Abstract
Background:
Umbilical cord blood hematopoietic stem cells are commonly used for hematopoietic system reconstitution in recipients after umbilical cord blood transplantation (UCBT). However, the optimal conditioning regimen for UCBT remains a topic of debate. The exact impact of total body irradiation (TBI) as a part of conditioning regimens remains unknown.
Objectives:
The aim of this study was to evaluate the impacts of TBI on UCBT outcomes.
Design:
This was a multi-institution retrospective study.
Methods:
A retrospective analysis was conducted on the outcomes of 136 patients receiving UCBT. Sixty-nine patients received myeloablative conditioning (MAC), in which 33 underwent TBI and 36 did not, and 67 patients received reduced-intensity conditioning (RIC), in which 43 underwent TBI and 24 did not. Univariate and multivariate analyses were conducted to compare the outcomes and the post-transplant complications between patients who did and did not undergo TBI in the MAC subgroup and RIC subgroup, respectively.
Results:
In the RIC subgroup, patients who underwent TBI had superior overall survival (adjusted hazard ratio [aHR] = 0.25, 95% confidence interval [CI]: 0.09–0.66, p = 0.005) and progression-free survival (aHR = 0.26, 95% CI: 0.10–0.66, p = 0.005). However, in the MAC subgroup, there were no statistically significant differences between those receiving and not receiving TBI.
Conclusion:
In the setting of RIC in UCBT, TBI utilization can improve overall survival and progression-free survival. However, TBI does not show superiority in the MAC setting.
Keywords
Introduction
Umbilical cord blood (UCB) has been used for over 30 years as an alternative source of hematopoietic stem cells for patients in need of an allogeneic hematopoietic stem cell transplantation (HSCT).1–3 As the total nucleated cell (TNC) dose of at least 2.5 × 107 nucleated cells per kilogram of body weight in adult recipients is known to be a critical factor in UCB transplantation (UCBT) success, the use of double cord blood units has greatly expanded the access to adult recipients. 4 The advantages of UCB include its rapid availability, reduced stringency in terms of human leukocyte antigen (HLA) match requirements, and subsequent increase in access to transplants for racial minorities.5–7 Both related and unrelated UCBTs with single or double units have been performed with high rates of success in pediatric and adult settings to treat a variety of medical conditions.8–22
Before undergoing UCBT, recipients often undergo a high-dose chemotherapy called ‘conditioning’ to create space in the bone marrow, suppress the host immune system to facilitate engraftment, and reduce the tumor burden in cases of neoplastic disease. 23 Total body irradiation (TBI) is commonly incorporated into conditioning regimens to aid in these efforts. Although conditioning regimens with higher intensity, such as myeloablative conditioning (MAC), are associated with a lower risk of relapse and graft rejection, there is an increased incidence of transplant-related mortality (TRM). The inclusion of TBI in conditioning regimens can result in heightened organ toxicity and risk of subsequent malignant neoplasm.24–27 Complications associated with MAC are also increased in the elderly and those with comorbidities. To this end, low-dose TBI or non-TBI and reduced-intensity conditioning (RIC) or nonmyeloablative regimens have been implemented as a means of increasing access to HSCT in those at-risk patient populations and benign hematological diseases patient populations.28–33
Despite advances in the use of conditioning regimens in HSCT, optimal conditioning in UCBT remains unknown. Furthermore, there are limited studies comparing conditioning regimens in UCBT, and the role of TBI has not been firmly established. We hypothesized that TBI in combination with RIC regimens may improve UCBT outcomes. In this multicenter retrospective study, we report the impact of TBI as a part of MAC or RIC regimens in patients undergoing UCBT for hematologic malignant diseases.
