Abstract
Background:
Waldenström’s macroglobulinemia (WM) is a B-cell neoplasia characterized by the infiltration of lymphoplasmacytic lymphoma cells in the bone marrow and abnormal secretion of IgM paraprotein. Ibrutinib, a Bruton tyrosine kinase inhibitor (BTKi), showed high efficacy in WM clinical trials. However, there is limited real-world data regarding its effectiveness and safety in routine clinical practice.
Objectives:
The MACRO study aimed to investigate the clinical, genetic, and demographic characteristics of WM patients treated with ibrutinib-based therapies in Spain. Key secondary objectives included describing effectiveness and safety profile.
Design:
Retrospective observational.
Methods:
This multicenter, observational, retrospective study included adult patients diagnosed with symptomatic WM treated with ibrutinib since its commercial approval in Spain in 2016. Data were collected from 19 hospitals through retrospective medical chart reviews.
Results:
Fifty-two eligible patients were recruited. The median age at the start of ibrutinib treatment was 74 years. Most of patients were male (65.4%) and had an Eastern Cooperative Oncology Group performance status of 0–1 (89.7%). Overall response rate was 92.2%, with a major response rate of 80.5%. Median progression-free survival (PFS) was 57.2 months, and the estimated 2-year overall survival rate was 89.2%. No significant differences in PFS were identified based on the parameters defining risk subgroups, nor did they vary according to treatment line, initial dose, or treatment schedule. Most common adverse events included bleeding (30.8%), diarrhea (23.1%), and infections (15.4%), with most of them being grades 1–2. No new safety signs were identified.
Conclusion:
This study presents real-world evidence on the characteristics and outcomes of WM patients treated with ibrutinib in Spain, showing it to be effective with a manageable safety profile consistent with clinical trial results. These findings support ibrutinib as a valuable treatment option for WM in real-world settings.
Introduction
Waldenström’s macroglobulinemia (WM) is a B-cell neoplasia characterized by the presence of a lymphoplasmacytic lymphoma in the bone marrow and the abnormal secretion of IgM paraprotein.1,2 This malignant disease is most commonly diagnosed in adults between 63 and 75 years and is more prevalent in men. 3 The incidence of WM is approximately three cases per million inhabitants per year. 4 The pathophysiology of the disease is typically driven by genetic alterations in myeloid differentiation factor 88 (MYD88) and C-X-C chemokine receptor type 4 (CXCR4) genes. MYD88 mutations are present in over 95% of the patients, the most common being the L265P mutation. CXCR4 mutations are more heterogenous and are present in approximately 35% of the patients. The mutational status of those two genes can affect disease presentation and response to treatments.5,6 Clinically, patients with WM can be asymptomatic and may not require treatment for several years. However, nearly 50% eventually develop symptomatic progression, requiring treatment initiation. 7 Clinical and laboratory indicators to initiate treatment typically include anemia, constitutional symptoms, lymphadenopathy, organomegaly, neuropathy, hyperviscosity, cryoglobulinemia, amyloidosis, thrombocytopenia, and IgM levels over 60 g/L. 8
Treatment options for managing WM are heterogeneous and their use may be influenced by variations in clinical guidelines, as well as individual patient factors, such as age, clinical characteristics, and comorbidities. In addition, disease relapse is common, further complicating the management and requiring personalized therapeutic approaches.1,3 Among the treatment options for WM treatment, ibrutinib is a first-in-class, oral, covalent BTK inhibitor (BTKi) indicated for patients with relapsed/refractory WM or as front-line treatment in patients nonsuitable for immunochemotherapy, that has demonstrated high efficacy with long-term follow-up.9,10 The 59-month follow-up of the phase II pivotal trial, including 63 patients with relapsed refractory (R/R) WM with two prior lines of therapy who underwent ibrutinib treatment as monotherapy,9,10 showed a 5-year progression-free survival (PFS) rate of 54% and a 5-year overall survival (OS) rate of 87%. In addition, ibrutinib in combination with rituximab (I + R) also demonstrated similar high sustained efficacy in the 50-month follow-up of the phase III iNNOVATE clinical trial11,12 that also included treatment naïve patients, with estimated 54-month PFS and OS rates of 68% and 86%, respectively.
However, despite the efficacy of ibrutinib has been widely demonstrated in clinical trials, published studies in WM real-world cohorts are relatively uncommon.9,13–17 Consequently, clinical results in routine clinical practice in WM patients, many of whom would not be eligible for clinical trials, remain uncertain. Hence, the aim of our study was to describe the clinical, genetic, and demographic characteristics of WM patients treated with ibrutinib-based therapies, along with their management and treatment effectiveness in routine clinical practice in Spain. For this purpose, we designed the retrospective observational MACRO study.
