Abstract
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) stands as a cornerstone in the treatment of hematological malignancies, recognized for its remarkable efficacy. However, the persistent challenge of graft-versus-host disease (GVHD) continues to represent a significant barrier, often being the leading cause of nonrelapse mortality after allo-HSCT. To address this limitation, hematopoietic stem cell microtransplantation (MST) has emerged as a novel therapeutic strategy that synergistically combines chemotherapy, allo-HSCT, and cellular immunotherapy. This innovative approach is designed to retain the patient’s immune function, promote the establishment of microchimerism, and achieve a potent graft-versus-tumor (GVT) response, all while significantly minimizing the risk of GVHD. MST has primarily been applied in the treatment of hematological malignancies, where it has demonstrated promising outcomes, including marked improvements in complete remission rates, overall survival rates, and progression-free survival rates. Moreover, MST facilitates hematopoietic recovery, decreases the likelihood of infections, and reduces the incidence of GVHD, thus contributing to an improved quality of life for patients. A deeper and more comprehensive understanding of MST’s mechanisms could enhance its clinical utility and integration into standard treatment protocols. This review aims to explore the underlying mechanisms, current clinical applications, and challenges of MST, shedding light on its potential role in advancing the management of hematological malignancies.
ALL, acute lymphocytic leukemia; AML, acute myeloid leukemia; GVT, graft-versus-tumor effect; HLH, hemophagocytic lymphohistiocytosis; MDS, myelodysplastic syndrome; MM, multiple myeloma; MSC, mesenchymal stem cell; NK cells, natural killer cells; RVT, recipient-versus-tumor effect.
Introduction
In recent years, the incidence of hematological malignancies has risen significantly, particularly within the elderly population. This trend underscores the growing burden these diseases pose on healthcare systems globally.1–5 Hematological malignancies include leukemia, myelodysplastic syndrome (MDS), multiple myeloma (MM), and lymphoma. Among these, acute myeloid leukemia (AML) presents particularly poor outcomes in older adults: the 5-year overall survival (OS) rate is under 25% for patients aged 60–65 and drops to below 10% for those over 70. 6 Moreover, the 5-year OS rate for patients with MDS stands at approximately 31%, with around 80% of these cases occurring in individuals over the age of 6. 7 Elderly patients often experience a poor prognosis, influenced by a combination of factors such as driver mutations, diminished performance status, resistance to treatment, and adverse cytogenetic characteristics. 8
Hematopoietic stem cell transplantation (HSCT), cellular immunotherapy, and targeted therapy are among the primary treatment options for hematological malignancies. However, conventional chemotherapy often yields suboptimal outcomes in elderly patients, with poor prognosis and low remission rates.9,10 Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is currently considered a potential curative approach. However, its broader application is limited by significant complications, including severe graft-versus-host disease (GVHD), infections or bleeding, all of which contribute to nonrelapse mortality (NRM).11–14 Consequently, there is an urgent need for innovative therapeutic strategies to improve outcomes for these patients.
MST was initially introduced by Ai et al. in China in 2011. This strategy integrates cellular immunotherapy, allo-HSCT, and conventional chemotherapy to achieve a graft-versus-tumor (GVT) effect, while substantially reducing the incidence of GVHD. 15 In recent years, MST has shown marked improvements in complete remission (CR), OS, and progression-free survival (PFS) rates among patients with elderly acute myeloid leukemia (EAML).16–20 Clinical trials and case reports have explored the application of MST, an innovative immunotherapeutic approach, across various hematological malignancies, with particular focus on AML and, to a lesser extent, MDS.21–24 Initially, Ai’s group has focused on AML across all age groups.15,16 Furthermore, Ai expands the application of MST to include other hematological cancers, such as acute lymphoblastic leukemia (ALL) and MDS.22,25 Wang’s team has applied MST to hemophagocytic lymphohistiocytosis (HLH) since 2016. 26 In the same year, Chen et al. have initiated a clinical trial regarding the application of MST for MM 27 (Figure 1). Guo and her colleagues have initially conducted international multicenter research on the application of MST in EAML patients. 17 Moreover, the advantages of unrelated donors and the immunologic mechanisms of MST are first proposed by Anthony D. Sung et al. in 2020. 28 An expert consensus on MST for EAML is established by the International Microtransplant Interest Group 12 years after the birth of this technique. 29 This review aims to deliver an in-depth analysis of the current landscape and challenges surrounding MST, highlighting its evolving role and potential in treating hematological malignancies.