Methods
Patient population and data collection
This retrospective study included 136 consecutive patients with acute lymphoid leukemia (ALL)/lymphoma and acute myeloid leukemia (AML) who underwent UCBT at one of four institutions from June 2001 to July 2017. Penn State Hershey Medical Center, Medical College of Wisconsin, University of Indiana, and University of Alabama participated in this study. The decision of whether or not to incorporate TBI was determined by individual institutional UCBT protocols. Data were collected through each institution’s medical record system. Standardized data abstraction included demographic information, diagnosis, conditioning regimen, infection disease complications, development of graft-versus-host disease (GVHD), neutrophil engraftment, disease progression status, and cause of death if applicable. GVHD was diagnosed clinically, with histologic confirmation when appropriate. Cytogenetic information was also explored following National Comprehensive Cancer Network (NCCN)’s guidelines. Patients received a TBI dose of 1200–1320 cGy (myeloablative TBI) given as fractionated dosing in MAC or a dose of 200 cGy in RIC. MAC regimens include busulfan (Bu: 8–11 mg/kg intravenous) or cyclophosphamide (Cy: 120 mg/kg) plus thiotepa (Thio: 10 mg/kg) and fludarabine (Flu: 75–150 mg/m2) ± melphalan (Mel: 100 mg/m2) ± antithymocyte globulin (ATG, 8 mg/kg). RIC condition regimens include Flu (180–200 mg/m2) and/or Cy (50 mg/kg) ± Mel (100 mg/m2) ± ATG (6 mg/kg) (Table 1). All patients received granulocyte colony-stimulating factor (G-CSF) until the absolute neutrophil count reached 500 cells/μL or above. All transplant protocols strictly followed American Society for Transplantation and Cellular Therapy (ASTCT) guidelines. All patients provided consent before starting the treatment. All patient data were de-identified for the study.
The MAC and RIC condition regimens in this study.

ATG, antithymocyte globulin; Bu, busulfan; Cy, cyclophosphamide; Flu, fludarabine; MAC, myeloablative condition; Mel, melphalan; RIC, reduced-intensity condition; TBI, total body irradiation; Thio, thiotepa.
Cord blood cell dose, unit, and HLA match selection
Each UCB unit contained
GVHD prophylaxis
GVHD prophylaxis was administered per individual institution protocol. In brief, all patients received GVHD prophylaxis with tacrolimus oral twice daily with a target trough level of 6-12 ng/ml and mycophenolate mofetil (MMF) of 15 mg/kg IV q8 h from day 3. In patients with no active GVHD, MMF was discontinued after day 28, and tacrolimus taper was initiated at day 120 and continued to 180 in patients without GVHD. Those with active viral infections were permitted to undergo faster taper of MMF. GVHD prophylaxis, diagnosis, and management followed NCCN guidelines. 35
Endpoints
Endpoints included overall survival (OS), progression-free survival (PFS), and TRM. OS was defined as the difference in time from transplant to the time of death from any cause. PFS was defined as the difference in time from transplant to the time of disease relapse. TRM was defined as difference in time from transplant to time of death due to a transplant-related cause. Assessment of post-transplant complications included the occurrence of acute GVHD, chronic GVHD, and systemic infections.
Statistical analysis
Demographics, treatment, and clinical characteristics of all patients were summarized using descriptive analysis. Their associations with TBI treatment were tested using Chi-square test and Mann–Whitney U test for categorical and continuous variables, respectively. Kaplan–Meier methods were used to summarize OS and PFS after UCBT. Cox proportional-hazard models were used to estimate the effect of TBI on OS and PFS. Logistic regression was used to compare acute GVHD, chronic GVHD, and systemic infection rate. The rate of TRM was compared using competing risk methods where death due to other causes was a competing risk. Both time to neutrophil engraftment and time to platelet engraftment were analyzed using competing risk methods with death due to any cause as a competing risk. Univariate and multivariable analyses were conducted. The multivariable analysis was adjusted for gender, age at transplant (⩾21 versus <21 years old), diagnosis (AML, ALL, or lymphoma), donor type (unrelated versus related donor), TNC dose, and institution in the model. Missing observations of a variable were excluded from its analysis. All statistical tests were two-sided, and p values <0.05 were considered significant. All statistical analyses were performed using R version 3.5.3.
Results
Patient characteristics
There were 136 patients from four institutions that underwent UCBT from June 2001 to July 2017 (Table 2, Supplemental Table S1). Pretransplant performance scores (Karnofsky/Lansky) were ⩾80%. The median length of follow-up for survivors was 6.1 years. The median age of patients at the time of transplant was 18.5 years (range: 0.6-65.4 years), and 96 patients were under the age of 21 years at the time of transplant. The cohort had 75 (55%) male and 61 (45%) female patients. The study includes 52 (38.2%) ALL/lymphoma patients and 84 (61.8%) AML patients. All patients received chemotherapy and were in bone marrow and/or molecular remission before undergoing UCBT. Sixty-nine (50.7%) patients received MAC regimens, and 67 (49.3%) patients received RIC regimens. There were 62 (90%) pediatric patients in the MAC subgroup with a median age of 11 years (range: 1.5-21.0 years), while there were 34 (51%) pediatric patients in the RIC subgroup with a median age of 13.8 years (range: 1.0-21.0 years). In the subgroup of patients that underwent MAC, 33 received high-dose TBI, and 36 did not. In the RIC subgroup, 43 patients received low-dose TBI, and 24 did not. In the RIC subgroup, a higher TNC dose was found to be used in patients who received TBI. In both subgroups, the patients receiving TBI were more likely to have ALL/lymphoma than those with no TBI (Table 2).
