Abstract
In systemic mastocytosis, cytoreductive treatment is indicated for advanced systemic mastocytosis (AdvSM) variants. The treatment scenario is rapidly diversifying especially with the introduction of KIT tyrosine kinase inhibitors. Avapritinib is a second-generation potent and selective inhibitor of the mutant KIT D816V that, based on the results of pivotal clinical trials, was approved for the treatment of adults with AdvSM by the regulatory agencies US FDA and EMA. The present article reports the experience of treating SM patients with avapritinib in an Italian compassionate use program. The data from our case series confirm the drug as being active after multiple lines of treatment allowing rapid achievement of profound responses, making it also an effective bridging strategy to allogeneic transplant in eligible patients. However, the anticipated wider use of avapritinib in the near future will require careful monitoring of side effects, especially in heavily pretreated patients.
Introduction
The term systemic mastocytosis (SM) encompasses a heterogeneous group of disorders characterized by the abnormal expansion and accumulation of mast cells (MCs) in various organs.1–3 In most cases, SM presents with an indolent course that does not significantly impair life expectancy. Accordingly, the disease is approached conservatively.2,3 Conversely, cytoreductive treatment is indicated for advanced SM (AdvSM) variants to control manifestations of myeloproliferation and prevent damage to target organs. 4 The definition of AdvSM relies on the evidence of organ disfunction, as defined by the so-called C-findings. However, it gathers forms with highly variable clinical presentations and therapeutic requirements.5–7 The treatment landscape is rapidly evolving, with KIT tyrosine kinase inhibitors becoming progressively more potent and selective. These drugs now flank conventional cytoreductive drugs (cladribine, interferon-alpha). Consequently, accurately identifying patients more likely to benefit from a specific drug has become imperative.
Avapritinib is a potent and selective inhibitor of the mutant KIT D816V. 8 Based on the results of pivotal clinical trials,9,10 it gained approval from regulatory agencies US FDA (in June 2021; approved for use regardless of prior therapy) and European Medicines Agency (EMA) (in March 2022; approved for use after at least one prior systemic therapy) for the treatment of adults with AdvSM.
This article reports the experience of treating SM patients with avapritinib in an Italian compassionate use program (CUP), as they did not qualify for enrollment in registration trials. The cases described below, following CARE guidelines 11 and with summarized key features in Table 1, serve as real life examples of how the introduction of avapritinib can contribute to the clinical management AdvSM patients. Leveraging the clinical characteristics and course of these patients, we address pertinent clinical questions in a scenario where avapritinib was available for treatment.
Characteristics and outcome of patients.
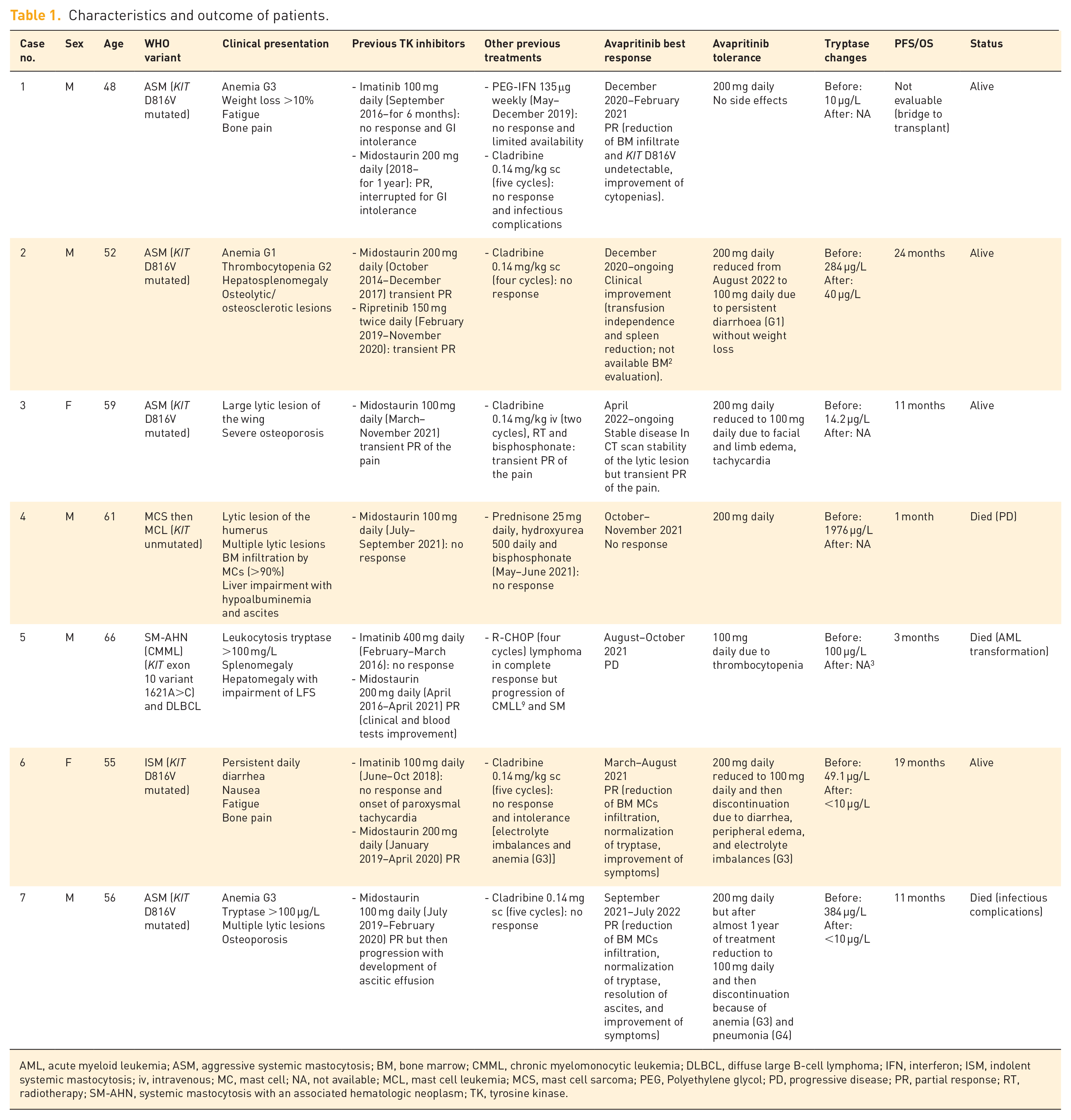
AML, acute myeloid leukemia; ASM, aggressive systemic mastocytosis; BM, bone marrow; CMML, chronic myelomonocytic leukemia; DLBCL, diffuse large B-cell lymphoma; IFN, interferon; ISM, indolent systemic mastocytosis; iv, intravenous; MC, mast cell; NA, not available; MCL, mast cell leukemia; MCS, mast cell sarcoma; PEG, Polyethylene glycol; PD, progressive disease; PR, partial response; RT, radiotherapy; SM-AHN, systemic mastocytosis with an associated hematologic neoplasm; TK, tyrosine kinase.
