Abstract
Background:
For more than 2 years medical practice has been dealing with the Covid-19 pandemic. Atypical symptoms, such as frostbites and acrosyndromes, have appeared, and autoimmune anemias (some of which with cold agglutinins) have been described.
Objectives:
We planned to study the prevalence of positive direct Coombs tests (DCTs) and hemolytic autoimmune anemia in patients infected with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) and its correlation with complications, and then investigate the impact of the infection on iron metabolism.
Design:
This is an observational, cross-sectional, single-center, exploratory study.
Methods:
We obtained Coombs tests in a population of 179 infected patients at the CHU of Liège. We then studied iron metabolism in some of these patients, by measuring serum ferritin, erythropoietin (EPO), erythroferrone and hepcidin.
Results:
We did not identify any case of autoimmune hemolysis. However, there was a 20.3% prevalence of positive DCT, mainly with IgG (91.7%). These patients, compared to DCT-negative patients, were not only more anemic and transfused, but also required more transfers to intensive care units and had longer hospital stays and mechanical ventilation. The pattern of anemia was consistent with the anemia of inflammation, showing elevated hepcidin and ferritin levels, while EPO and erythroferrone values were lower than expected at this degree of anemia. Erythroferrone was higher and Hb was lower in DCT-positive patients. Finally, we identified a correlation between iron parameters and complicated forms of infection.
Conclusion:
Covid-19 patients suffered from inflammatory anemia with more severe forms of infection correlated to positive DCT status. This could potentially be of interest for future clinical practice.
Introduction
According to the Johns Hopkins data center (as on 17 February 2023, https://coronavirus.jhu.edu/map.html) the Covid-19 epidemic is responsible for more than 673 million infections and 6.8 million deaths worldwide since its beginning. Belgium has been particularly affected with a total number of 4.7 million cases and 33,000 deaths by 17 February 2023 (https://covid-19.sciensano.be).
We were challenged, within our hospital, by the discovery of two cases of cold agglutinin hemolytic anemia in patients with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) during the first wave. Both cases were positive for C3d and IgM, with cold agglutinin titers of 1/64 and 1/2048 at 20°C, respectively (thermal amplitude was measured until 20°C in both cases). Hemolysis spontaneously improved within a few days during resolution of the infection, with IgM-to-IgG seroconversion and disappearance of cold agglutinins. The literature has described several cases of autoimmune cytopenia,1–4 as well as various skin disorders (acrosyndrome, frostbite) that could be attributed to cold agglutinins 5 in Covid-19 patients.
Cold agglutinin hemolytic anemia is an autoimmune disorder characterized by the presence of autoantibodies (majority of cases with IgM isotype and rare cases with IgG isotype) active at 0°C–4°C that agglutinate red blood cells. Then there is activation of the classical complement pathway, followed by extravascular hemolysis (mainly hepatic) through phagocytosis of red blood cells opsonized by C3d. Biologically, one can find a direct Coombs test (DCT) positive for C3d (rarely weakly positive for IgG) and a cold agglutinin titer ⩾1/64 at 4°C. The thermal amplitude, rather than IgM antibody titers per se, is more suggestive of clinical significance and severity of hemolysis. Cold agglutinin hemolytic anemias are divided into ‘cold agglutinin disease’ (a clonal lymphoproliferative pathology) and ‘cold agglutinin syndrome’ (polyclonal and secondary to infections, typically mycoplasma infections in adults and Epstein–Barr virus or cytomegalovirus in children). 6
Cold agglutinin-induced hemolysis involves complement. Several studies have shown an important role of the complement in the pathophysiology of SARS-CoV-2 infection.7–10 Interactions between the complement, inflammation and coagulation could account for some aspects that are not yet understood. In addition, a paper published in 2020 11 has shown a high prevalence (46%) of positive DCT among infected patients, correlating with more anemia and higher transfusion requirements.