Development history of MST.
Overview of the MST
MST is an advanced immunotherapeutic approach that combines conventional chemotherapy or targeted therapies with the infusion of granulocyte colony-stimulating factor (G-CSF)-mobilized, human leukocyte antigen (HLA)-mismatched peripheral blood stem cells (GPBSCs). This method promotes donor cell engraftment, leading to microchimerism and inducing a potent GVT effect, while effectively reducing the incidence of GVHD.21,30 Additionally, MST has demonstrated substantial benefits in facilitating the recovery of both the hematopoietic and immune systems, supporting improved patient resilience and post-treatment outcomes.15,31,32 Clinical trials have explored the use of MST across a range of malignancies, including acute leukemia, MDS, relapsed/refractory (R/R) lymphoma, MM, and even certain solid tumors. These studies aim to expand therapeutic options and improve outcomes for cancer patients facing limited treatment alternatives.
The advantages of MST are summarized as follows (Table 1): (1) MST allows for a broad spectrum of potential donors, including healthy related or unrelated donors with 0–7 out of 10 HLA mismatches, while fully matched (8–10/10 HLA) sibling donors are generally not preferred.33,34 (2) MST is associated with a low incidence of GVHD, largely due to the induction of transient or persistent donor microchimerism through repeated infusions of donor peripheral blood stem cells (PBSCs),29,35 significantly reducing the need for GVHD prophylaxis. (3) MST also enhances immune function recovery by activating anti-tumor responses via the infusion of HLA-mismatched cells.33,36 (4) The elimination of immunosuppressive drugs following cell transfusion facilitates faster hematopoietic recovery, which in turn reduces the risk of infections and bleeding complications.31,37
DLI, donor lymphocyte infusion; GPBSC, granulocyte colony stimulating factor (G-CSF)-mobilized peripheral blood stem cells; GVHD, graft-versus-host disease; GVL, graft-versus-leukemia; HLA, human leukocyte antigen; MNC, mononuclear cells; MST, microtransplantation; RVL, recipient-versus-leukemia.
