Abstract
Background:
Chimeric antigen receptor T-cell (CAR-T) cell therapies have been claimed to be curative in responsive patients. Nonetheless, response rates can vary according to different characteristics, and these therapies are associated with important adverse events such as cytokine release syndrome, neurologic adverse events, and B-cell aplasia.
Objectives:
This living systematic review aims to provide a timely, rigorous, and continuously updated synthesis of the evidence available on the role of CAR-T therapy for the treatment of patients with hematologic malignancies.
Design:
A systematic review with meta-analysis of randomized controlled trials (RCTs) and comparative non-randomized studies of interventions (NRSI), evaluating the effect of CAR-T therapy versus other active treatments, hematopoietic stem cell transplantation, standard of care (SoC) or any other intervention, was performed in patients with hematologic malignancies. The primary outcome is overall survival (OS). Certainty of the evidence was determined using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach.
Data sources and Methods:
Searches were performed in the Epistemonikos database, which collates information from multiple sources to identify systematic reviews and their included primary studies, including Cochrane Database of Systematic Reviews, MEDLINE, EMBASE, CINAHL, PsycINFO, LILACS, DARE, HTA Database, Campbell database, JBI Database of Systematic Reviews and Implementation Reports, EPPI-Centre Evidence Library. A manual search was also carried out. We included the evidence published up to 1 July 2022.
Results:
We included the evidence published up to 1 July 2022. We considered 139 RCTs and 1725 NRSI as potentially eligible. Two RCTs (N = 681) comparing CAR-T therapy with SoC in patients with recurrent/relapsed (R/R) B-cell lymphoma were included. RCTs did not show statistical differences in OS, serious adverse events, or total adverse events with grade ⩾ 3. Higher complete response with substantial heterogeneity [risk ratio = 1.59; 95% confidence interval (CI) = (1.30–1.93); I2 = 89%; 2 studies; 681 participants; very low certainty evidence] and higher progression-free survival [hazard ratio for progression or death = 0.49; 95% CI = (0.37–0.65); 1 study; 359 participants; moderate certainty evidence] were reported with CAR-T therapies. Nine NRSI (N = 540) in patients with T or B-cell acute lymphoblastic leukemia or R/R B-cell lymphoma were also included, providing secondary data. In general, the GRADE certainty of the evidence for main outcomes was mostly low or very low.
Conclusion:
So far, assuming important limitations in the level of certainty due to scarce and heterogenous comparative studies, CAR-T therapies have shown some benefit in terms of progression-free survival, but no overall survival, in patients with R/R B-cell lymphoma. Despite one-arm trials have already facilitated approval of CAR-T cell treatments, additional evidence from large comparative studies is still needed to better characterize the benefit-harm ratio of the use of CAR-T in a variety of patient populations with hematological malignancies.
Registration:
PROSPERO/OSF Preregistration:
10.17605/OSF.IO/V6HDX
Keywords
Background
Chimeric antigen receptors (CARs) are engineered receptors that insert a defined specificity onto an immune effector cell, typically a T cell. 1 This cancer immunotherapy enables enhancement of the immunological response against malignant cells. Although the development of CAR T-cell (CAR-T) therapy started >20 years ago, 2 the first steps in their transfer to clinical practice are now taking place, following the recent authorization of different CAR-T therapies by the U.S. Food and Drug Administration and the European Medicines Agency.
The earliest and most extensive research with CAR-T cell therapy has been carried out in hematologic malignancies, where it has pointed to high response rates in patients who generally have very poor prognoses such as relapsed/refractory acute lymphoblastic leukemia, diffuse large B-cell lymphoma, or multiple myeloma.3–5 Few therapeutic options are available in this setting, and CAR-T therapy is showing promising results. B-cell maturation antigen and CD19-targeting CAR-T cell therapies are more developed than others and have shown the best results so far. 6 At this time, CAR-T cells directed to other targets for treating solid tumors and infectious or autoimmune diseases are emerging.7,8
Clinical studies have suggested that CAR-T cell therapies can be even curative in responsive patients. However, response rates can vary in different pathologies or depending on the characteristics of the patient or the disease, and also recurrences and relapses may occur. Moreover, these therapies are associated with important adverse events such as cytokine release syndrome, neurologic adverse events and B-cell aplasia, which may lead to serious consequences. 1 To date, clinical trials have been mainly performed in terms of a single-arm design, whose encouraging results have allowed the approval of CAR-T therapies and their progressive introduction into clinical practice. However, it is still unclear what type of patients may benefit from CAR-T therapy with respect to other treatment options.