Inflammation is another well-known cause of anemia; it affects iron metabolism. Through its action with interleukin-6, inflammation increases the hepatic synthesis of hepcidin, which is the key hormone in iron metabolism. Its production by hepatocytes is also regulated by iron stores, anemia, erythropoiesis and hypoxemia. Hepcidin binds to ferroportin on enterocytes, macrophages and hepatocytes, and induces its degradation. This blocks the iron release by these cells into the blood. Inside cells, iron is bound to ferritin, which reflects the body’s iron stores. A recently identified protein, erythroferrone, links iron metabolism and erythropoiesis; it is produced by erythroblasts via the JAK2-STAT5 pathway. It inhibits hepcidin synthesis, thus making more iron available for erythropoiesis. 12
The aim of our study was to assess the different origins of anemia in patients infected by SARS-CoV-2.
More precisely, the objectives of this work were:
- To evaluate the prevalence of positive DCT and autoimmune hemolytic anemia, in particular cold agglutinin anemia, in patients hospitalized for Covid-19 at the University Hospital of Liège between September and December 2020.
- To search for a possible link between a positive DCT, anemia and the occurrence of complications and mortality.
- To study the origin of anemia and the impact of SARS-CoV-2 infection on iron metabolism, as well as the possible correlations between iron parameters and complications.
Methods
We have followed the guidelines for observational studies according to ‘Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement’. 13 The checklist is available in Supplemental Material.
Study population
The first part of our study focused on adult patients hospitalized at the CHU de Liège for SARS-CoV-2 infection (confirmed by Reverse transcription polymerase chain reaction or RT-PCR) during the second wave, between September and December 2020. Our cohort consisted of 179 patients, 72% of whom were men, with an average age of 66.4 years. In this population we studied the prevalence of positive DCT (including cold agglutinins) and its implication in biological and clinical issues.
The second part of our study aimed to document the impact of Covid-19 infection on iron metabolism and erythropoiesis. We used biobanked samples previously collected for studies on Covid-19. For some patients, we had several samples taken during their hospitalization. Of the 179 patients from our initial population, 52 had biobanked samples, 18 in the ‘positive DCT’ group and 34 in the ‘negative DCT’ group. We added 12 other patients with Covid-19 for whom we already had a minimum of three samples in the biobank and who formed a third group (DCT status unknown). The total cohort for the study of iron metabolism thus included 64 patients.
Laboratory measurements
The standard admission laboratory tests were measured on fresh blood and included complete blood count with reticulocytes count, mean corpuscular volume (MCV), haptoglobin, lactate dehydrogenases (LDH), ferritin, bilirubin, c-reactive protein (CRP) and autoantibody testing, that is, direct and indirect Coombs test with elution, testing for cold agglutinins.
Biobanked serum samples stored at −80°C until analysis were used to measure the parameters of iron metabolism and erythropoiesis [ferritin, hepcidin, erythroferrone and erythropoietin (EPO)]. Ferritin levels were quantified by chemiluminescence microparticle immunoassay and serum hepcidin-25 levels were quantified by a liquid chromatography method coupled with mass spectrometry.14,15 Erythroferrone assays were carried out using a specific ELISA kit, according to the protocol provided by the manufacturer (SKU # ERF-001, Intrinsic LifeSciences, The BioIron Company, La Jolla, CA, USA). The kit has a detection range of 0.16–10 ng/mL. EPO levels were determined by chemiluminescence (Access EPO kit, Beckman Coulter, Brea, CA, USA).
Statistical analyses
The clinical parameters and criteria for the severity of SARS-CoV-2 infection were compared within the initial cohort of patients according to the positivity status of the DCT. The binary variables are presented using frequency tables and compared using chi-squared tests (or Fisher’s exact tests if sample size was too small). When the normality assumption was valid, continuous variables were summarized by mean ± standard deviation and compared using Student’s t-tests. Otherwise, continuous variables were summarized by median (first and third quartiles) and compared using Student’s t-tests on log-transformed data.
The four variables of iron metabolism (ferritin, hepcidin, erythroferrone and EPO) were studied according to the positivity status of the DCT (positive versus negative, the third ‘unknown’ group was excluded) using Student’s t-tests on log-transformed data. The evolution over time of the different variables was analyzed using mixed models.