Methods
Study design and patient population
MACRO is a multicenter, observational, retrospective study conducted in 19 hospitals in Spain. Patients included in the study met the following inclusion criteria: aged ⩾18 years; diagnosed with WM; treated with ibrutinib, either as monotherapy or in combination with rituximab, from its commercial approval in Spain (2016) up to 2 months before signing the informed consent or death; and provided signed informed consent. Exclusion criteria comprised not providing informed consent, participation in an ibrutinib clinical trial prior to or during the study, and being pregnant or breastfeeding.
This study was approved by the Ethics Committee (EC) of the participating centers and was conducted following the Helsinki Declaration and national regulations. All patients gave written informed consent, except for those who were deceased at the time of study inclusion, for whom a waiver was granted by the EC.
Data collection and assessments
A retrospective medical chart review was conducted to collect data from patients’ medical histories, WM-related information, and their treatment sequences, focusing on ibrutinib. Data were entered into an electronic case report form (eCRF). The primary objective was to characterize the patient population with WM treated with ibrutinib in Spain in routine clinical practice. This included demographic, clinical, and genetic characteristics, comorbidities and concomitant therapies at the time of diagnosis and at the initiation of ibrutinib treatment, as well as detailed information on previous and, if available, subsequent treatments. Secondary objectives included the description of the ibrutinib effectiveness by assessing survival rates (PFS, OS), treatment response rates (overall response rate (ORR), major response rate (MRR), complete response (CR), very good partial response (VGPR), partial response (PR), minor response (MR), and stable disease (SD)), time to response, duration of response (DOR), and levels of hemoglobin (Hb) and IgM. Tolerability and ibrutinib management were addressed by collecting adverse events (AEs), dose modifications, and treatment interruptions and discontinuations.
Statistical analysis
The sample size for this study was calculated to estimate the distribution of the main patient characteristics with a 95% confidence level (CI) and an accuracy of ±15%. Based on these parameters, a minimum of 43 patients was deemed sufficient. Assuming an anticipated 10% rate of nonevaluable data during the study period, the final sample size was adjusted to 48 patients.
Descriptive statistics were used to summarize patient demographics, clinical characteristics, and safety data, including measures of central tendency and dispersion (median (interquartile range, IQR), or 95% CI) for quantitative variables and absolute (n) and relative (%) frequencies for qualitative variables. For dependent variables, parametric (paired t test) or nonparametric (Wilcoxon) statistical tests were performed according to sample distribution. All statistical analyses used two-tailed tests with a significance level of 95% (α = 0.05). Missing data for each parameter or variable were not included in the percentage calculations.
ORR was defined as the proportion of patients who had a response greater than or equal to MR, and MRR as the proportion of patients who had a response equal to or greater than PR. Time to first response was defined as the time (months) between ibrutinib start and the first objective response (PR or better). DOR was defined as the time between the date of the first objective response (PR or better) and the date of the first detection of progressive disease (PD) or death, whichever occurred first. PFS was determined as the time between the date of initiation of ibrutinib treatment and the date of disease progression or death from any cause, whichever occurred first, and OS as the time from the initiation of ibrutinib to the death of the patient (from any cause). The time-to-event endpoints (PFS, OS, DOR) were analyzed using the Kaplan–Meier method, with medians and 95% CIs. Patients were censored at the date of the last available follow-up if they were still alive or remained without disease progression at the time of the analysis. PFS and OS were stratified by ibrutinib treatment line, dosing, and treatment schedule (monotherapy or combination), and survival curves were compared using the Log-Rank test. Differences in PFS according to the categories defining the different WM risk groups (age, levels of Hb, β-2 microglobulin (B2M), IgM, lactate dehydrogenase (LDH), and serum albumin, platelet count, Eastern Cooperative Oncology Group performance status (ECOG PS), International Prognostic Scoring System for Waldenström’s Macroglobulinemia (IPSSWM)) at the start of ibrutinib treatment, the line of therapy, and the treatment scheme were also analyzed in a post hoc analysis. Hazard ratios and corresponding 95% CIs were calculated by Cox regressions. Analyses were performed with the IBM Statistical Package for the Social Sciences (SPSS), version 29.0.1.0.
Results
Fifty-eight patients with WM were recruited in 19 centers homogeneously distributed in Spain between March and June 2023, of which 6 were excluded for not fulfilling the eligibility criteria. Thus, 52 patients were eligible for analysis. At the time of WM diagnosis, median (IQR) age was 70 years (59–78), and 65.4% of patients were male. Most patients (64.4%) had an intermediate/high (⩾2) IPSSWM score and a baseline ECOG PS of 0–1 (96.2%). Most common clinical manifestations of the disease at diagnosis were fatigue (38.5%), weight loss (19.2%), and lymphadenopathy (19.2%). Regarding the laboratory findings, 38.5% and 21.2% of patients had Hb levels below 10 g/dL and IgM levels above 60 g/dL, respectively. MYD88 status was assessed at WM diagnosis in 29 cases, being positive MYD88L265P in 93.1% (n = 27) of them. Family history was available for 32 patients, and none had a family history of WM, although 2 patients had a positive family history of other lymphoproliferative disorders (Hodgkin’s lymphoma and gastric lymphoma).
