Abstract
Background:
Medical resources, especially blood products, were in short supply during the COVID-19. Less intensive therapy with hypomethylating agents/venetoclax (VEN) seems an effective treatment option for patients with acute myeloid leukemia (AML).
Objectives:
To retrospectively analyze the efficacy and safety of VEN combined with azacitidine (AZA) in young adult patients with newly diagnosed (ND) AML.
Design:
This was a retrospective study.
Methods:
The clinical data of 25 AML patients treated with the VEN + AZA regimen from January 2021 to December 2023 at our center were collected, compared with a randomized historical study cohort that was administered intensive chemotherapy (IC) from January 2018 to December 2019.
Results:
No rate of complete remission/complete remission with incomplete count recovery differences observed between the two arms reached statistical significance. Compared to traditional IC, minimal residual disease (MRD)-negative remission was achieved more quickly in patients treated with VEN + AZA regimens (after cycle 1: 8% in the IC group vs 56% in the VEN group, p = 0.0004; after cycle 2: 16% in the IC group vs 72% in the VEN group, p = 0.0001), especially in those AML patients who had a poor prognosis. The dependency of transfusion of red blood cell (RBC) and platelets during induction treatment was significantly lower in the VEN + AZA group (RBC: p = 0.0269; platelet: p = 0.0054). Compared with the standard IC, the incidence rate of non-hematological adverse events in VEN + AZA group was significantly decreased (infection: 100% vs 20%, p = 0.0001; gastrointestinal side effects: 48% vs 12%, p = 0.0055). The total hospitalization cost of the VEN group was significantly less than that of the IC group (p = 0.0395).
Conclusion:
In conclusion, our study indicated that VEN + AZA with a higher MRD-negative remission rate and less toxic appeared to be a therapy option for young patients with ND AML. However, further well-designed studies with larger numbers of patients are needed to confirm the benefits of VEN + AZA in this population.
Plain language summary
Study about a new treatment combination in young patients with newly diagnosed acute myeloid leukemia written by clinicians. Why was the study done? Acute myeloid leukemia (AML) is a hematological malignancy and the recommended standard induction regimen is intensive chemotherapy. A sufficient and safe blood supply is necessary for AML patients during the intensive chemotherapy. However, medical resources, especially blood products, were in short supply during COVID-19. Hence, doctors wanted to find a less intensive therapy with less blood products whereby preserving efficacy in this patient group. What did the researchers do? The research team collected medical records from patients who received this less intensive therapy and compared their clinical indicators with those of patients receiving intensive chemotherapy. What did the researchers find? A total of 50 people were included in the study, 25 in each group. There was no difference between the two groups in terms of complete remission rates. For those with a poor prognosis, the low-intensity regimen resulted in higher rates of remission and deeper remissions. Patients in the low-intensity therapy group had a lower incidence of adverse reactions than the intensive chemotherapy regimen. Patients in the low-intensity regimen group also had fewer numerical amounts of blood products than in the intensive chemotherapy regimen. What do the findings mean? This new treatment regimen seems to be a promising option with less intensive and less toxic for young untreated AML patients. More research is needed to support these findings.