The association of iron parameters with biological values was studied in the whole cohort (whatever the Coombs status) using Spearman’s rank correlation and the relationship between the first measurements of iron parameters and the occurrence of complications during hospitalization was studied using logistic regression models. When an effect was demonstrated, a Receiver Operator Characteristic (ROC) curve was defined to study the effect of the cut-off value on the sensitivity and specificity of the variables. From this curve, Youden’s criterion was used to determine an optimal threshold for each parameter.
Results were considered significant at the 5% uncertainty level (p < 0.05). Calculations were performed using SAS (Version 9.4; Analytics Software and Solution, SAS Institute, Cary, NC) and R (Version 4.1.0; R Foundation for Statistical Computing, Vienna, Austria).
Results
Prevalence of positive Coombs tests
We obtained a total population of 179 patients over the predetermined period. Among them, 141 had a negative DCT and 38 had a positive test (among them, two were already known to be DCT positive before their Covid-19, both in IgG). The prevalence of new positive DCT was therefore 20.3% (36/177).
Of the 36 patients, 33 were positive for IgG (91.7%), 2 for C3d (5.5%) and 1 for C3d and IgG (2.8%). Only two patients presented traces of cold agglutinins (insufficient to establish the titer), one associated with IgG, the other with C3d. None of the 179 patients presented with biological hemolysis.
Then, comparing the two groups according to the DCT (Table 1), we noted more anemia in the positive patients, with both hemoglobin at admission and nadir hemoglobin values being statistically lower (12.4 versus 13.4 g/dL and 9.2 versus 10.8 g/dL, respectively). Positive DCT patients were more transfused (39.5% versus 11.3%), and the number of units transfused per patient was significantly higher. Maximum CRP, bilirubin and maximum bilirubin levels were significantly higher in positive DCT patients, while other hemolytic parameters (haptoglobin, reticulocytes, LDH) were similar in the two groups. We did not perform complement analysis.
Comparison of clinical and biological parameters for patients with negative versus positive DCT.

p Values based on Fisher’s exact test or Student’s t-test on log-transformed data.
The number is inferior to the total cohort due to missing data.
CRP, c-reactive protein; d, days; DCT, direct Coombs test; LDH, lactate dehydrogenase; M, mean; MCV, mean corpuscular volume; N, number of patients; Q1–Q3, first–third quartile; RBC, red blood cells; SD, standard deviation.
Regarding complications, positive DCT patients were more often transferred to the intensive care unit (ICU) (60.5% versus 41.8%) and the lengths of hospital stay for survivors (25 versus 12 days) and mechanical ventilation were also longer (26 versus 10.5 days) (Table 1).
Therefore, our results show more anemia in Covid-19 patients with a positive DCT, associated with a more severe form of the disease but without clinical hemolysis. To understand the origin of anemia and hypothesize that it is inflammation induced, we studied iron metabolism in the second part of this work.
Iron metabolism
For this part we worked, as previously explained, on a cohort of 64 patients. We first excluded the 12 patients whose Coombs status was unknown. We segregated the 52 remaining patients into two groups, positive and negative DCT. Erythroferrone differed significantly between the two groups, negative DCT patients having a lower value compared to positive ones (4.7 versus 12.9 ng/mL). EPO was also lower in negative DCT patients (26.3 versus 38.3 U/L). Hepcidin and ferritin did not differ between the groups. Results were similar for peak values (Figure 1).
