Abstract
Chronic graft-versus-host disease (cGVHD) is a potentially life-threatening complication of hematopoietic stem cell transplantation (HSCT). The global incidence of cGVHD remains high, despite prophylactic regimens for patients undergoing HSCT. Systemic corticosteroids are the standard first-line treatment for cGVHD, but most treated individuals will require second-line or further treatment because of suboptimal disease control or toxicity from long-term corticosteroid use. No standard treatment algorithm exists for steroid-refractory cGVHD, with similar response rates and poor outcomes across different pharmacologic regimens. The pathogenesis of cGVHD is driven by an imbalance in effector T helper 17/T follicular helper cells and regulatory T-cells that leads to an inability to reverse a proinflammatory environment and the loss of immune tolerance, which causes irreversible fibrosis in target organs. Rho-associated coiled-coil–containing protein kinase 2 (ROCK2) signaling plays key roles in regulating T-cell–mediated immune responses, promoting the differentiation of fibroblasts into myofibroblasts, and stimulating transforming growth factor β-induced fibrosis. Belumosudil, an oral selective ROCK2 inhibitor, targets the ROCK2 pathway, thus correcting disrupted immune homeostasis, downregulating proinflammatory cytokines, and reversing fibrosis in cGVHD. Clinical trials demonstrated rapid and sustained overall response with belumosudil, and a favorable adverse effects profile with a low risk of infection and cytopenia in pretreated and multiorgan involved cGVHD patients, resulting in its approval for use in cGVHD. This narrative review provides an overview of the pathophysiology of cGVHD and the limitations of current treatment, and describes the pharmacologic activity, clinical studies, and real-world data supporting belumosudil use in patients with cGVHD.
Introduction
Chronic graft-versus-host disease (cGVHD) is a potentially life-threatening complication among individuals who have undergone hematopoietic stem cell transplantation (HSCT). 1 The 3-year cumulative incidence of cGVHD post-HSCT is 42% in the United States 2 and 53.3%–57.9% in China.3,4 In the United States, diagnosis of cGVHD most commonly occurs 100–200 days post-transplantation. 2
The development of cGVHD is driven by multiple interactions between dysregulated and alloreactive T- and B-cells, neutrophils, macrophages, and dendritic cells that lead to an abnormal immune response and fibrosis. 1 The pathogenesis of cGVHD is caused by dysregulation of the Rho-associated coiled-coil–containing protein kinase 2 (ROCK2) signaling pathway.5,6 The clinical manifestations of cGVHD include chronic inflammatory changes (phase I), followed by dysregulation of immune cells (phase II), and ultimately progression to aberrant tissue repair and significant fibrosis (phase III), either in a single organ or throughout the body.1,7
The development of nonreversible fibrosis during the third phase of cGVHD is associated with substantial morbidity and has a negative impact on physical functioning.8–10 Fibrosis may present as deep cutaneous sclerosis or as ocular or pulmonary manifestations. 9 For example, patients with oral cGVHD develop severe sclerotic fibrosis of the perioral tissue that can restrict mouth opening, leading to oral hygiene issues and eating difficulties with subsequent development of oral infections and malnutrition.11,12 Ocular tissue fibrosis may develop in 60%–90% of patients with cGVHD, including subconjunctival fibrosis, which contributes to dry eye and ocular surface damage. 13 Moreover, patients with bronchiolitis obliterans syndrome (BOS), the most common pulmonary form of cGVHD, develop progressive lung fibrosis that ultimately leads to severe restrictive lung disease and interstitial fibrosis with irreversible loss of function. 1 Furthermore, pulmonary and gastrointestinal fibrosis can have a significant impact on mortality. 9
Conventional agents used in the first- and second-line treatment of cGVHD most commonly consist of systemic corticosteroids, calcineurin inhibitors, mycophenolate mofetil, and sirolimus.14–16 These immunosuppressive agents generally provide only suboptimal disease control, with ⩾70% of individuals becoming steroid-dependent or -refractory over time.2,14,15,17,18 Selection of second-line treatment for steroid-refractory cGVHD is a shared treatment decision based on published literature, physician experience, and the patient’s values/preferences. 19 Most of the current first-line pharmacotherapies for cGVHD are unable to directly address cGVHD-related fibrosis without compromising immune function and increasing the risk of severe and/or fatal infections.15,20 Therefore, new treatments that focus on the prevention or reversal of fibrosis in patients with steroid-refractory cGVHD are expected to improve the potential for development of effective individualized treatment regimens.
More recently, second-line targeted treatment options have become available for patients with steroid-refractory cGVHD, including ruxolitinib (a selective Janus kinase (JAK) 1/2 inhibitor), ibrutinib (a selective and irreversible Bruton tyrosine kinase (BTK) inhibitor), and belumosudil (a selective ROCK2 inhibitor), all of which have improved response rates and safety profiles compared with conventional agents.17,21 This has led to a new era of cGVHD treatment; however, in the absence of data from large-scale, randomized clinical studies, there is no consensus around the standard second-line treatment of cGVHD. Indeed, a 2024 expert consensus report on the diagnosis and treatment of cGVHD in China suggested that selection of second-line treatment should be based on the cGVHD organ involvement. 22 Consequently, the approach to second-line treatment varies considerably by treatment center. 23
Second-line targeted agents have different mechanisms of action. Ruxolitinib and ibrutinib each have a single mechanism of action. JAK1/2 inhibition with ruxolitinib blocks cytokine receptor-mediated signaling for several proinflammatory cytokines, which leads to suppression of T-cell activation and a downstream reduction in tissue fibrosis. 17 Ibrutinib acts via inhibition of the BTK protein, a nonreceptor tyrosine kinase involved in B-cell receptor signaling and B- and T-cell activation. 24 The ROCK2 inhibitor belumosudil has a unique, dual mechanism of action, through which it both downregulates proinflammatory responses and inhibits aberrant tissue fibrosis. 25 In preclinical studies, belumosudil reduced or reversed fibrosis in animal models of cGVHD or fibrosis-related disorders.26–28 Belumosudil has also demonstrated promising efficacy and good tolerability in phase II and III trials in patients with treatment-refractory cGVHD,15,29,30 and is indicated for steroid-refractory cGVHD in patients aged ⩾12 years in the United States, 31 China, 32 and other countries or regions.
