Abstract
Background:
Secondary failure of platelet recovery (SFPR) is a common complication that influences survival and quality of life of patients with β-thalassemia major (β-TM) after hematopoietic stem cell transplantation (HSCT).
Objectives:
A model to predict the risk of SFPR in β-TM patients after HSCT was developed.
Design:
A retrospective study was used to develop the prediction model.
Methods:
The clinical data for 218 β-TM patients who received HSCT comprised the training set, and those for another 89 patients represented the validation set. The least absolute shrinkage and selection operator regression algorithm was used to identify the critical clinical factors with nonzero coefficients for constructing the nomogram. Calibration curve, C-index, and receiver operating characteristic curve assessments and decision curve analysis (DCA) were used to evaluate the calibration, discrimination, accuracy, and clinical usefulness of the nomogram. Internal and external validation were used to test and verify the predictive model.
Results:
The nomogram based on pretransplant serum ferritin, hepatomegaly, mycophenolate mofetil use, and posttransplant serum albumin could be conveniently used to predict the SFPR risk of thalassemia patients after HSCT. The calibration curve of the nomogram revealed good concordance between the training and validation sets. The nomogram showed good discrimination with a C-index of 0.780 (95% CI: 70.3–85.7) and 0.868 (95% CI: 78.5–95.1) and AUCs of 0.780 and 0.868 in the training and validation sets, respectively. A high C-index value of 0.766 was reached in the interval validation assessment. DCA confirmed that the nomogram was clinically useful when intervention was decided at the possibility threshold ranging from 3% to 83%.
Conclusion:
We constructed a nomogram model to predict the risk of SFPR in patients with β-TM after HSCT. The nomogram has a good predictive ability and may be used by clinicians to identify SFPR patients early and recommend effective preventive measures.
Keywords
Introduction
β-thalassemia major (β-TM) is a monogenic hereditary disease. Except for promising gene therapy strategies that are available in the context of clinical trials, allogeneic hematopoietic stem cell transplantation (HSCT) is the only definitive curative therapeutic method for TM thus far. Our previous study demonstrated that the 3-year overall survival (OS) and thalassemia-free survival (TFS) of TM patients were 97.8% and 97.3%, respectively. 1 Although successful HSCT can cure TM, the procedure carries the risk of potentially fatal complications. Thrombocytopenia is one of the common complications that occur after HSCT and is mainly caused by prolonged isolated thrombocytopenia and secondary failure of platelet recovery (SFPR).2,3 As a condition of thrombocytopenia after primary engraftment of platelets, the cumulative incidence of SFPR is 20%. 4 SFPR has been reported to be a crucial risk factor that increases the risk of bleeding and transplant-related death and seriously affects the prognosis of patients undergoing HSCT. 5 Platelet transfusion and medication use are universal therapies for managing and preventing bleeding; nevertheless, these strategies are not always effective and greatly increase the strain on blood banks and the economic burden on patients. However, the pathogenesis and risk factors for SFPR are very complex, and the precise pathogenesis remains uncertain. Multiple risk factors for SFPR have been identified, including unrelated donors, preparative regimens, renal or liver dysfunction, source of stem cells, CD34+ cell dose infused, acute graft-versus-host disease (aGVHD), and infection.2,4,6 Additionally, thrombotic microangiopathy, immune-mediated thrombocytopenia, and drug use such as ganciclovir are potential causes of SFPR.2,4
Nomograms are tools that can be used to combine all independent risk factors to predict an outcome. Nomograms are considered concise and intuitive models and have been widely applied to predict the risk of various hematological diseases, for example, acute kidney injury after HSCT 7 and bleeding in a Chinese immune thrombocytopenia (ITP) population, 8 and the prognosis of patients with, for example, acute myeloid leukemia with biallelic CEBPA mutations. 9 To improve transplant outcomes, it is vital to focus on the characteristics of β-TM patients undergoing HSCT and construct a risk prediction model for early SFPR. Here, based on a comprehensive consideration of various factors that may affect the occurrence of SFPR, we established the first nomogram for predicting the risk of SFPR in thalassemia patients after HSCT.