Mechanisms
Anti-leukemia effect
The exact mechanism by which MST prevents leukemia relapse is not yet fully understood. However, current research indicates that graft-versus-leukemia (GVL) and the induction of recipient-versus-leukemia (RVL) responses may play critical roles (Figure 2). The primary mechanism in traditional HSCT is the GVL effect, which involves hematopoietic stem cells (HSCs; primarily CD34+ MSCs) and immune cells (including CD4+ T cells, CD8+ T cells, and NK cells) derived from PBSCs activated by G-CSF.41,42 In addition to traditional transplantation, NK cells play an independent and crucial role in MST due to their recurring GVL effects. These cells continuously contribute to anti-tumor responses, enhancing MST’s therapeutic impact. 43 When lymphocyte function is impaired, CD4+ and CD8+ T cells take on a pivotal role, becoming the primary defense against leukemia. These T lymphocytes exert anti-leukemia effects by directly targeting leukemia cells and releasing cytokines, including interleukin (IL)-4 and interferon-gamma (IFN-γ), which enhance the immune response.44–46
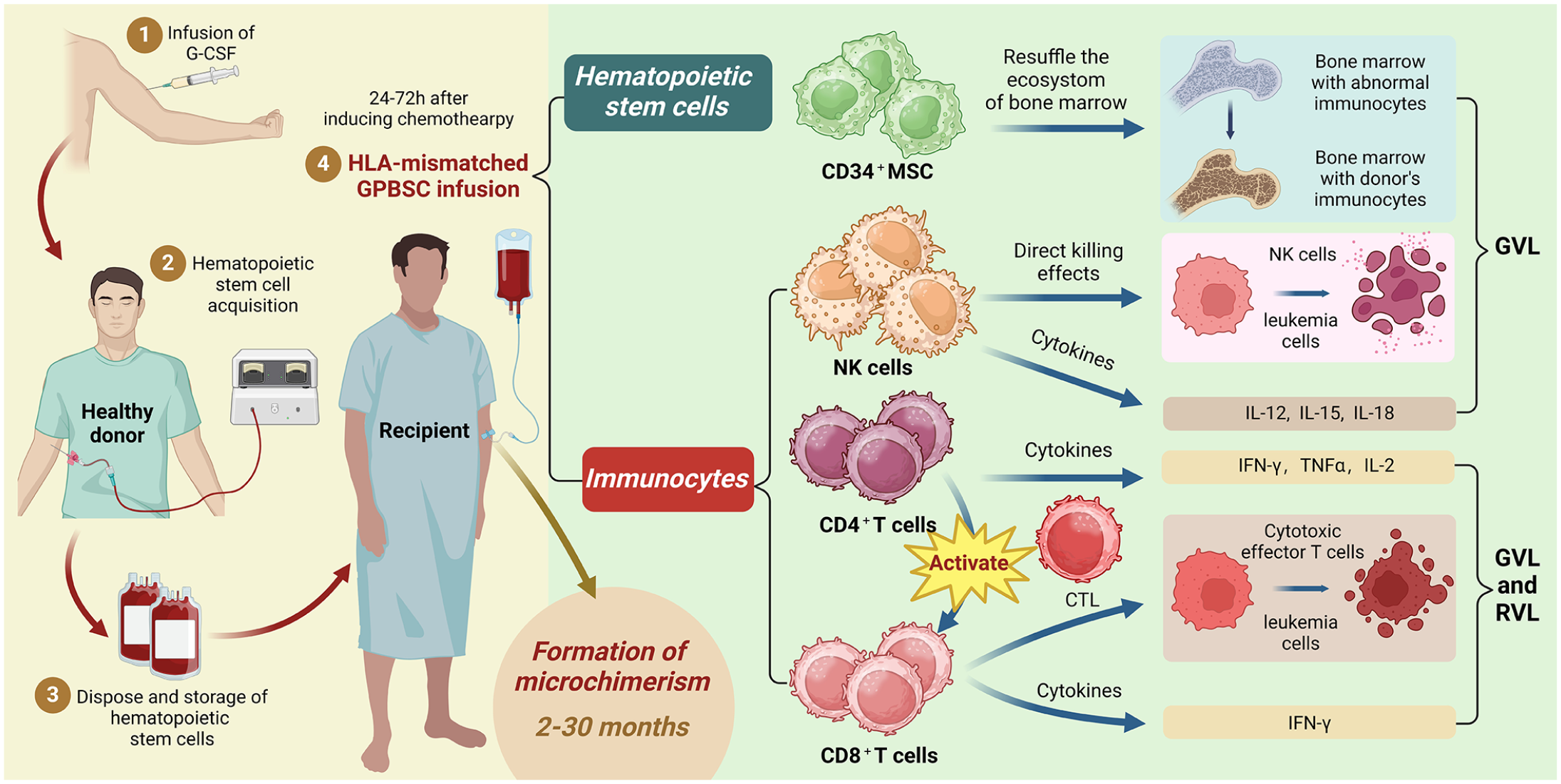
An overview of the progression and anti-leukemia effects of MST. Following induction therapy, GPBSCs are infused into the recipient, containing hematopoietic stem cells (such as CD34+ MSCs) and immune cells, including NK cells, CD4+ T cells, and CD8+ T cells. CD34+ MSCs contribute to the GVL effect by interacting within the reconstructed bone marrow microenvironment. Among immune cells, NK cells participate in GVL through direct cytotoxic actions and the release of cytokines like IL-12, IL-15, and IL-18. Both CD4+ and CD8+ T cells engage in anti-leukemia responses via GVL and RVL mechanisms; CD4+ T cells release cytokines, such as TNF-α, and activate CD8+ T cells, which then differentiate into CTLs that secrete IFN-γ to enhance the anti-leukemia response.
