Abstract
Background:
Antihuman T lymphocyte porcine immunoglobulin (p-ATG) has been the most common ATG preparation in immunosuppressive therapy (IST) in Chinese patients with severe aplastic anemia (SAA) since 2009.
Objectives:
This study aimed to evaluate the early hematologic response and long-term outcomes of a large cohort of patients with SAA who received p-ATG plus cyclosporine (CsA) as first-line therapy from 2010 to 2019.
Design:
This is a single-center retrospective study of medical records.
Methods:
We analyzed the data of 1023 consecutive patients with acquired aplastic anemia (AA) who underwent p-ATG combined with CsA as a first-line IST treatment from 2010 to 2019 at our department.
Results:
The median age of the patients was 24 (4–75) years, and the median follow-up time was 57.2 months (3 days–137.5 months). There was an early mortality rate of 2.8% with a median death time of 0.9 months (3 days–2.9 months). The overall response rates were 40.6% and 56.1% at 3 and 6 months, respectively. The 5-year cumulative incidences of relapse and clonal evolution were 9.0% [95% confidence interval (CI) = 4.2–16.0%] and 4.5% (95% CI = 1.4–10.6%), respectively. The 5-year overall survival (OS) and event-free survival rates were 83.7% (95% CI = 81.1–86.0%) and 50.4% (95% CI = 47.1–53.5%), respectively.
Conclusion:
p-ATG combined with CsA for the treatment of AA is effective and safe, and p-ATG can be used as an alternative ATG preparation for the standard IST regimen in areas in which h-ATG is not available.
Introduction
The first report of the beneficial effect of antilymphocyte globulin (ALG) on autologous hematologic recovery in severe aplastic anemia (SAA) was done by Methe et al. 1 in 1970. In a series of subsequent studies, antihuman thymocyte globulin (ATG) combined with cyclosporine (CsA), as a standard intensive immunosuppressive regimen, was found to be a well-established first-line treatment for patients with SAA who are ineligible for hematopoietic stem cell transplantation.2–9
ATG/ALG is obtained from the immunization of animals, generally horses or rabbits, with human lymphoid cells, such as thymus (ATG) and thoracic duct lymphocytes (ALG). 10 Previous prospective and retrospective studies demonstrated that horse antihuman thymocyte globulin (h-ATG) was superior to rabbit ATG in terms of both hematologic response (HR) and survival.11–15 Therefore, h-ATG is recommended as the standard preparation for first-line immune suppression in SAA. Nevertheless, rabbit antihuman thymocyte globulin (r-ATG) combined with CsA is still widely used as a first-line immunosuppressive regimen in Europe and Asia because h-ATG (lymphoglobulin) was withdrawn from the market in 2007 and replaced by r-ATG, which is the only available formulation of ATG.
As in Europe, h-ATG has been unavailable in China since the year 2000. The antihuman T lymphocyte porcine immunoglobulin (p-ATG, provided by Yujin Bio-Pharma Wuhan CNBG Co., Ltd., Wuhan, China) was made available in 1983 and approved for the treatment of aplastic anemia by the Sino Food and Drug Administration in 2004. It is a commonly used ATG preparation in immunosuppressive therapy (IST) for SAA in China, even exceeding the r-ATG usage since 2009. A few small retrospective cohort studies reported that p-ATG combined with cyclosporine (CsA) is a useful immunosuppressive regimen for SAA in China.16–19 This study aimed to evaluate the early HR and long-term outcomes of a large cohort of patients with SAA who received p-ATG plus CsA as first-line therapy from 2010 to 2019.
Methods
Patients
This study retrospectively analyzed the data of 1023 consecutive patients diagnosed with SAA admitted to the Anemia Therapeutic Center of the Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences, between January 2010 and December 2019. The study protocol was approved by the institutional ethics committee (IIT2021008-EC-1) and follows the Declaration of Helsinki. The reporting of this study conforms to STROBE (Strengthening the Reporting of Observational studies in Epidemiology) guidelines. 20
All 1023 patients with a confirmed diagnosis of transfusion-dependent nonsevere, severe, or very severe aplastic anemia (VSAA) enrolled in this study were ineligible for allo-hematopoietic stem cell transplantation (HSCT) due to a lack of a human leukocyte antigen (HLA)–matched sibling donor, being above 40 years of age, or for financial reasons. Therefore, they received p-ATG with CsA as first-line IST. AA was diagnosed based on the International Agranulocytosis and Aplastic Anemia Study Group criteria, 21 and AA severity was determined using the modified criteria described by Camitta and Bacigalupo.22,23 Patients with congenital bone marrow failure were excluded from the study. Patients diagnosed with aplastic anemia with del 20/20q, +8, and −Y were included in this study.