Methods
Patients
We estimated the lower sample size based on a value of 5–10 times that of the variables included in the model; we further estimated the total sample size based on the incidence rate of an end-point event to match the scale of the study. Patients eligible for this retrospective study were consecutive β-TM patients who consented to receive HSCT at the Stem Cell Transplantation Center, The First Affiliated Hospital of Guangxi Medical University, between January 2019 and July 2022. Our hospital is the largest general hospital in the Guangxi Zhuang Autonomous Region and the main HSCT center for thalassemia treatment in China. The entry criteria were as follows: (1) a diagnosis of TM with hemoglobin electrophoresis and a genetic diagnosis of β-TM by DNA analysis; (2) patients without thrombocytopenia before HSCT; and (3) patients with normal cardiopulmonary function. The exclusion criteria were (1) patients with incomplete data and a follow-up time of fewer than 3 months; (2) cytomegalovirus (CMV) or Epstein–Barr virus (EBV) DNA >400 copies/mL in blood by quantitative PCR before HSCT; and (3) patients positive for HIV serology, hepatitis C virus or hepatitis B virus. All procedures were carried out by the relevant guidelines.
Data collection
The clinical data of β-TM patients who underwent HSCT were extracted from electronic medical records; the data for 218 patients were used to build the training set, and those for another 89 patients represented the validation set. A total of 40 parameters were included in this retrospective study, which was based on previous studies and characteristics of thalassemia, including the general data of the patients, blood biochemistry assays, transplant characteristics, drugs used, postoperative complications, infections, and other information. Patients were divided into two groups: the SFPR group and the non-SFPR group. All patients were treated with the same conditioning regimen, which consisted of fludarabine, busulfan, cyclophosphamide, thyroglobulin, and hydroxyurea. Our center uses ganciclovir as the preemptive therapy for CMV reactivation, namely, the standard CMV antigenemia-guided preemptive approach.10,11 Additionally, cyclosporin, tacrolimus, mycophenolate mofetil (MMF) and basiliximab are used to prevent GVHD. Heparin sodium, nadroparin calcium, and ursodeoxycholic acid are applied to prevent hepatic veno-occlusive disease (VOD). All patient details already have been de-identified.
Definitions
The time to SFPR was calculated from the date of HSCT to the date of clinical diagnosis. SFPR was defined as the first of seven consecutive days of platelet counts <20 × 109/L or a requirement of platelet transfusions within 7 days after primary platelet recovery was achieved. 3 Neutrophil engraftment and platelet engraftment were defined as the first of three consecutive days with an absolute neutrophil count >0.5 × 109/L and an unsupported platelet count >20 × 109/L. 12 CMV and EBV reactivation was defined as DNA >1000 copies/mL of blood by quantitative PCR. 13 Human leukocyte antigen (HLA) mismatch was defined as ⩾1 mismatched antigen. Posttransplant biochemical tests were recorded on the first day of SFPR diagnosis in the SFPR group and 1 month after transplantation in the non-SFPR group.
Statistical analysis
All statistical analyses were completed with the Statistical Package for the Social Sciences 20.0 (IBM SPSS Inc.), R software (version 4.1.0), and auxiliary RStudio software (version 1.4.1717).
In this study, the continuous variables were represented by the mean ± one standard deviation or the median (range), and the categorical data were represented by frequencies (percentages). Continuous variables were compared using the Mann–Whitney U test or t test, and the chi-square test was used to compare categorical data. p Values (two-sided) <0.05 were considered statistically significant in all statistical analyses.
