Abstract
Background:
β-Thalassemia is an inherited blood disorder requiring lifetime management of anemia and its complications.
Objective:
This study aimed to determine the indirect costs and humanistic burden of β-thalassemia.
Design:
A systematic literature review was conducted.
Data sources and methods:
Searches were conducted in Embase, MEDLINE, MEDLINE In-Process, and EconLit (November 1, 2010, to November 25, 2020). Studies reporting indirect costs and health-related quality of life (HRQoL) for patients with β-thalassemia were eligible.
Results:
Seventy-five publications were included. Mean annual days lost due to transfusion-related absenteeism ranged from 15.6 to 35 days. Patients spent a mean of 592 min (standard deviation (SD): 349) daily on disease management on transfusion days and 91 min (SD: 221) daily on non-transfusion days. Patients with non-transfusion-dependent β-thalassemia (NTDT) showed worse HRQoL versus those with transfusion-dependent β-thalassemia (TDT) on the 36-item Short Form Health Survey (75.8 vs 66.5; p = 0.021). Caregivers of patients with TDT had more severe stress compared with patients (20.17 vs 18.95; p = 0.006), as measured by the standardized Cohen Perceived Stress Questionnaire.
Conclusion:
TDT is associated with substantial indirect costs and caregiver burden, and NTDT is associated with worse HRQoL. There is an unmet need for novel treatments in both TDT and NTDT that minimize patient and caregiver burden.
Introduction
Beta-thalassemia (β-thalassemia) is an inherited blood disorder characterized by anomalies in the synthesis of the β chains of hemoglobin. The pathophysiological hallmark of β-thalassemia is ineffective erythropoiesis, leading to anemia and abnormal iron metabolism. Severity ranges from clinically asymptomatic thalassemia to significant anemia requiring regular red blood cell (RBC) transfusions. 1
β-thalassemia can be broadly categorized into transfusion-dependent thalassemia (TDT) and non-transfusion-dependent thalassemia (NTDT). Patients with TDT receive regular RBC transfusions to prevent the life-limiting consequences of severe anemia but experience the side effects of chronic transfusions, namely transfusion-associated infections and damage caused by iron overload to end organs (including the liver, heart, and endocrine glands).1,2 Iron overload can lead to cardiac events, which are the primary cause of death in TDT patients in countries with insufficient monitoring and/or inadequate availability of iron chelation therapy (ICT). 3 In NTDT, compensatory extramedullary erythropoiesis is common, leading to enlargement of the spleen, bone deformities, osteoporosis, and formation of erythropoietic masses in the spleen, liver, lymph nodes, chest, and spine. Treatment is aimed at symptom management. As hypersplenism may worsen anemia, splenectomy is common in NTDT and may lead to an increased risk of thrombotic events. ICT may be ultimately needed due to iron overload from increased gastrointestinal absorption of iron or from occasional transfusions. 1
Significant progress has been made in the clinical care of patients with β-thalassemia, including treating patients earlier in the disease course, increased adherence to therapy, and improved management of iron overload and other complications.4,5 As a result, many patients experience prolonged complication-free periods and longer survival.6,7 However, this improvement in survival is accompanied by significant ongoing healthcare needs, specifically frequent blood transfusions and ICT used to manage iron overload. This can translate to a substantial impact on patient- and caregiver-related indirect burden of treatment and related complications, indirect costs related to hospital admissions, patients’ work productivity and absenteeism, and health-related quality of life (HRQoL).8,9 Treatments that reduce disease severity through decreasing transfusion and ICT burden may help improve HRQoL and indirect costs. To obtain a clear understanding of the indirect burden associated with β-thalassemia, a systematic literature review (SLR) of studies on indirect costs and overall humanistic burden was conducted. This is the first SLR investigating indirect burden in all β-thalassemia populations.
Methods
The SLR was conducted in accordance with the quality standards required by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 10 and the Cochrane Handbook for Systematic Reviews of Interventions; a completed PRISMA checklist can be found in the Supplemental Information. 11 The SLR was not registered. Electronic databases (Embase, MEDLINE and MEDLINE In-Process, and EconLit via Ovid SP) were systematically searched to identify suitable peer-reviewed records reporting on indirect costs and humanistic burden in β-thalassemia. The search was limited to records published from November 1, 2010 to November 25, 2020, in the English language, without geographical restriction. Search strategies included free-text and controlled vocabulary terms (Supplemental Information 1 and 2). Gray literature searches included proceedings from recent meetings (January 2018 to March 2020) of the Academy of Managed Care Pharmacy, American Society of Clinical Oncology, American Society of Hematology, European Hematology Association, and International Society for Pharmacoeconomics and Outcomes Research (last accessed on January 28, 2019). Researchers also examined the bibliographies of systematic reviews and/or meta-analyses that reported on β-thalassemia and were published between January 2018 and November 2020 to identify additional relevant studies.