Introduction
Acute myeloid leukemia (AML) is a heterogeneous neoplasm, which is characterized by the monoclonal proliferation of immature hematological progenitors. Age is one of the important factors in determining treatment for AML patients. For younger newly diagnosed (ND) patients who are defined as those being <60 years, the recommended standard induction regimen is intensive chemotherapy (IC), 1 which is composed of 3 days of anthracyclines and 7 days of cytarabine (Ara-C), the so-called 3 + 7 regimen. Although this regimen can achieve a high response rate, 2 high occurrence rates of life-threatening hematological adverse events (AEs) and non-hematological AEs often result in prolonged inpatient days, heavier economic burden, and even treatment-related death. 3
One of the challenges in the management of AML is treatment-related death mostly due to infection and bleeding. The induction-related mortality during the first month of treatment reported from population-based studies was ranging from 5% to 15%.4,5 Severe and prolonged infection can lead to life-threatening complications that may require intensive care unit (ICU) management. A large retrospective cohort study, including 6442 patients with AML from 313 hospitals during the contemporary period of 2010 to 2017, suggested practice patterns and outcomes with IC in real-world settings. It reported the median length of stay was 29 days (Inter-quartile range: 25–38), and 16.0% died or were discharged to hospice. During admission, 28.0%, 12.6%, and 4.0% of patients required treatment in ICUs, mechanical ventilation, and dialysis, respectively. 6
Retrospective analysis indicated that AML was associated with substantial healthcare resource utilization and cost in the USA (estimated to range from US$145,189 to $198,657 for induction therapy in each treatment episode across the course of the disease). Among these, supportive care cost (e.g., transfusion or intravenous antimicrobial therapy) was the primary cost driver in ND AML. 7 A sufficient and safe blood supply was necessary for AML patients treated with IC. Shortage of blood products as well as concerns about the safety of blood products had emerged and treatment options for ND AML patients were limited. The coronavirus 2019 (COVID-19) pandemic has imposed a worldwide challenge and has negatively impacted blood product management, with increasing morbidities (symptomatic anemia and thrombocytopenia) and induction mortalities, which has added stress to the already stressed healthcare system.8,9
Venetoclax (VEN), an oral BCL-2 inhibitor, has revolutionized the treatment of AML. VEN in combination with a hypomethylating agent has been approved by the Food and Drug Administration for ND AML adults who are 75 years of age or older or who have comorbidities and are not eligible to receive intensive induction chemotherapy. 10 This regimen targets leukemia stem cells and thereby achieves deep and durable response. 11 Notably, the safety profile of VEN-based induction regimens is favorable, with a low risk of early treatment-related mortality and less side effects compared with conventional IC.12,13
Given these promising results, during the outbreak of COVID-19, we treated ND young patients (<60 years) with VEN in combination with low-dose azacitidine (AZA). Then we assessed our single-institution experience with VEN + AZA, comparing outcomes with a historical study cohort that administered IC.
Methods
Patient cohort
A retrospective review of 25 ND AML cases who received first-line induction with VEN + AZA was performed between January 2021 and December 2023. As a comparison, we randomly searched for historical controls who were treated with IC as induction therapy during the past 2 years, revealing 25 patients between January 2018 and December 2019.
The inclusion criteria of patients were: (1) diagnosed as ND AML based on criteria 14 and have received no prior therapy for AML; (2) aged between 18 and 60 years; (3) actively accepted treatment and nursing as well as took medicine on time; (4) at least one bone marrow follow-up. Patients with incomplete or uncertain medical records were excluded.
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Second Affiliated Hospital of Xi’an Jiaotong University. Patient’s medical records were thoroughly reviewed and relevant data were collected, including baseline patient characteristics, treatment response, AEs, post-treatment regime, the time of death, the time of relapse, and the last time of follow-up. Genetic risk was defined using the European Leukemia Net (ELN) classification. 15 The reporting of this study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 16
Treatment
In the VEN + AZA group, VEN was given orally once a day with dosing starting at 100 mg on Day 1 of Cycle 1, escalating to 200 mg on Day 2, and reaching the target dose of 400 mg on Day 3. The 400 mg daily dosing was maintained until Day 28 of cycle 1 and on Days 1–28 in all subsequent cycles. AZA was administered to patients at the standard dose of 75 mg/m2 per day subcutaneously for the first 7 days of each 28-day cycle. Patients in the IC group received the standard induction therapy (3 + 7) using anthracyclines (Daunorubicin (DNR) or idarubicin (IDR)) and Ara-C. Anthracyclines were given intravenous injection (IV) for 3 days (DNR: 60–90 mg/m2/day or IDR: 12 mg/m2/day). Ara-C was given in a dose of 100–200 mg/m2 per day with continuous IV infusion for 7 days.
Patients in both study arms received alkalization and hydration during the treatment to mitigate the risk of tumor lysis syndrome. Red blood cell (RBC) transfusion and platelet transfusion were given when the amount of hemoglobin or platelet was less than 7 g/dL and 20 × 109/L, respectively.
According to ELN recommendations, 15 patients with favorable risk disease or who with non-favorable risk but refused allogeneic hematopoietic stem cell transplantation (allo-HSCT) were treated with high or intermediate-dose Ara-C as consolidation treatment. Meanwhile, patients with adverse risk AML and the majority of those with intermediate-risk disease, who achieved complete remission/complete remission with incomplete count recovery (CR/CRi), were recommended to receive allo-HSCT. Patients who were candidates for allo-HSCT were given the following bridge therapies, including high or intermediate-dose Ara-C or VEN + AZA. Salvage regimens for patients who failed to respond to the induction treatment of VEN + AZA included IC, FLAG (Fludarabine + Ara-C + granulocyte colony-stimulating factor), or MAE (Mitoxantrone +Ara-C + etoposide), etc.