In the same line, the vast majority of systematic reviews published until 2022 have focused on synthesizing overall response rates of CAR-T cell therapy, without providing results compared with a control group.9–12 A recently published systematic review focused on randomized controlled trials (RCTs) analyzing the efficacy and safety of CAR-T therapies versus high-dose salvage chemotherapy followed by autologous stem cell transplantation as second-line treatment of relapsed or refractory large B-cell lymphoma. 13 However, this review shows several limitations, such as the lack of diverse comparators and hematologic diseases, and methodological concerns with the pooled analysis strategy. A second systematic review showing remarkable differences with our living project was also identified, providing mixed results in adults and children, an updated search 6 months shorter than our study, less detail on pre-specified exploratory outcomes and no data available on complementary observational evidence. 14 In addition, numerous phase III RCTs are already underway that will shed light on these questions in the near future, and new CAR-T cell therapies will emerge. Thus, taking into account the wide variability of the available evidence, the shortcomings of the already published systematic reviews, and the potential availability of new studies, we proposed to carry out a living systematic review analyzing the evidence from comparative studies assessing the efficacy and safety of CAR-T therapies versus other treatment options in patients with hematologic diseases. Here we present the baseline report of this living systematic review.
Method
Protocol and registration
This systematic review is being developed as part of the Living Evidence to Inform Health Decisions project; 15 and complies with the ‘Preferred Reporting Items for Systematic reviews and Meta-Analyses’ (PRISMA) guidelines. 16 Considering the particular features of the present evidence on efficacy and safety of CAR-T therapy in patients with hematological cancers, the broad variability of the available systematic reviews, and bearing in mind that the evidence on this therapy is rapidly increasing, we have chosen a living systematic review as the preferred design to make it possible a sound and up to date synthesis of the most relevant literature. The protocol of this systematic review has been published somewhere else and currently is awaiting peer review. 17
Methods for identification of studies
The Epistemonikos-L.OVE platform 18 was used for evidence identification, screening, and selection. This platform has been validated as a repository for COVID-19, and proved to be a highly comprehensive source of evidence.19,20 The results of the literature searches were automatically incorporated into the L·OVE platform (automated retrieval) and organized in the corresponding L.OVE of Chimeric antigen receptor T cell therapy for hematological malignancies (https://cutt.ly/4XIYRMW).
The team maintaining the Epistemonikos-L·OVE platform devised the literature search. Searches were performed in the Epistemonikos database (https://www.epistemonikos.org), the main search source for the L.OVE platform that collates information from multiple sources to identify systematic reviews and their included primary studies, including Cochrane Database of Systematic Reviews, MEDLINE, EMBASE, CINAHL, PsycINFO, LILACS, DARE, HTA Database, Campbell database, JBI Database of Systematic Reviews and Implementation Reports, EPPI-Center Evidence Library. 21 Full search strategy is presented in Supplemental Appendix 1.
Additional searches aimed at identifying new RCTs and non-randomized studies of interventions (NRSI) were run in the MEDLINE database using validated high sensitive filters to identify these type of studies. We also carried out a manual search for reviewing the reference list of included studies, guidelines, narrative reviews and any other document of interest. We included the evidence published up to 1 July 2022. No publication status or language restriction was applied to the searches.
Types of studies
RCTs and NRSI (quasi-experimental studies, cohort studies, case-control studies) were considered for inclusion. Studies had to provide data for at least one primary or secondary outcome variable to be eligible.
Types of participants
Studies including adults (⩾18 years) or pediatric participants diagnosed with hematologic disease, such as multiple myeloma, leukemia, and lymphoma of any type were included. Untreated patients as well as patients previously treated were included, irrespective of the type of treatment or treatment line.
Type of interventions
Any CAR-T therapy type was considered regardless of the T-cell origin (allogenic or autologous), target antigen, or costimulatory domain. We did not restrict our criteria to any dose, duration, timing, or route of administration.
The comparator consisted of chemotherapy or any other active pharmacologic treatment, hematopoietic stem cell transplantation (HSCT), standard of care (SoC) or any other intervention.
Studies assessing CAR-T plus other therapeutic measures were eligible if the co-interventions were identical in both arms.
Type of outcomes
The primary outcome was overall survival (OS). Key secondary outcomes were complete response/remission (CR) rate, relapse from CR, progression-free survival (PFS), graft-versus-host disease (GvHD), total adverse events (TAE) (grade ⩾ 3), and serious adverse events (SAE). Other secondary outcomes were overall response rate (ORR), partial response/remission (PR) rate, time from CAR-T infusion to transplantation (TtT), incidence of cytokine-release syndrome (CRS; Grade 3 or higher), neurotoxicity (Grade 3 or higher), and quality of life.