Treatments
Antihuman T lymphocyte immunoglobulin administration
Patients received p-ATG for five consecutive days (day 1–5), at a dose of 20 mg/kg/day, as an intravenous infusion lasting 12–18 h. To prevent p-ATG-related serum sickness, corticosteroids were administered at a dose of 1 mg/kg/day for at least 7 days, tapered, and stopped within 2–3 weeks post-p-ATG treatment. For obese patients, p-ATG was dose adjusted according to the average between 1.3 × the ideal body weight and their actual weight.
CsA dosing and adjustments
CsA was administered orally from day 1 of p-ATG treatment at two separate doses starting from 3 to 5 mg/kg/day and adjusted to maintain the trough concentration at 150–250 ng/ml and the peak concentration at 800–1000 ng/ml. CsA was administered at the optimal dose until patients achieved the best HR for at least 3 months, and was then slowly tapered at a dose of 25–50 mg every 3 months.
Supportive care
Granulocyte colony-stimulating factor (G-CSF) was administered at a dose of 5–10 μg/kg when the absolute neutrophil count (ANC) was <0.5 × 109/l and discontinued when ANC was ⩾0.5×109/l. Red blood cell transfusion was administered to maintain hemoglobin levels ⩾70 g/l in elderly patients. Prophylactic platelet transfusion was administered to patients with stable AA with a platelet count of <10 × 109/l. Platelet transfusion was administered to patients with fever or sepsis when the platelet count was <20 × 109/l. During p-ATG treatment, the platelet count was maintained at 20 × 109/l. All patients were not given any antibacterial, antiviral, or antifungal prophylaxis after ATG administration.
Hematologic response evaluation
Complete response (CR) was defined as the fulfillment of all the following criteria without clonal evolution: hemoglobin >100 g/L, absolute neutrophils >1.0 × 109/l, and platelets >100 × 109/l. Partial response (PR) was defined as all the following: hemoglobin >70 g/l, absolute neutrophils >0.5 × 109/l, platelets >20 × 109/l, and transfusion independence. Good partial response (GPR) was defined as all the following: hemoglobin >80 g/l, absolute neutrophils >1.0 × 109/l, platelets >50 × 109/l, and transfusion independence. No response (NR) was defined as not meeting any of the response criteria defined above. The HRs were classified as NR if the patients died or were lost to follow-up within 6 months. Four and 14 patients were lost to follow-up within 3 and 6 months, respectively.
Follow-up and clonal evolution
The final follow-up in this study was in April 2022. Early death, delayed HR, overall survival (OS), event-free survival (EFS), clinical relapse, clonal evolution, and hemolytic paroxysmal nocturnal were evaluated during follow-up. Paroxysmal nocturnal hemoglobinuria (PNH) clones were screened every 3 months during the first year after IST and then every 6 months. Chromosomal karyotyping was performed by bone marrow aspiration at 3, 6, and 12 months during the first year after IST and then semiannually or annually to screen for clonal evolution. Definitions of these events are provided in the Supplementary Information. To assess relapse rate, patients were included regardless of the response time. Clinical relapse was defined as a decrease in blood counts to values requiring red blood cell and platelet transfusions or reinstitution of IST or HSCT. Thrombopoietin receptor agonist (TPO-RA), such as eltrombopag, was unavailable in China before 2019; thus, all nonresponse patients without rescue treatment, such as HSCT or HD-CTX, did not receive TPO-RA treatment.
Statistical analysis
All the included patients had a telephonic follow-up. The final follow-up date was 1 April 2022. SPSS version 22.0 (IBM Corp., Armonk, NY, USA) was used for the statistical analysis. GraphPad Prism 9 (GraphPad Software Inc., San Diego, CA, USA) was used to generate the figures. The chi-square test was used for categorical variables, and the Mann–Whitney U test was used for continuous variables. OS and EFS were estimated using the Kaplan–Meier method. The log-rank test was used to compare the OS and EFS values in the subgroups. The cumulative incidences of relapse and clone evolution events were estimated using a competing risk model. Statistical significance was set at p < 0.05.
Results
Patients
One thousand twenty-three aplastic anemia were included in this study, with a median age of 24 (4–75) years, with 550 males and 473 females. The median follow-up was 57.2 months (3 days–137.5 months). The clinical features were also noted (Table 1).
Characteristics of the patients at baseline.

PNH, paroxysmal nocturnal hemoglobinuria.
The category of abnormal karyotypes included 7 patients with deletion Y, 8 patients with trisomy 8, 3 patients with deletion 20 or 20q, 1 patient with deletion Y and trisomy 8, and 11 patients with other abnormal karyotypes (Supplementary Table S1).