Assessment of efficacy and safety
Bone marrow was collected at the completion of each cycle. Bone marrow morphological detection was used to judge whether the patient had achieved CR or not. Eight-color flow cytometry was performed to evaluate minimal residual disease (MRD). An MRD level <0.01% was considered negative in our center. Response was evaluated on the basis of the ELN recommendations. 15 Early death was defined as death from any causes within 8 weeks of the therapy being studied from the first diagnosis. Platelet recovery was defined as days from the course start of chemotherapy to the amount of platelet recovered to >20 × 109/L for twice evaluation without platelet transfusion. The endpoint of overall survival (OS) was death from any causes or last follow-up.
The incidence and severity of AEs were monitored and recorded. Toxicity was assessed and graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. 17
Statistical analysis
All statistical analyses were performed using SPSS software (version 24.0, Chicago, IL, USA). Categorical variables were assessed using the chi-square test or Fisher’s exact test, and continuous variables were assessed using the t-test or the Mann-Whitney U-test between the two groups. Survival was estimated using the Kaplan-Meier method. Differences with p < 0.05 were considered statistically significant.
Results
Patients’ characteristics
Fifty patients were included in this study, with 25 patients in the IC group and 25 in the VEN + AZA group. In the IC group, 13 patients (13/25, 52%) were treated with IA (IDR + Ara-C) regimen while 12 patients were treated with DA (DNR + Ara-C) regimen. In the VEN + AZA group, AZA was given starting with the standard dose (7 days, 75 mg/m2/day), while an initial dose reduction of VEN (100 mg/day) was made in three patients taking CYP3A inhibitors (voriconazole) for preventing fungal infection. After starting induction therapy, lower doses were administered in 16 patients because of the use of antifungals; hence, 10/16 patients receiving voriconazole were treated with VEN 100 mg (VEN dose was increased to 400 mg after withdrawal of voriconazole in nine of them) and 6/16 cases on fluconazole therapy received VEN 200 mg. None of the patients in either group who had FLT3 mutations received FLT3 inhibitors. There was no difference in the distributions for gender, age, or the FAB types, risk stratification, and bone marrow blast count between the two groups. Whereas, the median hemoglobin at first diagnosis in the IC group was higher than that in the VEN + AZA group (p = 0.0038). Baseline clinical characteristics of the different treatment groups are shown in Table 1.
Baseline clinical characteristics between IC and VEN + AZA.

ANC, absolute neutrophil count; AZA, azacitidine; ELN, the European Leukemia Net; IC, intensive chemotherapy; VEN, venetoclax; WBC, white blood cell.
Response rates for all patients or selected subgroups
All 50 patients survived to evaluate the response after the first induction chemotherapy. After the first cycle of treatment, in the IC group, the objective response rate (ORR) was 84% with 18 patients (72%) achieving CR/CRi and 3 patients (12%) achieving partial remission (PR). While for patients in the VEN + AZA group, the CR/CRi rate was 84% (21/25) after a single course. There was no significant difference between the two groups in ORR (84% in the IC group vs 88% in the VEN group, p = 0.5032). During the second course of induction therapy, one partial responder after the first course of induction chemotherapy treated with IC obtained CR, and one who did not respond to therapy achieved PR in the VEN group. Two cases that did not respond to treatment with VEN were AML with monocytic phenotype. The immunophenotypic information regarding individuals with M4 or M5 leukemia in each arm is presented in Supplemental Tables 3 and 4. In addition, patients No. 1 and No. 8 in the IC group, and patients No. 6 and No. 11 in the VEN group failed to respond to the induction therapy.
For patients in the IC group, CR/CRi was achieved in 19 patients (76%) after two courses of induction therapy. Compared to results achieved with IC, VEN + AZA induction therapy did not significantly improve CR rates. However, among the patients with CR/CRi, the number of patients in the VEN + AZA group achieving MRD-negative was significantly higher than those in the IC group before and after the beginning of cycle 2 (before: 8% in the IC group vs 56% in the VEN group, p = 0.0004; after: 16% in the IC group vs 72% in the VEN group, p = 0.0001). No early death was reported during the period of induction treatment.