In this narrative review, we provide an overview of the role of ROCK2 in immune response and fibrosis in cGVHD, and comprehensively describe the dual mechanism of action and pharmacologic activity of belumosudil, as well as clinical and real-world data supporting its use in patients with cGVHD.
Role of ROCK2 in regulating immune responses and fibrotic processes
The ROCK2 signaling pathway regulates the cellular processes responsible for abnormal immune responses and fibrotic processes; therefore, it has several key roles in cGVHD pathophysiology (Figure 1).
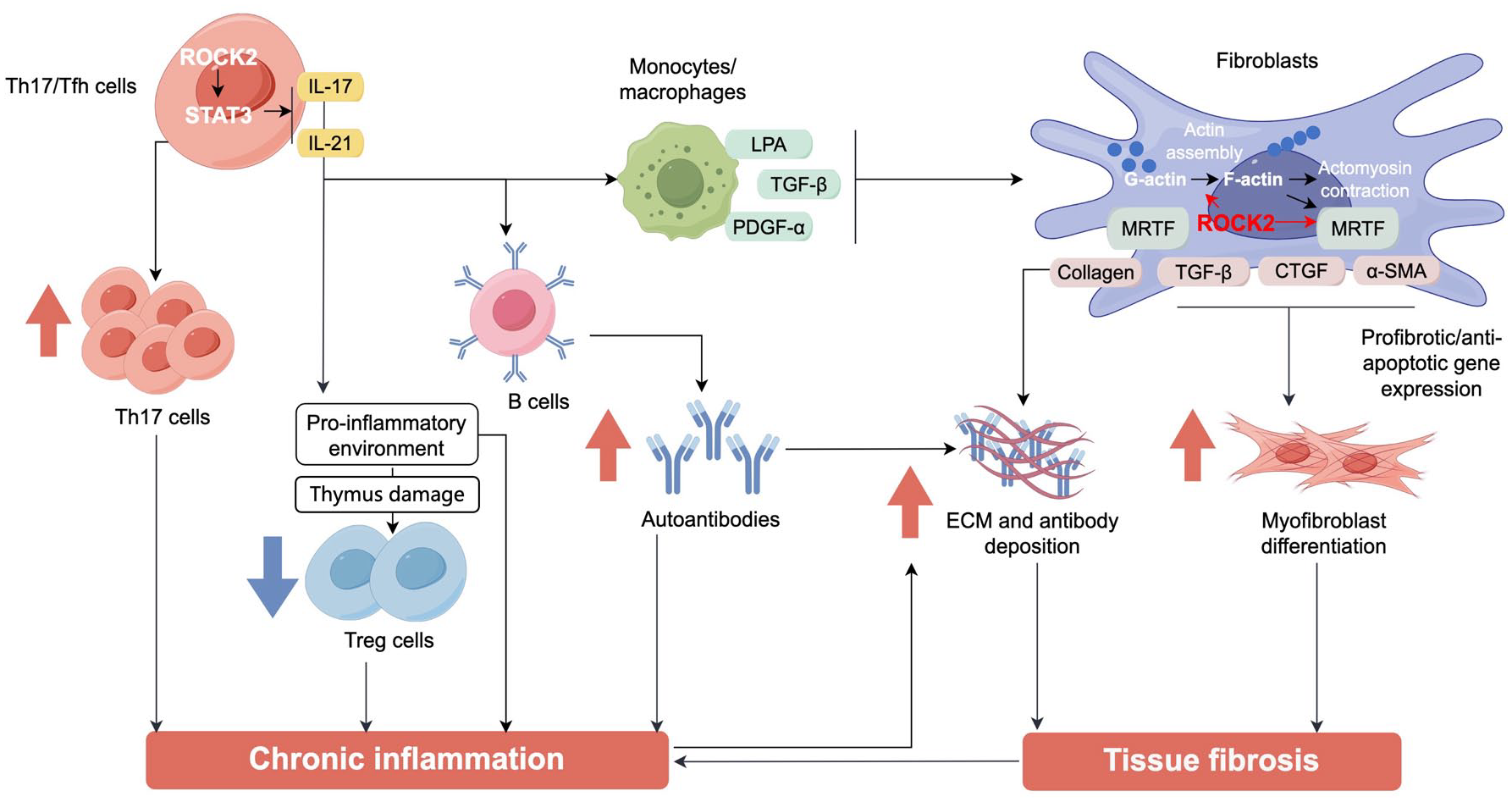
Overview of the ROCK2 signaling pathway and its role in regulating immune responses and fibrotic processes in chronic graft-versus-host disease. During an inflammatory immune response, ROCK2 activation stimulates secretion of the proinflammatory cytokines IL-17 and IL-21 from Th17/Tfh cells via phosphorylation of STAT3. IL-17 and IL-21 cause imbalanced activation of B-cells and monocytes/macrophages that leads to excessive production of autoantibodies and secretion of profibrotic factors (e.g., LPA, TGF-β, and PDGF-α), respectively. The proinflammatory environment (i.e., IL-17 and IL-21) causes downregulation of Treg cells, thereby sustaining chronic inflammation. Profibrotic factors also activate ROCK2 in fibroblasts, promoting polymerization of G-actin to F-actin and contractile fiber formation. This leads to release of MRTF, which causes increased expression of collagen, α-SMA, CTGF, and TGF-β. This results in increased ECM deposition and stimulates fibroblast differentiation into myofibroblasts, thereby causing target organ fibrosis, which further contributes to sustained chronic inflammation.
