Abstract
Background:
Hemophilia A is caused by coagulation factor VIII (FVIII) deficiency and increases bleeding risk during invasive procedures.
Objectives:
To investigate FVIII concentrate use and bleeding outcomes for invasive procedures after valoctocogene roxaparvovec gene transfer.
Design:
This manuscript presents post hoc analysis of the phase III GENEr8-1 trial.
Methods:
A post hoc analysis was performed for GENEr8-1, a global, single-arm, open-label, phase III trial that enrolled 134 adults with severe hemophilia A. FVIII activity and bleeding were evaluated after 2 years of follow-up. Invasive procedures were reviewed and categorized as major or minor. FVIII activity was measured with a chromogenic assay. Bleeding was self-reported by participants. Principal investigators completed questionnaires about perioperative management.
Results:
In total, 111 invasive procedures were performed in 65 participants during GENEr8-1 as of the data cut. Procedures performed with FVIII treatment included 33 minor and 11 major procedures. The remaining 67 invasive procedures were minor and performed without FVIII treatment. When considering these 67 minor procedures, 43/46 investigators completing the questionnaires reported that the gene-therapy-derived FVIII activity was sufficient for the type of procedure. Minor procedures performed without FVIII treatment were associated with participants’ higher mean endogenous FVIII activity (50.5 IU/dL) compared with major procedures (14.2 IU/dL) or minor procedures (16.4 IU/dL) performed with concomitant FVIII. Fourteen participants experienced 18 procedure-related bleeds (13 co-occurring with FVIII use). Participants who received FVIII treatment for procedure-related bleeds had numerically lower mean endogenous FVIII activity than those who did not receive FVIII treatment.
Conclusion:
Invasive procedures were safely performed in participants following treatment with valoctocogene roxaparvovec. The questionnaire responses from investigators generally suggest they used endogenous FVIII activity derived from valoctocogene roxaparvovec to inform clinical decisions in a manner comparable to exogenously administered FVIII, and more commonly prescribed supplementary FVIII concentrate in the peri-procedural period for participants with lower FVIII activity levels.
Introduction
Hemophilia A is an X-linked recessive bleeding disorder caused by coagulation factor VIII (FVIII) deficiency. 1 Severe hemophilia A, defined as FVIII levels <1 IU/dL, affects approximately 9.5 out of 100,000 males globally and is associated with an increased risk of spontaneous bleeding. 2 Currently, the standard of care for people with severe hemophilia A is prophylactic treatment with FVIII concentrate or bispecific antibodies that mimic activated FVIII function. 1 Despite the use of prophylaxis, on-demand treatment is still necessary for breakthrough and trauma-related bleeds or in advance of high-risk bleeding activities such as contact sports and some surgical procedures. 1 The need for frequent intravenous infusions can be burdensome and can have a negative impact on quality of life, and FVIII concentrate use is complicated by the risk of inhibitor formation.1,3,4
Valoctocogene roxaparvovec is an approved gene therapy for severe hemophilia A in the United States and Europe5,6 that employs an adeno-associated virus serotype 5 vector to transfer a B-domain-deleted FVIII gene cassette under the regulatory control of a liver-selective promoter. 7 Valoctocogene roxaparvovec was evaluated in phase I/II (NCT02576795)8 –12 and phase III (GENEr8-1; NCT03370913)13 –15 clinical trials. The GENEr8-1 trial was a single-arm, open-label study of 134 participants with severe hemophilia A designed to evaluate the safety and efficacy of valoctocogene roxaparvovec. Collectively, results at 1, 2, and 3 years post gene therapy demonstrated that a single infusion of valoctocogene roxaparvovec supported endogenous FVIII production and reduced bleeding and FVIII use compared with FVIII prophylaxis.13,14
As life expectancy has increased among people with hemophilia A, treatment for age-related complications—both related and unrelated to hemophilia—have become more frequent, increasing the need for invasive procedures.16,17 FVIII levels are an important consideration when individuals with hemophilia A need to undergo invasive procedures. 1 The World Federation of Hemophilia (WFH) has set generalized lower-limit guidelines for peak plasma FVIII levels according to procedure category. Recommendations for major procedures include preoperative FVIII levels of 60–80 IU/dL and postoperative FVIII levels of 30–40 IU/dL for 1–3 days with tapering of FVIII thereafter for up to 2 weeks. For minor procedures, WFH recommends preoperative FVIII levels of 40–80 IU/dL and subsequent FVIII levels of 20–50 IU/dL to maintain perioperative hemostatic support for an additional 1–5 days. 1 However, lower FVIII levels may be sufficient to enable hemostasis for certain procedures, and these guidelines may be conservative estimates that do not sufficiently acknowledge challenges with the availability of hemophilia A medication in some parts of the world. 18
Generally, the risk of bleeding for individuals with hemophilia A is inversely correlated with circulating FVIII activity. 1 Whether valoctocogene roxaparvovec may provide hemostatic control during and after invasive medical procedures is therefore of key interest to clinicians and people with hemophilia A. To address this need, we performed a post hoc analysis of the 134 participants from the GENEr8-1 trial to characterize invasive procedures that were performed after participants received gene therapy, including whether exogenous FVIII was used and if bleeding occurred.
