Abstract
Background:
Venetoclax is the first representative of a new class of targeted therapy, that inhibits selectively B-cell lymphoma-2 (BCL-2), an anti-apoptotic protein, frequently overexpressed in hematological malignancies. Venetoclax was approved by the Food and Drug Administration for chronic lymphocytic leukemia and for acute myeloid leukemia in 2016 and 2021, respectively. Because of its promising role in many hematological malignancies, several clinical trials are in progress and other extensions of indication are expected. An analysis of its long-term safety profile in real life is necessary.
Objectives:
The aim of our study was to evaluate all adverse events (AEs) reported to the French national pharmacovigilance database since its approval in France.
Methods:
We performed a retrospective study of all cases of AEs occurring under venetoclax recorded in the French national pharmacovigilance database since its market approval until March 2022 in France.
Results:
During the period study, a total of 209 AEs were spontaneously reported in 123 patients, of which 173 (82%) were serious. We confirmed that the most frequent toxicities described by the summary of product characteristic (SPC) and literature data on it, including hematological (21%), gastrointestinal (11%), dermatological (9%), infectious (8%) AEs, and tumor lysis syndrome (3%). Seventy-six (36%) AEs were not listed in the SPC for which the causal relationship of venetoclax could not be excluded including autoimmune hemolytic anemias (2%) or cardiac AEs (7%).
Conclusion:
These data especially in cardiac events provide important information on the safety of the venetoclax in a real-world setting.
Plain language summary
Keywords
Introduction
Apoptosis plays a crucial role in tumor development and in the response to anticancer treatment. 1 In hematological malignancies, the alteration of this process is often the result of the overexpression of an anti-apoptotic protein: B-cell lymphoma-2 (BCL-2).2,3 Targeting this protein in hematological malignancies has been recognized as an interesting approach and led to the development of venetoclax, the first and only oral drug in the market, a potent selective BCL-2 inhibitor. By binding to protein BCL-2, it restores apoptotic activity that triggers cell death. 4 Venetoclax was first approved by the Food and Drug Administration (FDA) in 2016. Nowadays, venetoclax is indicated in patients with chronic lymphocytic leukemia (CLL) 5 and acute myeloid leukemia (AML) 6 meeting certain criteria (ineligibility for chemotherapy, previous treatment failure, and/or mutation status). In the United States, since May 2019, the use of venetoclax has been generalized to all patients with CLL regardless of their mutation status or line of treatment. 7 In addition, venetoclax has also been shown to be clinically effective as monotherapy in many malignant hemopathies. 8 Other trials involving the use of venetoclax in combination therapies are also still in progress in acute lymphoblastic leukemia (ALL), diffuse large BCLs, and multiple myeloma (MM) especially with t(11;14) translocation.9–11 Given its promising role, further extensions of therapeutic indications and its use on a larger scale should be considered in the next few years. Thus, safety profile analysis in a real-life setting is needed to help hematologists prescribe the drug as safely as possible. Short-term clinical trials have reported significant adverse events (AEs) related to venetoclax including tumor lysis syndrome (TLS), neutropenia, thrombocytopenia, infections, and other common AEs including anemia, diarrhea, nausea, and musculoskeletal pain. 7 Most of the efficacy and safety data of newly marketed drugs come from clinical trials. However, due to the relatively small sample size and eligibility criteria, these studies cannot fully describe the impact of drugs in the real world, especially in terms of safety. Our study aimed to evaluate all AEs of venetoclax reported to the French national pharmacovigilance network, since its approval in France.
Methods
Data collection
We conducted a retrospective study using data recorded in the French national pharmacovigilance database (FNPVD) between January 1, 2016, and March 1, 2022, for the suspected substance “venetoclax.” This database includes all declarations of AEs submitted through spontaneous reports by healthcare professionals, which are then validated and analyzed by the 31 French regional pharmacovigilance centers. Each report contains information on patient demographics, medical history, drug therapy, treatment indication, and the nature, seriousness, and outcome of the AEs. The study was performed in accordance with the Declaration of Helsinki. The institutional review board of each center approved the study, and anonymous data collection was declared to the appropriate authorities (authorization protocol number: CNIL-1922081).
We included all AEs reported in association with venetoclax in the FNPVD. Exclusion criteria, based on chart review, were: duplicate reports, absence of venetoclax treatment, or absence of AEs related to venetoclax.