Current research highlights MST’s dual role in leukemia prevention through both GVL and RVL effects. In RVL cases, MST may help restore the anti-leukemia functions of the recipient’s immune system via an allogeneic response. A retrospective analysis of 45 Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) patients has examined circulating T cells in patients and/or donors with WT1+HLA-A*02:01 and/or WT1+HLA-A*24:02. 30 Notably, WT1+CD8+ T cell responses persisted up to 644 and 580 days post-MST in two pairs with patient-only HLA-A*02:01 or HLA-A*24:02, underscoring the RVL effect. Similarly, at 518, 644, and 858 days post-MST, three of four pairs with donor-only HLA-A*02:01 or HLA-A*24:02 exhibit a strong WT1+CD8+ T cell response, indicative of GVL effects. 25 Research into the RVL effects of MST suggests that donor CD4+ T cells may play a crucial role in reactivating the recipient’s CD8+ T cells, thereby enhancing MST’s anti-leukemia benefits. In a study by Wang et al., 41 higher IFN-γ levels are associated with improved anti-leukemia effects; the IFN-γ release in recipients correlates positively with CD8+ T cell counts, but not CD4+ T cells. This implies that CD4+ T cells significantly impact MST outcomes by activating leukemia-specific CD8+ T cells and the recipient’s antigen-presenting cells. Additionally, CD4+ T cells are essential for sustaining CD8+ T cell activity and enabling memory formation within MST.41,47,48
Recovery promotion of hematopoietic cells
Hematopoietic recovery is a crucial determinant of therapeutic efficacy, as it directly impacts the likelihood of severe infections, life-threatening bleeding, and other treatment-related toxicities. MST is widely acknowledged for its ability to accelerate hematopoietic restoration, thereby reducing the risks of bleeding and infection, factors essential for improving overall treatment outcomes.15,21
Infusing GPBSCs, which are rich in hematopoietic stem and progenitor cells, has been shown to significantly enhance hematopoietic recovery. Studies indicate that GPBSCs not only improve the pace of hematologic restoration but also stimulate cytokine production. Patients receiving MST experience faster hematologic recovery, allowing for a more adaptable and flexible treatment approach that mitigates damage to the hematological system. Research further suggests that hematologic recovery time is shorter in MST recipients compared to those undergoing HLA-matched sibling donor transplantation, with MST patients benefiting from a less intense conditioning regimen. 49
Microchimerism
Microchimerism refers to the presence of a small number of cells or DNA from a genetically distinct individual within a host, arising through both natural and artificial mechanisms. MST is desired to create short- or long-term donor microchimerism (donor cells <1%–5%) in recipients, but complete donor chimerism and mixed chimerism similar to classical transplantation are discouraged or even avoided after MST as a way to achieve an effective GVL effect while avoiding GVHD. 50 The duration of microchimerism exhibited a wide range, from a few days to 34 months. Donor microchimerism is observed on day 2 post-transplantation, reaching its peak on days 7–14 post-transplantation, with a maximum duration of 1020 days. 16 It is as yet unclear whether microchimerism is capable of exerting sufficient GVL effects or whether they are a necessary component of the treatment of MST.
Current clinical applications of MST
Acute myeloid leukemia
Elderly acute myeloid leukemia
AML stands as the most common and aggressive hematological malignancy. While intensive chemotherapy and allo-HSCT have improved cure rates for adult AML patients under 60, the outlook for EAML patients remains grim. Studies show that the average survival time for EAML patients is only 8–12 months. 30 This subgroup faces significant challenges, including high-risk genetic mutations and low CR and overall response rates (ORR).31,51,52 Given these challenges, there is an urgent need for safer and more effective treatments tailored to the EAML population.
Experts have reached a consensus regarding MST induction therapy for EAML patients (Table 2). Generally, the infusion of donor GPBSCs is administered 24–72 h after the final dose of cytotoxic medications, though in specific cases, this interval can be extended up to 96 h or more if needed. 29
Principles concerning MST induction therapy for EAML. 29

EAML, elderly acute myeloid leukemia; HMA, hypomethylating agents; MST, microtransplantation; PO, Peros.