In addition, we compared the two treatment responses in AML patients after cycle 2 by subgroup analysis based on risk stratification (Table 2). In the VEN + AZA group, 10 (77%) patients in the adverse subgroup had a CR to the treatment. Surprisingly, we observed that AML patients with intermediate or adverse risk in the VEN + AZA treatment group achieved higher MRD-negative rates compared with those in the IC treatment group (intermediate: 13% in the IC group vs 80% in the VEN group, p = 0.0149; adverse: 0% in the IC group vs 67% in the VEN group, p = 0.0070), indicating that VEN + AZA treatment may be more effective and could achieve deeper remission in those AML patients who had a poor prognosis. The treatment responses in patients who had adverse-risk cytogenetics and mutation status are shown in Figure 1.
IC and VEN treatment response after cycle 2 by genetic-risk classification.
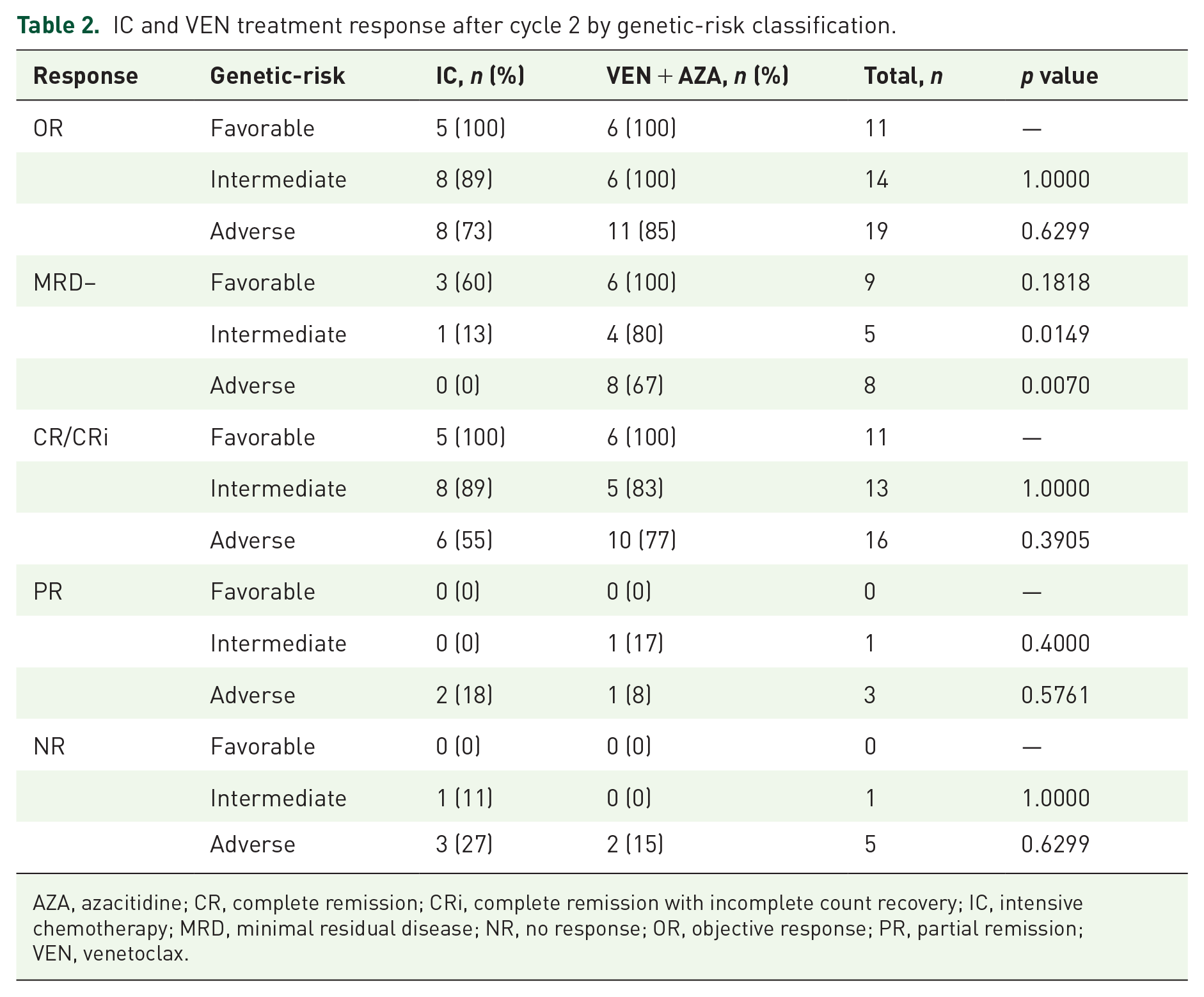
AZA, azacitidine; CR, complete remission; CRi, complete remission with incomplete count recovery; IC, intensive chemotherapy; MRD, minimal residual disease; NR, no response; OR, objective response; PR, partial remission; VEN, venetoclax.