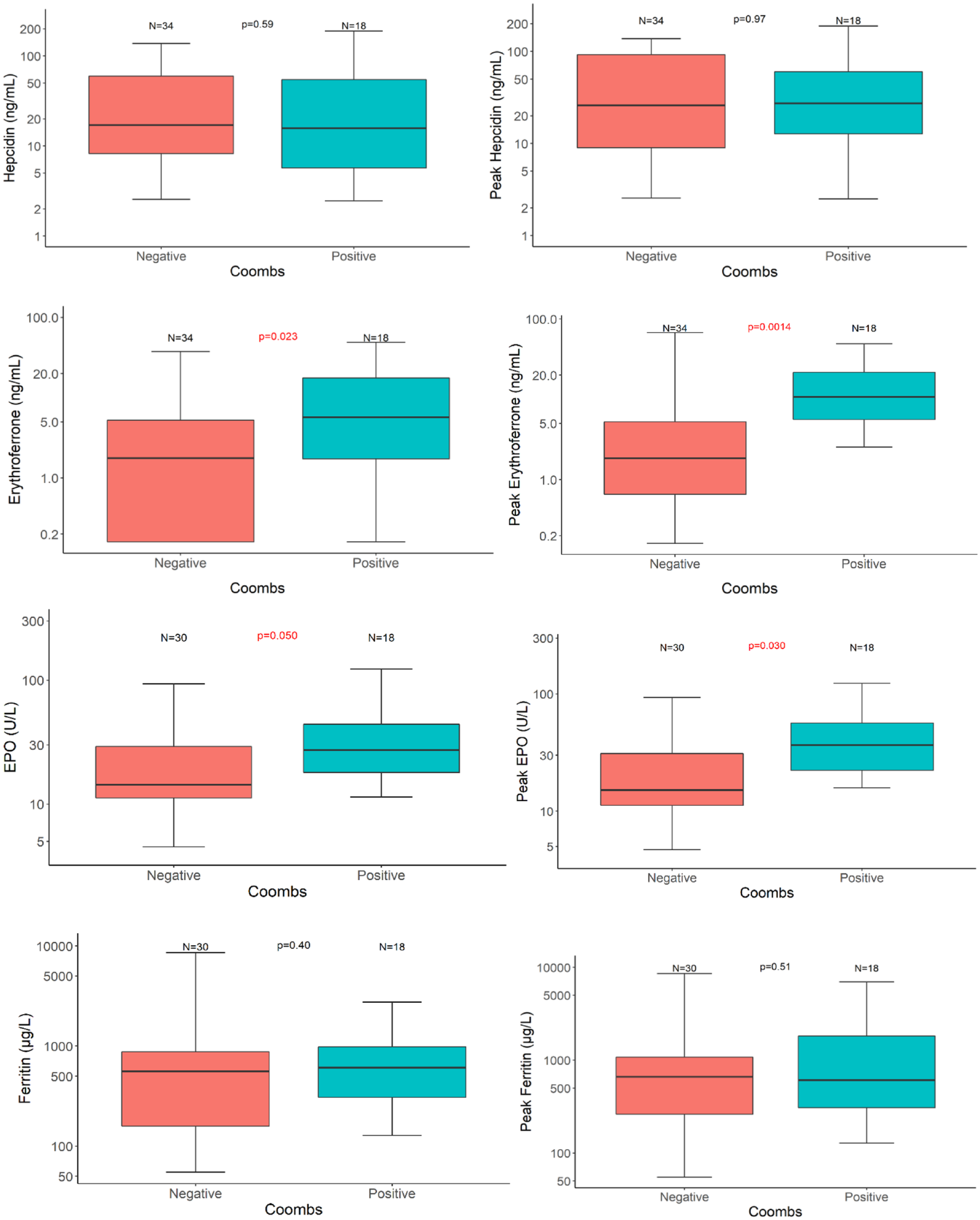
Initial and peak values of iron parameters (median, Q1–Q3) according to DCT (Coombs) status.
For the next analyses, we included back the 12 patients with no DCT result, thus working on the total population of 64 patients. Statistically significant correlations were evidenced between iron parameters, hemoglobin and CRP. Hemoglobin correlated inversely with erythroferrone, EPO and ferritin, while CRP correlated positively with all the four parameters. As expected, hepcidin and ferritin levels were strongly correlated (r = 0.69), as were EPO and erythroferrone values (r = 0.65) (Figure 2).

Correlations among biological parameters.
The temporal evolution of the variables, taking into account the repeated measures for each patient, showed that ferritin decreased significantly over time (p = 0.0002). No significant effect of time was demonstrated for other variables.
Finally, statistically significant correlations were identified between iron/biological parameters and clinical complications. Ferritin correlated with length of hospital stay, need for mechanical ventilation, transfer to ICU and mortality. Hepcidin levels were associated with admission to the ICU and mortality. Elevated erythroferrone correlated with the need for mechanical ventilation and transfusion, and EPO levels with the need for transfusion. High CRP values on admission predicted for referral to the ICU, while high CRP levels during hospitalization (represented by the maximum CRP) correlated with the risk of transfer to ICU, mechanical ventilation, need for transfusion and risk of death (Table 2). Thresholds, sensitivity and specificity of these parameters to predict complications are described in Supplemental Material.