Yao et al. 53 have reported an impressive 1-year OS rate of 89.8% among elderly patients receiving MST (Table 3). In a study by Hu et al., 30 involving 111 AML patients aged 70–88, 80 opted for MST, 15 for conventional chemotherapy, and 16 for supportive treatment. Relapse rates (RRs) differ significantly among these groups, with the MST group showing a reduced severe infection rate of approximately 16%, compared to 46.7% in the chemotherapy group (Table 3). Rapid neutrophil recovery considerably decreases the prevalence of infections in the granulocytosis phase, 54 further demonstrating the security of MST, according to Tao et al. In a pilot study of 23 newly diagnosed AML patients over 60, impressive outcomes have been observed: complete response is achieved in 90.9% of patients with normal cytogenetics and in 80.8% of those with unfavorable cytogenetics after just one or two treatment cycles 55 (Table 3). Similarly, a pilot study with 17 EAML patients has further supported the efficacy of MST, particularly in patients with high-risk cytogenetics 28 (Table 3).
The application of MST in AML.

AML, acute myeloid leukemia; ASCT, autologous stem cell transplantation; CR, complete remission; CSA, Cytarabine single agent; d, days; EAML, elderly acute myeloid leukemia; GVHD, graft-versus-host disease; m, months; MST, microtransplantation; N, the total number of patients; NA, not applicable; NST, nonmyeloablative stem cell transplantation; OS, overall survival; PFS, progression-free survival.
Young and middle-aged AML
Allo-HSCT is currently the only method with curative potential for young and middle-aged individuals diagnosed with AML. However, approximately 40% of AML patients are expected to experience relapse following allografts, with rates escalating to as high as 70% in high-risk AML patients.56–59 Guo et al. have reported on a cohort of 156 AML patients, with 57 undergoing nonmyeloablative stem cell transplantation (NST) while 99 receiving MST. The MST group demonstrates superior 10-year OS and leukemia-free survival (LFS) rates compared to the NST group, with rates of 70.7% versus 61.4% and 59.6% versus 57.9%, respectively. 60 The MST group also exhibits significantly lower NRM at 6.3% compared to 19.3% in the NST group (Table 3). Guo’s team has conducted a study with 101 AML-CR1 patients aged 9–65 in an MST trial. 16 The 6-year LFS rates are 84.4% for the low-risk group and 59.2% for the intermediate-risk group, indicating improved outcomes compared to prior treatment regimens 61 (Table 3). Allogeneic blood or marrow transplantation is typically not pursued for relapsed or R/R AML patients due to their generally poor prognosis. 62 However, Sidharthan et al. have reported cases involving three patients with R/R AML who have received MST as a bridge to allo-HSCT. 63 All patients have achieved CR after receiving MST without serious adverse effects, notably, one patient with the first relapse of AML has maintained CR after 506 days post-MST. In summary, MST offers superior overall therapeutic outcomes in AML compared to chemotherapy alone (Table 3).
Acute lymphocytic leukemia
Over recent decades, significant advancements in treatment approaches have greatly improved the prognosis for children with ALL. However, treatment-related mortality remains concerningly high, ranging from 11% to 25%, and RRs persist at 15% to 41%.64–66
The hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (hyper-CVAD) based MST protocol is the most widely adopted treatment approach, with mismatched GPBSCs infused 24 h after each cytarabine cycle.25,67 In 2015, Qian and Shen have first applied MST to a patient with R/R ALL. 68 Despite infusing a larger number of NK cells, the patient exhibited poor anti-leukemic effects and ultimately died from gastrointestinal hemorrhage 2 months later. Nevertheless, MST has demonstrated improved outcomes in terms of LFS and OS in subsequent clinical studies on ALL (Table 4). Cai et al. 25 have examined 45 patients with Ph+ ALL into three groups: 11 receiving MST with tyrosine kinase inhibitors, 17 undergoing allo-HSCT, and 17 receiving chemotherapy with TKIs. The OS rates for MST, allo-HSCT, and chemotherapy are 91%, 39%, and 80%, respectively. Statistically, the 4-year OS rate for MST (91%) is significantly higher than that of SCT (31%, p = 0.005) and chemotherapy (36%, p = 0.013; Table 4). In another study, Cai et al. 67 have studied 48 patients receiving hyper-CVAD-based MST. Among these patients, 26 (54%) patients di ed due to disease progression, with no NRM reported. There are no reports of early mortality or acute GVHD, and adults show better outcomes than adolescent and young adult (AYA) patients (62% vs. 26%, p = 0.058; Table 3). This trial indicates that hyper-CVAD-based MST is a promising treatment for adult ALL patients due to its low toxicity. While MST demonstrates strong efficacy in the patients with ALL, further clinical trials are needed to assess its benefits thoroughly.