Case 1: Young patient with ASM treated with avapritinib as a bridge-to-transplant
A 48-year-old male patient was diagnosed with aggressive systemic mastocytosis (ASM) in 2016 at a different medical institution. He presented with grade 3 anemia, weight loss >10% in the preceding 6 months, fatigue, and bone pain, associated to normal level of serum tryptase. A bone marrow (BM) evaluation revealed infiltration by multifocal clusters of MCs and the presence of KIT D816V mutation. No anti-mediator treatment could ameliorate his symptoms, and a previous trial with imatinib 100 mg daily was attempted in September 2016 (at that time midostaurin was not available in Italy). Imatinib was withdrawn after 6 months due to gastrointestinal intolerance and no response. The patient subsequently received management through steroid use and symptomatic medications. In late 2018, following Italian approval, the patient commenced second-line treatment with midostaurin at a daily dosage of 200 mg. After 1 year, however, the therapy was discontinued due to gastrointestinal intolerance, despite achieving partial disease control. A third-line treatment was initiated, involving PEG-Interferon 135 μg weekly. This regimen was administered intermittently from May to December 2019 due to drug unavailability. By the time of referral to our center in December 2019, the patient exhibited pancytopenia, extensive BM infiltration by abnormal MCs (60%), and reported bone pain and fatigue. A subsequent treatment phase involved the administration of five cycles of cladribine 0.14 mg/kg subcutaneously on a monthly schedule. This approach was terminated in June 2020 due to a lack of response and a major infectious complication (pneumonia). Considering patient’s age and the availability of an Human leukocyte antigen (HLA)-identical sibling donor, the option of Hematopoietic Stem Cell Transplantation (HSCT) was evaluated. In December 2020, avapritinib 200 mg daily was started. During the initial months of avapritinib therapy, no major complications were encountered. After 2 months, a BM assessment revealed sparse microaggregates of CD25+ MCs at core biopsy, and the KIT D816V mutation was undetectable at Real time - Polymerase chain reaction (RT-PCR). Concurrently, improvements in thrombocytopenia and neutropenia were observed, while partial alleviation of bone pain and fatigue was reported. In February 2021, 3 months after initiating avapritinib, the patient underwent allogeneic HSCT. Avapritinib was discontinued shortly before the conditioning regimen. Following HSCT, the patient experienced poor graft function, characterized by incomplete recovery of platelet count and hemoglobin, necessitating weekly transfusion. However, over time, BM function progressively improved, and full recovery was achieved 18 months after the transplant. Several BM evaluations were performed, with the most recent one in February 2023. All assessments confirmed complete donor chimerism and undetectable KIT mutation. Bone pain persisted as the only symptom after HSCT throughout the period of poor graft function. Tryptase levels remained stable within the normal range since HSCT (last determination in March 2023).