ROCK signaling plays a key role in T-cell–mediated immune responses, with the ROCK2 isoform being involved in maintaining the balance between proinflammatory and regulatory T (Treg) cell subsets. 6 The pathogenesis of cGVHD is driven by an imbalance in effector T helper 17 (Th17)/T follicular helper (Tfh) cells and Treg cells that leads to an inability to reverse a proinflammatory environment and the loss of immune tolerance. 25 ROCK2 is responsible for the balance between proinflammatory and Treg cell subsets, which can cooperate to induce the secretion of autoantibodies. These autoantibodies are then deposited in tissues and trigger a fibrotic reaction. 6 In Th17 cells, ROCK2 stimulates interleukin (IL)-17 and IL-21 production via phosphorylation of interferon regulatory factor 4 (IRF4). 33 These cytokines subsequently stimulate recruitment and differentiation of innate immune cells into pathogenic phenotypes, driving sustained chronic inflammation. 25
The ROCK2 pathway also drives pathologic remodeling of the extracellular matrix, with the abnormal deposition and/or orientation of collagen and fibronectin. The ROCK2-mediated release of IL-17 and IL-21 also leads to the activation of myofibroblasts by transforming growth factor-β (TGF-β) and subsequent collagen deposition in target organs. 25 ROCK2 inhibition has been shown to ameliorate this abnormal organization and improve macromolecular permeability of fibrotic tissues in a three-dimensional cell culture model of the fibrotic pancreatic tumor microenvironment. 34 In addition, activated ROCK2 phosphorylates c-Jun N-terminal kinases, extracellular signal-regulated kinases, and nuclear factor-kappa B, thereby facilitating activation of several profibrotic genes, including connective tissue growth factor (CTGF), fibronectin 1, collagen type I-α2, and collagen type IV-α1. 5 These profibrotic mediators promote differentiation of fibroblasts into myofibroblasts and increase collagen production. 5 Another role of ROCK2 in fibrosis is the regulation of monocyte chemoattractant protein-1 (MCP-1)–induced monocyte recruitment via promotion of lysophosphatidic acid-induced chemotaxis and E-selectin–induced adhesion of endothelial cells. 35 Furthermore, ROCK2 activity stimulates phagocytosis of fibronectin by fibroblasts and regulates cortical contractility. 36
Given the key roles of ROCK2 signaling in cGVHD, targeting this pathway is a critical component of any therapeutic strategy designed to modify disrupted immune homeostasis or aberrant fibrosis. 25
ROCK2 inhibition with belumosudil
Dual mechanism of action
Selective ROCK2 inhibition with belumosudil restores immune homeostasis between Th17 and Treg cells by downregulating the activation/phosphorylation of signal transducer and activator of transcription 3 (STAT3) and upregulating phosphorylation of STAT5.26,37 The downregulation of STAT3 phosphorylation inhibits formation of the JAK2/STAT3 complex in Th17 and Tfh cells. Inhibition of the JAK2/STAT3 complex decreases activation of several transcription factors, such as IRF4, retinoic acid receptor-related orphan receptor gamma (RORγt), and B-cell lymphoma 6 (Bcl6), and triggers significant downregulation of both Th17 and Tfh cells. 25 This shifts the balance between Th17 and Treg cells toward the Treg phenotype, inducing an immunomodulatory effect and decreasing chronic inflammation. 37
Belumosudil downregulates and reverses fibrosis via several molecular mechanisms: (1) inhibition of actin polymerization; (2) proinflammatory cytokine suppression; and (3) inhibition of profibrotic gene expression and TGF-β signaling. 25 Inhibition of actin polymerization (from G-actin to F-actin) reduces the development of contractile fibers, while suppression of proinflammatory cytokines (e.g., IL-17, IL-21) reduces the production of autoantibodies and profibrotic factors (e.g., α-smooth muscle actin, CTGF, and TGF-β). The inhibition of profibrotic gene expression and TGF-β signaling suppresses myocardin-related transcription factor-mediated transcription and blocks fibroblast differentiation into myofibroblasts and collagen production. 25
In contrast to the dual mechanism of action of belumosudil, ruxolitinib and ibrutinib each have a single mechanism of action. Ruxolitinib is an inhibitor of JAK1/2, which plays a key role in cytokine signaling and the initiation and progression of inflammation. 38 JAK1/2 inhibition via ruxolitinib is associated with a reduction in proinflammatory cytokines and decreased cell migration. The selective and irreversible BTK inhibitor ibrutinib acts via suppression of B-cell receptor signaling to inhibit B-cell proliferation and expression of co-stimulatory molecules. 38 Although ruxolitinib and ibrutinib have been used more extensively than belumosudil, its dual mechanism of action, may allow for improved outcomes with belumosudil among patients with cGVHD.
In vitro and preclinical data
In a series of studies involving human peripheral blood mononuclear cells from patients with active cGVHD, belumosudil decreased Tfh cells and reduced production of IL-21, IL-17, and interferon-γ. 26 In isolated human T-cells, belumosudil significantly reduced STAT3 phosphorylation, IRF4 production, and nuclear hormone RORγt protein levels, and blocked STAT3 binding to IL-17 and IL-21 promoters. 37
Preclinical studies have demonstrated that ROCK2 inhibition with belumosudil can ameliorate the pathology associated with cGVHD and other immune-mediated disorders by restoring immune homeostasis.26,28,37,39,40 In mice with collagen-induced arthritis, belumosudil markedly reduced the progression of inflammatory arthritis by targeting the Th17-mediated pathway, with an associated improvement in clinical arthritis scores. 37 Similarly, in the MRL/lpr mouse model of systemic lupus erythematosus, belumosudil effectively prevented kidney damage and disease progression, by reducing STAT3 phosphorylation and transcription regulator Bcl6 levels, and increasing STAT5 phosphorylation in the spleen; concurrent improvements were recorded in both histological and clinical scores. 39 Belumosudil markedly reduced intestinal mucosal T-cell activation and proinflammatory cytokine production in a mouse model of inflammatory bowel disease, which correlated with improvements in clinical and pathological scores. 40 Moreover, belumosudil administration induced a pronounced reduction in immune cell infiltrates and downregulated RORγt and Bcl6 expression in a thioacetamide-induced liver fibrosis mouse model that correlated with a significant reduction in IL-17 in the liver, splenic germinal center formation, and serum immunoglobulin G. 28 Regarding cGVHD, belumosudil effectively decreased the presence and activity of Tfh and Th17 cells, along with an increase in Treg cells, through a Stat3-dependent mechanism in murine models of cGVHD, including BOS and sclerodermatous cGVHD, establishing a restoration in the immune response. 26
Belumosudil has also been shown to reduce or reverse fibrosis in preclinical studies.27,28,41 For example, in the bleomycin-induced pulmonary fibrosis Fischer rat model, belumosudil inhibited glycolysis and endothelial cell migration, stimulated oxidative phosphorylation, and strengthened barrier integrity in rat pulmonary microvascular endothelial cells in vitro, and strengthened baseline rat lung barrier integrity independent of bleomycin injury. 27 These findings indicated that belumosudil has barrier-enhancing effects on pulmonary endothelium in vivo. 27 Belumosudil has also been shown to effectively mitigate cardiac dysfunction, fibrosis, and hypertrophy in a mouse model of transverse aortic constriction. 41 Using the thioacetamide-induced liver fibrosis mouse model, other research has shown that belumosudil prevents and reverses liver fibrosis by inhibiting macrophage function by disrupting STAT3 phosphorylation and cofilin signaling pathways, inhibiting proinflammatory cytokine production, and blocking macrophage migration. 28 In the BOS mouse model, belumosudil administration was associated with a significant decrease in lung fibrosis, with normalization of pulmonary function. 26 These functional improvements were correlated with a marked reduction in antibody and collagen deposition in the lungs of treated mice to levels comparable with non-cGVHD controls. 26
Clinical studies of belumosudil
Efficacy
Phase II and III clinical studies have assessed the efficacy and safety of belumosudil in patients with steroid-dependent and/or -refractory cGVHD (Table 1).15,29,30,42
Summary of outcomes from key clinical trials of belumosudil, ruxolitinib, and ibrutinib treatment in patients with chronic graft-versus-host disease who had received ⩾1 prior line of therapy.

