Abstract
Background:
Late acute graft-versus-host disease (aGVHD) is a complication of allogeneic hematopoietic stem cell transplantation (allo-HSCT) with little data regarding treatment and outcomes. There is no standard treatment for gastrointestinal (GI) late aGVHD, especially for steroid-refractory (SR) GI late aGVHD. Vedolizumab, a monoclonal antibody inhibiting the migration of both naive and activated lymphocytes into the GI endothelium, has been verified to be effective for SR GI aGVHD.
Methods:
We retrospectively analyzed the clinical efficacy and safety of vedolizumab as the second line for SR GI late aGVHD in seven patients after allo-HSCT.
Results:
Four patients received two doses of vedolizumab infusion, while three patients received only one dose of vedolizumab infusion. The complete response and partial response rates were 57.1% (4/7) and 42.9% (3/7), respectively. No patient progressed to chronic GVHD during the period of follow-up. There was no severe adverse event related to vedolizumab.
Conclusion:
Our data suggest that vedolizumab is expected to ameliorate SR GI late aGVHD. Further data on the treatment timing, efficacy, and safety of vedolizumab are warranted in prospective clinical trials.
Introduction
Acute graft-versus-host-disease (aGVHD) is a major cause of non-relapse rate (NRM) after allogeneic hematopoietic stem cell transplantation (allo-HSCT), especially severe gastrointestinal (GI) accompanied by prolonged and debilitating illness. The incidence of grade II–IV aGVHD ranges from 40% to 70% after allo-HSCT.1,2 The essence of aGVHD is the immune response caused by activation of alloreactive donor T lymphocytes against the recipients, and the ensuing host tissue damage. 3 Limited treatment options for patients with aGVHD consist of ruxolitinib, calcineurin inhibitors (CNIs), and glucocorticosteroids, but about half of patients responded to the therapy and one-third of patients maintained durable responses.4–6 Systemic aGVHD was mainly derived from inflammation and damage of intestinal system, 7 with high mortality due to steroid-refractory (SR). There is a critical need for novel effective therapies for the treatment of aGVHD, particularly in GI aGVHD.
Vedolizumab, a monoclonal antibody targeting α4β7 integrin expressed on the activated T lymphocytes and blocking its binding to the homing receptor, especially on GI mucosa,8,9 was developed for the treatment of inflammatory bowel disease, both in ulcerative colitis (UC) 10 and Crohn’s disease (CD). 11 It has been revealed that expression of α4β7 on immunocompetent donor T lymphocytes played a vital role during the process of GI aGVHD in murine allogeneic bone marrow transplantation models. 12 Hence, targeting α4β7 to block the homing of T lymphocytes to the inflamed GI tissue may be a promising therapeutic for the treatment of GI aGVHD. There have been several types of research about vedolizumab as second- to fourth-line therapy for SR GI aGVHD in recent years. Fløisand et al. 13 described a series of 29 patients who received vedolizumab for the treatment of SR aGVHD. The overall response rate (ORR) of 6–10 weeks and overall survival (OS) at 6 months were 64% and 54%, respectively. Another large retrospective multicenter research reported the outcome of 29 patients with SR GI aGVHD after the administration of vedolizumab. 14 The ORR was 79.3% (23/29), including 27.6% (8/29) complete response (CR) and 51.7% (15/29) partial response (PR). The results showed that early application of administration may be related to successful outcomes.
Late aGVHD, by definition, is defined as the persistent, recrudescent, or de novo manifestations of aGVHD after 100 days of allo-HSCT. 15 The pathogenesis of late aGVHD is still less clear, and we have limited knowledge of it. The study assessing the outcomes of 75 patients with late aGVHD after allo-HSCT revealed that the persistent late aGVHD was relative to chronic GVHD (cGVHD) and NRM, as well as poor OS. 16 The current treatment of late aGVHD remains to be steroid-based therapy. In view of the similarity of early aGVHD and late aGVHD, we therefore hypothesized whether vedolizumab would be a hopeful agent for late aGVHD.
Methods
Seven patients with late SR GI aGVHD at Affiliated Cancer Hospital of Zhengzhou University provided written informed consent forms for off-label use of drugs between May 2021 and June 2023. Vedolizumab has been approved for the treatment of UC and CD. The patients aged 18–70 years and diagnosed with late SR GI aGVHD after allo-HSCT were eligible. Patients with advanced primary disease, active infections, and dysfunction of multiple organs were excluded. All patients met the criteria of grade III–IV intestinal late aGVHD, as defined by the Gluckman criteria and 2014 National Institutes of Health Consensus Criteria (NIH-CC).15,17 Late aGVHD was further classified as de novo, recurrent, and persistent. 16 SR aGVHD was defined as progressive disease or lack of at least a PR after seven consecutive days of primary treatment with at least 1 mg/kg methylprednisolone or equivalent daily dose in combination with CNI, or lack of a CR after 14 consecutive days of primary treatment with at least 1 mg/kg methylprednisolone or equivalent daily dose for GI aGVHD. After initial dose of vedolizumab, patients were followed up for a median of 140 days (range, 62–550 days).