Case 2: AdvSM refractory to multiple prior therapies
A 52-year-old man was diagnosed with SM in 2013. Upon referral, his medical history included maculopapular cutaneous mastocytosis (with onset at 3 years of age), osteoporosis with vertebral fragility fractures (noted at 47 years old and treated with alendronate), and hymenoptera venom allergy.
During his initial examination, he presented with grade 1 anemia, grade 2 thrombocytopenia, a basal serum tryptase level of 175 µg/L, and palpable hepatosplenomegaly. Dual-energy X-ray absorptiometry and total skeleton X-ray showed mixed osteolytic and osteosclerotic lesions with multiple osteoporotic fractures. A BM assessment confirmed the presence of multifocal clusters of atypical MCs and the presence of KIT D816V mutation. Given the occurrence of one C-finding, his SM was classified as ASM according to the WHO classification. 12
Cytoreductive therapy was initiated in December 2013 with PEG-interferon alpha-2b at a dosage of 80 μg/week, which was discontinued in March 2014 due to hematological toxicity and ineffectiveness. Second-line therapy with midostaurin was started in October 2014 through a CUP, initially at a dose of 50 mg twice daily, then increased to 100 mg bid. This treatment achieved a minor partial clinical response based on IWG-MRT and ECNM criteria. 13 However, by December 2017, the patient’s anemia and thrombocytopenia worsened, accompanied by increasing serum tryptase levels, indicative of disease progression. Next Generation Sequencing (NGS) testing for myeloid genes on peripheral blood (PB) cells revealed an SF3B1 K666N mutation [Variant Allele Frequency (VAF) 12.6%]. In April 2018, a BM smear documented the presence of more than 20% atypical MCs thus leading to a diagnosis of mast cell leukemia (MCL). Third-line therapy involved cladribine 0.14 mg/kg subcutaneously for 5 days every month for four cycles, but there was no improvement. In February 2019, treatment with ripretinib 150 mg twice daily was initiated under a clinical trial protocol, resulting in notable clinical improvement, transfusion independence, and reduction of splenomegaly. However, in November 2020, the disease progressed with weight loss, grade 2 diarrhea, transfusion-dependent anemia (1–2 RBC units per month) and severe splenomegaly (13 cm below the left costal margin), accompanied by increased MCs infiltration (60–70% of total cellularity at BM biopsy).
The patient then began avapritinib as a fifth line of therapy, starting at 100 mg daily, then escalating to 200 mg/day after 2 weeks. After 2 months of treatment, transfusion independence was regained yet persistent diarrhea (grade 1) was observed, possibly drug related. Notably, a reduction in spleen volume was noticed after 6 months of treatment, with the best response occurring in October 2021 (10 cm reduction of the longitudinal spleen diameter).
Two years later, the patient remains on avapritinib treatment, with good control of clinical symptoms and laboratory parameters. Despite a reduction in the treatment dose to 100 mg/day to mitigate gastrointestinal side effects, a sustained clinical response is observed, with continuous drug exposure without interruption. This response has led to improved clinical outcomes, stabilization of skeletal involvement, decreased serum tryptase levels (from 284 to 40 µg/L), and increased hemoglobin levels. Though a full assessment of response per IWG-MRT-ECNM criteria was hindered by the lack of BM evaluation due to previous bleeding complications, the patient continues to experience clinical benefits from avapritinib treatment.
Question 1: Depth of response to available treatments: IS disease eradication possible?
AdvSM is a complex and diverse myeloid neoplasm that poses significant challenges in designing therapeutic strategies with a curative intent. Evaluating treatment response is often complicated due to the wide range of clinical manifestations, 14 and the stringent criteria required for defining complete response are rarely met (Table 2). 12 Patients can experience symptom improvement without substantial reductions in indicators of SM such as BM infiltration, KIT allele burden, and tryptase levels. This frequently leads clinicians to focus on achieving meaningful disease control through sequential treatments rather than aiming for disease eradication. Allogeneic transplant (HSCT) is possible for only a minority of patients, and is supported by limited published data (the largest series included 57 patients), 15 often affected by the pre-procedure MC burden.
Response criteria according to IWG-MRT-ECNM joint consensus.

For patients with ASM, MCL, and SM associated with a myeloid neoplasm;
Only valid as a response criterion if the pretreatment serum tryptase level is ⩾40 ng/mL;
Preservation of at least one CI finding permits a patient to maintain the response of ‘CI’ if one or more CI findings are lost but none meet criteria for PD. However, if one or more of the CI findings become PD, then the CI finding assignment is lost and the patient meets criteria for PD. The baseline value for evaluating PD is the pretreatment measurement(s).