AEs and drug identification
The screened AEs are standardized and classified through the system organ class (SOC) and preferred term (PT) of the international medical dictionary for regular activities (MedDRA 25.0 developed under the auspices of the International Council for Harmonisation (ICH) of Technical Requirements for Pharmaceuticals for Human Use). SOC and PT were taken as the objects of data analysis and research. All French regional centers of pharmacovigilance evaluate the relationship between the AE and the medication according to the French imputability method for each case. 12
We collected data about patient baseline characteristics (age, sex, medical history/comorbidities, hematological malignancy, stage, year of diagnosis, line of treatment), treatment with venetoclax (date of start, associated therapy, dose during onset of AE, treatment modifications due to AE, treatment discontinuation, reason for treatment discontinuation), and AE (number of AEs, grade, date of appearance, time to onset, SOC, death, intrinsic imputability, other suspected products with an imputability greater than or equal to that of venetoclax, concomitant administration with CYP3A4 or P-gp inhibitors). AEs were also categorized as listed or not in the summary of product characteristic (SPC) of venetoclax and according to the indication (CLL or AML). Thus, an AE reported in the context of an indication will be considered as not listed if it occurs within the part of the treatment of the other indication. In case of multiple AEs notified per patient, each AE was described in separate sheets. We also collected data relating to biological examinations when available (hematology, kidney, liver, and microbiological). In addition, we also provided information for each cytopenia according to version 5.0 of the Common Terminology Criteria for Adverse Events. 13
Statistical analysis
Continuous data were expressed as median with interquartile range (IQR, 25th–75th percentiles). Categorical data were expressed in numbers (n) and percentages. All data processing and statistical analysis were performed by GraphPad Prism 9 (GraphPad software part of Insightful Science) and R 4.3.3 (The R project for statistical computing, open-source software project supported by the R foundation for statistical computing).
Results
Overview
From the FNPVD, a total of 218 reports of AE occurring in 127 patients were collected during the study period between January 1, 2016, and March 1, 2022. After reviewing the reports by three hematologist physicians and one pharmacologist, we excluded a total of nine AEs in four patients (Figure 1). Then, we included a total of 209 AEs occurring in 123 patients (40 AML, 69 CLL, and 15 others in the present analysis). Figure 2(a) illustrates the evolution of the number of notifications relating to venetoclax according to the reporting year and venetoclax indication. The demographic and clinical characteristics of the patients are presented in Table 1. More than a third of identified patients (n = 47, 38%) had at least cardiovascular comorbidity. The most reported were hypertension for 33 (27%) followed by dyslipidemia, diabetes, and atrial fibrillation (AF) for 13 (10%), 12 (10%), and 10 (8%) patients, respectively. Also, 12 (10%) patients were active smokers, and 9 (7%) had a BMI > 30 kg/m2. In addition, 13 (10%) patients had a history of solid cancer, and 7 (6%) had another blood disease including essential thrombocythemia (n = 2), primary polycythemia (n = 1), Hodgkin lymphoma (n = 1), diffuse large BCL (n = 1), beta-thalassemia minor (n = 1). Finally, 8 (6%) patients had previous stem cell transplantation including 6 allografts and 2 autografts. No differences were observed in the characteristics of patients treated with venetoclax across hematological malignancies, except for the frequency of diabetes (Table 2).

Flowchart of the study.

Number of adverse events related to venetoclax reported to the French pharmacovigilance network each year (a) and per patient (b).