The titles and abstracts from the searches were imported to EndNote X9 software (Clarivate, London, UK), and duplicates were removed. The search results were uploaded to Distiller Systematic Review software (Evidence Partners, Ottawa, ON, Canada), and the titles and abstracts were evaluated using predefined inclusion and exclusion criteria (Supplemental Information 3). Full-text articles from the relevant abstracts that met these criteria were retrieved and examined. Title, abstract, and full-text screenings were conducted by two independent researchers, and any discrepancies were resolved by a third researcher. Extraction was performed by one researcher using an approved template and validated by a second researcher. Any discrepancies were resolved by a third, senior researcher. All costs were converted to United States dollars (USD or $) using conversion rates from August 2021 (Supplemental Information 4). For costs not reported in USD, purchasing power parity (PPP) was calculated using the PPP Calculator (https://pppcalculator.pro/) and used to assess the economic impact on the patient household as a percentage of per capita gross domestic product (GDP) (Supplemental Information 4).
In our analysis, we categorized all patients with transfusion dependency as having TDT, alongside those with β-thalassemia major undergoing regular blood transfusion. Patients categorized as having NTDT included those who were transfusion independent or those who had β-thalassemia intermedia. We also included a mixed group that included patients with TDT and NTDT who were regularly and intermittently transfused, and an unclear group composed of patients without a clear transfusion dependency status (Supplemental Information 5).
Indirect burden was defined as the economic cost endured due to work productivity loss, absenteeism/presenteeism, transportation cost and time burden due to scheduling medical/transfusion appointments, traveling for appointment(s) and work impairment. Time burden was reported in terms of the amount of time spent in a healthcare setting or with other activities to facilitate regular treatments. Indirect burden due to transfusion was reported as time lost due to treatments and other treatment-related indirect expenses.
Results
Study and patient characteristics
A total of 2387 records, including 865 duplicates and 1522 abstracts and titles, were identified. Of these, 250 publications were selected for full-text review; 185 records were excluded, and 10 from the gray literature searches were included. Following screening, 75 records were included, of which 69 were full-text publications and 6 were conference abstracts (Figure 1; Supplemental Information 6).

PRISMA diagram.
Most studies had a cross-sectional study design (n = 55 studies); nine were economic studies reporting cost data, six were prospective studies, four were retrospective studies, and one reported patient-reported outcomes from a randomized controlled trial.
Data sources across studies largely included medical records (n = 34), followed by questionnaires (n = 17), interviews (n = 13), interviews combined with questionnaires (n = 3), survey data (n = 1), and registry data (n = 1). The remaining studies did not report their data sources (n = 6).
The review identified studies conducted in various geographical locations, including Europe (n = 14; Greece, n = 7; Italy, n = 3; United Kingdom, n = 3; and France, n = 1), Asia (n = 15; including Pakistan, n = 6; India, n = 5; Thailand, n = 3; and Sri Lanka, n = 1), North America (n = 3; including the United States, n = 1; Canada, n = 1; and the United States and Canada, n = 1), and the Middle East (n = 33; including Turkey, n = 3; Iran, n = 24; Saudi Arabia, n = 3; Egypt, n = 1; and Lebanon, n = 2). Nine studies were conducted in multiple locations, and one study did not report its location. Studies were conducted in adults and adolescents ⩾10 years of age (n = 43), a mixed population of adults and children (n = 27), caregivers/parents (n = 3), or unspecified populations (n = 2).
Across studies, transfusion dependency was categorized as TDT (n = 57) and NTDT (n = 7). Patients were also categorized in our study as a mixed group (n = 7) and an unclear group (n = 8). Of the publications that focused on TDT populations (n = 57), not all publications specified patients’ status of transfusion dependency. Therefore, our review provides information on TDT status in cases where TDT was reported by the authors (n = 44) or could be inferred by the context (n = 13). NTDT was defined as individuals who received ⩽5 RBC units during the last 24 weeks (n = 2), those who were transfusion and iron chelation independent (n = 2), patients who had undergone hematopoietic stem cell transplantation (HSCT) and were thus transfusion free (ex-β-thalassemia; n = 2), and patients who were β-thalassemia minor (n = 1; Supplemental Information 7).