If valued with computed tomography or magnetic resonance imaging progression in splenomegaly is defined as an increase in spleen volume of at least 25%.
ANC, absolute neutrophil count; ASM, aggressive systemic mastocytosis; BM, bone marrow; CI, clinical improvement; CR, complete remission; Hb, hemoglobin; IWG-MRT-ECNM: International Working Group-Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) & European Competence Network on Mastocytosis (ECNM); LOR, loss of response; MCL, mast cell leukemia; PD, progressive disease; PR, partial remission; SD, stable disease; SM, systemic mastocytosis.
Avapritinib has demonstrated the potential to induce profound responses, including molecular remissions.9,11,16 This makes it reasonable to consider the drug as a bridge to HSCT for a larger group of potentially eligible patients compared to earlier eras. Even for patients not suitable for HSCT, achieving a deep reduction in disease burden can contribute to long-term disease control. Avapritinib has shown efficacy in overcoming resistance to prior therapies, including first-generation KIT inhibitors 17 .
The two cases presented highlight a common challenge in SM setting: patients who are refractory or only transiently responding to multiple lines of treatment, experiencing prolonged constitutional symptoms, and organ-related toxicities, including cytopenias. Conventional chemotherapy and first-generation KIT inhibitors have proven ineffective in reducing BM infiltration, clearing KIT mutation or alleviating clinical symptoms in these cases. Additionally, treatment interruptions occurred due to varying degrees of intolerance. Despite the failure of previous treatments, avapritinib led to a rapid reduction in MC infiltration and amelioration of symptom burden in both cases. As suggested by preliminary data, 16 the efficacy of avapritinib is not significantly affected by adverse risk genotypes (i.e. mutations in SRSF2, ASXL1, RUNX1, S/A/R), a finding needing confirmation in larger series. As in case 1, achieving profound responses with a relatively safe profile has become a crucial pre-requisite for considering allogeneic HSCT, historically featured by dismal outcome and high relapse rate. 15 For eligible patients, avapritinib promises to provide an important contribution to therapeutic strategies with curative intent.
Case 3: A patient with severe bone manifestations
A 59-year-old woman with a history of severe osteoporosis and multiple vertebral fractures, managed with bisphosphonates and denosumab, was referred to our center in November 2021. An MRI in September 2020 revealed a lytic lesion on the right iliac wing. Biopsy of the lesion demonstrated infiltration by atypical MCs, and established the presence of KIT D816V mutation on biopsy-extracted DNA. The patient underwent two cycles of cladribine followed by radiotherapy on the symptomatic lesion (20 Gy/five fractions in November 2020 and then 800 cGy in a single session in September 2021). Despite these interventions, persistent lumbosacral pain radiating to the right leg led to the initiation of midostaurin 50 mg bid from March 2021 to November 2021, yielding only temporary relief.
A subsequent BM biopsy revealed multifocal infiltrates of spindle-shaped MCs, with 0.07% aberrant (CD25+/CD2+) MCs at flow cytometry and KIT D816V mutation on RT-PCR. Tryptase level was 14.2 µg/L. The clinical presentation was consistent with ASM due to a C-finding (large osteolytic lesion), without apparent impairment of other organs. Due to a lack of response to the prior two lines of therapy, avapritinib was initiated at a daily dose of 200 mg. Within 3 weeks, the patient experienced facial and bilateral lower limb edema and tachycardia, prompting a dose reduction to 100 mg daily, with rapid resolution of these manifestations. The patient reported a rapid improvement in bone pain associated with the right iliac wing lesion, while dorso-lumbar spine pain persisted, consistent with the longstanding severe osteoporosis. Subsequent CT scans in June 2022, after 2 months of avapritinib treatment, demonstrated multiple vertebral fractures associated with osteoporosis (L1, L2, L3) and stability of the right iliac wing lesion, without cortical remodeling or contrast enhancement. The patient is still on treatment for more than 2 years with maintained clinical response.
Question 2: What role for KIT inhibitors in the bone involvement of SM?
Bone involvement is a frequent manifestation in SM, exhibiting a wide range of clinical presentations, from osteoporosis with fragility fractures to diffuse bone pain and the presence of osteolytic and/or focal sclerotic lesions and osteosclerosis. 18 The sites and the characteristics of bone involvement are influenced by various mechanisms, including the direct colonization of BM by MCs, and the effects of mediators on bone metabolism. 19 Osteoporosis, the most frequent bone complication of SM, can be easily detected through Dual-energy X-ray absorptiometry (DXA) scan and is not considered indicative of disease aggressiveness. However, the symptom burden might not align with radiological findings. Conversely, the rare occurrence of large osteolytic lesions suggests diagnosis of an ASM variant, and a histologic confirmation by a focused biopsy is generally recommended in order to exclude solid tumor metastases. 20 Among available therapeutic modalities, interferon has been suggested as a potential option due to its presumed beneficial effects on osteoporosis.19,21,22 Evaluating the response of bone disease presents its own challenges, as indicators of response, whether clinical or laboratory-based, can exhibit slow improvement.