We presented primary and key secondary outcomes in a GRADE Evidence Profile format. 22
Selection of studies
The results of the literature search from all databases were automatically incorporated into the L·OVE platform, and de-duplicated by an algorithm comparing unique identifiers (database ID, DOI, trial registry ID), and citation details (i.e. author names, journal, year of publication, volume, number, pages, article title and abstract).
In the first step, two researchers independently screened the titles and abstracts of the identified references yielded by the searches in L.OVE. Following this step, we screened all RCTs and NRS yielded by searches against the inclusion criteria. We obtained the full reports of all potentially eligible titles and of those requiring further analysis to decide their inclusion. In the second stage, two independent reviewers screened the full texts to decide on their inclusion. We recorded the study selection process in a PRISMA flow diagram and recorded the reasons for exclusion.
Extraction and management of data
Two reviewers independently extracted data from each included study using standardized forms. We resolved disagreements by discussion. We contacted authors of the primary studies in case of missing information in the retrieved studies.
Risk of bias assessment
The risk of bias for each RCT was assessed using the Cochrane Risk of Bias Tool, which includes the domains: selection bias, performance bias, detection bias, attrition bias, reporting bias, and other bias. 23
In the case of NRSI, the ROBINS-I tool was used. 24 This tool evaluates the domains: bias due to confounding, selection of participants, classification of interventions, deviation from intended interventions, missing data, measurement of outcomes, and bias in selection of the reported result. The overall risk of bias was judged as ‘low’, ‘moderate’, ‘serious’, ‘critical’, and ‘no information’.
Measures of treatment effect
For binary outcomes, results were presented as the proportion of patients who suffered the corresponding analyzed event. We used hazard ratio (HR), risk ratio (RR), and odds ratio (OR) for summarizing the results as appropriate. Absolute risk reduction (ARR) or absolute risk increase (ARI), and number needed to treat for an additional beneficial outcome (NNTB) or number needed to treat for an additional harmful outcome (NNTH) were also calculated when applicable, with 95% confidence interval (CI).
For quantitative data, mean difference or standardized mean difference were estimated as appropriate. We also determined the 95% CI.
Data synthesis
Results from the different studies were analyzed separately according to the specific study design. Evidence from RCTs was prioritized. The information from NRSI was considered as complementary when there was no RCT for main outcomes or when the evidence from RCTs was rated as ‘low’ or ‘very low’ certainty according to the GRADE approach. 25 A fixed-effects model was used to combine results for each outcome across trials when applicable. Data were analyzed following an intention-to-treat approach.
To test for heterogeneity of treatment effect among trials we used I² statistic. We considered I² > 60% as indicative of substantial heterogeneity, whose possible reasons were planned to be explored by carrying out sensitivity and subgroup analyses.
When only one study provided data for an outcome or when data obtained from different studies showed substantial heterogeneity (I² > 60%), we provided a narrative description of the results (aggregated estimates are presented only for verification purposes).
We used Review Manager Software (RevMan® version 5.4) for carrying out the meta-analyses. 26
Subgroup and sensitivity analysis
We planned to perform subgroup analysis according to type of CAR-T therapy, type of hematologic disease, type of comparator, age group (⩾18 years and <18 years), treatment line, tumor burden, or cancer stage.
We planned to perform sensitivity analyses excluding high risk of bias studies and excluding industry-sponsored studies.
Assessment of certainty of evidence
Two reviewers independently assessed the certainty of the evidence for each outcome following the GRADE approach, which considers five aspects: risk of bias of included studies, directness of the evidence, consistency among trials’ results, precision of effect estimates, and risk of publication bias. 25 Overall certainty of evidence was downgraded starting from a high quality in the case of RCTs, and evidence was upgraded starting from a low quality in the case of NRSI, when appropriate. 27 Assessment of certainty of evidence was performed when a meta-analysis of results was possible.
Discrepancies were resolved by discussion and by involvement of a third author.
Living evidence synthesis and future updates
We will keep a living search in the L·OVE platform, in which an artificial intelligence algorithm will provide instant notification of articles with a high likelihood to be eligible. All potentially eligible studies will undergo the selection process described above and in the review protocol. 17 The living process for this question will end when the certainty of the evidence on the updated estimates for the desirable and undesirable effects becomes high or after 12 months of surveillance whatever is reached first. An updated report at this stage will be published in this or any other peer-reviewed journal when findings from the new evidence differ substantially from the baseline report.