Methods
GENEr8-1
Briefly, male participants ⩾18 years of age were eligible to enroll in GENEr8-1 if they had severe congenital hemophilia A (residual FVIII activity level ⩽1 IU/dL), had been on a prophylactic FVIII concentrate regimen ⩾1 year prior to enrollment, and were negative for FVIII inhibitors. Participants were given a single infusion of 6 × 1013 vg/kg of valoctocogene roxaparvovec via peripheral vein and continued their prophylactic regimen of FVIII for at least 4 weeks following infusion. After discontinuing prophylaxis, FVIII concentrate was used as needed. Endogenous FVIII activity was measured with a chromogenic substrate assay (CSA) throughout the study. The CSA lower limit of quantitation changed from 3.0 to 1.5 IU/dL between years 1 and 2. The study protocol was approved by the independent ethics committees or institutional review boards of all study sites, and all participants provided written informed consent prior to enrollment. The reporting of this study conforms to the TREND statement, 19 and further details about GENEr8-1 have been published previously.13,14
Post hoc analysis
Throughout the GENEr8-1 trial, principal investigators documented all medical procedures for the trial participants after valoctocogene roxaparvovec infusion. For this post hoc analysis, the medical procedures as reported by the principal investigators were then categorized as either non-invasive (e.g., dental cleaning or tattooing) or invasive by the study authors. Invasive procedures were further subcategorized as major (e.g., joint debridement) or minor (e.g., dental extraction, depending on the number and type of teeth) according to previously published criteria. 16 Briefly, major procedures included general invasive surgical procedures, orthopedic surgical procedures except for chemical- or radio-arthroscopic synovectomies, and dental procedures involving ⩾3 teeth. Study participants self-reported all bleeding episodes by submitting bleed logs, in which they additionally noted if the episode was related to a medical procedure. The endogenous FVIII activity measurement most proximal to the medical procedure or bleeding event was used for this analysis. Decisions regarding the management of invasive procedures with exogenous FVIII were made at the discretion of the principal investigator, and no criteria were defined or included as part of this study. In addition to the information that was captured in the electronic database, principal investigators were then sent a questionnaire to obtain further information on clinical decision-making regarding the use of exogenous FVIII in these cases. For some types of minor procedures in which there was minimal risk of bleeding, such as phlebotomy or SARS-CoV-2 antibody tests, principal investigators were not sent questionnaires regarding decisions on FVIII use. All results are described as mean ± standard deviation unless otherwise noted; no hypothesis testing was performed.
Results
Study population and procedures
There were 134 participants in the GENEr8-1 intent-to-treat population, defined as those who received a single dose of 6 × 1013 vg/kg valoctocogene roxaparvovec. 14 Of these, 77 participants underwent a total of 260 procedures prior to the 2-year data cut. Imaging procedures and electrocardiograms, which carry minimal bleeding risk, were excluded from the analysis, and of the remaining 148 procedures, 37 were categorized as noninvasive procedures (Figure 1). The presented analysis focused on the remaining 111 invasive procedures that were performed in 65 participants (Table 1). Concomitant FVIII treatment took place for 44/111 (39.6%) invasive procedures, 11 of which were categorized as major procedures and 33 as minor procedures. The 11 major procedures were performed for 7 participants (Table 2). The remaining 67/111 procedures were performed without concomitant FVIII treatment, and all were categorized as minor. The types of minor procedures performed with (Supplemental Table 1) or without (Supplemental Table 2) FVIII infusion are reported in the supplement. Minor dental surgeries, endoscopic procedures, and intra-articular joint injections were among those minor procedures performed both with and without FVIII treatment.

Procedure categorization and concomitant FVIII concentrate use. There were 77 participants who underwent a total of 260 procedures prior to the 2-year data cut. From these, 111 invasive procedures were performed in 65 participants.
Demographics of participants who underwent procedures after gene therapy with valoctocogene roxaparvovec.