Best organ-specific ORR for belumosudil and ibrutinib, organ-specific ORR at Week 24 for ruxolitinib and best available treatment.
Grade ⩾3 AEs reported in ⩾5% of participants.
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BID, twice daily; cGVHD, chronic graft-versus-host disease; CI, confidence interval; CNIs, calcineurin inhibitors; FFS, failure-free survival; GGT, γ-glutamyl transferase; GI, gastrointestinal; N/A, not available; ORR, overall response rate; QD, once daily; SAE, serious adverse event.
A phase IIa dose-finding, open-label study was conducted in US patients with cGVHD who had received a median of three prior lines of systemic treatment. 29 Over a median follow-up of 29 months, participants had an overall response rate (ORR) of 65%, 69%, and 62% with belumosudil 200 mg once daily, 200 mg twice daily, and 400 mg once daily, respectively.
The failure-free survival (FFS) rate was 76% at 6 months and 47% at 12 months, and the 2-year overall survival (OS) rate was 82%. During belumosudil treatment, 67% of participants reduced their corticosteroid dose and 19% successfully discontinued corticosteroid therapy. This dose-finding study determined that belumosudil dosages of 200 mg once daily or 200 mg twice daily were effective and safe in patients with treatment-refractory cGVHD. 29
In the phase II, randomized ROCKstar study in US patients with persistent cGVHD after a median of three prior lines of systemic treatment, the ORR for belumosudil 200 mg once daily (n = 66) and 200 mg twice daily (n = 66) in the 12-month analysis was 74% and 77%, respectively, with all affected organs showing complete response (CR). 15 Median duration of response (DOR) was 54 weeks, and 59% and 62% of participants in the respective dose cohorts reported a clinically meaningful improvement (i.e., ⩾7-point reduction) in the 7-day Lee Symptom Scale (LSS) score. The overall FFS rate was 75% at 6 months and 56% at 12 months, the 12-month OS rate was 89%, and the rates of relapse and non-relapse mortality were low (3% and 7%, respectively). 15 Of note, a subsequent combined analysis of data from the phase IIa and ROCKstar studies indicated that the risk of treatment failure with belumosudil was increased in participants with progressive cGVHD at baseline. 45 Indeed, when ORR was assessed by duration of cGVHD in ROCKstar, the ORR with belumosudil was greater in participants with shorter (⩽50th percentile) versus longer (>50th percentile) duration of disease (83% vs 68%). 15 During belumosudil treatment, 65% of participants reduced their corticosteroid dose; the mean corticosteroid dose was reduced by 45% in the modified intent-to-treat cohort and by 54% in responders. 15 In addition, 21% of participants discontinued corticosteroid therapy, while 22%, 20%, and 21% of participants discontinued calcineurin inhibitor therapy, sirolimus, and mycophenolate mofetil, respectively. 15 Preliminary data from a 3-year follow-up of this study indicate that belumosudil provided sustained response and good tolerability with long-term treatment. 46 The 3-year ORR in the modified intent-to-treat population, which included an additional 20 patients, was similar to that reported in the 12-month primary analysis (74% for belumosudil 200 mg once daily (n = 77) and 76% for belumosudil 200 mg twice daily (n = 75)). Moreover, organ-specific ORRs were also consistent with the primary analysis. The 3-year FFS rate was 44% in the overall population and 52% for responders. No new safety concerns were identified. After ⩾36 months’ follow-up, belumosudil treatment was ongoing in 8% of participants in the 200 mg once daily arm and 7% of those in the 200 mg twice daily arm; 23% and 25% of participants, respectively, had successfully discontinued all immunosuppressants. 46
An open-label phase II study in China in patients with cGVHD, who had received a median of three prior lines of systemic treatment, reported an ORR of 73% with belumosudil 200 mg once daily (median follow-up 12.9 months), with all responders achieving partial response (PR). 30 Median time to response (TTR) was 4.3 weeks and the median DOR was not reached. Clinically meaningful improvement in symptoms (i.e., ⩾7-point reduction in LSS score) was reported by 15 participants (50%), with a mean duration of 16 weeks. Dose reductions for corticosteroids and calcineurin inhibitors were reported in 57% and 35% of participants, respectively; the proportion of participants with a corticosteroid dose reduction was higher among responders than in non-responders (64% vs 38%). 30
Data from an open-label phase III study in Japanese patients with steroid-dependent or -refractory cGVHD (median of one prior line of systemic treatment, median of two organs involved) reported that belumosudil 200 mg once daily was associated with a best ORR of 85.7% at 24 weeks after enrolment of the last patient, with all responders having PR (median follow-up 9.9 months). 42 At 24 weeks, the median TTR was 4.1 weeks and the best ORR was 80.0% in the joints and fascia, 66.7% in the mouth, 54.5% in the skin, and 20.0% in the eyes. However, none of the six patients with lung involvement had CR or PR during the observation period (five had stable disease and one had progressive disease). Clinically meaningful symptom improvement and corticosteroid dose reduction were each reported in 12 participants (57.1%). The median maximum percent change in corticosteroid dose was −23.3%, but discontinuation of corticosteroids or calcineurin inhibitors was not reported. 42
A pooled analysis of data from the phase IIa 29 and ROCKstar 15 studies demonstrated a strong positive correlation between clinical organ response to belumosudil and improvements in patient-reported outcomes (PROs). 47 Among participants with BOS from the phase IIa 29 or ROCKstar 15 studies, belumosudil was associated with lung-specific clinical responses, and the ORR was higher in those with less advanced disease. 48 The ORR was 37% for participants with BOS and a clinically meaningful symptom improvement (i.e., ⩾10-point reduction in lung-specific LSS score) was achieved in 68% of participants.48,49
Although no head-to-head studies between second-line belumosudil and ruxolitinib or ibrutinib have been conducted, an indirect comparison of their key clinical studies suggests that the best ORR is similar in patients with steroid-dependent or -refractory cGVHD (Table 1).23,43,44