Characteristics of UCBT recipients who did and did not undergo TBI, stratified by conditioning regimen.

AML, acute myeloid leukemia; ATG, antithymocyte globulin; Bu, busulfan; Cy, cyclophosphamide; Flu, fludarabine; HLA, human leukocyte antigen; MAC, myeloablative condition; Mel, melphalan; RIC, reduced-intensity condition; TBI, total body irradiation; Thio, thiotepa; TNC, total nucleated cells; UCBT, umbilical cord blood transplantation.
Transplant outcomes after conditioning regimens
Among patients treated with RIC, those who received TBI had a median OS of 7.9 years versus 0.5 years in patients who did not receive TBI [hazard ratio (HR) 0.51, 95% confidence interval (CI): 0.27-0.97, p = 0.04; Figure 1(a)]. In a multivariable analysis adjusting for gender, age at transplant, diagnosis, donor type, TNC dose, and clinical centers, the difference remained significant [adjusted HR (aHR) 0.25, 95% CI: 0.09-0.66, p = 0.005; Supplemental Table S2]. The risk of TRM was significantly lower in TBI group than that in the non-TBI group (HR 0.43, 95% CI: 0.21-0.88, p = 0.02; Figure 2(a)). The magnitude of effect maintained after the adjustment in the multivariable analysis (aHR 0.44, 95% CI: 0.11-1.69, p = 0.23) although the comparison became nonsignificant (Supplemental Table S2).

Kaplan–Meier estimate of overall survival comparing patients who did and did not undergo TBI as a part of the pre-UCBT conditioning regimen. (a) Reduced-intensity conditioning (RIC) subgroup (p = 0.04). (b) Myeloablative conditioning (MAC) subgroup (p = 0.67). TBI, total body irradiation; UCBT, umbilical cord blood transplantation.

Cumulative incidence of transplant-related mortality of patients who did and did not undergo TBI as a part of pre-umbilical cord blood stem cell transplantation (UCBSCT) conditioning regimen. (a) RIC subgroup (p = 0.02). (b) MAC subgroup (p = 0.88). MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; TBI, total body irradiation.
In the RIC subgroup, PFS was longer in the TBI group than that in the non-TBI group (median 2.4 years versus 0.4 years, HR 0.56, 95% CI: 0.31-1.08; p = 0.09; Figure 3(a)). Multivariable analysis revealed that the risk of disease progression was significantly lower in the TBI group than that in the non-TBI group (aHR 0.26, 95% CI: 0.10-0.66, p = 0.005; Supplemental Table S2). None of the patient characteristics such as gender, age, diagnosis, donor type, and TNC dose had significant association with OS, TRM, or PFS in the multivariable analysis (Supplemental Table S2).