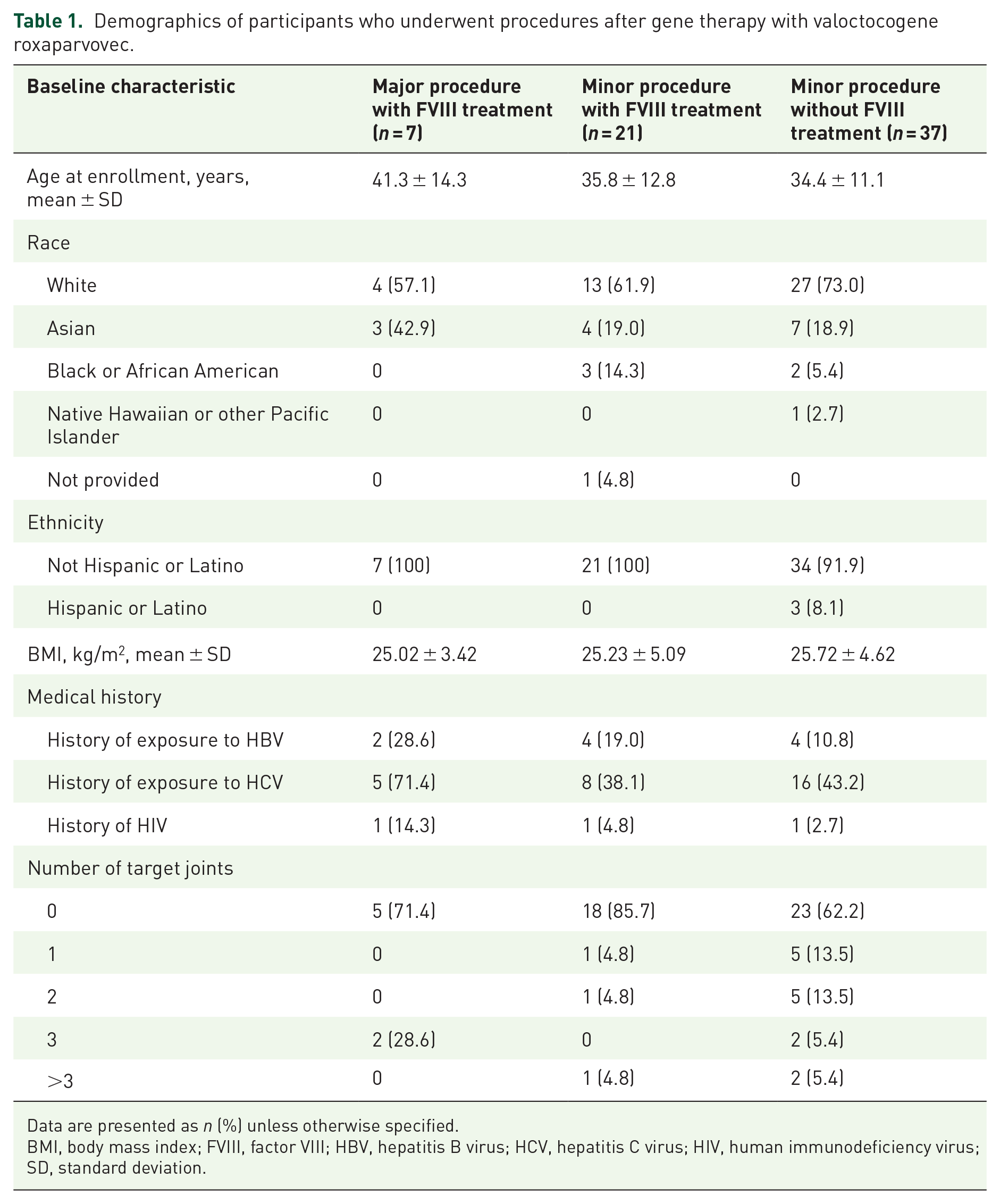
Data are presented as n (%) unless otherwise specified.
BMI, body mass index; FVIII, factor VIII; HBV, hepatitis B virus; HCV, hepatitis C virus; HIV, human immunodeficiency virus; SD, standard deviation.
FVIII activity, reported bleeds, and concomitant medications associated with major procedures.

The FVIII activity reported is the closest assessment recorded prior to the indicated procedure. The LLOQ for the CSA used to assess FVIII activity was 3.0 or 1.5 IU/dL.
CABG, coronary artery bypass graft; CSA, chromogenic substrate assay; FVIII, factor VIII; IV, intravenously; LAD, left anterior descending; LLOQ, lower limit of quantitation; OM2, obtuse marginal 2; PDA, posterior descending artery; PLB, posterior lateral branch; PO, orally; RSVG, reversed saphenous vein graft; tid, three times a day.