CR/CRi rate in cytogenetics and mutation status.
Safety and toxicities
During induction therapy, the VEN + AZA regimen was generally safer compared with IC (Table 3). All patients in the entire cohort suffered from grade III or IV myelosuppression, but no patient died in the first course of induction chemotherapy. There was a remarkable difference in platelet recovery between the two groups, with a median recovery time of 17 days in the VEN + AZA group and 24 days in the IC group (p = 0.0394). Notably, patients treated with VEN + AZA regime were less dependent on RBC transfusion (7 units per cycle vs 10 units per cycle, p = 0.0269) and platelet transfusion (2.5 units per cycle vs 6 units per cycle, p = 0.0054) during the induction therapy compared to patients treated with IC. Among the non-hematological AEs, the incidence rates of both infections and gastrointestinal side effects in the VEN + AZA group were significantly lower than those in the IC group (infections: 100% in the IC group vs 20% in the VEN group, p = 0.0001; gastrointestinal side effects: 48% in the IC group vs 12% in the VEN group, p = 0.0055). Specifically, in the VEN + AZA group, infections occurred in five patients, with 4% of cases classified as grade >2. These rates were significantly lower than those observed in the IC group (p = 0.0001). Abnormal liver function was only observed in one patient receiving voriconazole in the VEN + AZA group, only grade 1. All patients were hospitalized for induction treatment, and patients in the VEN group tended to have shorter stays than those in the IC group, though there was no statistical significance between the two groups (p = 0.0612). The total hospitalization costs for the VEN group were significantly lower than those for the IC group (p = 0.0395) (Table 4). ICU transfers were initiated for two patients who developed respiratory failure and hypotension in the IC group. Fortunately, they passed through the crisis of illness and were re-admitted to a regular room receiving subsequent treatment.
AEs during cycle 1 therapy between IC and VEN + AZA.

AE, adverse event; ANC, absolute neutrophil count; AZA, azacitidine; IC, intensive chemotherapy; PLT, platelet; VEN, venetoclax; WBC, white blood cell.
Comparison of healthcare resource utilization between two groups.

AZA, azacitidine; IC, intensive chemotherapy; ICU, intensive care unit; VEN, venetoclax.
Transplantation and prognosis
Among 21 complete responders in the VEN group, 6 patients with favorable risk features received intensive Ara-C–based consolidation chemotherapy after 2 cycles of VEN. One patient lost follow-up. Of 14 cases who were eligible for allo-HSCT, 3 patients received VEN + AZA treatment only before transplantation and were still alive by the end of follow-up. No severe graft versus host disease (GVHD) and relapse was observed. Seven patients received the following consolidation chemotherapies (high-dose chemotherapy with Ara-C with a total dose of 16–18 g/m2). However, five of these patients developed serious infections during the preparation phase prior to transplantation, including three patients who died from septicemia due to the uncontrollable infections and two patients who relapsed and subsequently lost the opportunity for allo-HSCT because of the discontinuation or delay of their chemotherapeutic regimen caused by severe infections. Among the 19 patients with CR status who received IC, there were three patients unable to receive allo-HSCT due to their willingness and economic reasons. Eleven cases were willing to receive allo-HSCT and 10 of them received high-dose Ara-C as consolidation chemotherapy. Unfortunately, half of them (five cases) experienced serious infections during the post-remission treatment, three of whom died from septicemia and disease progression due to deferred chemotherapy. Eventually, 17 patients (VEN + AZA: n = 9, IC: n = 8) underwent allo-HSCT successfully. The details regarding the treatment processes and follow-up of patients enrolled in this study are shown in Figure 2.