Characteristics of patients treated by venetoclax with a declaration of adverse events, subdivided according to fatal or non-fatal outcome of adverse events.

Others hematological malignancies were B-cell lymphoma (n = 5), T-cell lymphoma (n = 3 including two T-cell prolymphocytic leukemia), multiple myeloma (n = 2), myelodysplastic syndrome (n = 2), chronic myelomonocytic leukemia (n = 1), and unknown (n = 2).
AML, acute myeloid leukemia; BMI, body mass index; CLL, chronic lymphocytic leukemia; IQR, interquartile range.
Characteristics of patients treated by venetoclax with a declaration of adverse events, subdivided according to hematological malignancies.
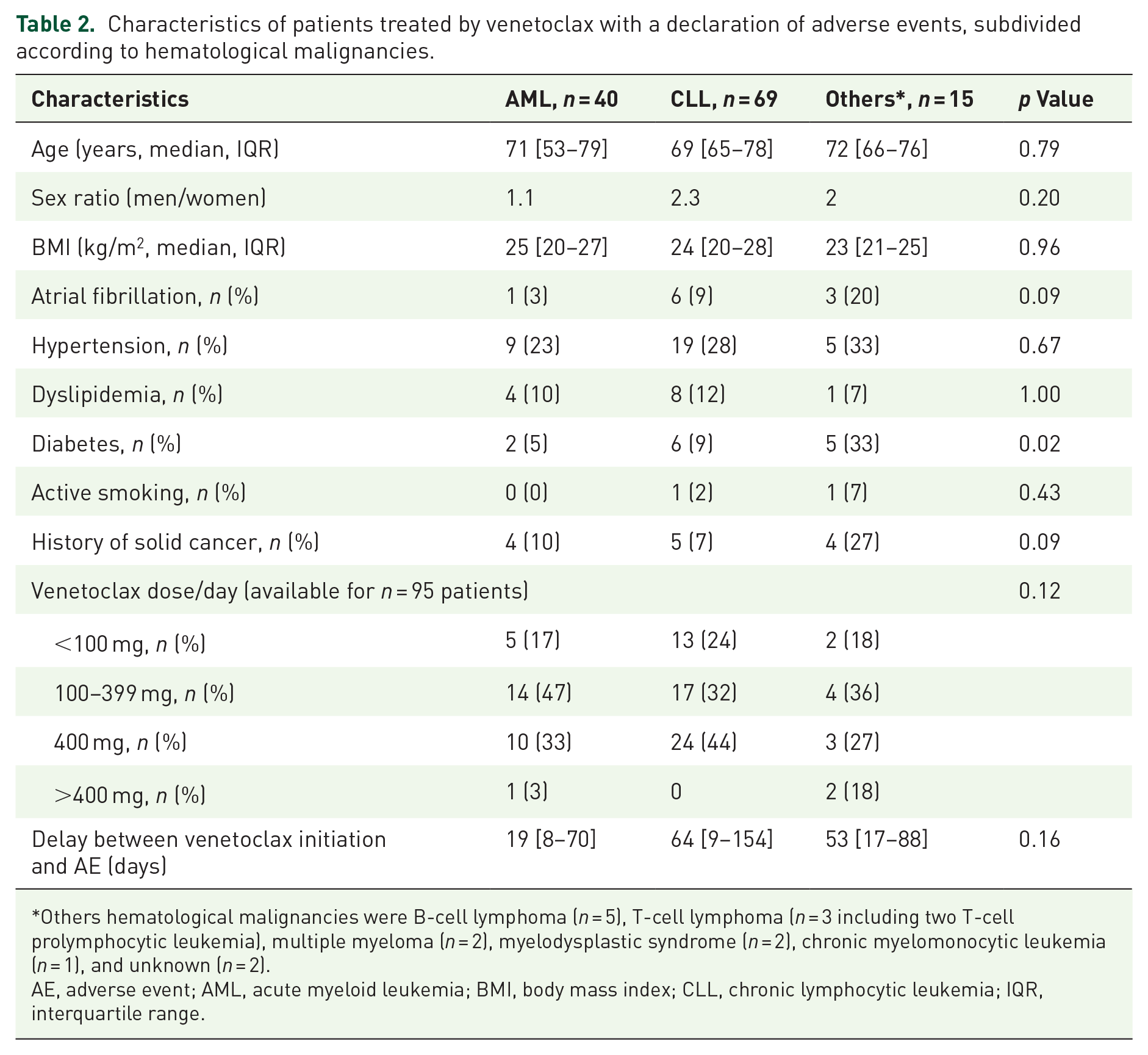
Others hematological malignancies were B-cell lymphoma (n = 5), T-cell lymphoma (n = 3 including two T-cell prolymphocytic leukemia), multiple myeloma (n = 2), myelodysplastic syndrome (n = 2), chronic myelomonocytic leukemia (n = 1), and unknown (n = 2).
AE, adverse event; AML, acute myeloid leukemia; BMI, body mass index; CLL, chronic lymphocytic leukemia; IQR, interquartile range.
A large majority of patients (n = 108, 87%) were treated with venetoclax as part of the two therapeutic indications: CLL (56%) and AML (32%). Other hemopathies were B lymphoma (n = 5), T-cell lymphoma and leukemia (n = 3), MM (n = 2), myelodysplastic syndrome (n = 2), chronic myelomonocytic leukemia (n = 1), and unknown (n = 2). More than half of patients (n = 63) were treated with venetoclax monotherapy while the others received it in combination with another treatment (azacitidine for AML and rituximab for CLL). Some patients were taking off-label combinations, in particular the combination of venetoclax with ibrutinib, cytarabine, or lenalidomide and one patient a combination venetoclax-ibrutinib-obinutuzumab. Among the 37 (30%) patients with data available, 36 (97%) received venetoclax as second line treatment or more.
Adverse reaction profile
The median number of AEs by patient was 1 [IQR 1–2]. The median delay between venetoclax initiation and AE was 48 days [IQR 7–92] with no difference between hematological malignancy (p = 0.16). Most patients (55.3%) experienced a single AE related to venetoclax and a maximum of six AEs occurred in one patient (Figure 2(b)). Similar repartition of SOC was observed between diseases (Figure 3(a)). As expected according to the SPC of venetoclax, the SOC “blood disorders” was the most represented with 44 (21%) AEs followed by gastrointestinal disorders (11%) and infections (8%; Table 3). Off note, 18 (9%) patients experienced AE for the SOC “skin and subcutaneous tissue disorders” that were mainly not clinically significant and including rash, itching, and erythema. Seventy-six (36%) AEs were not listed in the SPC for which the causal relationship of venetoclax could not be excluded including dermatological (n = 18, 9%), hepatobiliary (n = 15, 7%), cardiac (n = 14, 6%) and renal (n = 10, 4%) AEs. The most frequent AEs of all grades and all indications combined in patients who treated with venetoclax (monotherapy or in combination) were neutropenia or febrile neutropenia (n = 20, 10%) followed by thrombocytopenia (n = 7, 3%) and TLS (n = 6, 3%). In addition, we observed a greater number of AEs reported in patients treated with venetoclax for CLL (n = 114, 54%) compared to those treated for AML (n = 71, 34%). The most common AEs in patients with CLL were hematologic: neutropenia (n = 10), TLS (n = 5), thrombocytopenia (n = 5), and autoimmune hemolytic anemia (AIHA, n = 3). The most common AE reported in patients with AML was neutropenia/febrile aplasia (n = 4).

Subanalysis of SOCs and AEs related to venetoclax reported to the French pharmacovigilance network. (a) Proportion of SOCs according to venetoclax indication; (b) proportion of SOCs according to venetoclax dose; (c) proportion of hematological AEs according to venetoclax indication.