Managing bone disease in SM patients is complex. On one side, a rapid reduction of MCs causing infiltration and bone lysis is desirable to obtain local control of such severe, albeit rare, manifestations of AdvSM, as occurred in the case 3. On the other hand, it may be rational to expect an improvement of much more common SM-related osteoporosis from KIT inhibition. However, the regression of severe, consolidated osteoporosis is quite difficult to appraise and likely requires long periods of drug exposure in order to prevent fragility fractures, as they were actually experienced by patient no. 3 while on therapy. As avapritinib and other second-generation KIT inhibitors are increasingly explored in non-AdvSM setting, it will be interesting to investigate whether early utilization of targeted agents can slow down and eventually revert bone demineralization. In non-AdvSM, such approach clearly needs unequivocal reassurances in terms of safety profile.
Case 4: KIT negative MCL
A 61-year-old man presented with a pathologic fracture of the left proximal humerus in 2019 and underwent prosthetic surgery for the underlying lytic lesion. The affected bone segment displayed newly formed tissue, with undifferentiated features. Subsequent total body imaging tests, including 18FDG PET/CT, bone scintigraphy, and total body CT, did not identify additional lesions. Therefore, a watchful waiting approach was adopted, with periodic radiological monitoring. However, in May 2021, his clinical conditions worsened, prompting hospitalization. A total body fluorodeoxyglucose (FDG)/Positron emission tomography (PET) indicated diffuse skeletal involvement with increased uptake noted in left mastoid, cranium, and right iliac crest. This was confirmed by total body CT scan which revealed diffuse lytic lesions, with the largest located in the right iliac crest. BM biopsy showed grade 2 fibrosis and infiltration by atypical MCs accounting for over 90% of total cells. KIT D816V mutation was absent in both BM and PB samples. Initial treatment included steroids (prednisone 25 mg daily), hydroxyurea (500 mg daily), and bisphosphonate (zoledronic acid). Due to the diagnosis of MCL, potentially evolving from a previous, undiagnosed MC sarcoma in the left humerus, cytoreductive therapy with midostaurin was started in July 2021 at an initial dose of 50 mg bid. After 1 month of therapy, the patient reported improved bone pain but also experienced weight loss and peripheral edema. A chest radiography revealed massive right pleural effusion, and abdominal ultrasound showed rapidly progressive splenomegaly (bipolar diameter 17.5 cm) and ascites.
Upon referral to our Center, the patient was in poor clinical conditions [Eastern Cooperative Oncology Group Performance Status (ECOG PS = 3)], with diffuse skin rash, superior displacement of the left humerus prosthesis, severe ascites unresponsive to diuretics, bilateral pleural effusion, and bilateral edema of the lower limb. A subsequent BM evaluation showed metaplasia of atypical MCs, and further testing confirmed the absence of KIT D816V mutation. The NGS myeloid panel did not detect any mutation in the studied genes, including KIT hot spots (exons 1, 2, 8–11, 13, 17). Blood tests showed mild leukocytosis, neutrophilia without MCs at PB smear or other abnormal counts, normal coagulation parameters, hypoalbuminemia, elevated serum tryptase (1976 µg/L), and alkaline phosphatase (244 U/L).
A CUP for the KIT inhibitor avapritinib was obtained, and the patient began treatment at a daily dose of 200 mg. After 1 month of treatment, the patient experienced disease progression, characterized by an increase in ascites and pleural effusion, confusion due to metabolic acidosis, and deceased shortly after.
Case 5: Non-canonical KIT mutation
A 66-year-old man was diagnosed with systemic mastocytosis with an associated hematologic neoplasm (SM-AHN) (namely chronic myelomonocytic leukemia, CMML type 1, without excess of blasts and marked eosinophilia) in February 2016. The patient’s medical history prior to this diagnosis was unremarkable. At diagnosis, his PB counts exhibited WBC 25.5 × 109/L with absolute monocytic, and eosinophils count equal to 2.3 and 9.3 × 109/L, respectively. Tryptase level was measured at 100 µg/L. In terms of genetic analysis, the KIT gene was analyzed for mutations by DNA analysis with PCR amplification and direct sequencing. Analyzed gene regions included exons 8, 9, 10, 11, 13, 14, and 17. The DNA analysis showed a mutation 1621A>C on exon 10 causing amino acid substitution p.M541L. In the Single Nucleotide Polymorphism Database (dbSNP) this KIT mutation is identified as rs3822214. No other mutations were found.