Early death
Twenty-nine patients died within 3 months after IST, with an early death rate of 2.8% (29/1023); the median death time was 0.9 (0.1–2.9) months. There were 24 patients with VSAA and 5 with SAA, with infection and hemorrhage being the main causes.
Adverse events associated with p-ATG
The common adverse events were infusion-related reactions and serum sickness. The incidence of infusion-related reactions was 3.3% during the course of p-ATG infusion, including 12 cases of fever and 21 cases of mild rashes. The incidence of serum sickness was 55.4%, including 25.7% with fever, 16.1% with rashes, and 41.0% with muscle and joint pain. The incidence of other abnormal conditions was 7.9%, including hepatotoxicity (3.0%), renal insufficiency (1.4%), arrhythmia (0.4%), hoarseness (0.9%), and hemolytic anemia (2.2%). These events were controlled by a sufficient dose of glucocorticoid treatment.
Hematologic response
The overall hematologic response rate (ORR) was 40.7% (416/1023) at 3 months, including 5.9% (60/1023) patients with CR and 12.6% (129/1023) with GPR. Of the 608 patients who did not respond at 3 months, 129 had an HR at 6 months. The ORR was 53.2% (544/1023) at 6 months, including 13.5% with CR and 18.9% with GPR (Figure 1).

Hematologic response. The bar graphs showed the overall and complete responses at 3, 6, and 12 months, respectively.
The overall response rate at 3 months was lower in patients with VSAA (24%, 91/379) than in those with SAA (46.6%, 225/483) and tansfusion-dependent nonsevere aplastic anemia (TD-NSAA) (61.5%, 99/161) (both p < 0.000). The percentage of patients with CR at 3 months was 3.7% for VSAA, 6.2% for SAA, and 9.9% for TD-NSAA (p = 0.096, 0.000; Figure 2(a)). At 6 months, the ORRs were 38.8%, 59.4%, and 68.3% for VSAA, SAA, and TD-NSAA, respectively. The ORR was significantly lower in the VSAA group than in the other severity groups (p < 0.000, 0.000). The CR rate at 6 months was 9.8% for VSAA, 15.1% for SAA, and 17.4% for TD-NSAA (Figure 2(b)).

Hematologic response rate at 3 and 6 months (a and b) in different severity of aplastic anemia group. The bar graphs showed OR and CR at 3 and 6 months for 1023 patients in three severity groups (379 VSAA, 483 SAA, and 161 TD-NSAA), respectively.
The ORR of very young (<10 years old) patients at 3 months was 60.9%, which was significantly higher than that of the other four age groups (Table 2, p < 0.05). The CR rate at 3 months was 13.0% in the younger group and 7.1%, 4.6%, and 6.1% in the other three age groups (<65 years). No patient achieved a CR in the oldest age group (⩾65 years) at 3 months. At 6 months, the ORR was 71.7% in the younger group, which was higher than that in the other age groups (50.9%, 57.8%, 46.2%, and 26.3%). The CR rate at 6 months was not different among age groups, except for the oldest age group (⩾65 years) (Table 2).