Indirect burden was reported across 14 publications, all reporting on TDT only. Most full-text publications reported HRQoL outcomes (n = 59), of which the majority were reported in TDT (n = 42), followed by NTDT (n = 9), and mixed transfusion dependency (n = 7). Five reported unclear dependency, and four reported HRQoL outcomes for multiple transfusion dependency (TDT, NTDT, and mixed) (Supplemental Information 6).
Indirect burden
The SLR identified 14 unique studies that reported indirect burden for patients with β-thalassemia (Table 1).12–24 In children, mean (standard deviation (SD)) school days lost per patient annually due to treatment-related sick leaves ranged from 13.5 (1.7) 13 to 29.9 (18.5) days/year 23 as reported in India. A study from the Middle East reported a difference in mean time lost to treatment in pediatric patients (35 (48.1) h/month) and adult patients (19.0 (31.4) h/month). 20 Among caregivers of patients with β-thalassemia, an average of 19 days (12.7) were lost to absenteeism due to TDT annually in India. 23 The following sections describe in more detail the studies focusing on productivity loss, lost opportunities, absenteeism, and time burden.
Indirect burden results—transfusion-dependent population.

BT, blood transfusion; BTIC, blood transfusion and iron chelation; EOS, end of study; HSCT, hematopoietic stem cell transplantation; ICT, iron chelation therapy; INR, Indian rupee; IQR, interquartile range; IV, intravenous; NR, not reported; PKR, Pakistani rupee; PRO, patient-reported outcome; RBC, red blood cell; SD, standard deviation; SE, standard error; TDT, transfusion-dependent thalassemia; THB, Thai baht; WPAI, Work Productivity and Activity Impairment.
Productivity loss due to lost opportunities
Productivity loss was reported as the average cost of lost opportunities (definition not provided in the source study) across geographical locations and as costs of productivity loss stemming from absenteeism for management of disease. One study from Iran estimated total indirect annual mean costs in TDT as $573.5/person (cost base year: 2015 18 ; Table 1). This indirect mean cost included $356.2/patient as the average annual cost of lost opportunities due to TDT. The cost of lost opportunities due to premature death was found to be the highest in the 31–40 years of age group ($457.9) for patients and the 0–10 years of age group ($436.7) for patients’ families (cost base year: 2015). The cost of lost welfare or distress caused by TDT due to pain and suffering was $1360.5/patient/year (cost base year: 2015). 18
Similarly, one study from Thailand reported $229.6 as the average annual cost due to productivity loss, which accounted for 24% of the total treatment cost ($950; cost base year: 2005). 19
Productivity loss due to absenteeism
A study from Thailand estimated the indirect costs of absenteeism due to HSCT compared with blood transfusions combined with ICT. 15 In the first 2 years following HSCT, the total productivity loss due to absenteeism for patients with TDT was $2315.68. However, the productivity losses due to absenteeism for blood transfusion and ICT were reported to decrease to $573.06 in the follow-up years after HSCT (cost base year: 2005). 15 A higher average productivity loss for HSCT was reported in a study from France that estimated indirect costs of HSCT versus gene therapy: $16,394.7 versus $8,853.9 per patient per 2 years (cost base year: 2017). 22 Another Thai study reported productivity losses due to doctor’s visits as $820.15/year (cost base year: 2009 17 ; Table 1).
Time lost due to absenteeism
In addition to work productivity loss due to absenteeism, studies reported treatment-related average time lost across different patient groups (Table 1). In children, the mean school days lost per patient annually due to TDT treatment-related sick leave ranged from 13.5 to 35 days (as reported in India 13 and the Middle East 20 ); in adults, the mean time lost ranged from 17.7 to 19 days (as reported in India 13 and the Middle East 20 ). Among caregivers of patients with β-thalassemia, an average of 19 days were lost to absenteeism due to TDT annually in India. 23
Time burden
Patients with TDT spent 592 min (SD: 349) daily (estimated in the United Kingdom, the United States, Italy, and Germany) on transfusion days, and 91 min (SD: 221) daily on non-transfusion days, on activities including undergoing transfusion, cross-matching blood, arranging childcare, and arranging insurance payments. 24 Patients in the United States spent more time (mean: 164 min) each day on TDT management than patients in the United Kingdom (mean: 107 min), with a major difference due to managing insurance payments in the United States. 24 Another study in Middle Eastern patients with TDT reported a decrease in time lost to therapy for daily activities from 30.1 mean hours/month at baseline to 3.2 mean hours/month at the end of treatment with deferasirox (vs other ICT treatments 20 ; Table 1).