Due to the peculiar genotype, a treatment challenge with imatinib at a daily dose of 400 mg was initiated, without achieving any evidence of a response. Subsequently, in March 2017 midostaurin was introduced at 100 mg bid dose as part of a CUP. The patient exhibited a rapid response, with normalization of PB counts and liver function tests within a few months. Tryptase level decreased albeit remaining abnormal (54 µg/L at nadir after 9 months of treatment). Notably, the response in PB markers did not mirror the disease burden, as the spleen volume remained enlarged (17 cm of bipolar diameter) and BM infiltration by MCs persisted at a similar level as the pretreatment baseline. While on midostaurin treatment, an NGS analysis was carried out on PB in June 2020 revealing mutations in SRSF2 (c.284C>T, VAF 31%), SH2B3 (c.962C>G, VAF 81%), TET2 (c.2428C>T, VAF 42% and c.4367dup, VAF 44%), and RUNX1 (c.508+1G>A, VAF 41%).
Despite this, the patient’s clinical condition remained relatively stable until April 2021. At that time, the patient, still on midostaurin, was hospitalized due to an episode of gastric bleeding. A gastroscopy unveiled esophageal varices attributed to portal hypertension. Concurrently, two liver nodules were detected through a CT scan, and they exhibited high metabolic activity on PET scan. Subsequent biopsies of the liver nodules documented a diagnosis of diffuse large B-cell lymphoma (DLBCL), while random biopsies of the liver tissue highlighted diffuse MC infiltration. The KIT mutation was detected in samples from both sources: the liver tissue and the DLBCL nodules. A BM assessment via core biopsy confirmed the presence of CMML and SM, with MC infiltration around 50% of overall cellularity and without any evidence of infiltration by DLBCL. Molecular analysis of the PB detected the previously identified KIT (allele burden not available).
Clinically, the patient reported fatigue and the picture was mainly featured by portal hypertension with ascites. PB counts were within normal ranges, while Alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), and lactate dehydrogenase (LDH) were mildly elevated. After considering the clinical impact of the concurrent disease manifestations, a lymphoma-targeted immuno-chemotherapy was first started in May 2021, with rituximab and CHOP (three cycles). Following the completion of the third cycle, disease restaging with CT-PET scan showed a complete remission of the lymphoma. However, due to the persistence of SM-related manifestations (portal hypertension and ascites), a CUP for avapritinib was requested and approved in June 2021, although treatment initiation was delayed due to thrombocytopenia. A fourth R-CHOP cycle was administered, which led to a slow and incomplete recovery of platelet count. Additionally, there was the emergence of leukocytosis (70 × 109/L) with monocytosis (40 × 109/L), indicating progression of CMML.
Lymphoma-directed treatment was thus halted and avapritinib dose was started at 100 mg daily.
However, an overt progression to acute myeloid leukemia was documented and treatment with decitabine started. The patient died due to disease progression after the completion of one cycle.
Question 3: Is avapritinib effective on KIT negative/non-canonical mutations?
KIT is a member of the type III tyrosine kinase receptors which holds a critical role in regulating differentiation and survival of MCs. 23 In SM, KIT is constitutively activated due to somatic point mutations located at exon 17 of the gene in more than 90% of patients. 24 Additionally, there are rarer activating KIT alterations that encompass mutations in the extracellular, trans-membrane, or juxta-membrane domains. 25 In a minority of cases, collectively representing 5–10% of cases, no KIT mutations are detected. 26 Further complexity is added to the genotypic picture by the fact that KIT mutations are not exclusively confined to MCs. The Spanish Network on Mastocytosis (REMA) demonstrated the multilineage involvement of the mutation in virtually all patients with AdvSM. 27 Overall, the efficacy of KIT inhibition approach depends on the potency and selectivity of the targeted agent, in combination with the degree to which the MCs rely on KIT-related pathways for their survival. In cases where the KIT D816V mutation is absent, treatment with imatinib has shown a response rate of around 30%, likely related to certain underlying trans-membrane (F522) and juxta-membrane (V560) mutations. 26
Avapritinib is a potent type 1 KIT inhibitor with proven clinical efficacy across various SM variants, provided that one sensitive mutation (i.e. exon 11, and 17 activation loop mutants) is present.9,11
The key to avapritinib’s efficacy lies in its ability to strongly inhibit the activity of KIT D816V mutant. While this peculiarity predisposes to rapid and deep responses in the majority of SM cases, it raises concern in some specific subsets. One notable concern pertains to its effect on concomitant hematological neoplasm (i.e. AHN), as in case 5. Preliminary data from clinical trials are promising from this standpoint.28,29 Additionally, although relatively infrequent, cases of SM lacking the typical KIT D816V mutation need attention. Both cases described in this report (no. 4, 5) presented with severe clinical impairment and were not under treatment for a sufficient duration to experience any amelioration of disease manifestations. Nevertheless, these cases raise the issue whether avapritinib will show the same rapid activity and extent of response rate as in patients with the canonical KIT D816V mutant. A single case report of avapritinib treated KIT-negative form is available in the literature, reporting evidence of a response (tryptase reduction and symptom improvement). 30 These considerations reiterate the need for an extensive genetic characterization of SM, especially in advanced and D816V-negative variants, which should include comprehensive NGS panels and entire KIT gene sequencing to gain insights into the actual efficacy of the drug in this context.