The least absolute shrinkage and selection operator (LASSO) regression algorithm, which is suitable for the reduction of high-dimensional data, was applied to select the optimal predictive features related to risk in patients with SFPR in the training set.14,15 Features without nonzero coefficients were excluded from the LASSO regression model. 16 Then, a predictive model was built based on the results of multivariate logistic regression analysis, which also incorporated the results of the LASSO regression model. A nomogram is based on calculating the score of each parameter in the model and then adding the scores of each parameter to obtain a total score, which corresponds to the probability of the outcome event. The C-index reflects the consistency between the prediction probability and the observation results and can be used to assess the prediction accuracy and discriminant ability of the nomogram. 17 The C-index value ranges from 0.5 to 1.0, and the higher the C-index value is, the higher the consistency between the prediction and the observed result. The calibration curve was used to assess the consistency between the actual and predicted risks of the nomogram. The predictive ability of the nomogram was further assessed by the AUC curve. 18 Decision curve analysis (DCA) was used to evaluate the clinical usefulness of the nomogram by quantifying net benefits, which was calculated by subtracting the proportion of all patients with false positives from the proportion with true positives. 19 To verify the accuracy of the risk nomogram, internal validation was completed using the bootstrap validation method with 1000 replications, and external validation was completed in the independent validation set.
Results
Patient characteristics
A total of 221 β-TM patients received HSCT from July 2020 to July 2022. According to the entry and exclusion criteria, 218 patients comprised the training set, and the median follow-up after HSCT was 12.4 months (range 3–24). The sex ratio of the patients was 64.7% male/35.3% female. Fifty-seven (26.1%) patients developed SFPR, 161 (73.9%) patients did not develop SFPR, and 57 patients with SFPR were diagnosed at a median time of 27 days (range 17–123) after HSCT.
The clinical characteristics of the two groups are summarized in Table 1, including baseline characteristics, transplant characteristics, drugs to prevent GVHD and VOD, posttransplant-related factors, and complications. The median age of 218 patients was 8 years (range 2–19). Twenty-eight (12.84%) patients underwent splenectomy and 125 (57.3%) patients with hepatomegaly. The mean hemoglobin level before HSCT was 104.0 ± 188.4 g/L. The median serum ferritin level was 3464 (348–18,205) ng/mL. Sixty-five (29.8%) patients received HLA-mismatched transplants, and 89 (40.8%) patients received unrelated donor transplants. aGVHD was diagnosed in 80 (36.7%) patients. Bacterial bloodstream infection was documented in 63 (28.9%) patients. CMV and EBV reactivation were observed in 101 (46.3%) and 30 (13.8%) patients posttransplantation, respectively.
Clinical data of 218 β-TM patients who underwent HSCT.

aGVHD, acute graft-versus-host disease; ALT, alanine transaminase; AST, aspartate transaminase; BM, bone marrow; BMI, body mass index; CB, cord blood; CMV, cytomegalovirus; EBV, Epstein–Barr virus; Hb: hemoglobin; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplantation; MNC, mononuclear cell; PBSC, peripheral blood stem cell; SFPR, secondary failure of platelet recovery.
In this study, only two (1.4%) patients developed VOD, and both patients were in the non-SFPR group. Twenty-eight patients with hypersplenism had undergone splenectomy before transplantation. Among the 190 patients without splenectomy, 124 patients had splenomegaly, including 35 (68.7%) in the SFPR group and 89 (64.0%) in the non-SFPR group, and the difference was not statistically significant. Twelve (5.5%) patients had sepsis, including 2 (3.5%) in the SFPR group and 10 (6.2%) in the non-SFPR group, and the difference was not statistically significant. No diffuse intravascular coagulation occurred in these 12 patients. Due to the small number of patients with sepsis, this study included these patients in the analysis of bacterial bloodstream infection. In this study, only one patient died because of intracranial hemorrhage due to SFPR.