Results
Results of the search
Literature searches retrieved a total of 1864 articles to the L·OVE platform. We considered 139 RCTs and 1725 NRSI as potentially eligible, out of them 4 and 43 studies respectively were reviewed in full text. After a full-text review, we excluded 35 NRSI that did not fulfill our eligibility criteria. One RCT was reclassified as NRSI and another one was classified as an ongoing RCT and was not still included because of its preliminary status only providing immature data for its primary outcome. 28 Therefore, two RCTs29,30 and nine NRSI (two quasi-experimental studies31,32 and seven cohort studies)33–39 assessing the use of CAR-T in 1221 participants with B-cell lymphoma or T or B-cell acute lymphoblastic leukemia were included in the review. A detailed list with reasons for exclusion is presented in Supplemental Appendix 2. The study selection process is summarized in Figure 1.

PRISMA flowchart.
Description of the included studies
Patients included in the studies have either R/R B-cell lymphoma (two RCTs and two NRSI, 830 participants) or T/B-cell ALL (seven NRSI, 391 participants) and received a CAR-T therapy or one of the following comparators: HSCT, chemotherapy, donor leukocyte infusion (DLI), polatuzumab or SoC. Table 1 presents the characteristics of the included studies and baseline characteristics of the participants.
Characteristics of the included studies and baseline characteristics of the participants.

ASCT, autologous stem cell transplantation; BKCSCPP, Beijing Key Clinical Specialty Construction Project-Pediatrics; C, cohort study; CAR, chimeric antigen receptors; CAR-T, chimeric antigen receptor T-cell; CD, cluster of differentiation; CH, chemotherapy; CIFMS, Chinese Innovation Fund for Medical Science; CoI, conflicts of Interest; CR, complete remission; CRS, cytokine-release syndrome; DLI, donor lymphocyte infusion; GvHD, graft-versus-host disease; HSCT, hematopoietic stem cell transplantation; MC, multicenter; NA, not applicable; NR, not reported; OS, overall survival; ORR, overall response rate; PFS, progression-free survival; PR, partial remission; QE, quasi-experimental study; R/R B-ALL, relapsed/refractory B-cell acute lymphoblastic leukemia; RCT, randomized controlled trial; SAE, serious adverse events; SD, standard deviation; SoC, standard of care; UC, unicenter; TtT, time from CAR-T infusion to transplantation.
Risk of bias of included studies
Both RCTs29,30 were rated as serious overall risk of bias. The main concerns arose from the performance bias domain (open-label design, particularly relevant in case of subjective outcomes) and ‘other bias’ domain (crossover allowed). Selection and reporting bias were estimated as low risk and attrition bias as unclear. Detection bias was rated as low risk in ZUMA-7 29 and unclear in BELINDA. 30
The two quasi-experimental studies were both globally rated as ‘no information’, due to the lack of data on two domains: confounding31,32 and selection of participants. 31 Regarding most of the estimated outcomes (OS, ORR, CR, PR, relapse from CR, PFS, CRS, TtT, GvHD, neurotoxicity, SAE), the overall risk of bias in the cohort studies was judged as moderate33,34,37–39 or serious,35,36 mainly due to concerns related to the first domain (confounding). However, when other outcomes with a higher subjective profile were considered (e.g. TAE with ⩾3 grade), risk of bias was rated as serious, 31 taking into account the lack of blinding of the outcome assessment.
Supplemental Appendix 3 presents the detailed assessment by subdomain for all included studies.
Effects of interventions
Primary outcome
Overall survival
Meta-analysis of RCTs29,30 shows no significant difference in OS between CAR-T and SoC in R/R B-cell lymphoma patients [681 participants; HR 0.81, 95%CI = (0.63–1.05); I2 = 18%] (Figure 2). The certainty of evidence for this outcome is low due to crossover allowed and imprecision on estimates of the effect (Table 2).
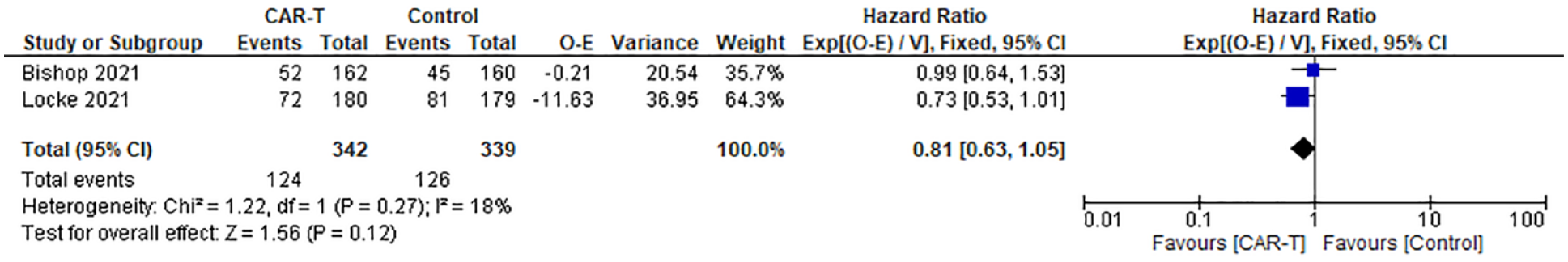
Overall survival comparing CAR-T with SoC therapy in RCTs including patients with R/R B-cell lymphoma.