Diagnosis of GI aGVHD was made by biopsy or clinical basis. Histological grading was based on the system proposed by Lerner et al. 18 Clinical staging was determined using the criteria proposed by Glucksberg et al. 17 on the basis of both stool volume and abdominal pain.
The intravenous doses and schedule of vedolizumab were in accordance with the treatment of UC and CD. The patients were administrated with 300 mg vedolizumab at week 0, followed by infusions at weeks 2 and 6 on the basis of response and disease status. But no patient in the series took an additional 300 mg at week 6. Assessment of response was categorized as CR, PR, and no response. A CR to vedolizumab was defined as resolution of signs and symptoms of aGVHD, that is, the grade of aGVHD reduced to 0 at 4 weeks from the administration of the first dose. A PR was defined as at least one stage improvement of aGVHD by 4 weeks after the first dose of vedolizumab.
Adverse effect of vedolizumab was assessed by common infectious complications based on clinical microbiological testing. The evidence of infection involved in the culture of blood, bronchoalveolar lavage, and tissue, as well as routine virus testing, including cytomegalovirus (CMV), Epstein-Barr virus (EBV), BK virus (BKV), and adenovirus by polymerase chain reaction or next-generation sequencing from the initial treatment of vedolizumab to 30 days after the treatment.
Survival data, including date of death and cause of death, was collected and Kaplan–Meier survival curves were calculated at 6 and 12 months following the first dose of vedolizumab by SPSS 18.0 (PASW Statistics 18.0).
The ethical approval was obtained by the Institution Review Board of the Affiliated Cancer Hospital of Zhengzhou University (2022-174-002), in agreement with the Declaration of Helsinki of 1975.
Results
Demographics and disease characteristics
Seven female patients who received vedolizumab for treatment of SR GI late aGVHD were included, with a median age of 24 years (range, 16–53 years). The baseline characteristics of the patients in the study are summarized in Table 1. Four patients underwent allo-HSCT for acute myeloid leukemia, three from haploidentical HSCT (haplo-HSCT) donors and one from a 10/10 unrelated donor HSCT. Two patients were indicated for haplo-HSCT due to acute lymphoblastic leukemia, while one patient underwent haplo-HSCT for myelodysplastic syndromes (MDSs). All the patients received myeloablative conditioning regimens, followed by peripheral blood stem cells (PBSCs). The seven patients were administrated with posttransplant cyclophosphamide and CNI, half-dose ruxolitinib, and mycophenolate mofetil, as well as antithymocyte globulin for GVHD prophylaxis.
Baseline characteristics of patient population.

ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ATG, antithymocyte globulin; CR, complete remission; CsA, Cyclosporin A; GVHD, graft-versus-host disease; FK-506, tacrolimus; MDS, myelodysplastic syndrome; MMF, mycophenolate mofetil; MUD, matched unrelated donor; NR, non-remission; PBSC, peripheral blood stem cell; PTCy, posttransplant cyclophosphamide.
GVHD characteristics
The GHVD characteristics are shown in Table 2. Late aGVHD appeared at a median of 143 days (range, 113–420 days) after allo-HSCT. All patients experienced grade III–IV GI late aGVHD with watery stool, and one patient had hematochezia. One patient had recurrent GI late aGVHD with a history of grade II GI aGVHD, while six patients had de novo GI late aGVHD. Six of seven patients had sign of abdominal pain. The reasons for GI late aGVHD were insufficient concentration of CNI in three patients, donor lymphocyte infusion (DLI) after relapse in two patients, and intestinal infections induced by improper diet in two patients, respectively. Three patients had only isolated intestinal involvement without the symptoms of other organs. One patient had skin grade I aGVHD and liver grade II aGVHD and three patients had liver grade II aGVHD. All patients received glucocorticosteroids as first-line therapy of GI late aGVHD based on CNI and mycophenolate mofetil. Vedolizumab was administrated after a median 8 days of systemic glucocorticosteroid treatment and as second-line therapy to all patients.
GVHD characteristics and vedolizumab therapy as second line.