Prior lines of therapy before ibrutinib initiation
Thirty-nine patients (75%) had received any therapy before ibrutinib: one line in 22 cases (56.4%), two lines in 9 cases (23.1%), and three or more in 8 cases (20.5%). The most common front-line regimens were dexamethasone, rituximab, and cyclophosphamide (DRC; 38.5% of patients), chlorambucil-based regimens (17.9%), and other cyclophosphamide-based regimens (15.4%). Most patients responded well to front-line treatment, with an ORR of 67%. Cyclophosphamide-based regimens were the most frequently used in patients receiving a second line before ibrutinib (41.2% of patients), followed by DRC (11.8%). The ORR for second-line therapy was 81.3%. For third-line therapy and beyond, most patients received chemotherapy-based regimens, with cyclophosphamide-based regimens being the most frequent in both third- (25% of patients) and fourth-line therapy (60%). These patients predominantly achieved PRs (57.1%, 75%, and 100% for third-, fourth-, and fifth-line therapy, respectively), and refractoriness was common (42.9%, 75%, and 50%). All patients receiving ibrutinib in the relapse setting were previously exposed to rituximab.
Ibrutinib treatment
The median time from WM diagnosis to ibrutinib treatment initiation was 42 months (range: 5–131), and the median age at the start of ibrutinib was 74 years (range: 48–90), with 44.5% of patients being older than 75 years. Characteristics of the patients at ibrutinib treatment initiation are summarized in Table 1. Most of the patients (89.7%) had an ECOG performance status of 0–1. MYD88 was mutated in 91.3% (n = 21/23) of patients, and CXCR4 in 20% (n = 2/10) of the patients, when the test was available. Considering the previous line of therapy, 27% of patients were primary refractory, while 29% had progressed in less than 3 years after their last line of therapy. At the time of the visit for treatment initiation, 65.9% (n = 27/41) of patients had hypertension, and only 71.4% (n = 25/35) exhibited regular sinus rhythm, when available. Furthermore, 21.4% of patients had extra nodal disease.
Characteristics of the patients at the start of the ibrutinib treatment.

Data available were indicated as denominators when some information was missing or not recorded in the clinical charts for the entire cohort.
ECOG PS, Eastern Cooperative Oncology Group performance status.
Forty-four patients (84.6%) had comorbidities at the time of ibrutinib initiation, the most common being cardiovascular disorders (32.7%), endocrine disorders (26.9%), other previous nonlymphoproliferative malignancies (21.2%), and respiratory disorders (21.2%). Patient comorbidities at the start of the ibrutinib treatment are detailed in Table 2.
Comorbidities presented in the patients at the time of ibrutinib initiation.

Forty-four (84.6%) out of the 52 patients presented at least one comorbidity at the time of ibrutinib initiation. Percentages calculated on the whole cohort (n = 52).
Seventeen patients presented a total of 19 cardiovascular disorders.
Accordingly, most patients (96.2%) were receiving concomitant medications, being the most common proton pump inhibitors in 53.8% of cases, antihypertensive drugs in 42.3%, antiviral medications in 36.5%, and antidepressant drugs in 21.2%. Additionally, 17.3% and 15.4% of patients were receiving anticoagulants and antiplatelets, respectively.
The most common clinical manifestations prompting therapy initiation were fatigue in 30.8% of cases, peripheral neuropathy in 21.2% and lymphadenopathy in 17.3%. Moreover, 32.7% of patients had Hb levels below 10 g/dL, 17.3% had IgM levels above 60 g/dL, and 11.5% presented platelet count under 100 × 109/L.
Among the entire cohort, 25% of patients received ibrutinib as first-line therapy, 42.3% as second-line therapy, and 32.6% in third-line or later. The median number of lines of therapy prior to ibrutinib treatment in R/R patients (n = 39) was 1 (range: 1–8). Most of the patients (90.4%) were treated with ibrutinib as monotherapy, while five of them received it in combination with rituximab. Forty-six patients (88.5%) received the recommended starting dose of 420 mg, while the remaining received a starting reduced dose of 280 mg due to various reasons. At the time of database lock, 42 patients (80.8%) were alive and ibrutinib treatment was still ongoing in 36 of them (85.7%).