Description of main adverse events and case of interest according to system organ classification with their seriousness and fatal outcome.

Reported non-fatal case more than once that was not reported in SPC.
AIHA, autoimmune hemolytic anemia; GGT, gamma-glutamyl transferase; HHV-6, human herpesvirus-6; RSV, respiratory syncytial virus; SPC, summary of product characteristic.
Venetoclax was the only suspected therapy in 94 (45%) of the cases and for 116 (55%) of the 209 reported AEs, other drugs had an equal or greater imputability than venetoclax. Due to the combination of venetoclax with other cytotoxic agents, or with other treatments for comorbidities, the onset of AE could also be attributable to the latter.
Among all AEs reported, 173 (83%) were considered serious with the most common severity criterion being hospitalization or prolongation of hospitalization (63%). The main AE leading to hospitalization of patients regardless of indication was febrile aplasia or neutropenia (n = 10), followed by thrombocytopenia (n = 5). A fatal outcome was recorded in 13 (10%) patients. The median age of deceased patients was 74 years [65–82] with a majority of men. Only two AEs leading to death (TLS and cardiogenic shock) can probably be attributed to venetoclax.
Concerning the timing and venetoclax dose of AEs by SOC following venetoclax initiation (Table 4), AEs in the skin and subcutaneous tissue disorders category appear to occur the fastest (median 8 days), followed by nervous system disorders (median 13 days) and metabolism and nutrition disorders (median 16 days). In contrast, the slowest-onset AEs were observed in renal and urinary disorders (median 76 days), infections and infestations (median 70 days), and other disorders (median 66 days).
Time and dose at onset of adverse events by system organ class following venetoclax initiation.
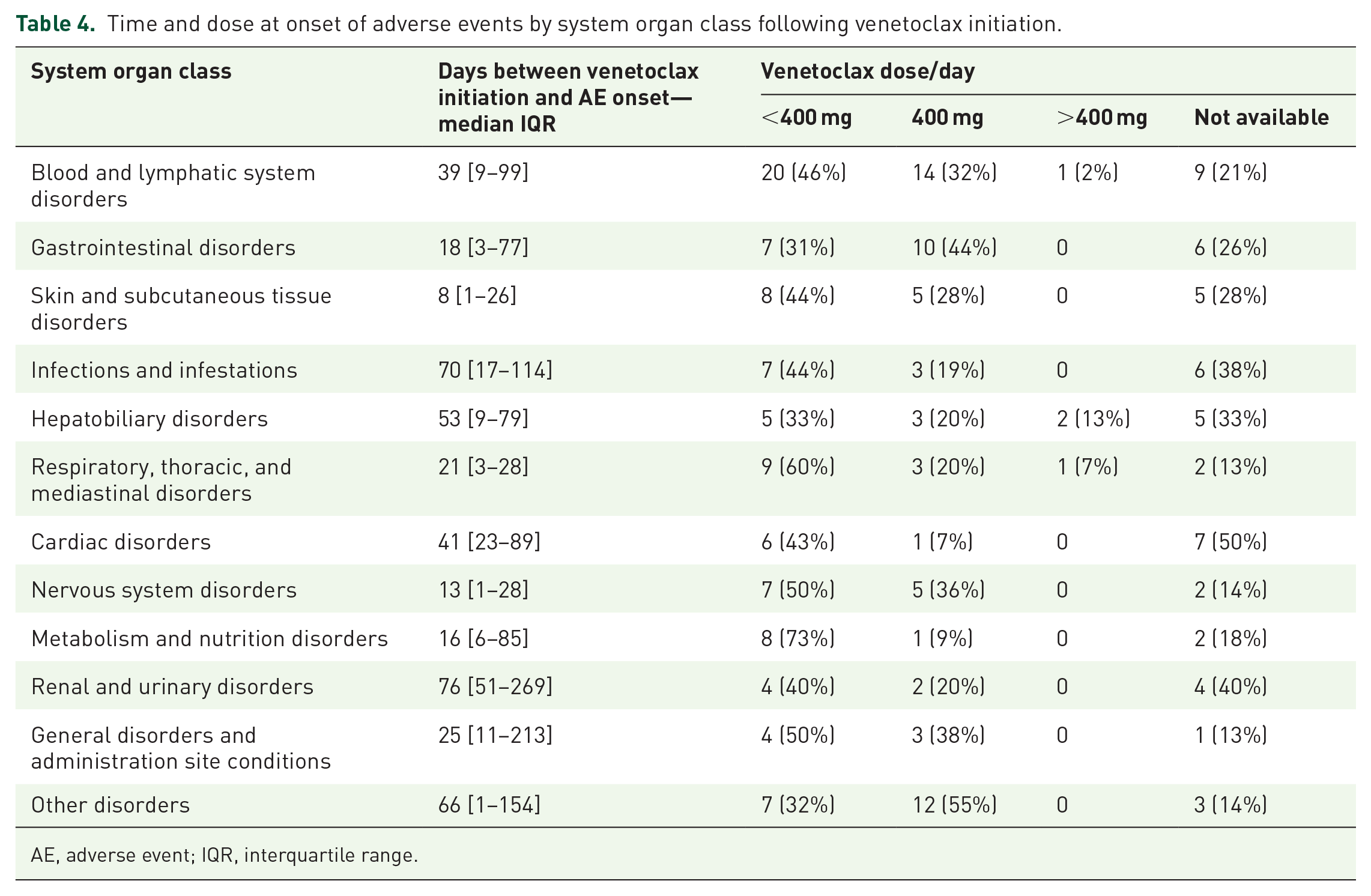
AE, adverse event; IQR, interquartile range.