Feature selection
We applied a LASSO regression algorithm based on each feature for feature selection in the training cohort. Dotted vertical lines were drawn at the optimal values by using the minimum criteria and the one standard error of the minimum criteria (the 1-SE criteria). According to fivefold cross-validation, we chose the best λ for LASSO regression analysis when the partial likelihood binomial deviance reached its minimum value [Figure 1(a) and (b)]. Four variables with nonzero coefficients were selected in the LASSO regression analysis, namely, pretransplant serum ferritin, hepatomegaly, MMF use, and posttransplant serum albumin (Table 2). Then, multivariate logistic regression was also performed for further verification. The results were shown in Table 2. Except for pretransplant serum ferritin, the p value of the other three features is below 0.05. However, pretransplant serum ferritin is meaningful to SFPR, we still use the above four features for further forecasting.

Clinical feature selection using the LASSO regression model.
Predictive factors for risk in SFPR in the training set.

SFPR, secondary failure of platelet recovery.
Nomogram development
According to the results of the LASSO regression analysis and multivariate logistic regression analysis, the forecast model that included the four independent predictors was developed and is presented as the nomogram [Figure 2(a) and (b)]. The nomogram showed that the predicted risk of SFPR in β-TM patients ranged between 0.02 and 0.98.

Nomogram of the SFPR risk prediction model for a patient with SFPR (a) and a patient without SFPR (b).
The nomogram that we developed is shown in Figure 2(a) and is described as follows:
Consider β-TM patients who received HSCT; the red dot for each parameter is specific information for the patient: pretransplant serum ferritin of 3844 ng/mL (34 points), use of MMF (3 points), hepatomegaly (52 points), and posttransplant serum albumin of 20.1 g/L (100 points). In summary, the patient received a total score of 189 points, and the corresponding predicted probability of SFPR was 0.935 (93.5%). In terms of overall morbidity, the patient had an extraordinarily high risk of SFPR.
Nomogram validation and assessment