GRADE evidence profile for randomized controlled trials.

CAR-T, chimeric antigen receptor T-cell; CI, confidence interval; GRADE, Grading of Recommendations, Assessment, Development and Evaluation; HR, hazard ratio for progression or death; NR, not reported; RCTs, randomized controlled trials; RR, risk ratio.
Two RCTs allowed crossover from standard care to CAR-T after insufficient response.
95%CI is consistent with the possibility for relevant benefit and harm exceeding a minimal important difference.
Unexplained and substantial heterogeneity, with point estimates widely different and confidence intervals not overlapping significantly.
RCTs allowed crossover from standard care to CAR-T after insufficient response and outcomes highly subjective, especially affected by the open-label design.
The two quasi-experimental studies assessing OS31,32 showed longer OS with CAR-T versus DLI therapy in R/R B-ALL patients (Wang et al. 31 DLI median 3.7 months (range 0–65); CAR-T median 12 months (range 3–29); p = 0.049. Hua et al. 32 DLI median 5.5 months (range 1–25); CAR-T median 9.5 months (range 3–25); p = 0.030), although it was not possible to meta-analyze them.
Finally, five cohort studies comparing CAR-T in T or B-ALL and R/R B-cell lymphoma with different control groups have reported OS results33,36–39 When examined individually, no statistically significant differences were found in four studies,33,36,38,39 while one study showed longer OS in the CAR-T group [HR 0.28, 95% CI (0.13–0.64)] 36 (Supplemental Appendix 4, Table S2), although it was not possible to perform a meta-analysis.
Key secondary outcomes
Complete CR rate
The meta-analysis of two RCTs29,30 showed a higher CR with CAR-T compared with SoC therapy in R/R B-cell lymphoma patients [60 participants; RR 1.59, 95% CI (1.30–1.93); I2 = 89%]. However, substantial heterogeneity was detected, which greatly reduces confidence in the estimate. The NNTB leading to an additional CR with CAR-T therapies was 6 (95% CI 4–12). The certainty of evidence for this outcome is very low, downgraded because the crossover was allowed and inconsistency (Table 2).
Evidence from two quasi-experimental studies31,32 showed a higher CR with CAR-T compared with DLI therapy in R/R B-ALL patients [60 participants; OR 4.12, 95% CI (1.04–16.37); I2 = 0%] (Figure 3). The NNTB leading to an additional CR with CAR-T therapies was 4 (95% CI 3–100).

Complete response/remission rate comparing CAR-T with DLI therapy in patients with R/R B-ALL.
The certainty of evidence for this outcome is low due to lack of information on adjustment for confounding variables and imprecision (Supplemental Appendix 4, Table S1).
Regarding the three cohort studies that reported CR results,37–39 no differences between CAR-T and control group were found in one study, 37 while the remaining two result in favor of the CAR-T group [CAR-T 91%; chemotherapy 71%, p = 0.036; 37 CAR-T + ASCT: 71%; ASCT: 33%, p = 0.003 38 ] (Supplemental Appendix 4, Table S2).
Relapse from CR
Evidence on relapse from CR from three cohort studies33,36,38 did not show differences between CAR-T and control group, although with high heterogeneity [244 participants; OR 1.24, 95% CI (0.69–2.24); I2= 90%]. The certainty of evidence for this outcome is very low due to lack of adjustment for important factors, serious inconsistency and serious imprecision (Supplemental Appendix 4, Table S2).
Progression-free survival
The only RCT reporting PFS 29 showed a higher rate of PFS with CAR-T compared with SoC in R/R B-cell lymphoma patients [HR for progression or death 0.49, 95% CI (0.37–0.65)]. The certainty of evidence for this outcome is moderate for the RCT due to crossover allowed (Table 2).
When analyzed individually, two cohort studies37,39 showed statistically significant PFS results in favor of the CAR-T [HR 0.49, 95% CI (0.25–0.98), p = 0.045; 36 CAR-T+ ASCT: 80% (95% CI; 60–98); ASCT: 44% (95% CI; 25–64), p = 0.036 38 ], while the remaining did not show differences. 34 The average certainty of evidence for this outcome could not be assessed (Supplemental Appendix 4, Table S2).