The application of MST in ALL.

ALL, acute lymphoblastic leukemia; Allo-HSCT, allogeneic hematopoietic stem cell transplantation; AYA, adolescent and young adult; CR, complete remission; d, days; GVHD, graft-versus-host disease; MST, microtransplantation; NA, not applicable; N, the total number of patients; OS, overall survival; Ph, Philadelphia chromosome; PFS, progression-free survival; R/R, relapsed/refractory.
Myelodysplastic syndrome
Patients with MDS face heightened risks of bone marrow failure and progression to AML, resulting in low OS rates, especially among those at intermediate to high risk. 69
Currently, decitabine (DAC) is a key component of induction therapy, although the optimal timing for GPBSC infusion remains uncertain.18,22,37 The effectiveness of MST was first demonstrated in a study by Ai’s team, which included 72 MDS patients divided into three groups: 28 patients receiving MST, 27 receiving standard-dose DAC, and 17 receiving low-dose DAC. The results show notable differences in ORR and CR rates, with ORRs of 82.14%, 51.85%, and 35.29%, and CR rates of 42.86%, 14.81%, and 29.41%, respectively, highlighting MST’s beneficial impact on response outcomes 22 (Table 5). The TP53 mutation is the only known molecular marker linked to poor survival and high RRs, and MDS patients with TP53 mutations generally have a poor prognosis even when receiving optimal allo-HSCT. 70 However, in a study by Song et al., 71 an MDS patient with a TP53 mutation who has undergone MST shows clinical improvement without severe complications, suggesting MST’s potential as a viable alternative in high-risk MDS cases (Table 5).
The application of MST in MDS.

CR, complete remission; DAC, decitabine; GVHD, graft-versus-host disease; m, month; MDS, myelodysplastic syndrome; MST, microtransplantation; N, the total number of patients; NA, not applicable; OS, overall survival; PFS, progression-free survival.
The safety profile of MST in treating MDS is promising. Cytokine release syndrome is a common side effect observed in MDS patients undergoing MST. In a study involving patients with de novo and R/R AML/MDS, all 25 recipients exhibit symptoms of haploimmunostorm following MST infusion. However, only nine patients (13%) have required specific intervention with corticosteroids or anti-IL-6 therapy, and the 28-day post-infusion mortality rate is relatively low at 7% (Table 5). 72 These results align with findings from Munoz et al., who have studied 12 AML/MDS patients undergoing MST, 73 further supporting its favorable safety profile (Table 5). In summary, existing data suggest that combining MST with chemotherapy can improve treatment response in MDS while potentially reducing side effects, indicating MST’s potential as a safe and effective therapeutic option.
Lymphoma
Chemotherapy regimens such as hyper-CVAD, dose-augmented methotrexate, and cyclophosphamide are frequently administered before MST, although the ideal timing for MST infusion remains unclear.74–78 In a study by Zhao et al., MST’s effectiveness in reducing the clinical impact of lymphoma is examined in 10 patients with R/R malignant lymphoma. Notably, six patients who achieved CR maintained PFS until the study’s conclusion. Among three patients with T-cell lymphoblastic lymphoma, favorable PFS durations of 41, 31, and 28 months are observed. The 1-year OS and PFS rates are both 60%. Additionally, a case study on Burkitt lymphoma has shown promising outcomes, with one patient sustaining CR and PFS for 40 months after three MST treatments. 75 These findings underscore MST’s potential as an innovative approach to improve clinical outcomes and survival rates in patients who have relapsed or are resistant to conventional treatments.
Multiple myeloma
Clinical trials exploring MST’s application in MM are limited. In one study focusing on MST for MM patients with end-stage renal disease, seven patients are enrolled; after 12 years of follow-up, five survived, with four achieving CR. 79 Additionally, a case study by David et al. has documented an 80-year-old MM patient with renal failure who attained CR through MST, which persisted for 8 years. 80 In 2016, Chen’s team has launched a clinical trial 27 to compare the efficacy, safety, and 2-year PFS and OS outcomes of MST versus autologous transplantation in MM patients who have achieved partial remission or better. Reports on MST treatments for MM remain sparse, underscoring the need for further research in this area.