In the phase III REACH3 study of ruxolitinib in patients with moderate or severe steroid-refractory or -dependent cGVHD who had failed prior treatment with corticosteroids (± calcineurin inhibitors), the best ORR was 76.4% with ruxolitinib 10 mg twice daily versus 60.4% with best available treatment at Week 24 (median follow-up 57.3 weeks). 23 Ruxolitinib was associated with longer FFS than control treatment (median FFS >18.6 vs 5.7 months), with an 6-month FFS rate of 74.9% versus 44.5%. OS data were not mature; the estimated 12-month OS rate was 81.4% with ruxolitinib versus 83.8% with control treatment. 23
In the single-arm phase Ib/2 PCYC-1129 study of ibrutinib in patients with steroid-dependent or -refractory cGVHD (median of two prior lines of systemic treatment, ⩾2 organs involved in 85% of participants), 44 ibrutinib 410 mg once daily was associated with a best ORR of 69% after a median follow-up of 26 months. 43 The FFS rate was 51% at 18 months and the estimated OS rate at 24 months was 71%. There were clinically meaningful improvements in LSS score in 43% of participants and 64% had decreased their corticosteroid dose to <0.15 mg/kg/day. 43
Of note, belumosudil was associated with promising efficacy (best ORR >70%) in the aforementioned phase II studies, despite the participants being heavily pretreated (median of three prior lines of systemic treatment) and having advanced disease (50%–53% of patients had ⩾4 organs/systems involvement).15,29,30 In contrast, clinical studies of ibrutinib and ruxolitinib reported similar best ORRs (69% and 76%, respectively) among patients with less advanced cGVHD and limited organ involvement.23,43
With regard to organ-specific response, the ROCKstar study reported high response rates with belumosudil in joint/fascia (71%), lower gastrointestinal tract (69%), and upper gastrointestinal tract (52%), as well as a promising ORR of 26% in the lungs, the most difficult organ to treat (Table 1). 15 The promising response rate in the lungs was most likely because of its dual mechanism of action on immune responses and fibrotic processes. 25 There is little evidence for the efficacy of ibrutinib in pulmonary cGVHD. In a retrospective study of ruxolitinib treatment in patients with less advanced BOS, patients with early-stage inflammation were more likely to achieve treatment response than those with late-stage fibrosis due to the anti-inflammatory effects of ruxolitinib; however, reversal of BOS in patients with a rapid decline in lung function was not observed. 50
Safety
Belumosudil was well-tolerated in these clinical studies, with adverse events (AEs) being consistent with those expected among patients with advanced steroid-dependent or -refractory cGVHD.15,29,30 The frequency of cytomegalovirus (CMV) infection remained low, and most reported infections were considered unrelated to belumosudil.15,29,30 Moreover, these phase II studies reported low rates of grade 3–4 hematologic AEs.15,29,30 Indeed, the European Society for Blood and Marrow Transplantation (EBMT) consensus guidelines consider belumosudil as a potential treatment option, particularly among patients with an increased risk of infection or cytopenia, because of its low associated risk of toxicity. 21
As belumosudil is an immunomodulatory drug, rather than an immunosuppressant, the incidence of infections and hematologic toxicities in the phase II studies was low.15,29,30 In contrast, clinical studies reported a high incidence of infections with ibrutinib43,44,51 and ruxolitinib.23,52,53 Ibrutinib was associated with a high risk of invasive fungal infections, 51 possibly caused by the ibrutinib-induced loss of multiple neutrophil functions that lead to an impaired antifungal immune response. 54 Ruxolitinib was associated with CMV reactivation, mainly due to its immunosuppressive effects in the bone marrow. 55 Severe anemia and thrombocytopenia were also reported during ruxolitinib treatment,23,52,53 most likely because of its selective JAK1/JAK2 inhibitory mechanism, which indirectly suppresses STAT-mediated signaling of cytokines and growth factors involved in hematopoiesis and immune function (Table 1). 56
Real-world evidence of belumosudil use in cGVHD
In real-world studies, patients with steroid-dependent or -refractory cGVHD often have more complex and varied disease than those treated in clinical studies. Preliminary data from one Canadian and five US retrospective studies indicate that belumosudil is an effective treatment option and well-tolerated by patients with advanced cGVHD treated in routine clinical practice including those previously treated with ruxolitinib.57–63
The Canadian multicenter study reviewed the medical records of 35 patients with treatment-refractory cGVHD who had started belumosudil treatment. 57 Of the 25 patients evaluable for response after ⩾3 months, all had moderate-to-severe cGVHD, with a median of three organs involved, and had received a median of five prior lines of therapy. Belumosudil was predominantly combined with corticosteroid therapy (72% of patients) or ruxolitinib (44%). After a median follow-up of 4.8 months, add-on belumosudil was associated with an ORR of 28% at 3 months and 31.3% at 6 months; corresponding clinical benefit rates were 64% and 52%. At 6 months, the FFS rate was 72% and the OS rate was 77%. Following belumosudil initiation, the corticosteroid dose was markedly reduced in 40% of patients and no grade ⩾3 AEs were reported. Of the five patients who discontinued belumosudil treatment, the reasons for discontinuation were intolerance (muscle spasm and increased transaminases; n = 2), additional cGVHD treatment required (n = 2), and death due to a pre-existing infection (n = 1). 57