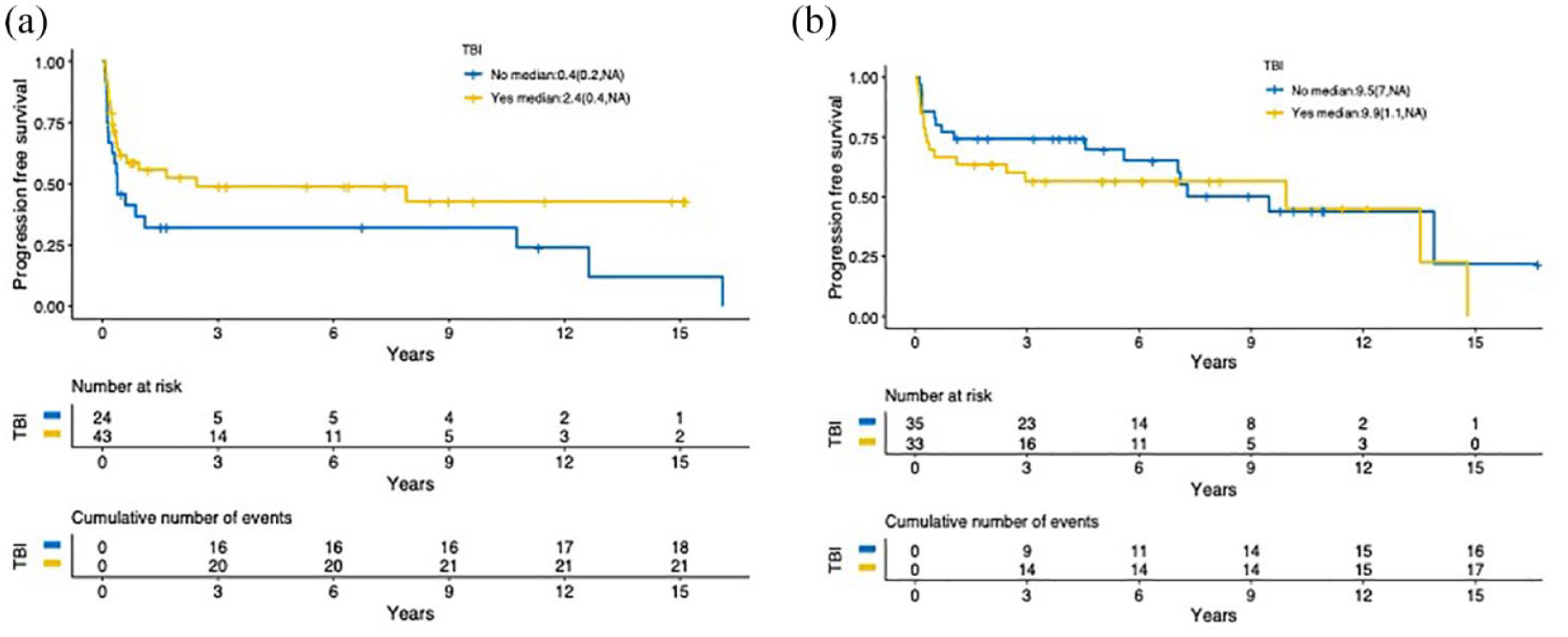
Kaplan-Meier estimate of progression-free survival comparing patients who did and did not undergo TBI as a part of the pre-UCBT conditioning regimen. (a) RIC subgroup (p = 0.09). (b) MAC subgroup (p = 0.41). MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; TBI, total body irradiation; UCBT, umbilical cord blood transplantation.
Among the patients in the MAC subgroup, OS did not show significant difference with and without TBI in the treatment regimen. The median OS was 13.5 years in the patients with TBI and 7.3 years in those with no TBI (HR 1.17, 95% CI: 0.58-2.37, p = 0.67; Figure 1(b)). In a multivariable analysis adjusting for patient baseline characteristics and institution, the effect of TBI remained nonsignificant on OS (aHR 1.68, 95% CI: 0.73-3.88, p = 0.22; Supplemental Table S3). TRM also did not differ significantly in patients with and without TBI in the univariate (HR 1.07, 95% CI: 0.47-2.42, p = 0.88; Figure 2(b)) and multivariable analyses (Supplemental Table S3). PFS did not differ between TBI and non-TBI groups. The median PFS was 9.9 years and 9.5 years with and without TBI (HR 1.33, 95% CI: 0.67-2.56, p = 0.41; Figure 3(b)), respectively.
Engraftment
In the RIC subgroup, time to neutrophil engraftment was shorter among the patients who received TBI than that among those without TBI although it did not reach statistical significance (HR 1.19, 95% CI: 0.71-1.99, p = 0.51; Figure 4(a)), and the difference remained in the multivariable analysis adjusting for gender, age at transplant, diagnosis, donor type, TNC dose, and institution (aHR 1.44, 95% CI: 0.7-2.97, p = 0.33; Supplemental Table S2). Time to platelet engraftment of patients receiving TBI was significantly longer (HR 0.63, 95% CI: 0.37-1.08, p = 0.09; and aHR = 0.45, 95% CI: 0.2-1.0, p = 0.049; Figure 5(a), Supplemental Table S2). Two of 42 patients with TBI had engraftment failure, while full engraftment was noted in all 23 patients with no TBI.

Cumulative incidence of neutrophil engraftment comparing patients who did and did not undergo TBI as a part of the pre-UCBSCT conditioning regimen. (a) RIC subgroup (p = 0.51). (b) MAC subgroup (p = 0.68). MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; TBI, total body irradiation.