Hemophagocytic lymphohistiocytosis
HLH is a life-threatening condition marked by the unchecked activation of CTLs, NK cells, and macrophages, resulting in severe organ damage due to immune dysfunction. Among the types of secondary HLH, Epstein–Barr virus-associated HLH (EBV-HLH) is particularly prevalent and is linked to a poor prognosis. Standard treatments for this critical condition include dexamethasone and etoposide; however, the clinical outcomes remain bleak due to high mortality and RRs.81,82
To date, only a single clinical study has been reported on EBV-HLH. Song et al. 26 have analyzed 37 cases of EBV-HLH, with 18 patients receiving MST (GPBSC group) and the remaining 19 undergoing salvage therapy alone. The chemotherapy regimens include the DEP protocol (liposomal doxorubicin, etoposide, and methylprednisolone) and the liposomal doxorubicin, etoposide, and methylprednisolone (L-DEP) protocol (which adds polyethylene glycolated native Escherichia coli-derived L-asparaginase (PEG-asparaginase)). Results show significant improvements in several laboratory markers in the GPBSC group 2 weeks after MST, including ferritin, fibrinogen (Fbg), and white blood cell counts. The ORR in the GPBSC group was 66.7%, with a CR rate of 22.2% and a partial response rate of 44.4%.
In cases of EBV-associated HLH, EBV-DNA levels significantly decrease following MST treatment—dropping to 5.0 × 10³ [<5.0 × 102, 5.7 × 105] after 2 weeks and to 4.1 × 10³ [0, 4.4 × 104] after 4 weeks. However, survival outcomes 3 months post-therapy show no significant difference between the GPBSC group (72.2% mortality) and the control group (78.9% mortality). Among the GPBSC-treated patients, seven (38.9%) developed acute GVHD; of these, six recovered with symptomatic treatment, while one patient succumbed to GVHD despite intervention. These findings suggest that while GPBSC treatment significantly reduces EBV-DNA viral load compared to standard chemotherapy, it does not markedly improve hematological recovery. This implies that MST’s viral load reduction is closely tied to the GVL effect, with CD8+ T cells playing a key role in GPBSC infusion. Clearance of the EB virus primarily relies on the activation of CD8+ T cells, underscoring their essential role in this therapeutic approach.83,84 HLA-mismatched donor GPBSCs have shown potential benefits in treating EBV-HLH, despite the differing treatment objectives for leukemia and HLH in the context of MST. Further clinical studies are needed to determine whether GPBSC treatment could act as an effective bridging therapy to allo-HSCT for EBV-HLH patients who do not have HLA-matched donors.
Other diseases
Clinical trials have also explored MST’s application in rare hematological conditions and solid tumors. In a study by Zhang et al., two patients with myeloid sarcoma (MS) have received chemotherapy combined with MST. The first patient, a woman with primary MS, undergoes induction chemotherapy with a DA regimen (daunorubicin 45 mg/m2 on days 1–3; cytarabine 100 mg/m2 on days 1–7), followed by MST 24 h after her third DA cycle. During MST treatment, neither GVHD nor transformation to AML occurs, though her MS has eventually relapsed. Despite this, she achieves a PFS of 66 months. The second patient, diagnosed with MS and central nervous system leukemia, has received one cycle of the IDA regimen (idarubicin 10 mg on days 1–3; cytarabine 150 mg on days 1–7) in conjunction with MST, resulting in sustained CR and a PFS of 55 months. Both cases demonstrate minimal adverse effects, allowing ample time to seek HLA-matched donors and other therapeutic options. These findings suggest MST as a promising treatment for MS, though randomized clinical trials are needed to confirm its efficacy. 23 Qiyun et al. presented a case series of 17 patients with malignant solid tumors who underwent MST treatment. The cases included patients with a variety of malignant solid tumors, including breast cancer, lung cancer, gastric cancer, pancreatic cancer, seminoma of the mediastinum, colorectal cancer, thyroid carcinoma, ovarian carcinoma, small renal cell carcinoma, prostate carcinoma, smooth muscle sarcoma, endometrial carcinoma, and cervical carcinoma. The 2-year PFS and OS rates are 25% and 31.5%, respectively. 85 The preliminary results of MST in the treatment of malignant solid tumors are encouraging, but the sample size is limited and larger cohort studies are required to validate these findings.