Treatment discontinuation and dose reductions
Discontinuations of treatment due to AEs were reported in 53 (43%) of patients treated with venetoclax. Dose reductions were reported in 11 (9%) patients. The most common AE leading to discontinuation of venetoclax were febrile neutropenia (n = 5), TLS (n = 4) and thrombocytopenia (n = 4). Median doses of venetoclax at the onset of the reported AE was 200 mg/day [100–400]. Most AEs (30%) occurred at the maintenance dose, that is, 400 mg/day. Regarding the distribution of AEs by venetoclax dose, skin and subcutaneous tissue disorders (17%) and nervous system disorders (14%) were more frequent in patients receiving low doses (<100 mg) of venetoclax (Figure 3(b)). The six cases of TLS appeared at a dosage ⩽200 mg/day. Three patients (2%) were reported at a supratherapeutic dose: two cases of liver injury at a dose of 600 mg/day in one AML and a case of pneumonitis and a case of AIHA in two patients treated at 800 and 1000 mg/day for prolymphocytic T-cell leukemia. AIHA occurred 1.5 months after the dose increased of venetoclax to 1000 mg.
Co-administration with CYP3A4 inhibitors
Among the 24 patients with available data, 11 (46%) were also taking an azole antifungal, potent CYP3A4 inhibitor, including posaconazole (n = 8), voriconazole (n = 1), and ketoconazole (n = 1). In 8 (73%) of these 11 cases, venetoclax was prescribed at a reduced dose (100 mg) when co-administered with a CYP3A4 inhibitor, including 9 cases of AML, 1 case of CLL, and 1 case of MM. These 10 patients experienced 18 different AEs, including electrolyte disorders of different etiologies (1 hypokalemia on diarrhea, 1 hypokalemia and hypomagnesemia on renal tubulopathy, and 1 hyponatremia due to excessive release of antidiuretic hormone), hepatobiliary disorders and cardiac disorders (n = 3, heart failure, hemopericardium, pericarditis). One patient died of fulminant hepatitis 27 days after ketoconazole was introduced to boost venetoclax.
Targeted descriptive analysis of AEs by SOC
Hematologic and infections AEs
Concerning blood and lymphatic system disorders AEs (n = 44, 21%), venetoclax was the only suspected drug in 19 (42%) cases. The same repartition of hematologic AEs was observed between hematological malignancies (Figure 3(c)). Isolated or combined cytopenias (neutropenia, anemia, thrombocytopenia, pancytopenia, aplastic anemia) accounted for 70% (n = 31) and occurred within a median of 22 days [IQR 8–92]. The majority of cytopenias reported under venetoclax (85%, n = 28) were grade 3 or more. Febrile neutropenias occurred in a median time of 11 days [9–54] following initiation of venetoclax. Documented infections associated with these febrile neutropenias were Escherichia coli cellulitis (n = 1), dental abscess (n = 1), septic shock due to Streptococcus oralis and disseminated candidosis (n = 1), and viral pneumonitis due to respiratory virus syncytial (n = 1). Most patients recovered after antibiotherapy and G-CSF support (granulocyte colony-stimulating factor). Among the unexpected toxicities (not listed in the SPC), three cases of AIHA were reported. In all three cases, the only suspect drug is venetoclax. Patients were 65 years of age or older, treated for CLL (n = 2) or prolymphocytic T-cell leukemia (n = 1). The lowest recorded hemoglobin was 35 g/L, which resulted in the death of one CLL patient. The other two patients recovered under corticosteroid therapy. Five cases of thrombocytopenia in patients with CLL have been reported. The severity of the thrombocytopenia varied (grade 4, n = 2; grade 3, n = 2; and grade 1, n = 1). Among them, two patients had symptomatic thrombocytopenia: one had a melena, epistaxis, and hematemesis, and the other had cutaneous hemorrhagic syndrome. The median time to onset of thrombocytopenia was 50 days [IQR 22–58]. For two cases, the imputability of venetoclax was assessed as likely.
Concerning infections (n = 16, 8%), seven patients experienced neutropenia grade 4. Two deaths were reported: a case of septic shock in a patient with grade 4 neutropenia and a case of human herpesvirus 6 (HHV-6) encephalitis in a patient with CLL. The microbiological documentation was available for 9 (56%) of the 16 infectious AEs: 3 E. coli, 2 HHV-6 encephalitis, 1 pneumocystis, 1 SARS-CoV-2, 1 respiratory syncytial virus, and 1 Pseudomonas aeruginosa. Among the cases of unexpected infectious toxicity, venetoclax had an imputability considered possible for three cases.