Transport and food costs for therapy
The annual travel expenses for transfusions accounted for 21.6% of all treatment expenditures incurred by patients with TDT in India. 13 In Iran, the cost of travel to and from clinics per year for patients with TDT ranged from $155.8 14 to $205. 18 In Pakistan, the mean transport and food costs per TDT treatment session were estimated at $4.6 and $1.9, respectively 9 (Table 1).
HRQoL
Overall, 66 studies reported HRQoL outcomes for patients with β-thalassemia; 60 were full-text publications, and 6 were conference abstracts (Table 2).9,16,21,24–43 The 36-item Short Form Health Survey (SF-36) was the most frequently reported HRQoL measure (n = 29), and studies reported considerable impairment across all domains, especially physical functioning, emotional functioning, vitality, and general health.
HRQoL results—TDT/NTDT/mixed transfusion dependency population. a
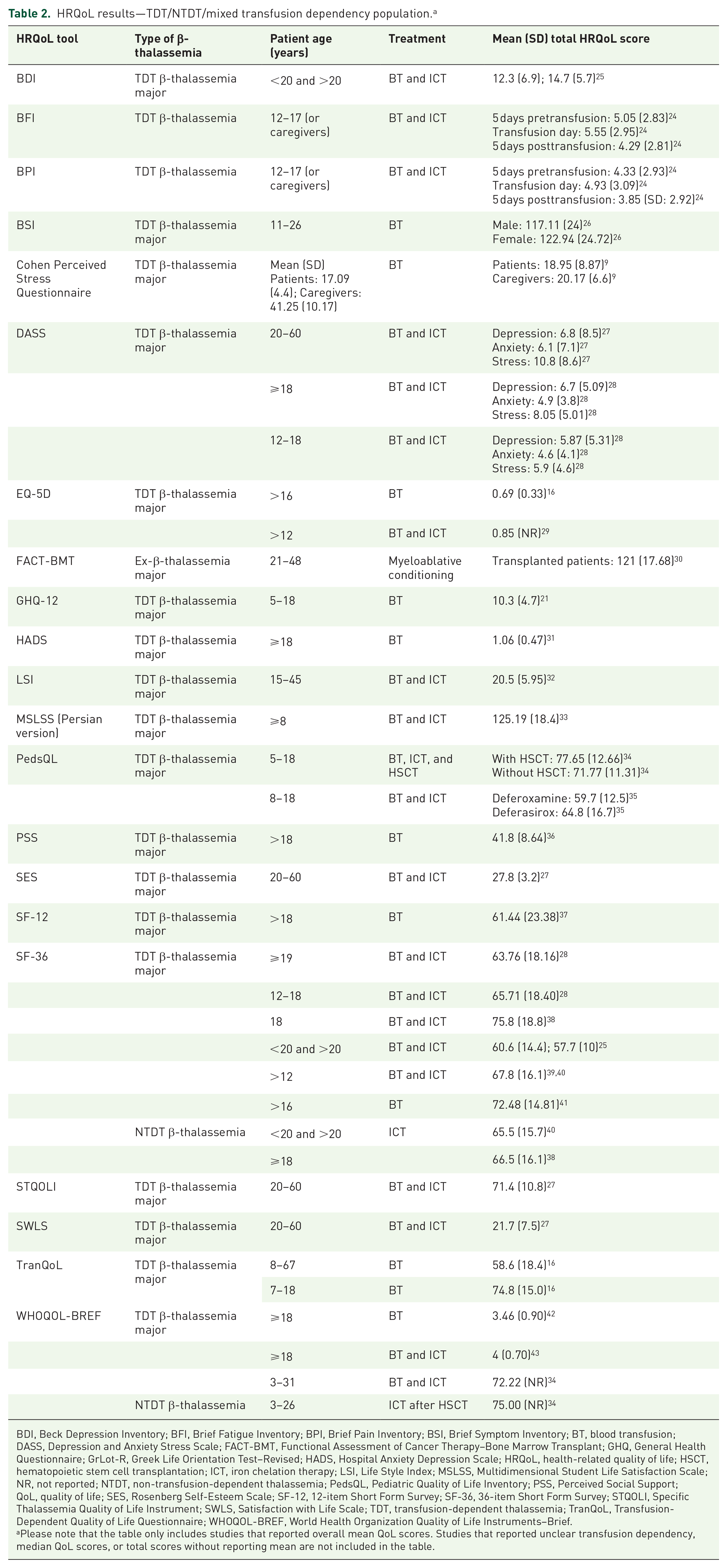
BDI, Beck Depression Inventory; BFI, Brief Fatigue Inventory; BPI, Brief Pain Inventory; BSI, Brief Symptom Inventory; BT, blood transfusion; DASS, Depression and Anxiety Stress Scale; FACT-BMT, Functional Assessment of Cancer Therapy–Bone Marrow Transplant; GHQ, General Health Questionnaire; GrLot-R, Greek Life Orientation Test–Revised; HADS, Hospital Anxiety Depression Scale; HRQoL, health-related quality of life; HSCT, hematopoietic stem cell transplantation; ICT, iron chelation therapy; LSI, Life Style Index; MSLSS, Multidimensional Student Life Satisfaction Scale; NR, not reported; NTDT, non-transfusion-dependent thalassemia; PedsQL, Pediatric Quality of Life Inventory; PSS, Perceived Social Support; QoL, quality of life; SES, Rosenberg Self-Esteem Scale; SF-12, 12-item Short Form Survey; SF-36, 36-item Short Form Survey; STQOLI, Specific Thalassemia Quality of Life Instrument; SWLS, Satisfaction with Life Scale; TDT, transfusion-dependent thalassemia; TranQoL, Transfusion-Dependent Quality of Life Questionnaire; WHOQOL-BREF, World Health Organization Quality of Life Instruments–Brief.
Please note that the table only includes studies that reported overall mean QoL scores. Studies that reported unclear transfusion dependency, median QoL scores, or total scores without reporting mean are not included in the table.
Based on SF-36 data, the presence of improved HRQoL scores for patients with TDT was attributed to compliance with ICT (p = 0.004, 39 p < 0.01 28 ), a higher level of income (p < 0.001), 39 employed status (p = 0.02), 28 a higher education level (p < 0.001), 28 younger age (inversely associated with age, i.e., better HRQoL among those ⩽14 years of age, p = 0.008), 44 lower serum ferritin levels (p = 0.05), 42 and male sex (p = 0.035 45 ; role-physical (p = 0.035), vitality (p = 0.037), and bodily pain (p = 0.044) scores were lower in female patients 46 (p = 0.041, 39 p = 0.009, 39 p = 0.037 47 )). Aspects of cardiovascular function, including stage of symptomatic heart failure (p < 0.001), 46 articular diseases (p = 0.01), 28 and heart diseases (p = 0.007) 28 were associated with worse HRQoL scores.