Serious adverse events
The meta-analysis of two RCTs29,30 did not show statistically differences in SAE with CAR-T compared with SoC in R/R B-cell lymphoma patients [660 participants; RR 1.00, 95% CI (0.85–1.17); I2= 16%] (Figure 4). The certainty of evidence for this outcome is low due to crossover allowed and imprecision on estimates of the effects (Table 2).

Serious adverse events in RCTs comparing CAR-T with SoC therapy in patients with R/R B-cell lymphoma.
Graft-versus-host disease
Two quasi-experimental studies reported the effect of interventions, after HSCT, in incidence of acute or new GvHD.31,32 The meta-analysis showed a lower incidence of GvHD with CAR-T compared with DLI in B-ALL patients [60 participants; OR 0.11, 95% CI (0.02–0.69); I2= 0%]. The NNTB to avoid an additional case of GvHD with CAR-T therapies was 4 (95% CI 3–13). The certainty of evidence for this outcome is low due to imprecision in estimates of the effect and lack of information on adjustment for confounding variables (Supplemental Appendix 4, Table S1).
In contrast, one cohort study 33 comparing CAR-T + HSCT versus chemotherapy + HSCT in B-ALL patients reported a higher incidence of acute GvHD (grade II or higher) in the CAR-T group [HR 2.36 (95% CI 1.18–4.75); p = 0.016]. The NNTH leading to an additional case of GvHD with CAR-T therapies was 5 (95% CI 3–26). The certainty of evidence for this outcome is low due to lack of adjustment for important factors and imprecision (Supplemental Appendix 4, Table S2).
TAE (grade ⩾ 3)
The meta-analysis of two RCTs29,30 did not show significant differences in TAE with CAR-T compared with SoC in R/R B-cell lymphoma patients [RR 1.01, 95% CI (0.95–1.07); I2= 86%]. However, a substantial heterogeneity was detected, which greatly reduces confidence in the estimate. The certainty of evidence for this outcome is very low due to crossover allowed, inconsistency and imprecision (Table 2).
The summary of findings on the key secondary outcomes in quasi-experimental and cohort studies is presented in Supplemental Appendix 4.
Other secondary outcomes
Overall Response rate
According to evidence from two RCTs,29,30 CAR-T therapy was associated to a higher ORR compared with SoC in R/R B-cell lymphoma patients [681 participants; RR 1.41, 95% CI (1.23–1.62); I2= 88%]. However, a substantial heterogeneity was detected, which greatly reduces confidence in the estimate. The NNTB leading to an additional ORR with CAR-T therapies was 6 (95% CI 4–10). The certainty of evidence for this outcome is very low due to crossover allowed and substantial heterogeneity.
No differences in ORR were found between CAR-T and control group in two cohort studies.37,39
Partial Response/Remission (PR)
According to the evidence from two RCTs,29,30 there were no statistically differences in PR with CAR-T therapy compared with SoC therapy in R/R B-cell lymphoma patients [681 participants; RR 1.10, 95% CI (0.79–1.52); I2= 0%]. (Figure 5). The certainty of evidence for this outcome is low due to crossover allowed and imprecision in estimates of the effect.

Partial response in RCTs comparing CAR-T versus SoC therapy in people with R/R B-cell lymphoma.
The only quasi-experimental study that reported results on PR 32 found no statistical differences when comparing CAR-T versus DLI in R/R B-ALL patients [OR 0.23, 95% CI (0.02–2.38)].
The only cohort study that reported PR results showed statistically significant results in favor of the control group [CAR-T + ASCT 19% (95% CI 5–42) vs. ASCT 56% (95% CI 41–71); p = 0.004]. 39
Time from CAR-T infusion to transplantation
One cohort study reported that median time from CAR-T infusion to HSCT was 67 days. 38
Incidence of CRS (grade ⩾ 3)
The meta-analysis of 2 RCTs29,30 showed a higher rate of CRS with CAR-T compared with SoC in R/R B-cell lymphoma patients [660 participants; RR 19.76, 95% CI (2.67–146.39); I2= 0%]. The certainty of evidence for this outcome is low due to crossover allowed and imprecision in estimates of the effect.
In addition, the meta-analysis of the two quasi-experimental studies reporting data on CRS31,32 showed a lower incidence of CRS with DLI compared with CAR-T in R/R B-ALL patients [60 participants; OR 0.08, 95% CI (0.01–0.84); I2 = 0%], assuming zero events in the comparator arm if not specifically reported. The certainty of evidence for this outcome is low due to imprecision in estimates of the effect and lack of information on adjustment for confounding variables.