Metabolism and nutrition disorders
Concerning the 11 (5%) metabolic AEs, there were 6 TLS, 2 hypokalemia, 1 hyponatremia, 1 hypomagnesemia, and 1 anorexia. Venetoclax was the only suspected drug in 7 cases including 6 TLS (2 biological only and 4 serious with multi-organ failure). The median delay to onset of TLS after venetoclax intake was 6 days [IQR 1–14] with doses ⩽200 mg/day. TLS evolution was fatal for 2 (33%) CLL patients. The imputability of venetoclax was plausible or probable in all cases of TLS.
Kidney and urinary tract disorders
Concerning the 10 (4%) kidney AEs, venetoclax has an equal or lower causality than other suspected drugs (n = 9, 90%). For two of the patients, impairment of renal function appeared in a context of TLS. For one of the patients, TLS had occurred the day after the introduction of venetoclax for the treatment of sixth-line CLL which resulted in the patient’s death. For the other, the patient presented with TLS complicated by acute renal failure following the first administration of venetoclax 50 mg and requiring 5 weeks dialysis, with residual renal impairment. The reintroduction of venetoclax caused a further worsening of renal insufficiency. One case occurred in the context of liver injury. Two cases were unexpected. One case of drug-induced vasculitis was documented by renal biopsy during venetoclax treatment combined with azacitidine and one case Fanconi syndrome also confirmed by renal biopsy under venetoclax and ciprofloxacin. The precise chronological analysis of the patient’s kidney function tests and the administered drugs led to suspect venetoclax as the primary cause for Fanconi syndrome. Moreover, in another case of hypomagnesemia and hypokalemia, a tubulopathy was suspected.
Respiratory, thoracic, and mediastinal disorders
Among the pulmonary AEs (n = 15, 7%), none of AEs except one case of dyspnea was listed in the SPC, and imputability to venetoclax was doubtful for the majority (n = 11, 73%). Venetoclax was the only suspected drug in six cases. Three cases of non-fatal pulmonary embolism were reported in patients older than 55 years treated for CLL (n = 2) and AML (n = 1). The imputability to venetoclax was doubtful for two and likely for one of the three cases. In addition, three cases of lung interstitial disease were collected in association with drugs already involved in pneumonitis (azacitidine, ruxolitinib) and the relationship with venetoclax was considered doubtful in two cases and possible in the last.
Hepatobiliary disorders
Among the 15 (7%) hepatobiliary AEs occurred in 9 patients, 5 cases were elevated liver enzymes (serum aminotransferases associated or not with an increase of gamma-glutamyl transferase or alkaline phosphatase). In two cases, the outcome was not favorable after venetoclax discontinuation; in one case, discontinuation led to a rapid decrease of cytolysis, then venetoclax reintroduction did not lead to the recurrence of liver injury; in the fourth case, withdrawal of voriconazole led to recovery, and in the last case, the outcome was unknown. One case of hyperbilirubinemia was recorded, with an appearance of giant cell hepatitis on biopsy, compatible with autoimmune hepatitis. One patient developed cholestatic hepatitis during the fifth line venetoclax therapy for refractory aggressive lymphocytic lymphoma, but the role of lymphoma progression was also considered. Fulminant hepatitis had occurred 55 days following the intake of venetoclax and 27 days after the addition of ketoconazole in a patient treated for MM. Off this patient took acetaminophen for bone pain 2 days before the onset of fulminant hepatitis. After positive results of adenovirus polymerase chain reaction (PCR) and liver biopsy, the most likely etiology of this fulminant hepatitis was adenovirus-induced hepatitis in a febrile aplastic patient undergoing treatment for MM. Finally, only one case of hepatocellular injury associated with acute renal failure was reported 9 days after venetoclax began in a patient with CLL, with a favorable outcome after discontinuation of treatment.
Nervous system disorders
The 14 neurologic AEs (6%) occurred in 12 patients mainly treated for CLL (67%). There were two cases of cerebellar damage that developed rapidly after the start of treatment, at D12 and D14, in association with cytarabine, with no CT-scan abnormalities, and which regressed spontaneously or with dexamethasone. There were also two cases of meningitis with no bacteriological documentation, which appeared at D1 and D3 after the introduction of venetoclax and progressed favorably on discontinuation of treatment and antibiotic treatment with dexamethasone. In addition, four cases of signs of peripheral neuropathy were reported, with the imputability of venetoclax remaining doubtful.
General disorders and administration site conditions
Among the 8 (3%) general disorders AEs, there were 2 case of SARS-CoV-2 vaccination failures that had fatal outcomes (imputability I2 to venetoclax). Those two patients were male patients (69 and 85 years old) treated with venetoclax for CLL and AML who deceased of severe SARS-CoV-2 infection (delta variant) 172 days and 4 months, respectively, after three doses of BNT162b2 and two doses of ChAdOx1/nCov-19 and a booster dose of BNT162b2 vaccines.