Treatment outcomes and survival rates
Median time to first response (⩾PR) was 4 months (95% CI: 4.7–10.3). ORR was 92.2% (2%, n = 1 CR; 21.1%, n = 11 VGPR; 55.7%, n = 29 PR; and 11.8%, n = 6 MR), with a MRR of 80.5%. Three patients had SD, and one had PD as best documented response. Response rates are shown in Figure 1.

Response rates to ibrutinib treatment. Response rates to ibrutinib were presented for 51 patients based on available data.
With a median follow-up of 23.7 (4.1–82.3) months, the median DOR was not reached (NR; Figure 2), and the estimated 2-year DOR rate was 62.8% (95% CI: 42.6–83).

Duration of response to ibrutinib treatment in responding patients with Waldenström’s macroglobulinemia (n = 41). Censored data includes patients who received post-ibrutinib therapy without experiencing disease progression during ibrutinib treatment and those who had reached their last follow-up date.
The median levels of Hb and IgM improved significantly during ibrutinib treatment (Figure 3). At best response, median Hb levels increased from 10.8 (4.7–14.5) to 13.1 (8.3–16.4) g/dL (p < 0.001) and remained above 12 g/dL for up to 48 months. Additionally, median IgM levels decreased from 3235 (range: 208–7470) to 727 (range: 46–3680) mg/dL at best response (p < 0.001), with levels remaining below 1000 mg/dL from 18 months of treatment onward.

Changes in hemoglobin (a) and IgM (b) levels during ibrutinib treatment. Median values (depicted as squares) are shown alongside error bars indicating the range (minimum to maximum values) for each assessment time point. In panel B, a Y-axis break was applied between 5500 and 7000 mg/dL.
Regarding survival outcomes, median PFS was 57.2 months (95% CI: 42.4–72.0; Figure 4(a)). No differences were found in median PFS in patients who received reduced dose initially (280 mg) compared to patients who received the recommended dose by label (420 mg; p = 0.534; Figure 4(b)), as well as for those who had dose reductions over the treatment versus those who did not (p = 0.568; Figure 4(c)). Similarly, no significant differences were found in patients who received ibrutinib as first line, second line, or third line or later therapy (p = 0.483; Figure 4(d)) or those who received ibrutinib as monotherapy or in combination with rituximab (p = 0.965).