To verify the accuracy of the nomogram, we conducted internal and external validation through C-index and calibration curve assessments. The calibration curve of the SFPR risk nomogram was close to the ideal curve, demonstrating good concordance between the training set and validation set [Figure 3(a) and (b)]. The C-index of the training set was 0.780 (95% CI: 0.703–0.857) and was confirmed to be 0.766 by internal bootstrapping verification, while the C-index of the verification set was 0.868 (95% CI: 0.785–0.951), which indicated that the model had good discrimination. Furthermore, the AUC of the predictive nomogram was 0.780 in the training set and 0.868 in the validation set [Figure 4(a) and (b)], which also showed that the model had a good prediction ability. All these results indicated that the nomogram shows significantly superior predictive ability.

Calibration curves of the SFPR risk nomogram predictions in the training set (a) and validation set (b).
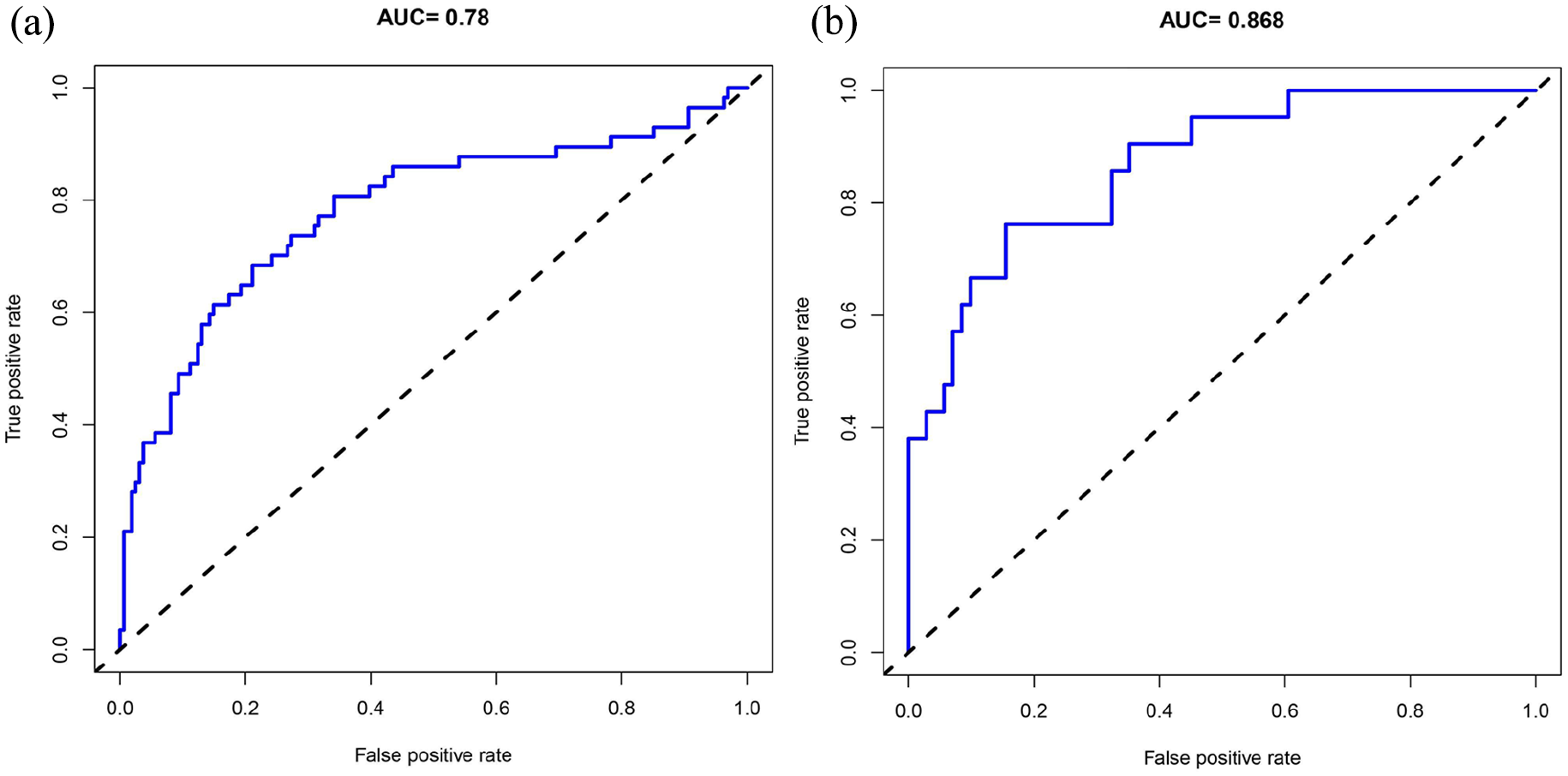
ROC curves of the SFPR risk nomogram in the training set.
Clinical application
DCA of the SFPR risk nomogram is presented in Figure 5, which shows that if the threshold probability of a patient was from 3% to 83%, using this risk nomogram to predict the risk of SFPR would greatly benefit patients.

Decision curve analysis of the SFPR risk nomogram.
The present study exhibited bone marrow cell images of a typical case of SFPR (Figure 6).

Bone marrow cell images of the patient with SFPR.
Discussion
SFPR is a common and serious complication after HSCT that severely influences the survival and prognosis of HSCT patients. 5 Therefore, it is extraordinarily important to identify the risk of SFPR in advance. The pathogenesis of SFPR is complicated and affected by various factors. Nevertheless, the specific mechanism of SFPR remains unclear. Impaired platelet production is a common cause of SFPR in HSCT patients. Complications such as poor transplantation function, viral infection, and adverse drug reactions often cause bone marrow hematopoietic dysfunction and immune disorders, which ultimately result in insufficient differentiation of bone marrow megakaryocytes and dysfunction of platelet maturation.3,20,21 In the past, a method of evaluating the risk of SFPR in β-TM patients has not been reported. This study was the first to construct a model to predict the risk of SFPR in β-TM patients after HSCT. Pretransplant serum ferritin, hepatomegaly, the use of MMF, and posttransplant serum albumin were shown to be predictive factors of SFPR in β-TM patients.