Current challenges in HSC MST
How to reduce the recurrence?
Although MST techniques have made significant strides in treating hematological malignancies, clinical studies indicate that their anti-tumor efficacy may fall short of that achieved with allo-HSCT. This limitation has resulted in higher RRs for MST compared to traditional bone marrow transplantation.26,28,49,60,67,68 Relapse remains the leading cause of treatment failure and mortality in MST-treated patients.49,86 For EAML patients treated with MST, the median time to relapse is approximately 8.5 months, with 1-year cumulative RRs between 23.2% and 69.0%, and 2-year rates ranging from 39.7% to 79.3%. 30 Similarly, the 2-year RR for MDS patients following MST is as high as 45.5%. 18 Although MST reduces the risk of GVHD, managing the high RR remains a significant challenge in advancing MST therapy.
Combination with more effective targeted drugs
Drugs play a critical role in influencing therapeutic outcomes and RRs in patients undergoing MST. For example, certain immunomodulatory agents, such as lenalidomide, have proven both safe and effective in treating advanced AML. 87 In a small cohort trial, Fathi et al. 88 have explored lenalidomide’s potential to enhance anti-leukemia effects, yielding promising results that call for further large-scale studies. Additionally, the selection and combination of drugs are essential in screening both donors and recipients; immunosuppressive agents like fludarabine are pivotal in assessing the recipient’s immune function. 30 Overall, optimizing drug combinations is crucial for improving recipient selection and therapeutic outcomes, ultimately reducing recurrence risk.
HSC infusion optimization
Accurate HSC infusion is essential for minimizing RRs, requiring both the timely administration of G-PBSCs and an optimal cell ratio within the infusion. Extended intervals between induction therapy and infusion may adversely affect treatment outcomes, whereas a more strategic infusion approach can strengthen MST’s anti-leukemia effects. Ai et al. have developed specific criteria for cell infusion.15,16 However, further guidelines are needed to refine HSC infusion practices across different patient populations, ensuring tailored approaches that maximize therapeutic efficacy.
Appropriate receptor selection
MST is generally more appropriate for patients with compromised baseline health, those intolerant to myeloablative conditioning, and individuals lacking HLA-matched donors. For patients with diminished autologous hematopoietic function and high-risk leukemia, conventional bone marrow transplantation remains the preferred choice. A study on MST in EAML patients has reinforced this perspective, showing limited therapeutic efficacy in those with high-risk profiles. 30 Importantly, not all groups are ideal candidates for MST. Cai et al. have reported that adult patients with ALL have a lower 4-year RR compared to AYA patients (38% vs 74%), indicating that hyper-CVAD-based MST may be more suitable for adults than for younger individuals. 67
Conclusion
MST, an innovative cellular immunotherapy, has demonstrated promising efficacy in treating hematological malignancies, particularly in EAML patients. This review provides an in-depth overview of MST’s current global application and the clinical challenges it faces. MST leverages GVL and RVL effects by inducing microchimerism, significantly reducing the risk of GVHD, enhancing immune recovery, and lowering infection rates. However, a major obstacle to MST’s advancement lies in refining the technology to reduce RRs. Additionally, the exact mechanisms by which MST exerts its anti-tumor effects, supports hematopoiesis, and enhances immune recovery remain unclear and warrant further investigation.
Large-scale clinical trials are essential to deepen understanding of MST’s potential in treating various hematological malignancies, including lymphoma and MM, as well as solid tumors, expanding its therapeutic scope. Unlike commercial CAR-T cells, MST is not as widely available, limiting its adoption across hospitals nationwide. Developing unified technical standards for HSC processing and guidelines for MST could support its broader commercialization. Ultimately, MST is positioned to become a safe and effective treatment strategy for hematological malignancies, solid tumors, and potentially even nonmalignant disorders.