FVIII activity levels prior to medical procedures
For procedures that were managed with FVIII treatment, the participants’ mean endogenous FVIII activity level prior to the procedures was 14.2 IU/dL (range, <3.0–52.1 IU/dL) for major procedures and 16.4 IU/dL (range, <3.0–93.2 IU/dL) for minor procedures (Table 3). For procedures that were managed without FVIII treatment, the participants’ mean endogenous FVIII activity level prior to the procedures was 50.5 IU/dL (range, <3.0–255.7 IU/dL).
Perioperative FVIII management for invasive procedures.

FVIII, factor VIII; max, maximum; min, minimum.
Procedure-associated FVIII administration
As expected, the mean total dose of FVIII concentrate administered for major procedures (255.4 IU/kg; range, 102.8–538.2 IU/kg; Table 3) was numerically higher compared with minor procedures (67.2 IU/kg; range, 13.7–324.3 IU/kg). Furthermore, major procedures were associated with a mean of 8.8 FVIII concentrate infusions (range, 3–21 infusions) over a mean of 6.9 days (range, 2–14 days). Minor procedures were associated with a mean of 2.2 FVIII infusions (range, 1–13 infusions) over a mean of 1.8 days (range, 1–7 days). Of the 33 minor procedures managed with FVIII treatment, 25 (75.8%) were managed with a single dose. Adjuvant therapies such as antifibrinolytic agents (tranexamic acid and aminocaproic acid) were also used during 10 procedures, including 2 minor procedures without FVIII administration, 5 minor procedures with FVIII administration, and 3 major invasive procedures with FVIII administration.
Bleeding episodes and associated FVIII activity
There were 18 acute procedure-related bleeding episodes (16 from minor procedures and 2 from major procedures) reported by 14 participants. From these, 13 were managed with FVIII treatment, and 5 were untreated (Figure 2). Participants reported that most bleeding episodes (14/18) occurred within 48 h of the procedure, whereas 2/18 were delayed, and there were insufficient data to determine the timing of the remaining two bleeding episodes. The maximum number of bleeds associated with any single procedure was three bleeds that occurred for a minor procedure performed with FVIII treatment. The mean endogenous FVIII activity for participants who did not receive treatment for a bleeding episode was 60.4 IU/dL (range, 14.1–117.9 IU/dL; N = 5 bleeding episodes). The mean endogenous FVIII activity for participants who did receive treatment for a bleeding episode was 10.4 IU/dL (range, <3.0–45.2 IU/dL; N = 13 bleeding episodes). In these cases of procedures managed with exogenous FVIII, the FVIII activity levels reported refer to FVIII activity derived from only the gene therapy, measured by the CSA, most proximal to the bleeding episode and do not include any contribution of FVIII activity derived from the exogenous FVIII administration.

Mean FVIII activity and procedure-related bleeds by procedure category and concomitant FVIII concentrate use.
Investigator rationales for treatment decisions
Study investigators were given questionnaires to determine the reasons underlying their decision to supplement individuals undergoing minor or major invasive procedures with FVIII. While there were a total of 67 invasive procedures performed without FVIII treatment, questionnaires were not sent for some procedures that carried minimal bleeding risk. Study investigators completed 46/52 questionnaires that were sent for minor procedures with bleeding risk that were performed without FVIII treatment. In 43/46 questionnaires, the clinician deemed endogenous FVIII activity derived from valoctocogene roxaparvovec treatment to be sufficient for the given procedure type. In the 11 major procedures, 6 questionnaires were completed, with 5/6 investigators indicating that the participant’s FVIII levels achieved by the gene therapy were considered when making decisions about exogenous FVIII treatment and 1/6 suggesting that they were not considered.
We sought to examine these findings in the context of WFH guidelines for procedures performed in people with severe hemophilia A. The WFH recommends for minor procedures a minimum peak plasma FVIII level of 40 IU/dL preoperatively and 20 IU/dL postoperatively. During the GENEr8-1 trial, the participants’ FVIII activity level met WFH preoperative guidelines for 24/67 (35.8%) minor procedures, and they met postoperative guidelines for 42/67 (62.7%) minor procedures. 1 Notably, 22/67 minor procedures were performed for participants with a FVIII activity level below 20 IU/dL (range, 0–19.7 IU/dL), and from these, only one participant reported a bleeding episode with an associated FVIII level of 14.1 IU/dL. The 13 investigators who completed questionnaires related to these procedures indicated that the participant’s endogenous FVIII expression was sufficient for the procedure. For the remaining procedures, there were no completed questionnaires available.