Finally, five cohort studies also reported incidence of CRS, ranging from 5% to 15% in the CAR-T groups33,35,37–39 None of the above studies but Wang et al. 39 provided explicit information on the incidence of CRS in their comparator groups, being assumed as zero events for these arms. We did not rate the certainty of evidence for this outcome due to insufficient information for the assessment.
Neurotoxicity (grade ⩾ 3)
The meta-analysis of two RCTs29,30 showed a higher rate of neurotoxicity with CAR-T compared with SoC in R/R B-cell lymphoma patients [660 participants; RR 26.02, 95% CI (5.14–131.67); I2= 0%]. The NNTH leading to an additional case of neurotoxicity with CAR-T therapies was 14 (95% CI 3–77). The certainty of evidence for this outcome is moderate due to crossover allowed.
Incidence of neurotoxicity in three cohort studies ranged from 0% to 15% in the CAR-T groups.33,37,39 Only Avivi et al. 37 provided information on the incidence of 1 case of peripheral neuropathy (2%) in the comparator group.
Quality of life
The included studies did not assess this outcome.
There was not enough information to perform pre-planned subgroup analyses. All the outcomes remained unchanged after carrying out sensitivity analyses excluding studies with serious risk of bias34–36 (Supplemental Appendix 3). Both RCTs and none of the quasi-experimental/cohort studies were industry-sponsored. Thus, it was not possible to explore potential differences on this aspect within each particular design.
Discussion
To date, this living systematic review has identified two RCTs evaluating the use of CAR-T compared with SoC for hematologic malignancies, namely R/R B-cell lymphoma.29,30 Beyond that, 9 NRSI31–39 in T or B-ALL and R/R B-lymphoma with different comparators (chemotherapy, HSCT, DLI, polatuzumab) have been retrieved on this topic. One ongoing RCT with interim results 28 was identified and its data will only be considered for inclusion when the trial is completed.
No comparative studies analyzing CAR-T therapy in participants with other hematological malignancies such as multiple myeloma were identified.
CAR-T therapy showed benefits in terms of a longer CR and PFS. On the other hand, no benefit in OS was observed, nor in the rest of secondary outcomes evaluated. However, the follow-up period was probably rather short to assess results in mortality. Regarding the safety outcomes, no differences between CAR-T and alternative therapies have been identified concerning SAE and TAE (grade ⩾ 3). As expected, serious CRS was strongly linked to CAR-T use regardless of the comparator, whereas GvHD showed conflicting results between quasi-experimental and cohort studies.
The results of CAR-T therapy obtained so far may be considered as modest. In the case of BELINDA, 30 authors claimed that treatment groups were not balanced at baseline. In the CAR-T group, there was a higher proportion of patients with high-grade lymphomas compared with the SoC group (24.1% vs. 16.9%, respectively). In addition, they also pointed out that the CAR-T group had a higher percentage of patients with an International Prognostic Index score of ⩾ 2, indicating a worse prognosis (65.4% vs. 57.5%, respectively). According to the authors, this could have biased results against the CAR-T cell group. However, in ZUMA-7, 29 both groups were balanced at baseline and results were similar.
Both RCTs included patients with aggressive B-cell non-Hodgkin’s lymphomas not responding to or progressing within 12 months after first-line therapy. It would be important to learn about the potential benefits of CAR-T therapy in less aggressive lymphomas. Likewise, we have other challenges ahead, like identifying patients who may have a better risk-benefit profile, improving CAR-T cells, testing the effects of CAR-T therapy in other tumors, and defining the risk-benefit balance in patients at earlier stages of disease. Also, the status of allogeneic or autologous CAR-T might also be relevant in terms of patient severity and adverse event profile marker. Remarkably, RCTs and NRSI in this review differed in this parameter, using autologous and allogeneic therapies respectively.
Evaluating the certainty of evidence
As for meta-analyses with acceptable heterogeneity, all assessed outcomes presented imprecision concerns and were evaluated with an insufficient sample size. Also all estimates in the cohort studies were judged as substantially imprecise and led to downgrade the certainty of this evidence.
Despite the apparently identical design, the two included RCTs29,30 showed divergent results and substantial heterogeneity (I2 > 80%) in key secondary outcomes such as CR or TAE (grade ⩾ 3). Apart from differences in CAR-T cell products, costimulatory domains or gene-transfer method, BELINDA 30 allowed participants with bridging chemotherapy and impending organ-compromising disease, which likely would lead to a worse prognosis and less benefit from the experimental intervention. 40 All these aspects, among others, might explain the observed inconsistency.