Further to the effectiveness of belumosudil in heavily pretreated patients with advanced cGVHD, as demonstrated in the abovementioned Canadian study, there are also data indicating that it is effective in patients with fewer lines of therapy. Several US real-world studies have reported a marked improvement in ORR among patients with a median of three prior lines of treatment. In a US single-center study of 45 patients with treatment-refractory cGVHD, who had received a median of three prior lines of therapy (including ruxolitinib), approximately half (49%) of the patient population received concomitant ruxolitinib and belumosudil. 58 At 12 months, the ORR was 47%, including CR in 7% and PR in 40%; the 1-year FFS rate was 46% and corticosteroid dose was reduced in 29% of patients. 58 A single-center chart review described belumosudil treatment in 26 patients with steroid-refractory or -dependent cGVHD, who had received a median of three prior lines of therapy. 59 In this study, belumosudil was associated with an ORR of 77% (median follow-up 213.5 days), with response reported in 85%, 53%, 33%, 30%, 13%, and 11% of participants with skin, joint/fascia, eye, pulmonary, liver, and mouth cGVHD, respectively. 59 In another single-center study of 66 adults with steroid-refractory cGVHD, 85% of whom had moderate-to-severe cGVHD with multiorgan involvement, most had previously received ruxolitinib before starting belumosudil. 60 Belumosudil was associated with a 12-month ORR, FFS rate, and OS rate of 64%, 72%, and 92%, respectively, with no reported grade ⩾3 AEs. 60
A multicenter study described the effectiveness and safety of belumosudil in combination with ruxolitinib in 14 patients with steroid-refractory or -dependent cGVHD. 61 At the start of belumosudil treatment, the ruxolitinib dosage was 10 mg twice daily in eight patients, 5 mg twice daily in five patients, and 5 mg once daily in one patient. Most participants had severe cGVHD (median of four organs involved) and participants were receiving a median of 1.5 additional immunosuppressive therapies, including prednisone, sirolimus, tacrolimus, mycophenolate mofetil, and extracorporeal photopheresis. Belumosudil was commenced at 200 mg once daily; the dose was 200 mg twice daily for participants on proton-pump inhibitor therapy. Concomitant treatment with belumosudil and ruxolitinib resulted in PR in approximately 43% of patients and stable disease in 50% (median follow-up 10.7 months), with an accompanying reduction in prednisone dose in several patients. After starting belumosudil, the ruxolitinib dose was reduced by 50% in eight patients (57%) and eventually discontinued in four patients (29%). The belumosudil dose was reduced in four patients due to worsening renal function, fatigue, and rash. AEs were consistent with those expected for patients with cGVHD receiving immunosuppressive therapy. 61
Combination therapy with belumosudil and ruxolitinib has also been reported in another single-center study of patients with steroid-refractory cGVHD after ruxolitinib treatment failure, most of whom had multiorgan involvement. 62 Preliminary data from this study indicated that belumosudil plus ruxolitinib was well-tolerated and provided a robust treatment response, with a 6-month ORR of 71% that was maintained at 12 months (70%). 62
One single-center US study described the effectiveness and safety of belumosudil treatment in eight adolescents and young adults (aged 12–20 years) with severe cGVHD and multiorgan involvement. 63 Seven of the study participants experienced significant improvements in cGVHD symptoms with belumosudil; the remaining patient had recently started therapy and could not be assessed. All study participants tolerated belumosudil well according to liver enzyme and renal function test results, without any major drug interactions. Of the four patients with elevated liver enzymes at the start of belumosudil treatment, all showed improvement during therapy. One patient successfully discontinued belumosudil after resolution of cGVHD. These findings indicated that belumosudil may be effective and safe for use in adolescents and young adults with cGVHD. 63
Regarding the real-world use of other second-line therapies, a retrospective study of ruxolitinib treatment in Chinese patients with steroid-refractory cGVHD (median of two prior lines of systemic treatment, ⩾3 organs involved in 25.0% of patients) reported that ruxolitinib was associated with a best ORR of 65.0% in those with severe cGVHD. 64 In a real-world US study of ibrutinib in patients with steroid-refractory cGVHD (as third-line or later systemic treatment in 72% of patients), the best ORR was 56%. 65 Real-world studies of ruxolitinib or ibrutinib in heavily pretreated patients have reported 6-month ORRs of 55% and 45%, respectively.65,66
Future directions
Currently, patients with moderate-to-severe cGVHD typically receive first-line treatment with corticosteroids plus a calcineurin inhibitor and/or sirolimus; however, <20% of these patients achieve CR or durable PR within 1 year. 67 According to consensus-based 2024 guidelines from the EBMT 21 and the Cell Therapy Transplant Canada (CTTC), 68 future studies on the treatment of cGVHD should focus on development of first-line steroid-free regimens. A survey of EBMT centers found that 51.9% of centers were using first-line steroid-free treatment in this setting. 69 Both belumosudil and ruxolitinib are considered to be promising agents for the first-line treatment of cGVHD as they are expected to minimize steroid exposure, prevent disability, and improve OS and quality of life (QoL). 17 A study in Chinese patients with cGVHD reported more impaired QoL in patients with more severe disease, with the overall symptom burden (including oral symptoms) and the severity of ocular GVHD having the greatest impact on QoL. 70 However, no studies to date have reported the use of belumosudil as first-line systemic treatment in patients with moderate or severe cGVHD. In addition, belumosudil resistance could potentially develop with long-term use, as previously reported with ruxolitinib,71,72 although resistance to belumosudil treatment has not yet been reported in the literature. Further research is warranted to determine potential mechanisms of drug resistance and possible strategies to overcome this resistance, including nanoparticles or phytochemicals, which are currently showing promise in managing patients with resistance to anticancer treatments. 73 Indeed, advanced drug delivery systems (e.g., liposomes and nanoparticles) have demonstrated success in overcoming poor bioavailability of plant-based alkaloids and could potentially be used to ensure targeted drug delivery. 74 The potential risks of prolonged use of belumosudil in immunocompromised patients are also unclear.