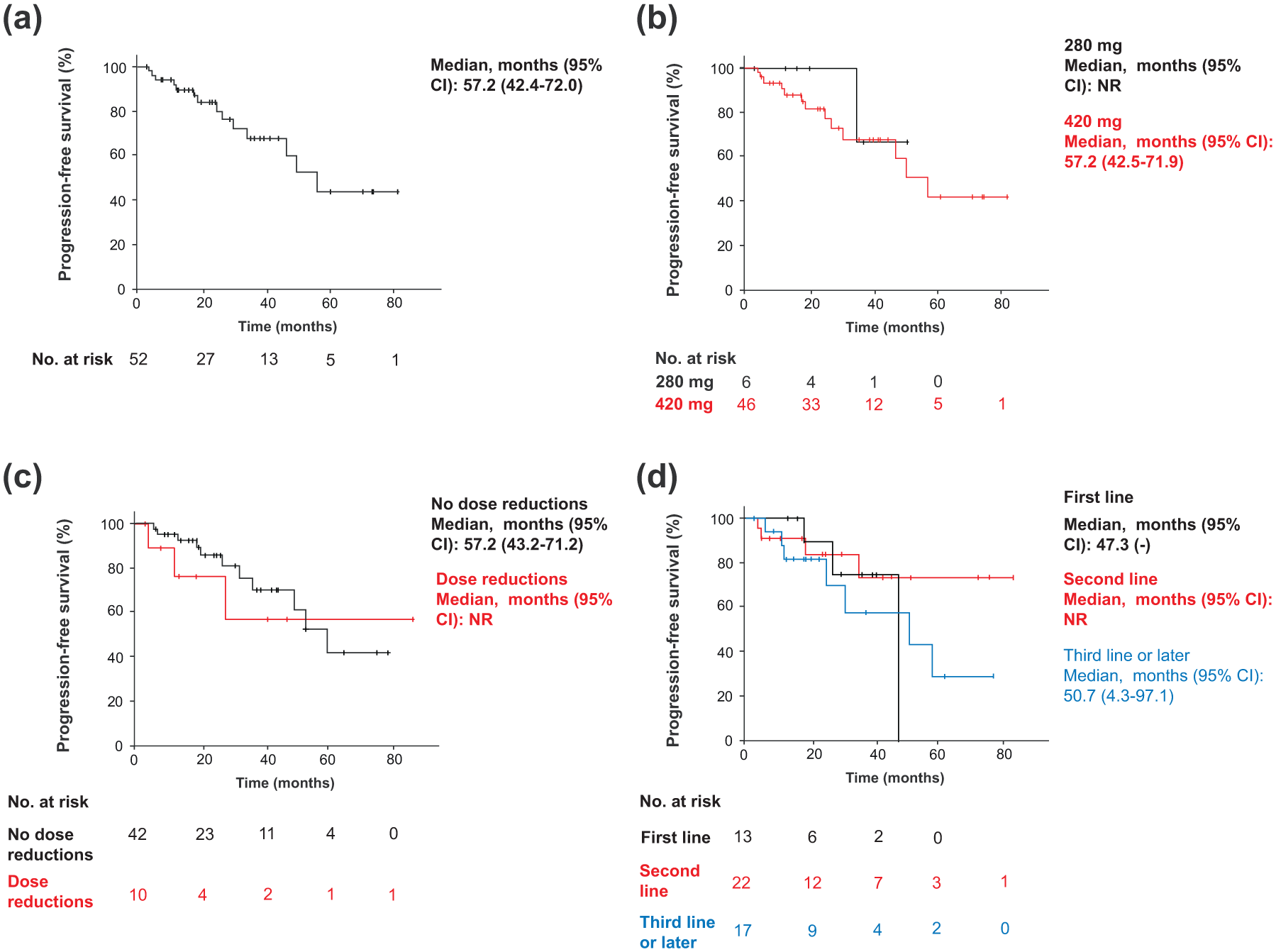
PFS for patients with Waldenström’s macroglobulinemia treated with ibrutinib. Kaplan–Meier curves for PFS in the entire cohort (a), according to the ibrutinib dose received (b), the need of dose reductions (c), and the line of therapy which ibrutinib was administered (d).
Moreover, we performed a post hoc analysis on PFS aiming to analyze the impact of the parameters defining the different WM risk subgroups. We did not find any differences according to age, hemoglobin, platelet count, B2M, IgM, LDH, serum albumin, ECOG, IPSSWM, line of therapy, and treatment scheme. However, age (66–75 years and >75 years) and LDH levels ⩾250 U/L, showed a trend toward poorer PFS outcomes (Supplemental Table 1).
Median OS for the entire cohort was NR (Figure 5) and the estimated 2-year OS rate was 89.2% (95% CI: 80.1–98.3). Similarly to PFS, no significant differences in OS were found considering the different subgroups studied.

OS for patients with Waldenström’s macroglobulinemia treated with ibrutinib. Kaplan–Meier curves for OS in the entire cohort.
Post-ibrutinib treatment
Six patients (11.5%) received a treatment after ibrutinib, which correspond to second line (n = 3), third line (n = 1), and fourth line (n = 2). The most common treatments used after ibrutinib were combinations of bortezomib, dexamethasone, and rituximab (BDR), DRC, and other chemotherapy combinations. After ibrutinib treatment, patients receiving subsequent second-, third-, and fourth-line therapies exhibited responses of PR or VGPR, SD, and VGPR or CR, respectively.
Safety
During the observation period, a total of 126 AEs were registered of which 9.5% were considered serious AEs. The most common AEs of any grade were bleeding (30.8%), diarrhea (23.1%), infections (15.4%), and atrial fibrillation (13.5%), most of them being grades 1–2 (26.9%, 23.1%, 9.6%, and 11.5%, respectively; Table 3). Fourteen patients (26.9%) had at least one grade 3–4 AE, being the most common infections (5.8%), bleeding (3.9%), and neutropenia (3.9%).
Most frequent AEs in patients treated with ibrutinib according to the grade.