Cardiac conditions
Among the 123 patients, 11 (9%) patients presented a total of 14 (6%) cardiac AEs. Five (45%) patients had previous cardiovascular risk factors. The median time to onset of these heart conditions was 41 days [range 23–89], and main AEs reported were heart failure (n = 3) and AF (n = 3). Most of the events were considered severe (93%) and leading to hospitalization in 64% of the cases. Among the 14 deaths potentially related to venetoclax, the cardiac effect directly contributed to the death of two patients: a case of cardiorespiratory arrest immediately after platelets transfusion and a case of cardiogenic shock. In the latter case, the patient had previous cardiovascular risk factors, including hypertension, obstructive heart disease, ischemic stroke, and AF. Two weeks following the initiation of venetoclax, the patient presented a cardiogenic shock with healthy coronary arteries. Venetoclax withdrawal and corrective treatment with dobutamine and norepinephrine provided initial stabilization. Following the reintroduction of venetoclax, a second cardio-circulatory deterioration appeared and was fatal. Given this positive “rechallenge,” a hypothesis of cardiac toxicity of venetoclax had been put forward by clinicians for this case. Venetoclax has an imputability equal to or less than that of other suspect medicinal products in the occurrence of six AEs and is the only suspected in the occurrence of 57% (n = 8) of AEs. Among the three cases of heart failure, two patients were treated for AML and were taking azacitidine in addition to venetoclax. Among the three cases of AF, the three patients were treated for CLL including one patient who concomitantly received ibrutinib, a well-known cause of AF. Imputability to venetoclax was low for the three cases.
Discussion
The present study described for the first time in France, all cases of AEs reported under venetoclax therapy reported to regional pharmacovigilance centers and registered in the FNPVD since its market introduction until March 2022 representing 209 AEs (83% serious) in 123 patients. In the VIALE-A study, at least one serious AE (SAE) was reported in 80% of patients 14 as observed in the intermediate results of the French prospective VERONE study. 15 Indeed, in this study, the patients received venetoclax in monotherapy or combination with rituximab and showed 46%/46% of SAE and 19%/17% of SAEs linked directly to venetoclax (neutropenia 7% and 5%), respectively. Finally, the results of the Italian pharmacovigilance study also showed that the majority of AEs were classified as SAEs (72%) and AE more frequently reported in AML patients (73%). 16
We observed that most patients (89%) received venetoclax within the two therapeutic indications: CLL and AML. Concerning the dose of venetoclax, we have noticed certain uses of venetoclax categorized as supratherapeutic dose especially for the treatment of AML, and for off-label treatment T-cell prolymphocytic leukemia for two patients. The use of venetoclax at dosages up to 1200 mg has been tested in phase I studies with CLL and NHL without identifying a maximum tolerated dose.8,17 Also, the BELLINI study evaluated the use of bortezomib and dexamethasone in combination with venetoclax 800 mg/day in patients with relapsed or refractory MM. 11 Regarding the type of AE most frequently reported, our analysis confirmed the main toxicities described in the SPC and the literature, 18 including hematological, gastrointestinal, infectious toxicity, and the occurrence of TLS and are coherent with results from the Italian database. 19
Concerning fatal outcomes, only five AEs (pneumopathy, pancytopenia, TLS, sepsis, cardiogenic shock) had been plausibly attributed or likely to venetoclax. For comparison, in the VERONE study, 15 four fatal venetoclax-related AEs were reported in three patients (ascites and hepatic failure, unspecified death). Surprisingly, a large proportion of reported AEs (n = 76, 36%) were not listed in the SPC. This could be partly explained by the under-reporting of AE, which is considered harmless or well-known. Some of these non-listed toxicities, some were plausibly or likely attributable to venetoclax, in particular heart, kidney, liver, and skin conditions.
Cytopenias are commonly observed in patients receiving venetoclax, particularly neutropenia, since BCL-2 promotes the survival of granulocyte precursors.7,20 We noticed that neutropenia was the most frequently reported AE (n = 13, 10% of patients). Although hematological AEs were generally expected, we still even noted that thrombocytopenia had appeared in five patients with CLL, while in the SPC of venetoclax, thrombocytopenia is a very common adverse effect described only in patients with AML. Unexpected thrombocytopenia was reported in 21% of patients, 24% of whom were grade 3 or 4. 20 The development of venetoclax, was also prioritized over navitoclax due to the significant platelet toxicity of the latter through the inhibition of BCL-XL, on which platelet survival is dependent. 7 In addition, a mice study interested in the role of BCL-2 in the survival of megakaryocytes and platelets have shown that BCL-2 was neither necessary for the production or platelet survival, unlike myeloid cell leukemia-1 (MCL-1) and BCL-XL which play a role important in maintaining the survival of megakaryocytes and platelets respectively. 21 Another hematological AE that appeared in three patients and not expected in the SPC was AHAI, including one fatal case. Cases of AIHA had also been described in clinical trial and case reports, 22 raising the possibility of a role for venetoclax, particularly in patients whose CLL did not progressed on venetoclax. Using data from three phase I/II studies, Davids et al. 20 observed in 350 patients with CLL treated with 400 mg daily venetoclax monotherapy that AIHA was reported in 10 patients (3%), 8 of whom had del(17p). Ibrutinib and venetoclax can inhibit autoantibody producing B-cells and act on T-cell homeostasis and cases of remission of AIHA have been reported during treatment with these molecules. 20