Association between clinical complications and biological parameters.
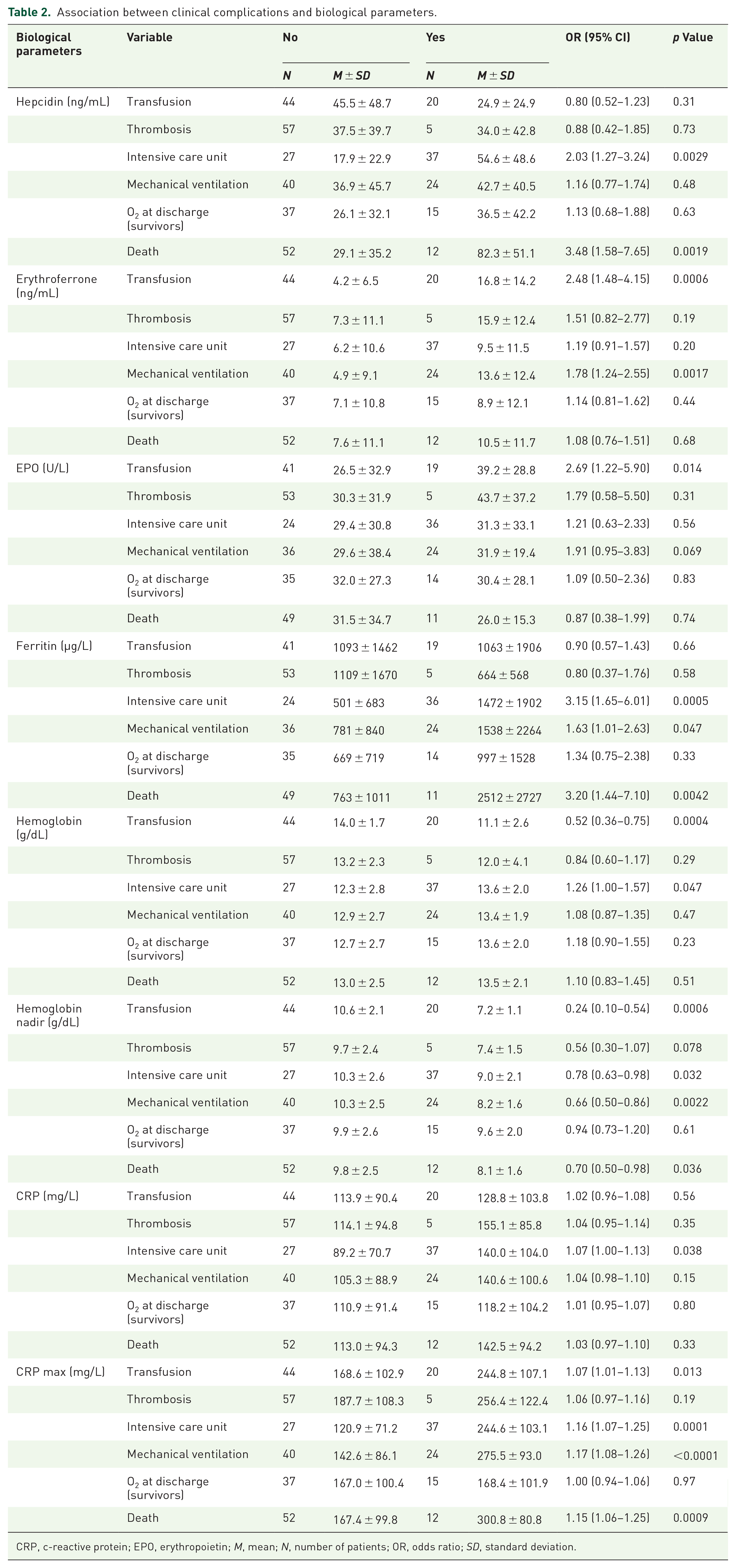
CRP, c-reactive protein; EPO, erythropoietin; M, mean; N, number of patients; OR, odds ratio; SD, standard deviation.