aGVHD, acute graft-versus-host disease; allo-HSCT, allogeneic stem cell transplantation; CNI, calcineurin inhibitor; CR, complete remission; DLI, donor lymphocyte infusion; GI, gastrointestinal; GVHD, graft-versus-host disease; IS, immunosuppression; PR, partial remission or partial response.
Vedolizumab response
Four patients received two doses of vedolizumab infusion, while three patients received only one vedolizumab infusion. No infusion reaction was observed. The response to treatment was evaluated for the patients at the 8 weeks after the first dose of vedolizumab. The median response time from the initiation of vedolizumab was 6 days (range, 5–16 days). An overall response was reported in seven patients on day 28. The CR and PR rates were 57.1% (4/7) and 42.9% (3/7), respectively (Table 2). One patient was discharged from hospital after 8 days of the first dose of vedolizumab. One patient experienced from hematochezia to yellow mushy stool at 6 days after the initial dose of vedolizumab. Glucocorticosteroids were discontinued in all seven patients with a median time of 21 days (range, 7–30 days). No patient progressed to cGVHD during the period of follow-up.
Infection complication
The most frequently reported complications were infections in all patients (100%), including four patients with CMV-related infections, of which two participants had CMV retinitis, two had CMV viremia, one had CMV enteritis, and three patients with BKV infections, of which two had BKV-related hemorrhagic cystitis and one was BKV viremia, and three patients with pulmonary infection, of which one was COVID-19 positive and two had fungal pneumonia. One patient experienced staphylococcal infection, while one patient experienced Candida guilliermondii enteritis (Table 3).
Infection complication and survival.
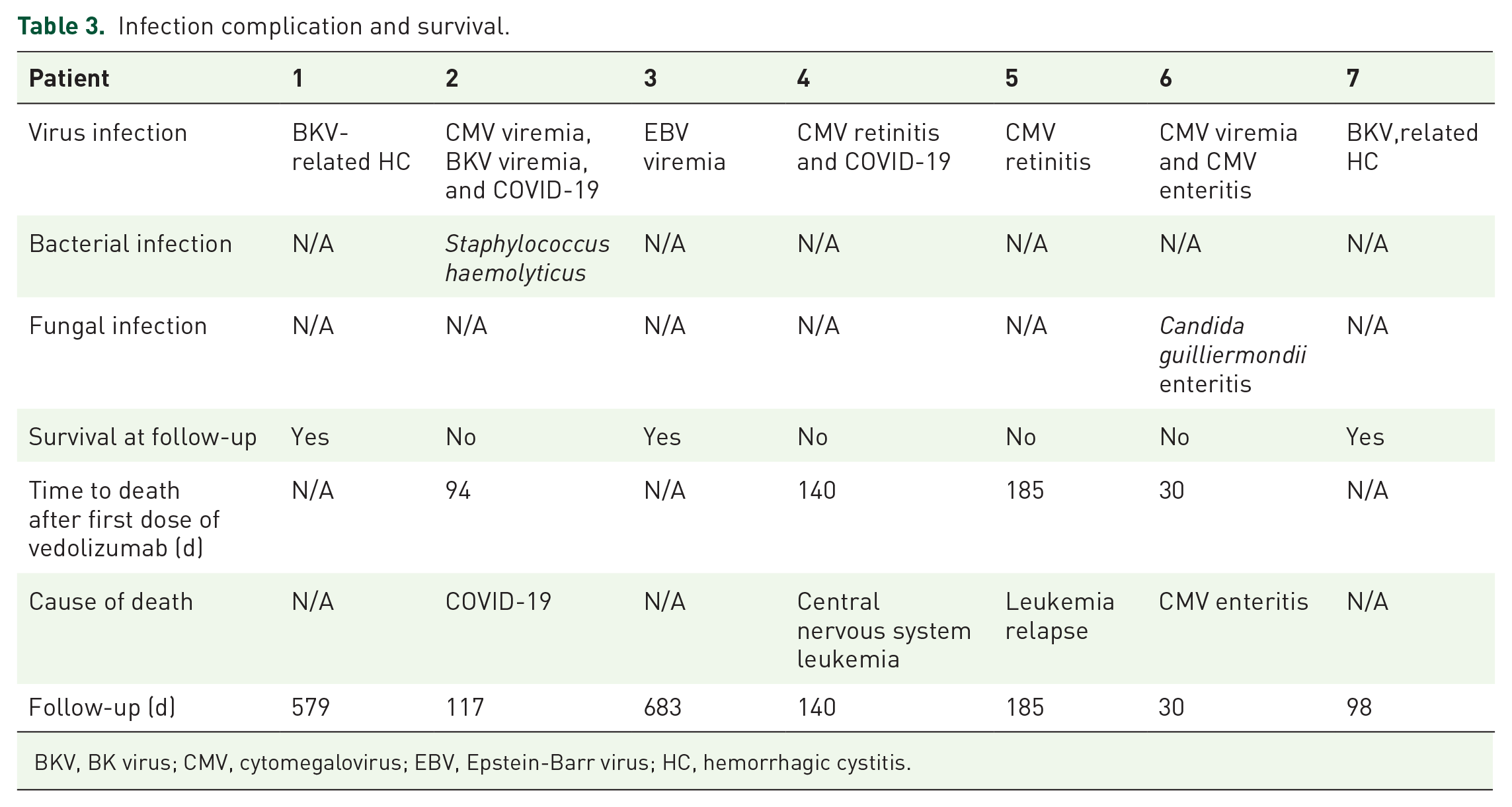
BKV, BK virus; CMV, cytomegalovirus; EBV, Epstein-Barr virus; HC, hemorrhagic cystitis.
