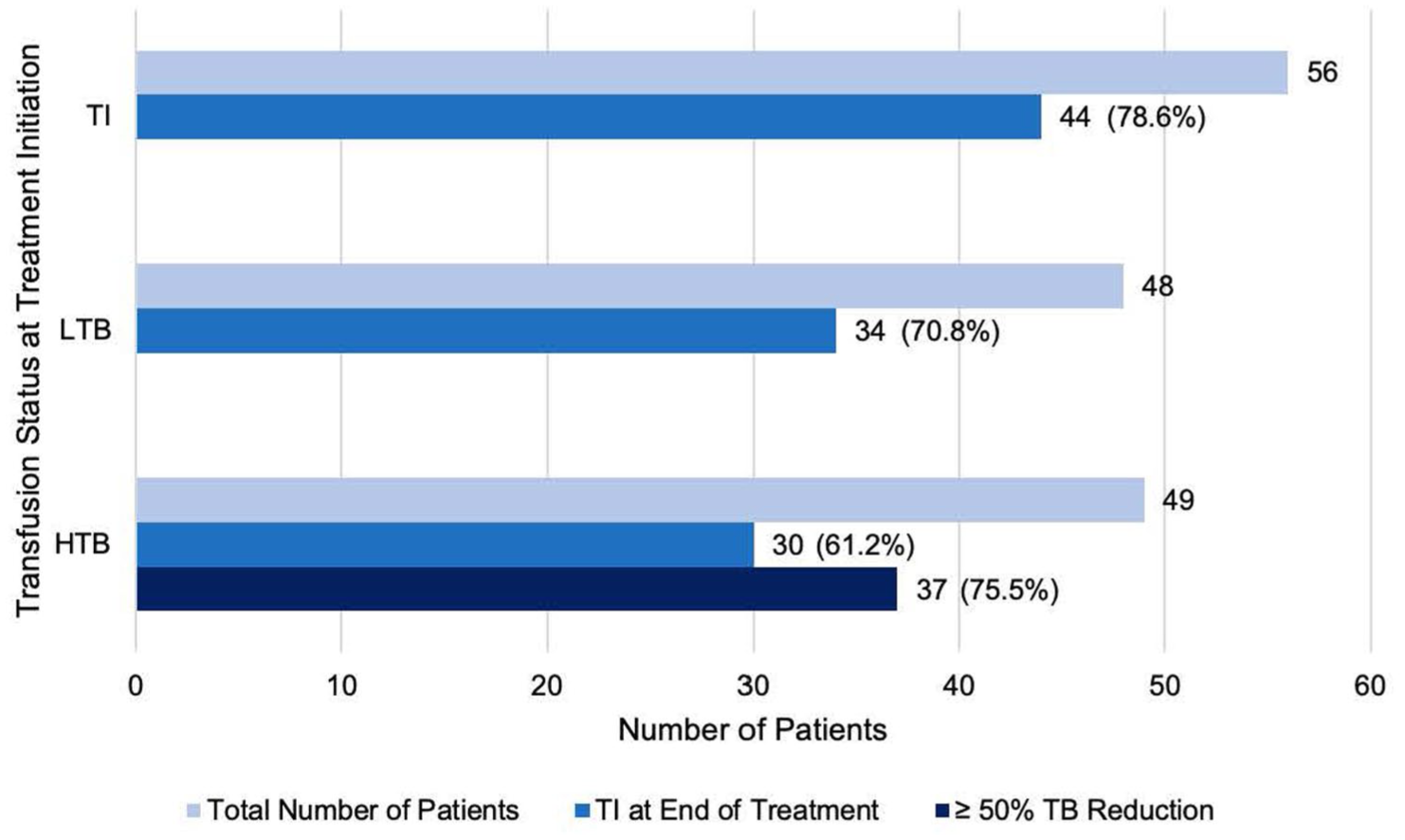
Transfusion status subgroups during the treatment period.
We analyzed baseline factors among the cohort associated with achieving TI at the end of the response assessment period in patients with HTB. Residence in the Northeastern US was significantly associated with a higher likelihood of achieving TI (odds ratio (OR = 0.004), 95% CI: 0.002–0.40; p < 0.01). In contrast, male patients were less likely to become TI by 6 months after treatment (OR = 12.66, 95% CI: 2.43–118.87; p < 0.01; Table 2). No other baseline characteristics were found to significantly impact the probability of becoming TI after 6 months of treatment.