Discussion
We were unable to find a correlation between Covid-19 infection and cold agglutinin hemolysis. Case reports have been published,16,17 testifying that anti-SARS-CoV-2 IgM can have cold agglutinin activity, but these cases seem isolated. However, our results based on the DCT broadly corroborate those of the study by Berzuini, with lower hemoglobin levels on admission, a greater need for transfusion and more erythrocyte units transfused per patient in DCT-positive patients. 11 We had longer hospital stays and longer mechanical ventilation in positive DCT patients. Unlike them we explored hemolysis parameters, and we found no evidence of hemolysis. The difference in the prevalence of positive DCT between their study (46%) and ours (20.3%) may come from the fact that their population consisted only of patients with a severe form of Covid-19 and required a pre-transfusion assessment. Algassim et al. 18 reported a prevalence of positive DCT of 14.7% for anemic patients in ICU and 9% for those in general hospitalization (they used an Hb threshold < 12.5 g/dL defining anemia). Hemoglobin levels were also statistically lower and the length of hospital stay was longer in DCT-positive patients. In the study by Algassim et al., LDH levels were higher (p = 0.005) and this, along with the presence of spherocytes in the DCT-positive patients, led them to conclude that the patients had autoimmune hemolytic anemia (with neither haptoglobin dosage nor significant difference in bilirubin levels). A recent publication, by Hafez et al., showed results very similar to ours, with a prevalence of 20% positive DCT in a cohort of 135 Covid-19 patients. These DCT-positive patients were indeed more anemic and presented with more severe forms of infection in multivariate analysis, but had no biological sign of hemolysis. 19 However, in our study we cannot exclude that comorbidities could induce an imbalance between the two groups, DCT-positive and -negative patients, as we did not collect comorbidity data. This might also contribute to a difference in the rate of anemia besides DCT status.
The possible etiologies of these positive DCT have been discussed by Hendrickson et al., including erythrocyte membrane modifications (based on the fact that elution with a conventional commercial reagent, initially negative, became positive after repeated elution with blood from Covid-19-positive patients citer Berzuini), effects of the complement system or drugs. 20 A French study demonstrated a decrease of CD35 (complement type 1 receptor, involved in the uptake of circulating immune complexes) and an increase of C4d expression on erythrocyte surfaces in more than 80% of their 52 patients with severe Covid-19. CD35 over-solicited by immune complexes is destroyed, leading to the accumulation of C4d deposits and loop activation of complement. 21 Drug-induced hemolysis is unlikely in our cohort because Coombs determinations were performed at admission and therefore were not impacted by the various treatments applied thereafter.
Because DCT-positive patients were more anemic but did not show features of hemolysis, we could not confirm that the anemia was immune induced. Concerning the etiology of anemia in Covid-19 patients, a study by Bergamaschi et al. on 126 patients indicated a strong preponderance of inflammation (65% of cases), followed by iron, vitamin B12 or folic acid deficiency (10% of cases). 22 Our study of iron metabolism aimed at better understanding the pathophysiology of anemia in Covid-19 patients and investigating the potential effect of DCT status. Our results highlighted inflammation and functional iron deficiency, with markedly increased ferritin and hepcidin levels, the two being highly correlated.
During inflammation, interleukins and tumor necrosis factor (TNF) interfere with erythropoiesis by reducing the production of EPO via several mechanisms, such as direct cellular toxicity on renal cells and inhibition of transcription and signaling factors. Relative EPO deficiency may reduce the production of erythroferrone, itself limiting the inhibition of hepcidin. Iron deprivation, further increased in the event of bleeding, accentuates the issue by down-regulation of EPO receptors via the Scribble regulator. 23 However, erythroferrone was not decreased and was rather well within the normal range in our patients, possibly as a result of more hypoxia in the DCT-positive group that had higher admission rates to ICU and longer duration of invasive ventilation. Moreover, the strong correlations between hemoglobin and epo/erythroferrone do not support this hypothesis. The inhibitory effect of erythroferrone did not compensate for the stimulatory effect of inflammation on hepcidin secretion, as shown by the strong correlation between hepcidin and ferritin and the weaker negative correlation between hepcidin and erythroferrone.