Overall survival
Up to October 14, 2023, three patients were alive without complication. One died from leukemia relapse and one died from central nervous system leukemia. One died from CMV enteritis. The patient with recurrent GI late aGVHD died due to COVID-19 during the pandemic (Figure 1). Four deaths occurred in 30, 94, 140, and 185 days of starting vedolizumab. OS of patients after the initial dose of vedolizumab at 12 months was 34.29% (Figure 2). At the last follow-up, one patient was on two immunosuppressive regimens.

Swimmers plot of patient outcomes following vedolizumab treatment (n = 7). Each line represents a patient analyzed. Overall response was defined as CR and PR, indicated with green and pink squares. Death and life of patients before the final follow-up are denoted with a solid black circle and red triangle at the end of each line, respectively. The causes of death are presented by colored diamonds: blue diamond as CMV enteritis, orange diamond as leukemia relapse, purple diamond as CNS leukemia, and yellow diamond as COVID-19.

Overall survival (OS) of patients after the initiation of vedolizumab, estimated by Kaplan–Meier. OS was defined as the time from the initiation of vedolizumab to the date of death or the last follow-up.
Discussion
There was rare report that vedolizumab was a second-line treatment of SR GI late aGVHD. In the previous reports, vedolizumab was administrated to patients with SR GI early onset aGVHD, and showed prompt response. In the cases, the dosage of vedolizumab was based on previous experience in SR GI aGVHD within 100 days after allo-HSCT. All seven patients responded to the treatment on day 28 after the initial dose of vedolizumab, four were CR, and three were PR. So, vedolizumab was a promising regimen for patients with SR GI late aGVHD.
Late aGVHD was defined as features of aGVHD after 100 days of allo-HSCT, limited to the symptoms of skin, liver, and GI without sclerotic change by NIH-CC. 15 The clinical presentation and alteration in angiogenic factors of late aGVHD were similar to aGVHD, distinct from cGVHD. 19 The treatment of late aGVHD is not very clear, though the management of aGVHD and cGVHD has been well-defined.20–25 Steroid, as the first-line treatment of GVHD, is ineffective in 30%–50% of patients with all types of GVHD. That hinted at the need for innovative therapeutics. Though the biology of early onset of aGVHD and cGVHD has been well researched, whether the biology of late aGVHD is similar to that of early aGVHD and cGVHD is still unknown. Univariate analysis found that malignant disease, human leukocyte antigen (HLA) mismatch, and a history of classical aGVHD were related to late aGVHD. Multivariable analysis showed a history of classical aGVHD grades II–IV and PBSC grafts had significant associations with late aGVHD. 26 What was consistent with the previous study was that the patient with recurrent late aGVHD had occurred grade II GI aGVHD within 100 days after allo-HSCT. Furthermore, six patients underwent haplo-HSCT and one patient underwent unrelated donor HSCT. A retrospective cohort evaluated the clinical manifestations and outcomes of 75 patients with late aGVHD. The results showed that compared with recurrent and de novo late aGVHD, persistent late aGVHD was associated with worse OS and NRM. That is, recurrent and de novo late aGVHD seemed to be more sensitive to therapy.