Anxiety and depression were significantly associated with poor HRQoL in TDT (p < 0.001). 36 A study conducted in Iran estimated that 23.7% and 19.8% of patients with TDT were susceptible to anxiety and depression, respectively (Persian version of Hospital Anxiety and Depression Scale: mean (SD) score of 7.47 (4.35) for anxiety, 7.42 (3.17) for depression). 36 The Depression, Anxiety, and Stress Scale 21 (DAS-21) showed high levels of depression, stress, and anxiety for patients with TDT (mean (SD) score: depression: 6.43 (5.16), anxiety: 4.86 (3.96), stress: 7.36 (4.99)), with ⩾50% of patients suffering mild-to-severe stress, anxiety, and depression. 28 Compared with healthy individuals, patients with TDT reported worse scores on the Beck Depression Inventory (BDI; mean (SD) score: TDT: 13.5 (6.4), controls: 6.1 (3.7); p < 0.05). 25 Deficits were reported across all SF-36 domains, with mental health having the worst score, in a study where HRQoL was compared between TDT and healthy peers (48.5 vs 96.6; p < 0.001). 48 Mental health deficits related to TDT may be also amenable to improved therapeutic regimens. One study reported significant improvements in the mental health domain of SF-36 (mental health scale: Z score mean difference +4.0; 95% confidence interval (CI) 0.4–7.5; p = 0.030; mental component summary scale: Z score mean difference +3.2; 95% CI 0.2–6.2; p = 0.039) alongside improved median ferritin levels and an increased use of ICT with deferasirox. 49
A high transfusion burden had a negative impact on HRQoL for patients with TDT across studies analyzed in this review. TDT patients reported lower SF-36 scores versus healthy peers across all domains 25 (TDT patients total score: 59.2 vs 75.7 for healthy peers).25,50 Patients with TDT reported worse pain and fatigue scores on the Brief Fatigue Inventory (BFI) and Brief Pain Inventory (BPI) scales during the 5 days preceding transfusion than in the 5 days following transfusion (mean (SD) BFI scores: 5.05 (2.83) pretransfusion, 5.55 (2.95) on transfusion, 4.29 (2.81) posttransfusion; mean (SD) BPI scores: 4.33 (2.93) pretransfusion, 4.93 (3.09) on transfusion, and 3.85 posttransfusion (2.92)). 24 In addition, HRQoL varied for patients receiving different ICT agents, with patients receiving deferoxamine reporting worse HRQoL on the World Health Organization Quality of Life Scale–BREF (WHOQOL-BREF) than those receiving deferasirox (psychological health: 65.9 vs 47.1; p < 0.001; physical health: 48.9 vs 63.9; p < 0.001; social relationship: 63.6 vs 74.2; p = 0.016; environmental: 40.6 vs 58.3; p < 0.001). 35 Patients with TDT receiving oral agents had better health utility values than those treated with injectable ICT (EQ-5D: 0.87 vs 0.81; p < 0.05). 29