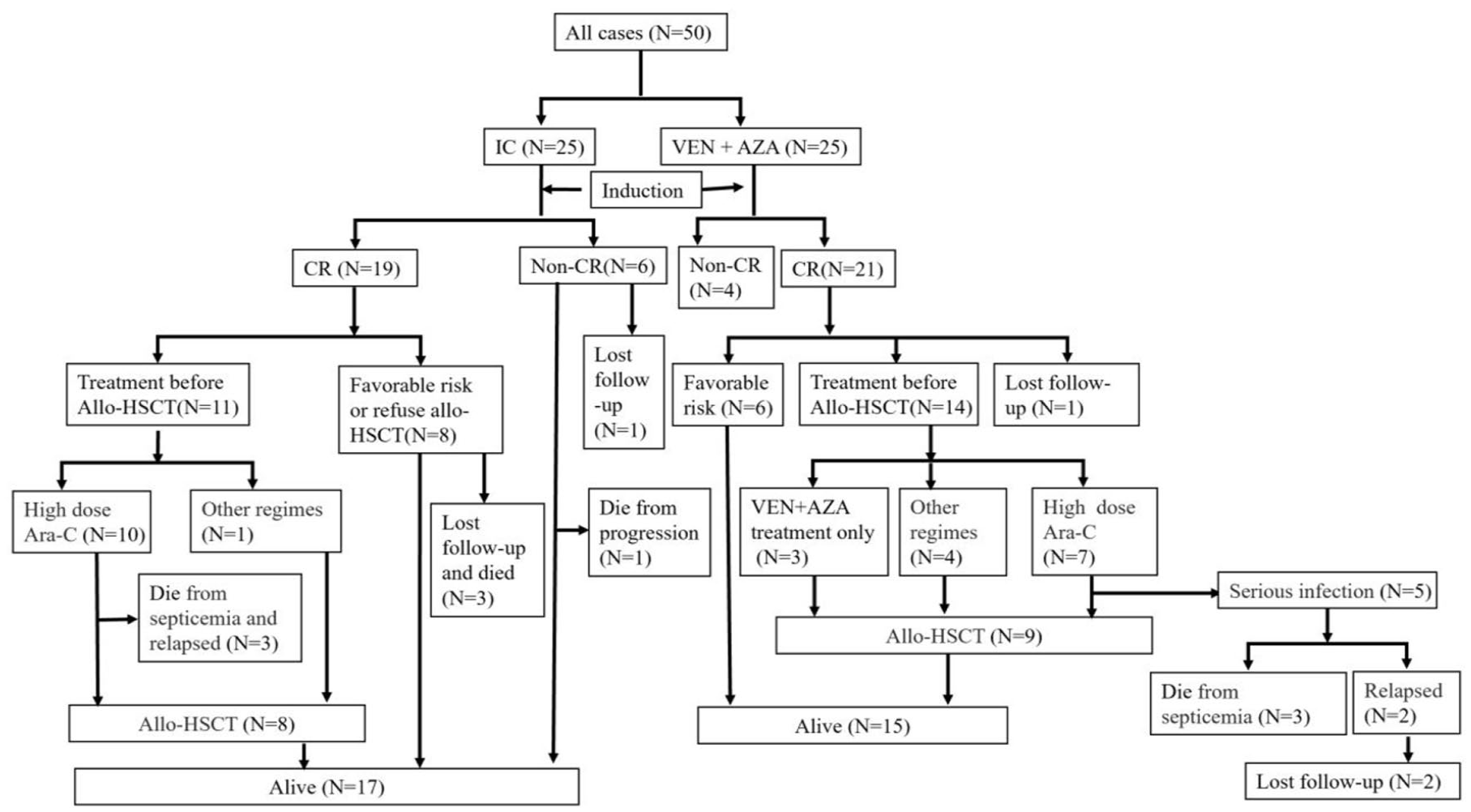
Treatment process and follow-up of patients enrolled in this study.
During 16 months of median follow-up (range from 1.8 to 35.4), 32 (64%) patients were alive (VEN + AZA: n = 15, IC: n = 17), while 5 (10%) patients were lost to follow-up. Among these patients, the median OS was 24.77 months in the IC group versus 17.63 months in the VEN group after median follow-up durations of 23.5 months (IC) and 9.7 months (VEN).