Effective prevention and treatment of aGVHD, especially for GI aGVHD, is a crucial aim for patients undergoing allo-HSCT, in particular with haplo-HSCT or unrelated donor HSCT. In past decades, some improvements have been achieved to reduce the risk and improve the prognosis of aGVHD, which is driven by donor selection, HLA typing, as well as T-cell depletion in vivo and ex vivo. However, what we need is actual innovation in agents to directly prevent or treat aGVHD. Ruxolitinib has been the first agent by the Food and Drug Administration (FDA) for treatment of SR aGVHD. In phase III trials REACH2 (NCT02913261), 309 patients underwent randomization to ruxolitinib group or the control group. The ORR at 28 days was 62% versus 39%, and durable ORR at day 56 was 40% versus 22% in the ruxolitinib and the control cohorts, respectively. 4 Furthermore, it revealed that the manifestations of aGVHD gave credit to donor T lymphocytes sequential relocation to host secondary lymphoid tissue followed by target organs in the mouse model. 27 The study had shown that α4β7 expressed on donor T-cells was responsible for the development of GI GVHD in comparison to patients without GVHD or with skin GVHD. 28 Mucosal addressing cellular adhesion molecule 1 (MAdCAM-1), the counter receptor of α4β7, plays a critical role in the homing of T lymphocytes to the intestinal tract. Therefore, targeting α4β7 could be a promising method for treatment of GI aGVHD. Vedolizumab, a humanized monoclonal antibody, inhibits alloreactive lymphocyte migration to the GI by specifically recognizing the α4β7 integrin expressed on lymphocytes followed by blocking the interaction of homing T lymphocyte with MAdCAM-1. The results of SR sever GI aGVHD treated with vedolizumab showed that the ORR was 79%, with a CR of 28% and a PR of 52%. 14 In the second-line therapy, an ORR rate was 100%, with 54% of patients achieving CR, but only 6.25% of CR for patients treated with vedolizumab in the later aGVHD process. What the findings suggested that early treatment with vedolizumab might be related to a higher likelihood of therapeutic response. In addition, a phase Ib, open-label study evaluated the tolerance, safety, and pharmacokinetics of vedolizumab for the prevention of GVHD in adults undergoing allo-HSCT. The results showed no safety concerns were observed when vedolizumab was used as GVHD prophylaxis, with a low incidence of overall grade III–IV aGVHD and lower-intestinal aGVHD. 29
As for previous pediatric experience with vedolizumab for GI late aGVHD in two patients, one patient did not respond to vedolizumab for second-line treatment, while the other one obtained CR after seven doses vedolizumab as third-line or later. At present, there is less experience in vedolizumab for SR GI late aGVHD. In our study, seven patients had GI late aGVHD and the major causes focused on insufficient immunosuppression, DLI, and intestinal infection. As second-line therapy, what was different from previous studies treating GI aGVHD was that the dose of vedolizumab was one or two in our study, less than in previous studies.14,30,31 The ORR of the study achieved 100% in the study, superior to those of previous studies and ruxolitinib.4,14 Small sample size might take the lead role. To my mind, the difference in treatment may be due to a later time of late aGVHD for the patient whose immunity was reconstituting. Moreover, early administration of vedolizumab was relevant to better effect. Another possibility was that additional immunosuppression, such as CNI and mycophenolate mofetil had contributed to the response in GI GVHD, hence resulting in requirement of a lesser dose of vedolizumab. However, whether it is related to different mechanisms deserves further study.
In terms of the infection, all patients had viral infections, including CMV, EBV, and BKV. But BKV-related hemorrhagic cystitis of two patients and EBV viremia of one patient occurred earlier than GI late aGVHD. CMV-related infection suffered in four patients and occurrence time of one patient prior to development of GI late aGVHD. In other words, the most common vedolizumab-related infection was CMV infection (3/7, 42.9%). A retrospective, multicenter study analyzed the outcomes of patients with SR GI GVHD treated with vedolizumab. The results showed that CMV reactivation was the most frequent (16/26, 61.5%). 14 A multicenter, randomized controlled trial to determine the efficacy and safety of mesenchymal stromal cells (MSCs) combined with basiliximab as the second-line therapy for SR aGVHD exhibited that CMV infection occurred in 82.3% of the MSC-basiliximab group and 76.9% of the basiliximab alone group. 32 Three patients were alive at the end of follow-up, two patients died from leukemia, while two died of infections from both COVID-19 and CMV enteritis. None was GVHD-related death. This study had several limitations, including lower number of samples and shorter follow-up. In addition, it was a retrospective study. Further studies with prospective studies and large sample sizes are needed to evaluate the feasibility of vedolizumab in the treatment of SR GI late aGVHD in the future. In conclusion, our experience provides valuable insights into the potential efficacy and feasibility of vedolizumab for SR GI late aGVHD after allo-HSCT.