Curative treatment options, such as HSCT, improved HRQoL scores across the included studies. HRQoL scores 20 years after HSCT were comparable to those of the general population (SF-36 change in HRQoL: −8.9; 95% CI −15.0 to 2.7; p = 0.005). 30 Patients with TDT who underwent HSCT had better physical, emotional, and social health on the PedsQoL scale, owing to less anger (non-HSCT TDT vs HSCT TDT: 47.79 vs 61.11; p = 0.031), better social ability (non-HSCT TDT vs HSCT TDT: 69.11 vs 83.33; p ⩽ 0.034), higher sports involvement (non-HSCT TDT vs HSCT TDT: 75.73 vs 89.58; p = 0.025), and better physical abilities (non-HSCT TDT vs HSCT TDT: 44.85 vs 72.91; p = 0.001) compared with non-HSCT patients. 34 On the WHOQOL-BREF, HSCT patients with TDT had higher quality of life (QoL) in the physical domain only (non-HSCT TDT vs HSCT TDT: 79.7 vs 66.6; p = 0.041). 34 Further, perceptions of overall health were significantly higher in HSCT patients with TDT compared with non-HSCT TDT patients (WHOQOL-BREF: non-HSCT TDT vs HSCT TDT: 80.5 vs 60.4; p = 0.034). 34
Although patients with NTDT have milder anemia and lower transfusion burden, which suggests better HRQoL, evidence from our analysis demonstrated worse QoL compared with patients with TDT. Across all domains of SF-36, patients with NTDT were reported to have worse scores compared with patients with TDT (mean (SD): 75.8 (18.8) vs 66.5 (16.1); p = 0.021). 38 Another study reported mean differences in SF-36 scores between NTDT and TDT patients for the mental component score (47.4 (8.4) vs 51.6 (7.2); p < 0.01), and the domains of general health (41.9 (8.2) vs 46.3 (10.0); p = 0.018), vitality (49.8 (8.1) vs 54.0 (7.1); p = 0.008), and mental health (47.8 (7.8) vs 51.7 (6.9); p = 0.011). 51 Patients with NTDT had significantly better scores on the physical functioning subscale of SF-36 alone, compared with patients with TDT (NTDT vs TDT mean (SD): 76.8 (26.6) vs 86.9 (12.9); adjusted mean difference: 12.5; 95% CI 5.6–19.3; p < 0.0001). 40 Functional Assessment of Cancer Therapy–Anemia (FACT-An) scale scores for patients with TDT showed moderately better health than for patients with NTDT (FACT-An General: TDT vs NTDT: 83.0 (11.3) vs 89.0 (10.7); p = 0.009). 51 Patients with TDT had more favorable changes in FACT scores for fatigue subscales (TDT vs NTDT: +1.8 vs −1.9; p = 0.057) over a 24-week study period compared with patients with NTDT. 51 There was high prevalence of depression (TDT vs NTDT: 29.2% vs 43.8%; p = 0.233; median BDI score: 6.5 vs 8; p = 0.244) and anxiety (18.8% vs 28.1%; p = 0.415; median State-Trait Anxiety Inventory: 34 vs 41; p = 0.035) for patients with TDT and NTDT, with patients with NTDT experiencing more anxiety compared with those with TDT. 52
Our findings showed high QoL impairment in caregivers of patients with TDT. Caregivers had severe stress compared with patients with TDT (mean score: 20.17 vs 18.95; p = 0.006), as measured by the standardized Cohen Perceived Stress Questionnaire. 9 Caregivers’ QoL, as measured by the Carer QoL 7D, was found to be significantly associated with the QoL of their children with TDT (adjusted odds ratio (AOR): 3.0; 95% CI 1.6–5.5), caregivers’ education level (AOR: 2.3; 95% CI 1.3–4.1), socioeconomic status (AOR: 2.3; 95% CI 1.2–4.3), patients’ age (AOR: 2.2; 95% CI 1.2–4.2), and their clinical/therapeutic profile (AOR: 2.1; 95% CI 1.1–3.9). 