Cumulative incidence of platelet engraftment comparing patients who did and did not undergo TBI as a part of the pre-UCBSCT conditioning regimen. (a) RIC subgroup (p = 0.09). (b) MAC subgroup (p = 0.05). MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; TBI, total body irradiation.
In the MAC subgroup, time to neutrophil engraftment did not differ significantly between patients who did and did not receive TBI (HR 0.90, 95% CI: 0.55-1.49, p = 0.68; Figure 4(b)), according to the univariate analysis. The multivariate analysis showed a similar difference (Supplemental Table S3). Patients who received TBI had longer time to platelet engraftment than those without TBI (HR 0.58, 95% CI: 0.34-1, p = 0.049; Figure 5(b)), and the difference was no longer significant after adjusting for patient characteristics (aHR 0.71, 95% 0.37-1.34; Supplemental Table S3). Two of 32 patients with TBI had engraftment failure, while one of 36 patients with no TBI failed to engraft.
Post-transplant complications
There were no statistically significant differences in GVHD or transplant-related infection rates between the TBI and non-TBI groups (Table 3). In the RIC subgroup, the rate of severe acute GVHD was 23.3% and 25% among the patients who did and did not receive TBI, respectively; 53.5% patients in the TBI group and 73.9% patients in the non-TBI group had chronic GVHD. The infection rate was lower among patients with TBI (65.6%) than that in the non-TBI group (89.5%); however, this was not significantly difference (p = 0.096; Table 3). In the MAC subgroup, the rates of acute GVHD and chronic GVHD were numerically higher (although not statistically significant) in the TBI group than those in the non-TBI groups, while the infection rate were similar for both groups. The differences remained nonsignificant after adjusting for patient characteristics and clinical centers in the multivariate analysis.
Development of GVHD and transplant-related infection rates between patient subgroups that did and did not undergo TBI as a part of their pre-UCBT conditioning regimen.

GVHD, graft-versus-host disease; MAC, myeloablative condition; RIC, reduced-intensity condition; TBI, total body irradiation; UCBT, umbilical cord blood transplantation.
ATG use and cause of death
There was a concern that imbalanced use of ATG might impact the incidence of systemic infection-caused death. To explore this issue, we collected data on the causes of death and the ATG use status in four cohorts (Supplemental Table S4). Although the data were incomplete because of the nature of the retrospective study, it showed that the most frequent cause of death was disease progression in RIC cohorts [7 (36.8%) and 7 (36.8%), respectively]. Organ failure in both RIC/TBI and MAC/TBI cohorts are the second frequent cause of death [5 (26.3%) and 5 (33.3%), respectively]. In the RIC/TBI cohort, 12 patients (27.9%) received ATG, and 28 patients (65.1%) did not receive ATG, with infection-related death of 4 (21.1% of death); in the RIC/without TBI cohort, 2 patients (8.3%) did and 15 patients (62.5%) did not receive ATG, with infection-related death of 1 (5.3% of death); in the MAC/TBI cohort, 1 patient (3.0%) received ATG, and 25 patients (75.8%) did not receive ATG, with infection-related death of 2 (13.3% of death); in the MAC without TBI cohort, 17 patients (47.2%) received ATG, with infection related death of 1 (6.2%).
Discussion
We showed that in combination with RIC regimens, the 200 cGy TBI significantly improved OS and PFS compared with RIC without TBI. In contrast, the addition of TBI as a part of the MAC regimen did not significantly impact UCBT outcomes. Although patients who received TBI did not show significant difference in neutrophil or platelets engraftment time in the MAC setting, 200 cGy TBI use in combination with RIC was associated with delayed platelet engraftment.
The OS data in our study are consistent with a retrospective study of a Japanese transplant registry database. 36 In that study, Nakasone et al. found that there was no difference in rates of GVHD and OS in patients who received TBI and non-TBI conditioning regimens before UCBT. However, in that study, there was no comparison between patients receiving TBI versus no TBI in an RIC subgroup. Furthermore, the TBI doses in that study were much higher than those in our RIC subgroup (less than 800 cGy versus 200 cGy). We also performed an overall analysis combining the two cohorts (MAC and RIC) and, similar to Nakasone et al., found no significant difference between the TBI and non-TBI groups in respect to OS (HR 0.82, 95% CI: 0.51-1.32), TRM (HR 0.69, 95% CI: 0.39-1.20), and PFS (HR 0.92, 95% CI: 0.58-1.46).