Case 6: Edema and electrolyte imbalances
A 55-year-old female patient was diagnosed with indolent SM in 2012. BM assessment revealed an infiltration by multifocal clusters of CD117/tryptase+ MCs, with the KIT D816V mutation detected. Her serum tryptase was measured at 27 µg/L. After being asymptomatic for 6 years, the patient developed bone pain, fatigue, and nausea. A treatment with imatinib 100 mg was initiated in June 2018. However, by October 2018, the patient ceased imatinib due to a deterioration in her condition. She reported increased fatigue and nausea, alongside the onset of paroxysmal tachycardia. Furthermore, the treatment’s efficacy appeared to be lacking in addressing her symptoms.
Upon referral in December 2018, she complained of persistent bone pain, nausea, and fatigue, and new episodes of diarrhea, unresponsive to anti-mediator therapy (antihistamines, membrane stabilizers, and steroids on demand). Subsequent BM evaluation confirmed MC infiltration; gastroscopy and colonoscopy reported a minimal CD117/tryptase+ MC infiltration, skeletal X-ray, and DEXA scan revealed osteoporosis but no focal lesions. Serum tryptase was measured at 31.3 μg/L. In January 2019, the patient started treatment with midostaurin 200 mg daily, which led to a partial benefit on symptom burden until April 2020. At that point, treatment was halted due to the progressive exacerbation of musculoskeletal and abdominal pain. The patient then began a regimen of cladribine in May 2020, administered subcutaneously at a monthly schedule. After administration of the third cycle, she experienced severe electrolyte imbalances (hypocalcemia, hypomagnesemia, and hypokalemia) necessitating hospitalization for recovery. The patient underwent two additional cycles of cladribine with no major complications, but a disease assessment in November 2020 unveiled grade 3 anemia, increase of serum tryptase to 49.1 µg/L, persistence of BM infiltration, and KIT D816V mutation. Nausea, fatigue, and diarrhea persisted despite a lack of evidence for increased MC infiltration in gastrointestinal random biopsies. Thus, in March 2021 the patient commenced avapritinib at a dosage of 200 mg daily. After 2 months on the treatment, she reported worsening of diarrhea, that severely impacted her daily life, accompanied by periorbital and lower peripheral edema. Biochemical analyses showed severe hypomagnesemia, hypokalemia, hypocalcemia, and hypernatremia. She underwent proper fluid and electrolytes correction and discontinued avapritinib for 1 month. After recovery, she resumed avapritinib at a reduced dosage of 100 mg daily, but once again worsening of diarrhea, electrolytes imbalances, and reappearance of peripheral edema led to permanent discontinuation of the drug in August 2021. Quite unexpectedly, upon re-evaluation, serum tryptase levels had normalized, and there was a significant reduction in BM MC infiltration. The patient also reported amelioration of bone pain, nausea, and fatigue. Moreover, diarrhea progressively lessened and eventually disappeared few weeks after discontinuing avapritinib and receiving electrolyte supplementation. Even at 19 months after cessation of treatment, the patient remains in partial response, with no significant diarrhea, peripheral edema, or nausea. Unfortunately, she reported worsening of diffuse bone pain since April 2022 and occurrence of a vertebral fracture after an accidental fall in August 2022, further impacting her overall quality of life.
Case 7: Refractory ASM experiencing an infectious complication
A 56-year-old male patient was diagnosed with ASM in 2019. BM biopsy showed infiltration of multifocal clusters of CD117/tryptase+ MCs and the KIT D816V mutation was detected. Serum tryptase levels were elevated at 155 µg/L. The patient presented diffuse cutaneous maculopapular lesions positive for CD117/tryptase+ MCs, grade 3 anemia, osteoporosis and multiple osteolytic lesions detected by DEXA scan and skeletal X-ray, respectively.