53 Another study conducted on patients with TDT in Pakistan reported that caregiver burden was negatively correlated with psychological well-being as measured by the Ryff Scale of Psychological Well-Being (p < 0.001; HRQoL scores not provided in the study). 54 Caregivers were reported to have mild-to-moderate depression and anxiety in a study conducted in India, where 37% reported mild and 38% moderate-to-severe depressive symptoms, and 50% reported mild and 17% moderate-to-severe anxiety symptoms (HRQoL scores not provided in the study). 55
Discussion
To the best of our knowledge, this study is the first SLR on indirect burden among patients with β-thalassemia. Significant productivity loss, lost opportunities, and absenteeism were reported among TDT patients, with differences across geographies and age groups. Indirect burden was high across geographical regions. For example, families of patients with TDT put between 10% and 39% of their annual income toward indirect costs related to blood transfusions, follow-up appointments, hospitalizations, and other associated expenses.12,13 This proportion of income is substantial even when put in the context of other chronic conditions such as diabetes (11%) and rheumatoid arthritis (22%). 56 In addition, large productivity losses were reported, owing to the need for regular blood transfusions, disease complications, and distress caused by the disease. Based on our evidence, the cost of productivity losses was 6.38% of the total indirect costs, compared with 19% reported in a recent thalassemia study that did not fit the inclusion criteria of the SLR. 57 These variations may be attributed to the heterogeneity in the populations included in the study and variation in study designs and methods used to estimate productivity losses. Additionally, costs should be viewed within the context of the economic wealth of a country; for example, productivity loss due to TDT represented 11.5% of Iran’s and 5.2% of Thailand’s GDP in 2015 (https://data.worldbank.org/indicator/NY.GDP.PCAP.KD?locations=IR).18,19 Furthermore, PPP values show that the euro, Thai baht (THB), Indian rupee (INR), and Pakistani rupee (PKR) are, respectively, valued at 1.2, 2.8, 3.2, and 3.6 less than the USD, reflecting differences in wages, purchasing power, and economic development between the countries included in this analysis. The proportion of income spent on indirect costs per household (as a percentage of per capita GDP) in each of these countries further supports the economic impact of β-thalassemia on individuals and their families.
Our results indicate a lack of published data quantifying indirect burden among NTDT patients. Given the impacts of NTDT on HRQoL, further discussed below, it is likely that a substantial indirect burden is experienced by these patients and their caregivers. There also remains a relative paucity of data on the effect of the indirect burden of TDT on families and caregivers, with one study reporting on lost opportunities 18 and another on time lost due to absenteeism. 23 Further research is required in both these areas to illustrate the full indirect burden of β-thalassemia.