Discussion
Individuals with severe hemophilia A have a high risk of bleeding when undergoing invasive procedures. While valoctocogene roxaparvovec supports endogenous FVIII production to prevent bleeding and reduce FVIII concentrate use in people with severe hemophilia A, it was unknown how these parameters affect invasive procedure-related outcomes. We used data from the GENEr8-1 trial population to investigate these relationships and present them in the context of current standard-of-care guidelines.
Decisions on whether to manage invasive procedures with FVIII treatment were left to the discretion of the principal investigators managing the participants. Most of the minor invasive procedures performed in participants of GENEr8-1 were managed without supplementary FVIII concentrate use, and similar types of procedures, such as dental, endoscopic, and joint-related procedures, were performed both with and without FVIII treatment. For the minor procedures that were managed with FVIII treatment, the respective participants generally had lower endogenous FVIII levels derived from the gene therapy. However, regardless of the participant’s gene-therapy-derived FVIII level, all major procedures were managed with FVIII treatment. Importantly, there were only 18 procedure-related bleeding episodes reported by the participants for all procedure types. Overall, the results suggest that after valoctocogene roxaparvovec infusion, invasive procedures can be safely performed with appropriate use of concomitant FVIII treatment as needed.
These findings on procedure-related bleeding outcomes in participants of GENEr8-1 contribute to the field by leveraging the continuous FVIII monitoring included as part of the GENEr8-1 study design to provide insight on real-world management decisions in relation to FVIII status. There were 43 minor procedures performed for participants with FVIII activity levels below 40 IU/dL. However, the majority of investigators still indicated that they felt the participant’s endogenous FVIII levels were sufficient for the respective minor procedure. Furthermore, of the 22 minor procedures performed for participants with FVIII activity levels below 20 IU/dL, only 1 experienced a procedure-related bleed. Of note, the FVIII levels reported in this manuscript were measured using the CSA; these values would be higher (approximately 1.5-fold) if measured with the one-stage assay. 13 Collectively, these outcomes reinforce that individualized decisions should take into account the participant’s health, medical history, FVIII level, procedure type, and bleeding risk.
Limitations
Whereas self-reported bleeding logs facilitated the large dataset analyzed here, the lack of clinician-reported bleeds introduces the possibility that some procedure-related bleeds were unreported or, conversely, that some nonrelated bleeds were misattributed to a procedure. The bleed logs also did not collect data on the type of bleed or the severity of the bleed. By extension, even though a bleed may have been related to a procedure, we cannot definitively attribute the cause to FVIII deficiency without clinician input. For example, some bleeds may have been expected based on the procedure type, such as the coronary artery bypass graft, even for individuals without hemophilia A. Finally, as the timing of FVIII infusions (pre- vs postoperative) was not included as part of our dataset, we are only able to report whether FVIII concentrate use occurred on the day of a procedure.
Conclusion
Invasive procedures can be safely performed with and without concomitant FVIII use in people with severe hemophilia A after treatment with valoctocogene roxaparvovec. Although most participants in the GENEr8-1 trial had sufficient endogenous FVIII activity to avoid additional FVIII supplementation for minor procedures, FVIII concentrate was used if necessary. Thus, valoctocogene roxaparvovec-induced endogenous FVIII production facilitates personalized healthcare. In most instances, the confluence of individual FVIII levels, participant-investigator discussions, and procedure type determined the use of FVIII concentrate for an invasive procedure.
Supplemental Material
sj-docx-1-tah-10.1177_20406207241304645 – Supplemental material for Outcomes and management of invasive procedures in participants with hemophilia A post gene therapy: a post hoc analysis of the GENEr8-1 phase III trial
Supplemental material, sj-docx-1-tah-10.1177_20406207241304645 for Outcomes and management of invasive procedures in participants with hemophilia A post gene therapy: a post hoc analysis of the GENEr8-1 phase III trial by Doris V. Quon, Jiaan-Der Wang, Michael Wang, Dominic Pepperell, Young-Shil Park, Gili Kenet, Johnny Mahlangu, Teh-Liane Khoo, Tara M. Robinson, Konstantia-Maria Chavele and Steven W. Pipe in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
We thank all study participants and their families, trial principal investigators, and study site personnel. We also thank members of all valoctocogene roxaparvovec development teams at BioMarin Pharmaceutical Inc. Medical writing support was provided by Tony Sallese, PhD, and M. Amin Ghane, PhD, of AlphaBioCom, a Red Nucleus company, and funded by BioMarin Pharmaceutical Inc. Jonathan Morton, PhD, of BioMarin Pharmaceutical Inc. provided project management support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