Factors associated with transfusion independence at the end of the response assessment period for high transfusion burden patients.
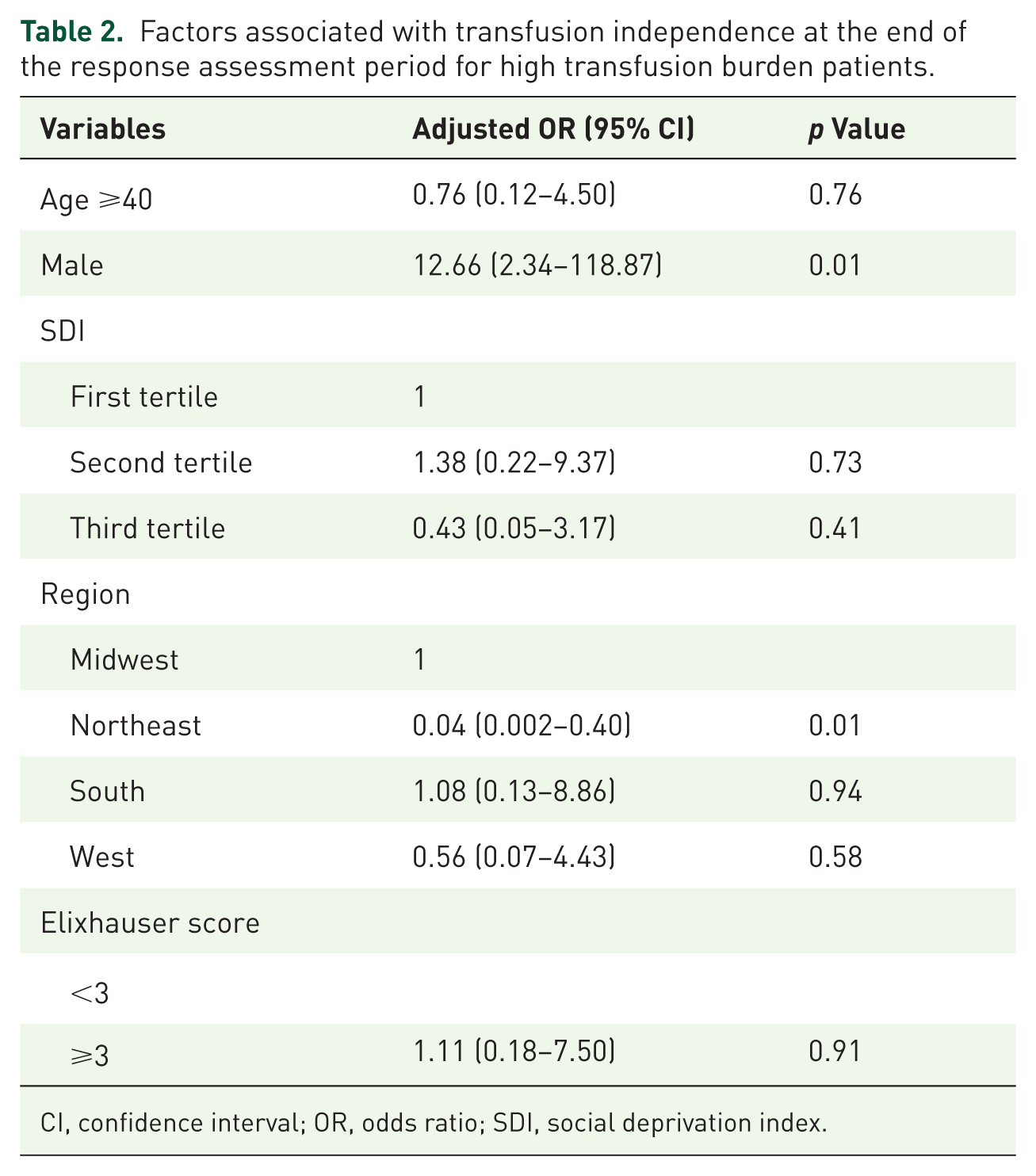
CI, confidence interval; OR, odds ratio; SDI, social deprivation index.