The CTTC guidelines also suggest implementing a pre-emptive approach to cGVHD treatment in high-risk patients to potentially mitigate disease progression and improve long-term outcomes and QoL in these patients. 68 Patients at high risk of cGVHD based on their underlying disease and donor/transplant characteristics should be closely monitored for early signs of cGVHD, with early initiation of appropriate treatment. 67 In a 2024 study of pediatric patients undergoing haploidentical CD34-enriched and CD45RA‑depleted HSCT followed by natural killer‑cell infusion, a short course of immunosuppressive therapy (sirolimus or mycophenolate mofetil) was used as GVHD prophylaxis. 75 According to an updated consensus statement from the Chinese Society of Hematology, patients with advanced leukemia who have undergone HSCT should receive modified donor lymphocyte infusion (DLI) and interferon-α as pre-emptive therapy when minimal residual disease is detected. 76 Cellular therapies, such as DLIs or umbilical cord-derived mesenchymal stromal cells (MSCs), may be promising options for cGVHD prophylaxis, particularly in patients undergoing haploid HSCT.77–81 MSCs have also shown potential in the treatment of steroid-resistant acute GVHD when combined with basiliximab and calcineurin inhibitor therapy. 82 Another emerging pre-emptive strategy for GVHD is optimized CD8+ Tregs, which may provide another form of cellular therapy for GVHD prevention that preserves graft-versus-leukemic effects. 83 Development of such pre-emptive strategies warrants further research to identify reliable predictors, including risk assignment biomarkers associated with cGVHD development and severity, such as IL-39 expression 84 and recirculating tissue-resident memory T-cells. 85 This may aid in the development of robust risk assessment models to accurately predict cGVHD in HSCT recipients.17,86
Several ongoing studies are evaluating the efficacy and safety of belumosudil as first-line or pre-emptive treatment for cGVHD (Table 2). A randomized, double-blind, placebo-controlled, phase III study is evaluating the use of belumosudil in combination with corticosteroids in patients with newly diagnosed moderate or severe cGVHD (ROCKnrol-1; NCT06143891). Another open-label, phase II study is investigating belumosudil plus rituximab as first-line treatment in patients with extensive cGVHD (NCT06046248). For pre-emptive therapy, an open-label, phase II study is assessing belumosudil (± standard of care) as treatment for new-onset/early-stage BOS (BEBOP; NCT05922761). A randomized, placebo-controlled, phase II study is evaluating whether belumosudil can be used as pre-emptive treatment in patients with at least one clinical manifestation of cGVHD (NCT05996627). Further studies are warranted on the impact of belumosudil treatment on biomarkers of response in cGVHD (i.e., ST2 and soluble B-cell activating factor (sBAFF)), and prognostic biomarkers (i.e., C-X-C motif chemokine ligand 9 (CXCL9) and CXCL10); however, data on biomarkers in cGVHD are limited. 87
Summary of current ongoing studies of belumosudil administered as first-line or pre-emptive treatment in patients with chronic graft-versus-host disease.
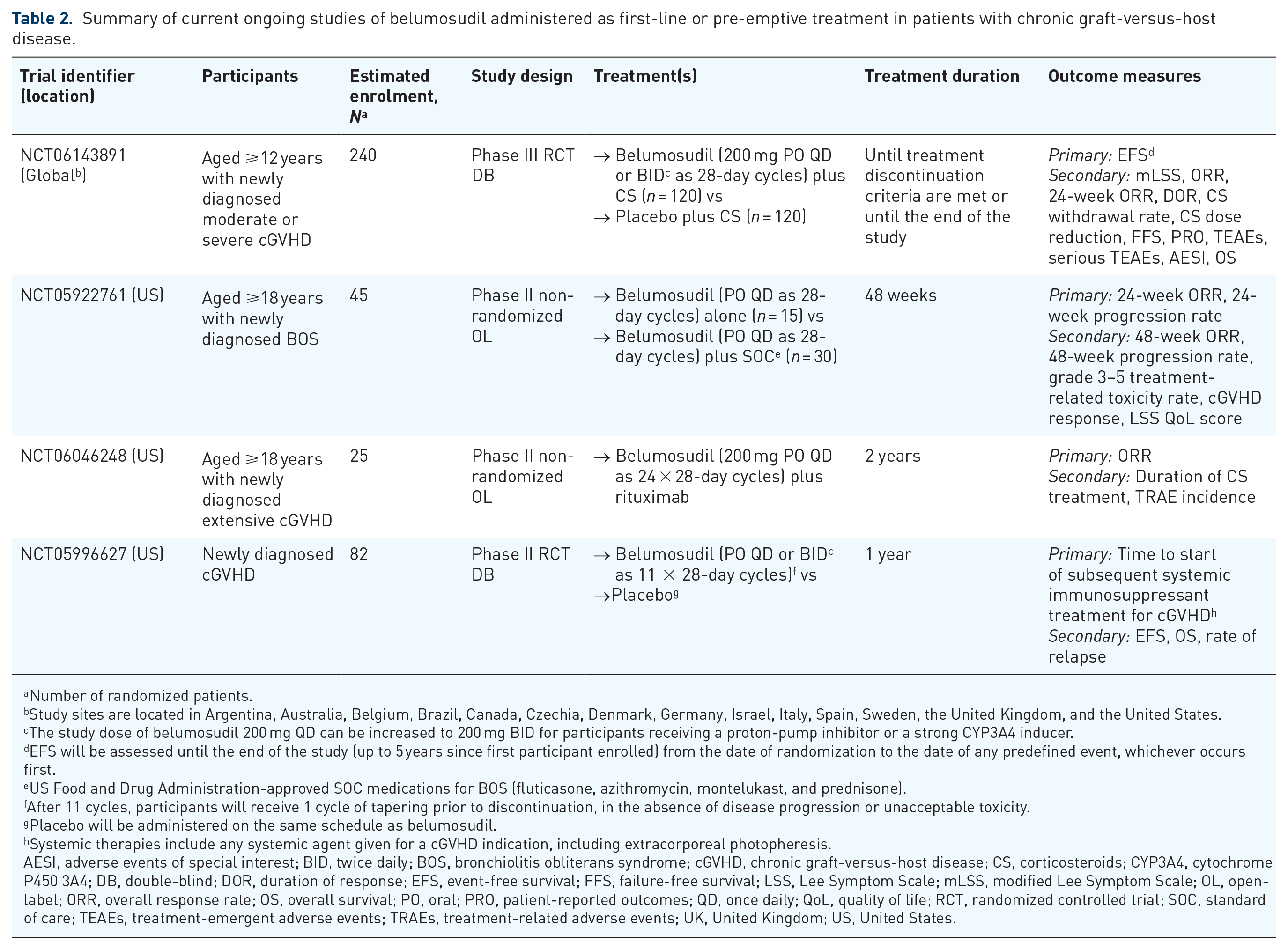
Number of randomized patients.