Discussion
The regimen of IC has been used as the first-line treatment for ND AML, but the management of toxicity is always a challenge for clinical practice. 18 Besides, patients with AML who receive intensive induction chemotherapy typically experience prolonged cytopenia upon completion of treatment, increasing the risk of serious infection complications, a major contributor to early death or ICU admission. These factors account for high healthcare resource utilization and costs. Nevertheless, it can hardly be denied that China’s medical resources were relatively insufficient during the COVID-19 outbreak. The rising cost of AML supportive care which was imposed by the COVID-19 pandemic has prompted physicians to reconsider the methodology behind induction therapy. Several small studies have suggested the efficacy and safety of VEN + AZA as frontline treatment in adults with ND AML.10,19 Given these considerations, along with the emphasis on lower-intensity therapies during the COVID-19 pandemic, we reviewed clinical data of young AML patients receiving VEN + AZA. We evaluated the efficacy and safety of VEN + AZA regimen compared with IC. Our findings demonstrated that treatment with VEN + AZA was more effective in achieving MRD-negative remission and less toxic than IC.
Our results showed that the VEN + AZA regimen yielded a higher CR/CRi rate (84%) than the CR rates of 61%–73% for IC induction regimens observed in a published study on patients aged <60 with de novo AML. 20 Moreover, the goal of modern therapies for AML is not only CR but also the deepest possible remission, such as MRD-negative remission. A variety of retrospective studies have demonstrated that AML patients with MRD + positive had a higher risk of relapse and shorter survival compared to MRD-negative patients. 21 Our study revealed that in the VEN group, the MRD-negative rate was more than 50% in the CR patients, while it was 16% in IC group. This finding indicated a deeper remission and more rapid clearance of MRD upon the therapy of the VEN + AZA regimen. Furthermore, it has been widely recognized that AML patients with adverse risk were less likely to achieve deep and durable remission following conventional IC. 22 In our study, we reported that VEN + AZA treatment could achieve as high as 67% MRD-negative rate in high-risk subgroups. Adverse risk cytogenetics (complex karyotype) and mutation status (RUNX1, TP53, FLT3-ITD) were also associated with higher CR rates for VEN + AZA when compared with IC, which was similar to those described by Cherry et al. 23 and Venugopal et al., 24 though the difference was not significant in our study. Despite the superior outcomes observed with VEN + AZA in patients with adverse risk, we noted poor outcomes for patients with secondary AML (s-AML). Two patients with s-AML (One was arising from myelodysplastic syndromes and the other developed AML after exposure to cytotoxic chemotherapy and irradiation) in the VEN + AZA group failed to have a response. Notably, one patient who was resistant to VEN was diagnosed with M5 according to FAB typing. Flow cytometry analysis indicated that he was monocyte-like AML with high expression of CD4, CD14, CD64, and CD11b and low expression of CD117. A retrospective cohort study conducted by Jin et al. 25 showed that ND AML with a monocytic immunophenotype had a poor prognosis with VEN/HMA treatment. In clinical practice, when determining induction therapy for AML, the expression of monocytic AML surface markers can be employed as a valuable indicator for the selection of the VEN/HMA regimen.
Additionally, three patients receiving VEN + AZA treatment as both induction and consolidation therapy were administered with allo-HSCT without severe GVHD and other complications, which indicated that VEN + AZA therapy as a bridge to allo-HSCT might be a promising option. In a retrospective study, short-term post-transplant outcomes in the VEN group appeared to be similar to those achieved after traditional IC, even though the VEN group exhibited several characteristics that were traditionally conferred inferior outcomes (older age, secondary AML, and adverse cytogenetic features). 26 In our study, five patients in the VEN + AZA group developed serious infections when they received high-dose Ara-C as consolidation chemotherapies, and they all lost the opportunity of allo-HSCT eventually. Ashby et al. 27 reported that lower-intensity VEN-combinations using VEN + AZA or VEN + LDAC as a bridge to allo-HSCT were associated with minimal treatment toxicities and hospital admissions. It appeared that post-remission VEN + AZA consolidation therapy could be considered before allo-HSCT for AML patients achieving CR. Moreover, potential toxicities resulting from intensive consolidation chemotherapy could be severe enough to delay subsequent transplantation or might lead to complications that could make patients lose the opportunity for allo-HSCT. 28 Therefore, whether lower intensive VEN regimens such as VEN + AZA could effectively and safely bridge patients to allo-HSCT after achieving CR is a novel debated issue in clinical practice. We will further expand the sample size or expect a large prospective study to further address this issue.