Our study introduced transplant features and pretransplant and posttransplant parameters to construct a novel risk prediction nomogram model of SFPR. The calibration curve of the SFPR risk nomogram revealed good agreement in the training set and validation set. The C-indexes of the training set and validation set were 0.780 and 0.868, respectively, which indicated that the prediction ability of the nomogram was highly accurate. 22 The study showed that the efficiency of discrimination increased with the C-index of external verification. 23 To further validate the accuracy of the nomogram, a ROC curve was utilized to calculate the AUC. In addition, we applied DCA to quantify the net benefit of the nomogram, and the results showed high clinical practicability and predictive effect. The nomogram model could be applied to predict the risk of SFPR in β-TM patients in advance, which is beneficial for clinicians to identify patients at risk of SFPR with easily available information and guide the management of HSCT patients.
Iron overload is a familiar consequence of transfusion-dependent thalassemia. Studies have shown that iron overload is a predictor of adverse outcomes in HSCT. 24 Chai et al. 25 found that iron overload significantly reduced the proportion of hematopoietic stem cells and progenitor cells and inhibited the cloning function of hematopoietic cells in mice by increasing intracellular reactive oxygen species (ROS) levels. Zhang et al. 26 evaluated the effect of iron overload on bone marrow transplantation in mice and found that excessive iron accumulation of ROS damaged the bone marrow microenvironment and led to delayed hematopoietic reconstruction, which was related to the decreased expression of chemokine stromal cell-derived factor-1, stem cell factor-1, and vascular endothelial growth factor-1. Additionally, iron overload can induce abnormalities in T lymphocyte subsets and destroy immune cells through the accumulation of ROS, leading to delayed immune reconstitution. 27 These data show that iron overload can damage bone marrow hematopoietic function and the bone marrow microenvironment, resulting in insufficient megakaryocyte differentiation and delayed hematopoietic reconstruction. Serum ferritin is considered the first choice for a biomarker of iron overload and has been widely used as a clinical indicator to measure body iron burden. 24 Malki et al. 28 indicated that elevated levels of pretransplant serum ferritin (SF > 2000 ng/mL) were closely associated with an increased cumulative incidence of delayed platelet engraftment (HR: 0.48; p = 0.044). Similar to what has been reported in the literature, our results showed that higher pretransplant serum ferritin was a risk factor for SFPR in β-TM patients who received HSCT. Iron overload is one of the important characteristics of thalassemia, and many studies have shown that iron overload severely affects the function of platelet production and implantation, therefore, we included pretransplant serum ferritin as a risk factor in the construction of the risk model.
Hepatomegaly is an important manifestation of extramedullary hematopoiesis caused by early irregular blood transfusion in patients with thalassemia and has been considered one of the independent risk factors for poor prognosis of HSCT in thalassemia patients. 29 Moreover, patients with thalassemia do not receive regular iron chelation therapy, which could aggravate the iron load of multiple organs in the body, including the heart, liver, spleen, and brain, leading to organ damage and dysfunction. Thrombopoietin (TPO) is an essential cytokine required for HSC proliferation and platelet production and is mainly produced by hepatocytes. Okabe et al. 30 confirmed that liver iron deposition increased significantly and TPO levels decreased significantly in iron overload mice, which seriously affected platelet production and implantation. The above findings support our study that thalassemia transplant patients with hepatomegaly may have a higher risk of SFPR.