Our study suggested a signal of early AEs related to cardiac disorders (7% of reported AEs). We reported one fatal cardiogenic shock attributable to venetoclax based on a positive rechallenge. We also reported three cases of heart failure and three of AF. SPC do not mention the cardiac toxicity of venetoclax. However, in the VIALE-A study, 13 (5%) and 6 (2%) patients with AML in the azacitidine–venetoclax group were reported to have AF and heart failure, respectively, compared to 2 (1%) and 3 (2%) patients the azacitidine plus placebo group. 14 One explanation was the selection of patients during clinical trials including younger patients with less comorbidity. 4 In real-life setting with FDA database, Yang et al. 23 described 1935 cases of cardiac AEs and among them, 238 cases of AF that conduct to a strong signal of AE. Cardiac AEs were described in 20% of AML patients treated with venetoclax + hypomethylating agents at the Mayo Clinic (n = 170). 24 Cardiac AEs, increased in patients with venetoclax exposure compared to hypomethylating agents alone, were associated with increased hospitalization (89%) and decreased survival. As we reported, most cardiac AEs were characterized by decreased left ventricular ejection fraction and AF, with an early onset typically during the first two cycles (median onset: 41 days). A similar toxicity profile was confirmed in a subsequent study involving 301 AML patients 25 treated with venetoclax combined with hypomethylating agents: 23 (7.6%) patients experienced major cardiac complications, including 15 cases of cardiomyopathy, 5 non-ST elevation myocardial infarction, and 7 pericarditis or pericardial effusions, with some patients experiencing more than one cardiac event. Cardiac toxicity was associated with shorter overall survival (7.7 months vs 13.2 months; p < 0.01). Additionally, troponin elevation was observed in 15% of patients, including four who experienced clinical cardiac AEs in the context of a cofactor such as sepsis or anemia. Notably, sepsis and anemia are common early-onset AEs of venetoclax, both in clinical trials and in our study, and can precipitate severe cardiac events. A meta-analysis of nine venetoclax studies confirmed in 2024 an increased risk of cardiovascular events (risk ratio: 1.726, 95% confidence interval: 1.088–2.737), particularly in patients with pre-existing cardiac conditions. 26 Concerning physiopathology, a preclinical study on rats showed that venetoclax treatment can effectively induce cardiotoxicity manifested by changes in cardiomyocyte repartition, an increase in cardiac enzymes, induction of apoptosis, oxidative stress, and inflammatory markers. 24 Based on these elements, cardiac monitoring may be helpful. We proposed to monitor the troponin level at the initiation and during the first cycles as presented during the anthracycline regimen by recent European Society of Cardiology guidelines (no mention of BCL-2 inhibitors in this guideline 27 ). Active monitoring has helped to save lives in the recent discontinuation of the study of venetoclax and ibrutinib in Waldenström macroglobulinemia based on the high incidence of ventricular arrhythmia. 9 These previous studies, coupled with our findings, suggest that cardiac complications are a notable concern in this treatment context and warrant further prospective investigation, especially in patients with pre-existing cardiovascular conditions.
We acknowledge several limitations in the present study. First, the retrospective design of the study based on reported cases limit data quality and the reported predominance of severe cases could be explained by an under-reporting of less severe AEs by physicians. 28 Nevertheless, our results remain consistent with the toxicity profile described in the SPC of venetoclax as well as the literature data.14–16 Second, there is a potential bias due to spontaneous reporting. Reported AEs may not be representative of all the AEs that occurred in France during the study period. Additionally, during the COVID-19 pandemic, there may have been under-reporting of AEs. However, the peak of reported cases in the pharmacovigilance database occurred in 2020 and 2021, though this may still underestimate the true number due to under-reporting or an excess of deaths in this immunosuppressed population of patients with hematological malignancies who are at risk of severe COVID-19. Moreover, the literature does not appear to show an excess in mortality among COVID-19 patients treated with venetoclax but does suggest an interaction with anti-CD20 therapies.11–13 Third, despite well-documented reports of AEs, reports suffered from a lack of standardization of the information recorded and a lack of follow-up. Finally, the role of venetoclax in the occurrence of the reported AEs remains difficult to assess due to its administration in combination with other anticancer and cytotoxic therapies. Moreover, venetoclax was the only suspect in just under half of the reported cases (45%).
Conclusion
Our French pharmacovigilance study confirmed the main toxicities associated with venetoclax treatment, as described by the SPC of venetoclax and literature but also, some rare or new AEs. Indeed, we observed thrombocytopenia in patients with CLL or AIHA. In addition, cardiac, hepatic, and renal toxicities strongly attributable to venetoclax have been reported. Our study provided important insights on the real-world safety of venetoclax that should be confirmed in further large-scale prospective studies, for further extensions of therapeutic indications.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251343116 – Supplemental material for Safety of venetoclax in real-world experience: data from the French national database of pharmacovigilance through all indications in hematological malignancies over 5 years
Supplemental material, sj-docx-1-tah-10.1177_20406207251343116 for Safety of venetoclax in real-world experience: data from the French national database of pharmacovigilance through all indications in hematological malignancies over 5 years by Alexis Talbot, Pierre-Edouard Debureaux, Agnès Lillo-Le Louet, Yasmine Derri, Marine Aroux-Pavard, Hélène Jantzem, David M. Smadja, Cyrille Touzeau, Christine Le Beller and Nicolas Gendron in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