In our study, time to neutrophil engraftment was shorter in patients receiving TBI in the RIC subgroup although the difference was not statistically significant. Interestingly, Nakasone et al. 36 also demonstrated that the use of TBI before UCBT was significantly associated with accelerated neutrophil engraftment in both the MAC and RIC cohorts, concluding that TBI may be more critical for engraftment than conditioning intensity. Similarly, in a 2011 study that only included children with myelodysplastic syndrome who underwent UCBT, neutrophil recovery was noted to be faster in patients that underwent TBI-containing conditioning regimens. 37 Furthermore, a 2014 UCBT study found that a regimen containing low-dose TBI, Bu, clofarabine, and Flu exhibited superior OS and NRM compared with non-TBI-containing regimens (Mel/Flu/Thio, or Bu/Flu). 29 The same study also demonstrated superior neutrophil engraftment in the patient cohort that received TBI. The authors of that study attributed these results to the early elimination of the host T-cell population accomplished by TBI. The lack of statistical significance in our study could be due to the small sample size and patient heterogeneity. A large study is warranted to further confirm the effect of low-dose TBI on neutrophil engraftment.
Despite limited data specific to UCBT, comparisons of conditioning regimens ± TBI have been performed in other types of HSCT. In adults with mature T-cell and natural killer cell lymphoma, one retrospective analysis also concluded that OS, PFS, and toxicities of patients receiving TBI-containing conditioning regimens were comparable to those of patients who underwent conditioning that did not include TBI. 38 In adults with ALL, another retrospective analysis comparing Bu to TBI-containing conditioning regimens determined similar disease-free survival and OS between the two groups following HSCT although there were higher rates of TRM in the TBI cohort (19% versus 25%, p = 0.04). 39 Conversely, children with AML experienced superior 5-year OS and PFS following bone marrow transplant after employing a conditioning regimen of Bu/Cy compared with TBI/Cy. 40 However, a study of the clinical significance of low-dose TBI in patients with hematologic malignancies undergoing HSCT found that low-dose TBI improved OS in patients with a high-risk disease and multi-HLA mismatch in comparing to Flu/Bu conditioning. 41 It seems that TBI dosage could play a crucial role in post-transplant outcomes. On the other side, it is well accepted that low-dose TBI is less toxic. We postulate that low-dose TBI suppresses recipient T-cell activity and opens up the bone marrow matrix space for UCB stem cell settling, renewing, differentiating, and proliferating; meanwhile, it does not cause significant harm and damage to the bone marrow microenvironment.
We also evaluated the impact of ATG use in the incidence of systemic infection via comparing various combinations of condition intensity (MAC versus RIC), TBI use, and ATG use. Interestingly, ATG use appeared to have no obvious impact on infection-caused death. It may be due to the relative naivety of lymphocytes in UCB in response to ATG treatment.
Our study has several limitations, in part related to the nature of retrospective data analyses. Selective use of TBI was based on several clinical factors that could introduce selection bias. The multivariable analysis may not be able to account for heterogeneity of the treatment entirely. The size of the study limited our ability to examine the differential effect of TBI with diagnosis. Cause of death and infection were not considered. In addition, the clinical significance of TBI in UCBT might further depend on patient-/donor-specific factors that were not accounted for in this study as previously stated. 42 In addition, differences could exist in dosing of chemotherapy and delivery among participating institutes. The recent omidubicel phase III trial data further demonstrated that the UCB hematopoietic progenitor expansion status plays a role in UCBT outcomes. 43
Conclusion
This study suggests that the combination of low-dose TBI and RIC regimens may improve OS and PFS for patients undergoing UCBT. TBI has no significant impact on the incidence or severity of acute/chronic GVHD and systemic infection. These findings support further investigation of low-dose TBI (200 cGy) containing RIC regimens in patients undergoing UCBT.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231170708 – Supplemental material for The impacts of total body irradiation on umbilical cord blood hematopoietic stem cell transplantation
Supplemental material, sj-docx-1-tah-10.1177_20406207231170708 for The impacts of total body irradiation on umbilical cord blood hematopoietic stem cell transplantation by Hao Wang, Kristin N. Berger, Elizabeth L. Miller, Wei Fu, Larisa Broglie, Frederick D. Goldman, Heiko Konig, Su Jin Lim, Arthur S. Berg, Julie-An Talano, Melanie A. Comito, Sherif S. Farag and Jeffrey J. Pu in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