MMF is a type II inosine monophosphate dehydrogenase inhibitor that has been extensively applied to prevent aGVHD in HSCT patients. 31 Apart from its synergistic effect with cyclosporine in preventing aGVHD, MMF also has the effects of reducing the generation of inflammatory cytokines and antibacterial activity.31,32 MMF is highly selective, particularly in the inhibition of lymphocytes rather than myeloid cells; therefore, it causes fewer cytotoxic effects, reduces interference with myeloid cells, and induces better recovery of platelets.33,34 The results of a meta-analysis involving 177 transplant patients showed that aGVHD prophylaxis with MMF compared with methotrexate appears to result in significantly faster platelet engraftment (HR: 0.87; p < 0.0001). 35 Notably, Chang 36 reported that MMF can also be used to treat ITP and improve platelet recovery. 36 Collectively, the mechanism by which MMF promotes platelet recovery may be its highly selective immunomodulatory effects. In this study, we found that the absence of MMF was a risk factor for SFPR in β-TM patients who underwent HSCT. In our study, the patients who did not receive MMF underwent cord blood transplantation. Previous studies have already confirmed that a low number of hematopoietic progenitor cells in cord blood increases the risk of delayed hematopoietic engraftment and poor immune reconstitution.37–39 Consequently, patients with cord blood transplantation are prone to SFPR. However, whether the absence of MMF in patients with cord blood transplantation is related to delayed engraftment needs further research.
Albumin is a crucial nutrient in the human body and has been used as a significant index to evaluate the nutritional status of patients. 40 Since thalassemia patients are chronically anemic and generally lag in growth and development, nutritional status is significantly important for the recovery of hematopoietic function, especially platelet recovery, in thalassemia transplant patients. Additionally, transplantation-related drugs can cause gastrointestinal side effects such as nausea, vomiting, diarrhea, and mucositis, leading to loss of appetite and insufficient nutritional intake. Hence, nutritional status is poor, and SFPR is likely to occur. A study evaluating the nutritional status in transplant patients revealed that low body mass index (BMI), as an indicator of nutritional status, was significantly associated with delayed platelet engraftment (p < 0.001). 41 More recently, another study illustrated that degraded levels of posttransplant serum albumin were a risk factor for delayed platelet engraftment (p = 0.015). 42 Similarly, our study showed that degraded levels of posttransplant serum albumin were a risk factor for predicting SFPR in β-TM patients.
By constructing a nomogram model, this study found that high levels of serum ferritin and hepatomegaly before transplantation are important risk factors for SFPR in thalassemia transplant patients, which may help clinicians to identify patients with a risk of SFPR before transplantation. The risk of SFPR could be reduced in thalassemia transplant patients and the prognosis of transplantation improved by strengthening the standardized management of patients before transplantation, including regular blood transfusion and iron chelation therapy. Low albumin levels after transplantation are also one of the risk factors for SFPR in thalassemia transplant patients. Clinicians may predict the risk of SFPR by closely monitoring the serum albumin level of patients and reducing the risk of SFPR by strengthening the nutrition of patients in the early stage. In addition, our results show that the use of MMF to prevent GVHD may reduce the risk of SFPR in thalassemia transplant patients. MMF is a unique regimen for the prevention of GVHD at our thalassemia HSCT center. The clinical application of this regimen has achieved remarkable results, that is, the 3-year OS and TFS are as high as 97.8% and 97.3%, respectively. 1 Therefore, we believe that MMF should be widely recommended for the prevention of GVHD in thalassemia transplant patients.
There are several limitations of our current study. First, this was a retrospective study, and more evidence is needed to prove the usefulness of the model from a prospective perspective. Second, our data were of limited size and from a single institution, which limits the generalizability and applicable scope of the nomogram. Although the stability of our nomogram was examined sufficiently with internal and external validation, which was intended for relatively strict validation, multi-institutional external validation would provide more convincing evidence.
Conclusion
Risk nomogram models are generally applied as prediction models for clinical decision-making. We found that some factors included in predicting the risk of SFPR in β-TM patients are serum ferritin, hepatomegaly, the use of MMF, and posttransplant serum albumin. These results are basically consistent with the clinical features of β-TM patients. Therefore, this nomogram may be used by clinicians to identify SFPR patients as early as possible and to make better individualized clinical treatment decisions, which is of great significance for managing β-TM patients after HSCT and preventing SFPR.