Supportive care such as transfused blood products during the initial intensive induction phase of therapy is of significance in alleviating severe cytopenia. 29 The average induction requires around 9–10.8 and 7–8.5 units of RBC and platelets, respectively.30,31 In our study, despite that the median hemoglobin level of patients in the VEN arm at first diagnosis was lower than that of IC group (6.2 g/dL vs 7.6 g/dL, p = 0.0038), the results demonstrated the number of both RBC and platelet transfusions was significantly lower in patients treated with VEN and AZA compared to those treated with conventional IC (RBC:10 units vs 7 units, p = 0.0269; platelets: 6 units vs 2.5 units, p = 0.0054). Moreover, it usually takes 29 ± 10 days for platelet recovery in traditional chemotherapy.32,33 The recovery time of platelets in the VEN + AZA group was significantly shorter than that in the IC group (24 days vs 17 days, p = 0.0394). In addition, the incidence rate and severity of non-hematologic AEs were lower in the VEN + AZA group, and no patients died within 8 weeks of the induction treatment. These also partially explain why patients receiving VEN treatment incurred lower total expenses in our study.
Overall, our retrospective study showed that VEN + AZA could achieve a higher MRD-negative rate and deeper remission in ND AML, with less transfusion need and fewer infections, compared to IC in young patients. Especially, this regimen proved effective in patients with adverse risk. Our observations in this study aligned with the recurrent reports of efficacy using VEN + AZA in AML. 34 Although this combination therapy presents a potential treatment option for young ND patients who are fit for IC, it has not been systematically tested in this setting.
Limitations
There are several limitations in this study. First, it was not a prospective randomized trial and there was potential confounding by information and selection bias. To illustrate the stability of our results, we made a comparison between all patients who received IC from January 2018 to December 2019 and cases in the VEN group. The findings did not alter the overall interpretation (Supplemental Tables 5–9). In recognizing the inherent constraints of this study, caution should be exercised when interpreting these results as patients without response assessment on both arms were excluded from analysis. Admittedly, we did not use the propensity score matching method to create a highly comparable control group due to the small sample size. Second, some patients who received VEN + AZA as induction chemotherapy underwent intensive consolidation, which may confound the interpretation of survival outcomes. It is challenging to ascertain whether the curative effect was due to VEN + AZA or the consolidative induction chemotherapy. Thirdly, the time of diagnosis for patients in VEN group was mainly in 2022 or 2023, and median OS between the two groups cannot be directly compared due to different follow-up time. Hence, a continued follow-up of the long-term prognosis is needed.
Conclusion
In conclusion, VEN + AZA seemed to be a promising option with a higher MRD-negative remission rate and less toxic for young untreated AML patients. However, the limited number of cases and the retrospective nature made it difficult for us to draw a solid conclusion. These findings should be confirmed or refuted through well-designed, large-sample, and randomized clinical trials with longer-term follow-up.
Supplemental Material
sj-docx-1-tah-10.1177_20406207241311776 – Supplemental material for Single-center experience of venetoclax combined with azacitidine in young patients with newly diagnosed acute myeloid leukemia
Supplemental material, sj-docx-1-tah-10.1177_20406207241311776 for Single-center experience of venetoclax combined with azacitidine in young patients with newly diagnosed acute myeloid leukemia by Xuezhu Xu, Rui Liu, Hongli Chen, Ruoyu Yang, Gongzhizi Gao, Aili He and Fangxia Wang in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-2-tah-10.1177_20406207241311776 – Supplemental material for Single-center experience of venetoclax combined with azacitidine in young patients with newly diagnosed acute myeloid leukemia
Supplemental material, sj-xlsx-2-tah-10.1177_20406207241311776 for Single-center experience of venetoclax combined with azacitidine in young patients with newly diagnosed acute myeloid leukemia by Xuezhu Xu, Rui Liu, Hongli Chen, Ruoyu Yang, Gongzhizi Gao, Aili He and Fangxia Wang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