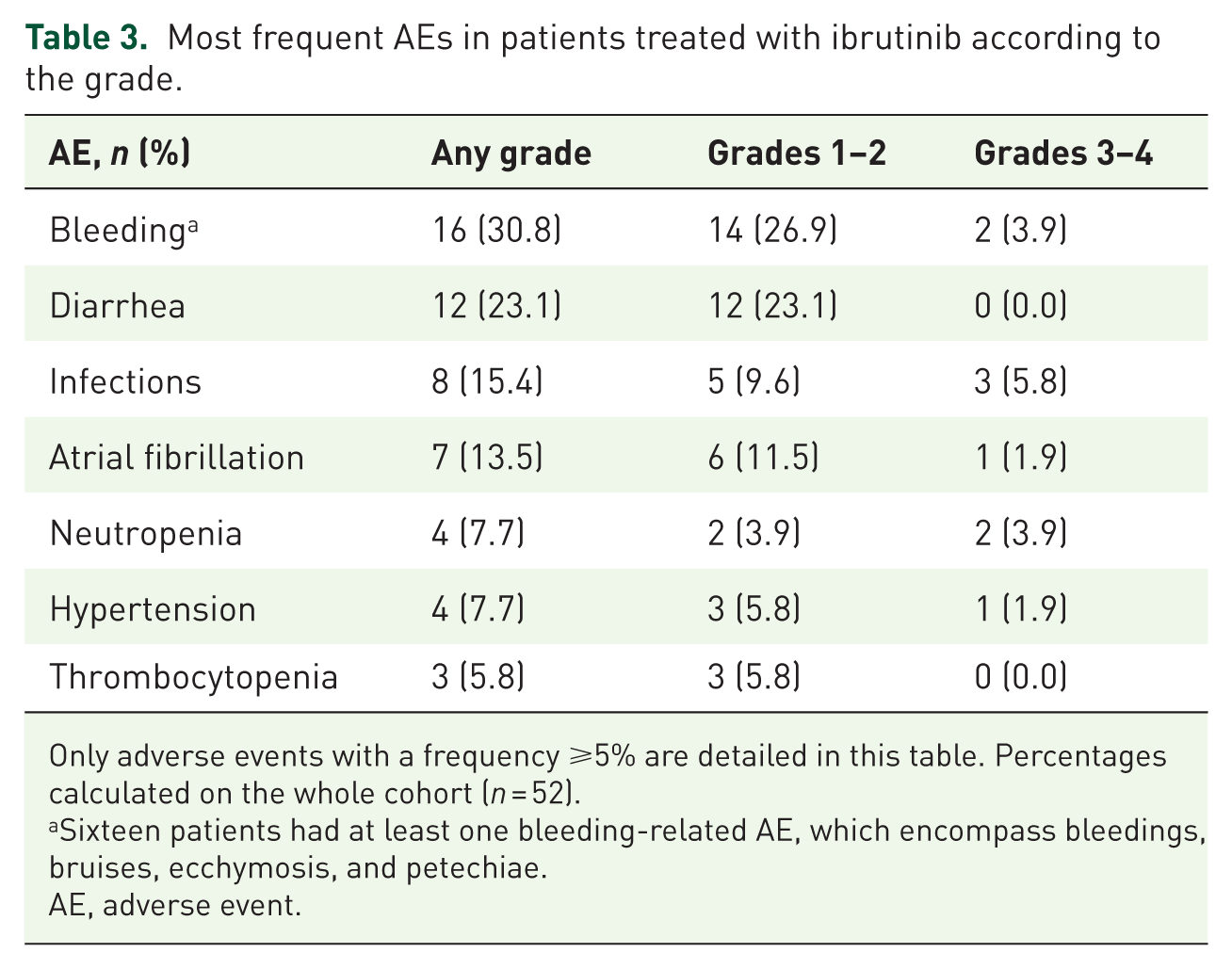
Only adverse events with a frequency ⩾5% are detailed in this table. Percentages calculated on the whole cohort (n = 52).
Sixteen patients had at least one bleeding-related AE, which encompass bleedings, bruises, ecchymosis, and petechiae.
AE, adverse event.
At the time of study inclusion, 74% of AEs had resolved. Most of AEs (64.3%) did not require pharmacological intervention, while 25% of AEs required temporary interruption, 5% required a dose reduction, and 7% treatment discontinuation (most frequently due to atrial fibrillation and bleeding). Six out of seven patients with atrial fibrillation required intervention with three of them discontinuing ibrutinib treatment.
A total of 11 dose reductions occurred in 10 patients (19%) due to hematological toxicities (18.2%), nonhematological toxicities (45.5%) and other reasons (36.4%; including possible signs of disease progression, a potential interaction with isoniazid, and patient-reported gastrointestinal discomfort and diarrhea). Fifty-two temporary interruptions occurred in 28 patients (53.8%), with 17.3% due to hematological toxicities and 48.1% due to nonhematological toxicities.
Discussion
Despite ibrutinib being approved for the treatment of WM several years ago, 18 real-world data with the patient characteristics who received it and their clinical outcomes in routine clinical practice remain limited. This study provides a comprehensive characterization of patients with WM treated with ibrutinib in routine clinical practice in Spain and evaluates its effectiveness and safety in a real-world clinical setting.