Evidence on CR coming from quasi-experimental studies showed a higher certainty (low) than that from RCTs (very low). Nonetheless, the direction of effect was similar with both designs, showing benefit from using CAR-T to some degree.
Some valuable data, too many uncertainties
The severity of hematologic malignancies, added to the lack of appropriate treatment strategies when a recurrent stage is reached, has promoted the search of new interventions from different perspectives, CAR-T therapies among them. 1 While promising, those strategies are currently short of interventional evidence and, as shown in this systematic review, are also in need of more extensive comparative data. Consequently, at this point, there is still wide uncertainty of the actual balance between efficacy and harms for this alternative.
The results of the included studies seem to confirm the known CAR-T toxicity profile in terms of an uncontrolled CRS and neurotoxicity. Regarding other key safety outcomes such as SAEs and grade ⩾ 3 TAE, no particular risk or benefit has been reported with CAR-T therapies. Quality of life data was not provided by the identified studies. Also, no benefit has been proved favoring CAR-T versus other alternatives related to PR or relapse from CR, while the evidence was too scarce to make a definite statement.
Ongoing evidence is strongly needed
A still-increasing amount of evidence assessing these strategies is underway, 41 so the question is yet to be answered. Beyond the 11 included studies,29–39 a growing number of ongoing interventional and observational studies with comparative designs can be located at platforms such as ClinicalTrials.gov. One systematic review 13 carried out by authors with declared conflicts of interest and including three RCTs involving 865 participants28–30 was identified after an in-depth screening. The review addressed only two hematologic conditions (acute B-cell lymphoblastic leukemia and B-cell lymphoma), which obviously reduces the generalizability of the conclusions to other hematologic malignancies. The performed meta-analysis showed that CAR-T therapy was superior to SoC in OS and event-free survival. However, the beneficial effect in OS has reached thanks to the inclusion of the ongoing TRANSFORM trial, 28 a study designed for a 3-year follow-up whose published results, covering a median follow-up of only 6.2 months, are still preliminary and clearly immature. 42 Another systematic review 14 provided non-pooled results of the same three mentioned RCTs.28–30 In this sense, long-term, well-designed, and solid evidence is still needed, particularly on other highly prevalent hematologic diseases for which CAR-T therapies are already being used such as myeloma.
The aforementioned uncertainties make the use of rigorous methodologies when designing new clinical trials on this field particularly relevant. In the same way, an adequate reporting of their results will be essential to better inform clinical decision-making. Otherwise, the consequences of relying solely on low-quality designs to drive the clinical practice, such as one-arm trials, may lead to hasty definite conclusions with no real net benefit for patients.
Strengths and limitations
Our living systematic review has some strengths to be emphasized: First, up to our best knowledge, this is the first living systematic review that focused on comparative studies assessing CAR-T in hematologic malignancies. Second, this work has benefited from advanced technological tools to facilitate the process of assessing evidence. Third, it has been able to show some meaningful benefit favoring CAR-T on relevant variables such as CR and PFS. And fourth, no statistical heterogeneity was found in the majority of estimated outcomes.
However, this work is not exempt of limitations, principally due to the fact that it lies on only two RCTs assessing one hematologic condition (R/R B-lymphoma), beyond sparse and diverse quasi-experimental and observational data focused on T or B-ALL and R/R B-lymphoma. Also, the total sample size was rather limited and differences in comparators were confirmed among retrieved studies. As previously mentioned, some concerns have been raised about potential reporting bias in NRSI in relation to adverse events in the CAR-T group. Finally, reported information on baseline characteristics was not homogeneous among the included studies.
Conclusion
Current limited data from RCTs show a meaningful benefit in PFS and complete response for T or B-ALL and R/R B-lymphoma patients. However, no definite conclusion can be drawn at this point. We expect this limitation to be overcome in the near future when new sound evidence can be added to the existing one, especially when more RCTs are published.
Supplemental Material
sj-docx-1-tah-10.1177_20406207231168211 – Supplemental material for Efficacy and safety of chimeric antigen receptor T-cell (CAR-T) therapy in hematologic malignancies: a living systematic review on comparative studies
Supplemental material, sj-docx-1-tah-10.1177_20406207231168211 for Efficacy and safety of chimeric antigen receptor T-cell (CAR-T) therapy in hematologic malignancies: a living systematic review on comparative studies by Luis Carlos Saiz, Leire Leache, Marta Gutiérrez-Valencia, Juan Erviti and María Ximena Rojas Reyes in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The members of the Epistemonikos Foundation have made it possible to build the systems and compile the information needed by this project. Epistemonikos is a collaborative effort, based on the ongoing volunteer work of over 1000 contributors since 2012.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