Most publications reported HRQoL data in the TDT population, with more limited data identified in the NTDT population. In a prospective observational study, baseline HRQoL scores for NTDT were worse than TDT scores for all domains, except Role-Physical on SF-36 and Physical Functioning, and across all domains of FACT-An. In addition, after 24 weeks, NTDT patients generally had less favorable changes in HRQoL, with decreased FACT-An scores observed on the Anemia Subscale, Fatigue Symptoms, Fatigue Impact, and Fatigue Experience. 51
Despite its presence as a cardinal symptom and the well-demonstrated relationships with reduced QoL in other chronic conditions such as cancer and chronic obstructive pulmonary disease (COPD),58,59 there is an apparent lack of focus on the effect of anemia on QoL in the literature on thalassemias, and in related disorders such as sickle cell disease.60,61 Yet, unlike in TDT, where treatment-related issues drive disease complications, chronic anemia is a major driver of disease complications in NTDT, 62 and such complications have been shown to be correlated with QoL in an international study of thalassemia, which did not meet our inclusion criteria. 63 An NTDT-specific patient-reported outcome measure aimed at assessing anemia-related dysfunction has recently been developed. 64 Further research using such measures is needed into the effect of anemia on QoL in NTDT, in particular. However, the existing deficiencies in QoL, their worsening over time, and the apparent relationship with disease-related complications already speak to an unmet treatment need in these patients.
SLR data suggest patients with TDT have increased susceptibility to anxiety and depression, with an association with reduced HRQoL. 36 This increased susceptibility was reflected in high scoring on the DAS-21, 22 and worse scoring on the BDI than healthy individuals, 25 in two separate studies. Increased incidence of anxiety and depression is common for patients with chronic health conditions, 65 and anxiety and depression are associated with worse outcomes for patients with coronary heart disease, for example. 66 Mental-health-impairment data speak to a further potential unmet need in the treatment of TDT. Indeed, a further study from the SLR showed that implementation of ICT may be related to significant improvements in the SF-36 mental health domain, 49 indicating that improved treatment options that reduce the need for RBC transfusions may also improve mental health and HRQoL in TDT.
Although limited, evidence found in the SLR on caregivers highlighted a substantial HRQoL burden. For example, caregivers of patients with TDT had significantly higher stress levels compared with the patients themselves. Reduced QoL in caregivers has been reported in other chronic conditions, such as COPD and rare inherited conditions.67,68 Notably, in our study, caregiver HRQoL was related to patient HRQoL, a relationship that has also been established in other chronic health conditions. 69 Given the potential for improved therapeutic strategies and reduced transfusion burden to increase patient well-being, as well as the direct effect of patient clinical/therapeutic profile on caregiver HRQoL, better treatment options may also ameliorate the impact of thalassemia on caregivers.
There are some limitations in our study. Few studies reported the methodology used in estimating productivity losses. The analysis of indirect costs of β-thalassemia relied on how cost data are reported in each study included in this SLR, which differed across the studies. Publications that reported indirect burden included data from various geographical locations with diverse health systems, costs of therapies, and patient backgrounds; therefore, a comparative analysis was not feasible. Many studies were from lower- and middle-income groups, thereby reducing the generalizability of the findings across broader β-thalassemia populations in high-income countries. Heterogeneity due to the inclusion of different study designs precluded formal study-quality assessment. The HRQoL scales used in the studies were mostly generic; therefore, this review may not give a complete understanding of the QoL of the condition or patients’ perception of their QoL. In addition, most studies presented findings based on a mixed population of adults and children. Finally, many of the publications in this SLR analyzed data collected from questionnaires and therefore may be limited by self- and proxy-reporting, and recall bias.
In summary, the indirect burden of β-thalassemia on patients and caregivers constitutes a significant cost due to lost opportunities, productivity loss, and loss of welfare. There is a need for global observational studies focused on the indirect burden of TDT and, particularly, NTDT. The use of thalassemia-specific HRQoL measures will facilitate a better understanding of QoL for TDT and NTDT populations and patients’ perception of their own HRQoL.
Supplemental Material
sj-docx-1-tah-10.1177_20406207241270872 – Supplemental material for Systematic literature review of the indirect costs and humanistic burden of β-thalassemia
Supplemental material, sj-docx-1-tah-10.1177_20406207241270872 for Systematic literature review of the indirect costs and humanistic burden of β-thalassemia by Yesim Aydinok, Sneha Purushotham, Aylin Yucel, Mrudula Glassberg, Sohan Deshpande, Barbara Potrata, Myrto Trapali and Farrukh Shah in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors received editorial support in the preparation of this manuscript from Patricia Fonseca, PhD, of Excerpta Medica, funded by Bristol Myers Squibb. The authors are fully responsible for all content and editorial decisions for this manuscript. The authors have authorized this support and approved the inclusion of all conflicting interests and funding disclosures.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