In July 2019, the patient initiated treatment with midostaurin, obtaining a partial reduction of skin lesions and improvement in anemia. The treatment was discontinued in February 2020 due to the progression of BM infiltration, reaching over 70% of cellularity, alongside an exacerbation of skin lesions. Since then, a progressive clinical deterioration occurred, with the subsequent development of ascitic effusion. By September 2020, he had started diuretics and hydroxyurea, with some clinical benefit. Upon referral to our center in October 2020, BM infiltration and KIT D816V mutation were documented, and serum tryptase level was 384 µg/L. CT scan showed multiple lymph nodes enlargements, hepatosplenomegaly, ascites, and pleural effusion. A lymph node biopsy confirmed the involvement by SM.
Considering progression of disease and worsening of symptoms, in November 2020 the patient commenced treatment with cladribine 0.14 mg/kg subcutaneously, administered every 2 months to minimize toxicity. Despite receiving five cycles of cladribine, the patient did not experience any noticeable clinical or hematologic improvement. Subsequently, in September 2021, the patient transitioned to avapritinib therapy 200 mg daily through a CUP. While there were sporadic episodes of gastrointestinal intolerance, the patient managed to consistently take the drug with no significant complications. Over a span of 6 months of treatment, the patient obtained a significant reduction of BM and lymph nodes involvement, normalization of serum tryptase, resolution of ascites, and pleural effusion. This period of treatment was sustained until July 2022. At that point, the patient reported subjective dyspnea and experienced severe anemia (Hb 6.4 g/dL), with normal platelet and absolute neutrophil count. BM evaluation did not show an increase of MC infiltration, and bleeding was ruled out as the cause of anemia. In response, avapritinib dosage was initially reduced to 100 mg daily and ultimately discontinued. The patient underwent blood transfusion and a chest X-ray documented pneumonia. He was therefore admitted to the hospital for antibiotic therapy. On physical examination he presented with hypotension, tachycardia, but no fever. Pseudomonas aeruginosa was detected on sputum examination and antibiotic therapy was adapted accordingly. Regrettably, the patient’s condition continued to worsen, and he passed away due to respiratory failure.
Question 4: The safety profile
The safety profile of avapritinib dose, as observed in the EXPLORER and PATHFINDER clinical trials, was overall consistent with the clinical context of advSM.9,11,16 It is conceivable that outside the setting of clinical trials, an even more advanced and pretreated patient population will have access to the drug, a figure that calls for optimization of safety precautions. The experience with CUP somehow reflects what we might expect from a wider use of avapritinib and can aid to develop the most appropriate working-up procedures and help manage some of the potential complications.
The published clinical trials have outlined certain nonhematological adverse effects that were relatively common among patients receiving avapritinib, including peripheral/periorbital edema, diarrhea, nausea, and cognitive effects. 31 Hematological toxicity observed encompassed neutropenia, anemia, and thrombocytopenia. Notably, intracranial bleeding, associated with thrombocytopenia, occurred in 13% of patients in the EXPLORER study. Consequently, the trial’s inclusion criteria were amended, and the FDA label recommends against using the drug with a platelet count less than 50 × 109/L.
In patient no. 6, the temporal occurrence of gastrointestinal symptoms (preceding avapritinib start but worsening during treatment), and peripheral edema suggest the adverse events to be directly related to the drug. The symptoms were not alleviated by dose reduction, ultimately leading to treatment withdrawal due to intolerance. It is interesting to note the concomitant disease response, maintained also after treatment discontinuation, once again underlying the potentiality of the agent in terms of response depth. Patient no. 7, on the other hand, experienced a different pattern of toxicity, with a delayed onset of transfusion-requiring anemia, and bacterial pneumonia, culminating in respiratory failure. However, we could not establish a direct correlation of the infectious events with ongoing treatment.
These cases underscore the necessity for thorough vigilance regarding potential complications of avapritinib when used in real-world clinical practice. In terms of hematological toxicity and infectious complications, a proper monitoring of blood counts, particularly in heavily pretreated patients, is strongly recommended. Implementing pre-emptive measures against opportunistic infections (i.e. trimethoprim/sulfamethoxazole for Pneumocystis jiroveci prevention, periodic viral monitoring) could be considered in high-risk subsets. Additionally, the use of growth factors to manage cytopenias could prove beneficial. A reduction of the dose, from the conventional 200 mg QD, can prove to be very useful in mitigating some adverse effects. 31
Conclusions and future perspectives
Indeed, this case series provides valuable insights into the challenges and opportunities associated with the real-world use of avapritinib for the treatment of AdvSM.
Data presented here confirm the drug as being active after multiple line treatments by providing rapid achievement of profound responses, thus standing also as a suitable bridging strategy to HSCT in eligible patients. The anticipated large use of avapritinib in the near future will require a careful monitoring of possible complications especially in heavily pretreated patients. Moreover, some issues remain to be elucidated, as the actual efficacy on challenging disease manifestations (i.e. portal hypertension, long-term osteoporosis) and in SM subsets (i.e. KIT D816V negative forms).