Study sites are located in Argentina, Australia, Belgium, Brazil, Canada, Czechia, Denmark, Germany, Israel, Italy, Spain, Sweden, the United Kingdom, and the United States.
The study dose of belumosudil 200 mg QD can be increased to 200 mg BID for participants receiving a proton-pump inhibitor or a strong CYP3A4 inducer.
EFS will be assessed until the end of the study (up to 5 years since first participant enrolled) from the date of randomization to the date of any predefined event, whichever occurs first.
US Food and Drug Administration-approved SOC medications for BOS (fluticasone, azithromycin, montelukast, and prednisone).
After 11 cycles, participants will receive 1 cycle of tapering prior to discontinuation, in the absence of disease progression or unacceptable toxicity.
Placebo will be administered on the same schedule as belumosudil.
Systemic therapies include any systemic agent given for a cGVHD indication, including extracorporeal photopheresis.
AESI, adverse events of special interest; BID, twice daily; BOS, bronchiolitis obliterans syndrome; cGVHD, chronic graft-versus-host disease; CS, corticosteroids; CYP3A4, cytochrome P450 3A4; DB, double-blind; DOR, duration of response; EFS, event-free survival; FFS, failure-free survival; LSS, Lee Symptom Scale; mLSS, modified Lee Symptom Scale; OL, open-label; ORR, overall response rate; OS, overall survival; PO, oral; PRO, patient-reported outcomes; QD, once daily; QoL, quality of life; RCT, randomized controlled trial; SOC, standard of care; TEAEs, treatment-emergent adverse events; TRAEs, treatment-related adverse events; UK, United Kingdom; US, United States.
The tear cytokine profile of patients with ocular GVHD is particularly complex, comprising IL-1, -2, -6, -8, -10, and -17, as well as CXCL10, intercellular adhesion molecule 1, tumor necrosis factor-α, epidermal growth factor, lymphotoxin-a, E-selectin, BAFF, and neuropilin-1, all of which may be potential biomarkers of ocular GVHD risk, diagnosis, and prognosis. 88 Due to the lack of biomarker panels with high sensitivity or high specificity, novel immune biomarkers for cGVHD may also be considered when monitoring for response to belumosudil, including IL-21, IL-17, and Tfh cells; a combination of such biomarkers may provide higher specificity and greater clinical value. 89
Belumosudil, together with other novel cGVHD agents or treatment strategies, are promising treatment options for patients with cGVHD. For example, belumosudil in combination with B-cell targeted therapy is a potential therapeutic approach that is currently under investigation, as described above (NCT06046248). In light of the potential role of extracorporeal photopheresis when combined with other drugs and the related corticosteroid-sparing effect, 68 an add-on belumosudil treatment strategy may be a rational approach in the management of cGVHD. Furthermore, combination therapy with certain phytochemicals may help overcome clinical translational challenges and provide synergistic efficacy,90–92 as previously demonstrated by in vitro studies of the plant phytochemical thymoquinone in combination with 5-fluorouracil-loaded calcium carbonate nanoparticles in CT26 colon cancer cells.93,94 Similarly, components of the medicinal herb Polygonum minus have synergistic anti-colon cancer effects when combined with doxorubicin or 5-fluorouracil.73,95 Further studies are needed to determine whether similar synergistic effects are achieved with belumosudil-based combination therapy in patients with cGVHD.
Evidence suggests that dose modification may be needed when using belumosudil with immunosuppressants. In a single-center retrospective US study, the concentration–dose ratio of tacrolimus and/or sirolimus was increased during concomitant treatment with belumosudil, indicating that dose reductions of tacrolimus and sirolimus may be needed to prevent over-immunosuppression in patients with cGVHD. 96 Further studies are needed to determine the systemic exposure and the need for related dose modification of other immunosuppressants in patients with cGVHD receiving belumosudil.
Extensive organ fibrosis severely impacts physical function and QoL in patients with cGVHD. 97 Current evidence suggests that belumosudil is associated with improved QoL, with a pooled analysis of phase II study data showing a strong correlation between organ-specific clinical response with belumosudil and clinically meaningful changes in the LSS score. 47 Although several PRO measurements that are commonly used in patients with cGVHD (e.g., the 36-item short-form health survey (SF-36), Human Activity Profile, and Functional Assessment of Cancer Therapy–Bone Marrow Transplant), 98 there are no reported data for these PROs in belumosudil-treated patients. Nevertheless, current guidelines include belumosudil among the available treatment options in patients with steroid-refractory cGVHD, particularly due to its low toxicity profile 21 and antifibrotic properties. 68 More evidence on the PRO improvements among patients receiving belumosudil will help further guide clinical treatment choices.
A novel oral suspension of belumosudil is currently under clinical development for pediatric patients or adults with dysphagia and/or upper gastrointestinal cGVHD involvement who may have difficulty swallowing belumosudil tablets. A phase I study in healthy male volunteers has reported that the belumosudil suspension formulation had more rapid absorption and similar bioavailability compared to the tablet formulation. 99 The taste profile of the oral suspension was improved with the use of a sweetener and/or flavor. 99
Conclusion
Belumosudil is an effective and well-tolerated treatment option in patients with steroid-refractory or -dependent cGVHD. Ongoing studies are investigating the use of belumosudil as first-line or pre-emptive treatment of cGVHD, in combination with other cGVHD treatments, and in special populations (e.g., patients with BOS), the results of which could potentially address some of the unmet therapeutic needs in the clinical management of cGVHD.
Footnotes
Acknowledgements
The authors would like to thank Sarah Greig, PhD, CMPP, and Iona MacDonald of Springer Health+, who provided medical writing assistance. This medical writing assistance was funded by Sanofi.
Authors’ note
The content published in this article solely represents the views and opinions of the authors, who had full control over the collection, analysis, and interpretation of data obtained from the literature search, the writing of the manuscript, and the decision to submit the final version for publication.