In our study, consistent with prior epidemiology data, patients with WM were older, with a median age of 70 years at diagnosis and 74 years at the start of ibrutinib treatment. At the start of ibrutinib treatment, most patients had an ECOG PS of 0–1. In line with previous findings,13–17,19,20 nearly all patients with genetic evaluation performed presented the WM characteristic MYD88L265P mutation. Furthermore, most patients had comorbidities, being the most common hypertension and cardiovascular and endocrine disorders. In addition, they were also receiving concomitant medications, such as anticoagulants and antiplatelets. This patient profile, frequently excluded form clinical trials, is consistent with the profiles described in WM populations across other real-life studies.13–17,19–22
In our cohort, patients were most commonly treated with ibrutinib in the second line of treatment. This is consistent with the real-world study of Riva et al., 13 who analyzed the treatment patterns in 159 patients with WM. In their cohort, ibrutinib was also most frequently prescribed as second- and third-line therapies, despite potential variations in clinical practice depending on the countries and availability. Moreover, nearly all patients in our cohort received ibrutinib as monotherapy at the recommended starting dose of 420 mg, consistent with the findings from other real-world studies,13–16 where ibrutinib was prescribed as monotherapy despite the combination therapy with rituximab being on label. However, it is noteworthy that the lack of reimbursement for the combination ibrutinib + rituximab in Spain may have influenced the number of patients receiving this combination therapy during the study period.
Treatment response in our real-world cohort is consistent with the clinical trial findings and other real-world studies.10,14,16,17,23 In our analysis, we observed an ORR of 92% and a MRR of 81% at best response, along with an estimated 2-year DOR of 63%. With regard to previous real-world studies, these results are similar to those reported by Abeykoon et al. 16 and Castillo et al., 17 whose studies included two cohorts of 80 and 157 R/R WM patients treated with ibrutinib outside of clinical trials, respectively. Furthermore, Cencini et al. 14 found an ORR of 92%, a MRR of 87.7%, and an estimated 2-year DOR of 89% in an Italian cohort of 49 R/R patients with WM, further supporting these response rates of ibrutinib in routine clinical practice in Europe. They also observed rapid and sustained improvements in Hb and IgM levels, similar to the findings reported in our cohort.
The effectiveness of ibrutinib in terms of survival rates was also consistent with findings previously reported.10,14,16,17,23 In our real-world cohort, median PFS was 57 months and the estimated rate of OS at 2 years was 89%, closely mimicking the results from several other real-world studies,14,16,17 as well as findings from clinical trials.10,23 In particular, among previous real-world studies, Abeykoon et al. 16 reported an estimated 18-month OS of 87%, Castillo et al. 17 observed a 4-year OS of 81%, Sarosiek et al. 15 reported a 4-year OS of 88%, and Cencini et al. 14 observed an estimated 2-year OS of 84.1%. In line with these findings, which show similar outcomes between real-world studies and clinical trials, Castillo et al. found no significant differences in response rates or survival outcomes between WM patients enrolled in clinical trials and those treated outside of them. 17 These findings underscore the effectiveness of ibrutinib in routine clinical practice, especially considering that a significant number of patients in those real-world cohorts would not have been eligible for clinical trials.10,14,16,17,23
Additionally, in the subgroup analysis, we did not find any factors significantly impacting PFS, including those defining the risk groups for WM (age, hemoglobin, platelet count, B2M, IgM, LDH, serum albumin, ECOG PS, and IPSSWM), although advanced age and high levels of LDH showed a trend toward reduced PFS. Likewise, the line of therapy and treatment scheme did show no significant effect. Similarly, Cencini et al. 14 did not observe an association between some of these risk group categories and PFS in their cohort. This lack of significant findings may be attributed to the limited sample sizes of both real-world cohorts or to a real lack of impact of these prognostic factors when treating with ibrutinib. However, Castillo et al. found thrombocytopenia and CXCR4 mutations to be two factors impacting effectiveness in a cohort including patients treated in and outside clinical trials. 24 In fact, CXCR4 mutations have been detected in 30%–40% of patients with WM and have been shown to impact ibrutinib response and survival outcomes. 25 Specifically, patients with CXCR4 mutations treated with ibrutinib have exhibited a delayed time to response, lower rates of VGPR and shorter PFS rates, with the magnitude of this impact depending on the mutation subtype. 25 However, the number of patients with CXCR4 mutation analyzed in our cohort was very small due to the limited use of the mutation testing in clinical practice in our country at the time the patients were treated. This precluded us from analyzing its impact in effectiveness outcomes and limited the generalizability of the results in patients harboring CXCR4 mutations. Therefore, future studies should further investigate this aspect in real-world settings in Spain, and potentially consider combination treatment strategies that may help mitigate the effect of those mutations.