The differences of iron parameters depending on DCT status were statistically significant for erythroferrone and EPO and we could interpret them in following manner: positive DCT patients, significantly more anemic and hypoxic, would produce more EPO and erythroferrone to compensate for anemia. The absence of a significant difference in terms of hepcidin and ferritin would suggest that this attempt is thwarted by the importance of inflammation, preventing the release of the iron necessary for adequate erythrocyte regeneration. The associations demonstrated between iron parameters and biological inflammatory values support this hypothesis. In this context, it would be interesting to assay the soluble transferrin receptor (sTfR) to confirm the insufficiency of erythropoiesis in relation to the degree of anemia, as sTfR levels, unlike ferritin, are not increased in inflammation.24,25 Maira et al. also found higher EPO levels in critically ill patients and EPO levels doubling after 7 days although sTfR values did not increase in this group during the same period. 26
Finally, several associations between iron parameters, hemoglobin or CRP, and Covid-19 complications have been identified. Meta-analyses on this topic are rare due to high heterogeneity between studies. Taneri et al. performed a meta-analysis of ferritin in Covid-19 patients. Hemoglobin was lower and ferritin was higher in elderly or comorbid patients. A significant difference in ferritin levels was observed between deceased patients and survivors (mean ferritin 1303 versus 651 ng/mL, p < 0.001). 27 Zhou et al. showed that the ferritin/hepcidin combination reliably predicted severity (defined by tachypnea > 30/min, desaturation at rest < 94%, PaO2/FiO2 ratio < 300 mmHg), with respective sensitivity and specificity of 95.7% and 94.6%. 28 Hippchen et al. showed a correlation between serum iron concentration and the risk of hospitalization with a threshold of < 6 µmol/L (sensitivity and specificity of 94.7% and 67.9%, respectively). 29 Hepcidin was found to be relevant for assessing the severity and mortality of Covid-19 infections in the Italian study by Nai et al., with increased mortality for patients with hepcidin above the median of 361.9 ng/mL. 30
Recently, Frost et al. studied iron metabolism in a cohort of 246 Covid-19 patients and found a significant correlation between serum iron levels and severity of the disease (p = 0.008), but this was not the case for erythroferrone. In their study, hemoglobin also did not correlate with disease severity, although non-survivors had low hemoglobin values at admission. In our study, high ferritin and low hemoglobin and erythroferrone levels were also significantly associated with complications. Frost et al. highlighted the predominance of interleukin-6 in the inflammatory process but surprisingly there was no significant correlation between hepcidin and ferritin. In our study, on the other hand, we established a strong hepcidin/ferritin correlation. Similar to our study, in their study erythroferrone was higher when hemoglobin was low, indicating a probable attempt to compensate for anemia. 31
Regarding the limitations and risks of bias in our study, we can cite the relatively small number of patients in the study cohort and the fact that iron parameters were not available for all patients and were not measured at standardized times.
Conclusion
We could not find a correlation between Covid-19 and cold agglutinin hemolytic anemia and such cases remain quite rare. However, there were more complications in patients with a positive DCT, namely more anemia and a greater need for transfusion, more transfers to the ICU and longer hospital stays and mechanical ventilation. Covid-19 patients develop severe inflammatory anemia, with high hepcidin and ferritin values. In addition, iron parameters correlated with complications, again suggesting the importance of inflammation in SARS-CoV-2 infections. It would be useful to pursue long-term studies in a larger population to consolidate these results and integrate them into clinical practice.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231199837 – Supplemental material for Alterations of erythropoiesis in Covid-19 patients: prevalence of positive Coombs tests and iron metabolism
Supplemental material, sj-docx-1-tah-10.1177_20406207231199837 for Alterations of erythropoiesis in Covid-19 patients: prevalence of positive Coombs tests and iron metabolism by Léa Schmitz, Michelle Pirotte, Alizée Lebeau, Marie Ernst, Marianne Fillet, Anais Devey, Justine Schmitt, Gaël Cobraiville, Marilène Binsfeld, Stéphanie Gofflot, Yves Beguin and Gaëlle Vertenoeil in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