Discussion
Our study represents one of the largest real-world analyses of adult patients with AA treated with CNI and EPAG and provides evidence that this combination can effectively reduce the transfusion burden when used in the front-line setting. Among the 97 patients who were TD at baseline, 64 patients (66%) were transfusion-free by the end of the 6-month treatment period, and 37 patients (75.5%) with a higher baseline transfusion requirement achieved a ⩾50% reduction in the number of transfusions. The good tolerability of double therapy is demonstrated by a high PDC rate of 93.9% and the accessibility of CNI level monitoring is supported by the frequent testing. Notably, 80.4% of patients had CNI levels measured at least once during the first month of treatment. These findings highlight the potential of double therapy as a practical and effective treatment alternative for patients who may not be eligible for ATG-based regimens or allo-HCT.
Our recent study utilizing the same dataset demonstrated that CNI and EPAG was the most frequently prescribed combination, with only 4.4% of patients using front-line triple therapy. 16 Due to limitations of the study design and utilized dataset, we were not able to investigate the reasons for ATG underutilization but provided plausible explanations. Administration of ATG requires specialized institutional and physician expertise, central venous access, and inpatient monitoring given its risk for serious adverse effects. These logistical challenges, along with cost and accessibility barriers, have prompted interest in an ATG-free combination of CNI and EPAG as front-line therapy for patients with severe AA. The SOAR trial reported a 70% transfusion independence rate by 6 months of treatment (38 of 54 patients), including 45% of patients (13 of 29 patients) who did not achieve a hematological response. 11 Our study, which likely included some patients with less severe disease, demonstrated a 66% transfusion independence rate after 6 months of treatment confirming the efficacy of this combination in the real-world setting. While our results support the use of double therapy to reduce transfusion burden, it is important to emphasize that triple therapy with ATG, cyclosporine A, and EPAG remains the preferred initial therapy for transplant-ineligible patients with newly diagnosed severe AA. Double therapy may, however, serve as a reasonable alternative for adults in whom ATG poses excessive risks or is unavailable. The high degree of adherence to the double regimen and CNI level monitoring observed in our cohort suggests that this approach is feasible, particularly in community settings with limited specialized hematologic care.
The study has several limitations. Our cohort had a limited number of patients of 60 years of age and older (n = 34, 22.2%) due to the nature of BCBS Axis claims database which includes mostly younger individuals, challenging the generalizability of its findings to an older patient population. The BCBS dataset does not include laboratory values, pathology, or molecular testing results. Due to these limitations, we were unable to independently verify the AA diagnosis, assess disease severity using established Camitta criteria, or evaluate hematologic responses. 3 Transfusion independence was selected as a pragmatic surrogate endpoint, as it was reliably captured across the cohort and reflects a meaningful clinical benefit in clinical practice. Additionally, data on patient demographics is incomplete, preventing further analysis based on race and ethnicity relevant for evaluating EPAG dosing. 18 Another limitation is that our response assessment period extended only to 8 weeks after 6 months of initial treatment, leaving the durability of response beyond the timeframe of our study unclear. Future prospective trials should assess the duration of responses to double therapy and help identify predictive biomarkers for treatment effectiveness. While our study provides valuable insights into the real-world use of CNI with EPAG in AA, these findings should be interpreted with caution as the absence of disease severity assessment at baseline and hematologic response at the end of the study period using standard criteria limits results interpretation. Despite these limitations, our findings suggest that CNI and EPAG combination therapy is feasible and effective in reducing the transfusion burden among patients with AA. Expanding access to this treatment strategy may help bridge critical gaps in care for patients who are unable to receive ATG-based therapy.
Conclusion
We conclude that, in the real-world setting, the CNI plus EPAG regimen was initiated promptly after AA diagnosis and was associated with high degree of patient adherence to treatment and CNI monitoring. The oral combination regimen effectively reduced the transfusion burden after 6 months of treatment, further supporting its role in the management of patients with AA.
Footnotes
Acknowledgements
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under Award Number T32CA233414 (Awarded to J.M.S.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