Regarding ibrutinib-related toxicities, treatment was generally well tolerated, with most AEs being grades 1–2 and no new safety signal being reported. The most common AEs were bleeding, diarrhea, infections, and atrial fibrillation, consistent with the findings from other real-world cohorts,14–16 and clinical trials.10,23,26 Notably, we observed a lower frequency of infections compared to the pivotal trial, 23 but a comparable frequency to that reported by Cencini et al., 14 which may also be influenced by limitations in reporting low-grade infections in retrospective studies. In addition, the frequency of low-grade atrial fibrillation observed in our cohort was higher than the one documented in the pivotal trial, 23 mirroring the findings from other real-world cohorts. 16 This could indicate that low-grade atrial fibrillation is more frequently reported in the real-world, possibly because patients are often in a vulnerable state, typically older and with multiple other health issues. Moreover, it also may be due to the awareness of the clinicians prescribing ibrutinib, who may be prone to detect low-grade AF faster in their current practice. Furthermore, preexisting cardiovascular comorbidities at the initiation of ibrutinib treatment may have contributed to the observed incidence of atrial fibrillation in our cohort. The proportion of patients in our cohort requiring dose reductions was comparable to that reported by Abeykoon et al., 16 but lower than the rates observed in other recent real-world studies, which reported a dose reduction rate of 27%.15,14 This discrepancy might reflect differences in the approaches to managing AEs in clinical practice.
Our study provides important insights into the real-world use of ibrutinib for WM in Spain; however, it has some limitations. As in most retrospective studies, there was missing data potentially affecting the robustness of certain variables. Moreover, the retrospective design inherently limits the control over data collection and can introduce potential biases, such as selection and reporting bias. Furthermore, the sample size is limited, which may have influenced some of the outcomes, including the identification of factors associated with survival outcomes. However, this study contributes to the characterization of population with WM receiving ibrutinib and their corresponding outcomes in a real-world setting in a different region of Europe, as only one real-world study was previously conducted in Italy, 14 while the others were conducted in the United States15–17 and Latin America. 13 Additionally, our findings support the effectiveness of this treatment in clinical practice, as previously described, and demonstrate a manageable toxicity profile, highlighting the low frequency of infections and higher frequency of atrial fibrillation in clinical practice.
Regarding future perspectives, several treatment gaps require further investigation, such as the optimal sequencing of therapies in WM, including combination regimens and BTKis, as well as the management of specific patient subgroups, such as the elderly, high-risk patients, those with refractory disease, or those with associated amyloidosis, which still represent challenging populations in terms of optimal treatment and management.
Conclusion
This study provides real-world evidence on the characteristics of patients with WM treated with ibrutinib in Spain, highlighting their commonly vulnerable status, often marked by comorbidities. Additionally, our findings show that the effectiveness of ibrutinib, as evidenced by treatment responses and survival outcomes, is favorable and its toxicity profile is manageable in everyday clinical practice in patients with WM.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251379670 – Supplemental material for Observational retrospective study of the treatment of Waldenström’s macroglobulinemia with ibrutinib in routine clinical practice in Spain
Supplemental material, sj-docx-1-tah-10.1177_20406207251379670 for Observational retrospective study of the treatment of Waldenström’s macroglobulinemia with ibrutinib in routine clinical practice in Spain by Carlos Fernández de Larrea, Javier Loscertales, Valentín Cabañas, Carmen Freiria, María Jesús Blanchard, Elham Askari, Marcos Lorenzo Pérez, Laura Abril, Jordi López Pardo, Sergio Pinzón, Javier Díaz Gálvez, Neus Amer, Ángel Ramírez Páyer, Ricarda García Sánchez, María Magdalena Alcalá, Francisco Javier Capote, David Moreno, Celia Bolumburu, Eva Rubio-Azpeitia and Ramón García Sanz in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251379670 – Supplemental material for Observational retrospective study of the treatment of Waldenström’s macroglobulinemia with ibrutinib in routine clinical practice in Spain
Supplemental material, sj-docx-2-tah-10.1177_20406207251379670 for Observational retrospective study of the treatment of Waldenström’s macroglobulinemia with ibrutinib in routine clinical practice in Spain by Carlos Fernández de Larrea, Javier Loscertales, Valentín Cabañas, Carmen Freiria, María Jesús Blanchard, Elham Askari, Marcos Lorenzo Pérez, Laura Abril, Jordi López Pardo, Sergio Pinzón, Javier Díaz Gálvez, Neus Amer, Ángel Ramírez Páyer, Ricarda García Sánchez, María Magdalena Alcalá, Francisco Javier Capote, David Moreno, Celia Bolumburu, Eva Rubio-Azpeitia and Ramón García Sanz in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
We would like to express our gratitude to all the participating centers, patients, and their families for their invaluable contributions to this research. We are grateful to the data entry personnel for ensuring the accuracy and quality of our results. We also acknowledge the significant contributions of researchers not listed as authors, whose dedication has been essential. Medical writing and editorial assistance were provided by Javier Rodríguez Cobos at Evidenze Health España S.L.U. Responsibility for opinions, conclusions, and interpretation of data lies with the authors. Finally, we extend our thanks to Johnson & Johnson Innovative Medicine for their funding, support, and collaboration in this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