Hematologic response, according to age.
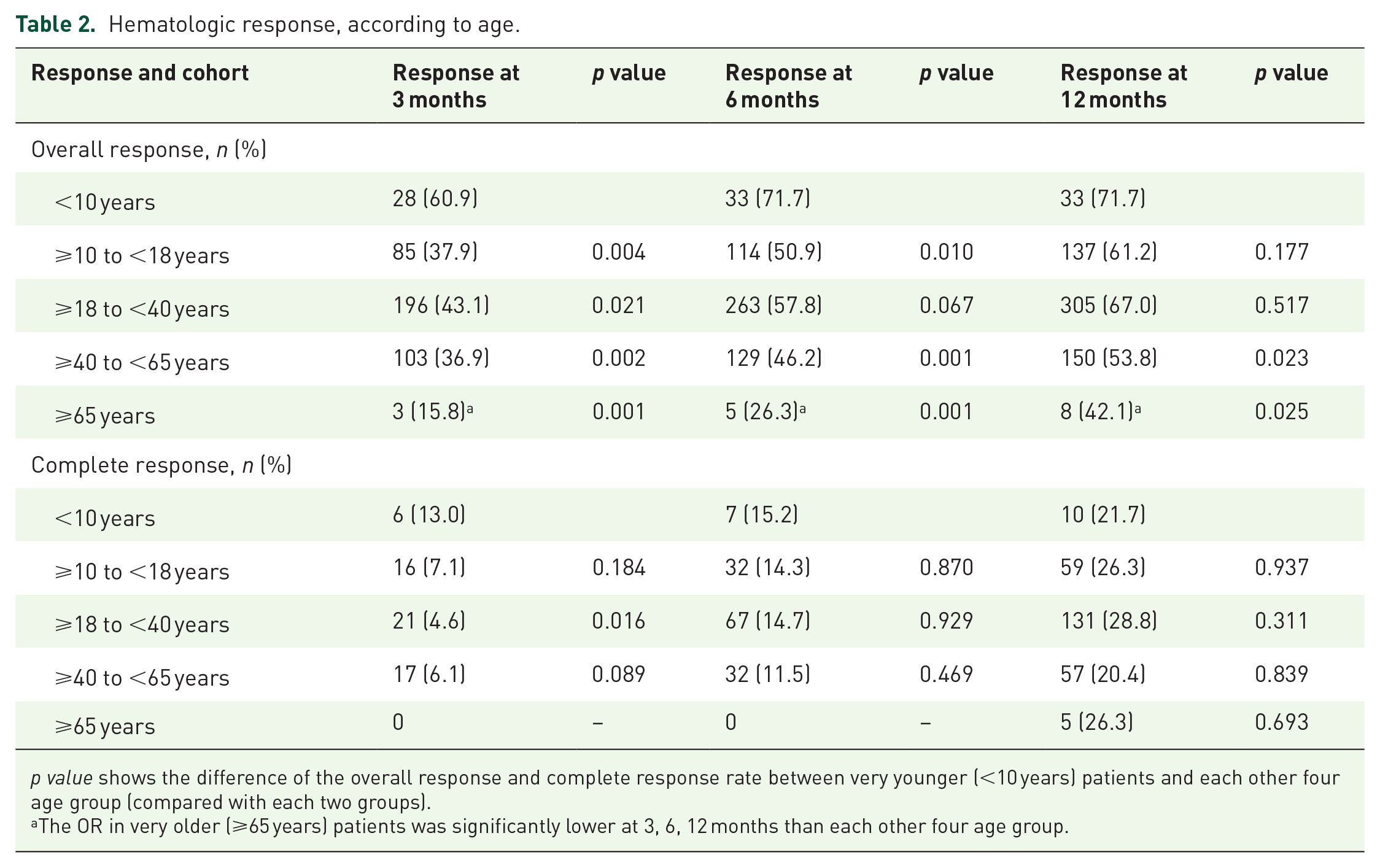
p value shows the difference of the overall response and complete response rate between very younger (<10 years) patients and each other four age group (compared with each two groups).
The OR in very older (⩾65 years) patients was significantly lower at 3, 6, 12 months than each other four age group.
The ORR of patients with PNH clones at 3 months was 42.3%, with CR + GPR rate of 21.2%, which were not different from those of patients without PNH clones (ORR of 40.3% and CR + GPR rate of 18.0% at 3 months). At 6 months, the ORR and CR + GPR were 54.5% and 39.7%, respectively, for patients with PNH clones, which were similar to those of patients without PNH clones (ORR of 52.9% and CR + GPR of 30.7%). According to the year of IST, the ORR and CR rates were comparable between 2010–2014 and 2015–2019 at both 3 and 6 months (Supplementary Table S2).
Delayed HR
Of the 395 patients without early relapse and clonal evolution and who did not respond at 6 months, 26.1% (103/395) had an overall response at 12 months without any rescue treatment (10 had a CR, 30 had a GPR, and 63 had a PR). Moreover, 61.5% (243/395) of the patients had persistent NR, without any rescue treatment or clonal evolution. The other 49 patients underwent HSCT, died, or had clonal evolution. Patients with a delayed HR at 12 months had more VSAA and lower baseline median ANC, hemoglobin (HGB) levels, platelet (PLT) counts, and absolute reticulocyte count (ARC) compared with patients who had an HR within 6 months (Supplementary Table S3).
Of the 243 patients without a response at 12 months, 37.0% (90/243) had an overall response at the final follow-up, including 29 with CR, 30 with GPR, and 31 with PR. Only 48 patients were alive, with NR or any other event. Therefore, the overall delayed response rate without rescue treatment was 48.9% (193/395) at the final follow-up.
Predicting factors for HR
Details of the univariate analysis are provided in Supplementary Table S4. The multivariable analysis showed that disease history, disease severity, and age were the only three factors associated with an HR at 6 months (listed in Supplementary Table S5). Patients with older age (⩾40 years), VSAA, and longer disease history had a lower overall response at 6 months.
Rescue treatment and relapse
At the final follow-up, 60 (12.5%, 60/479) patients with NR underwent allo-HSCT as rescue treatment (48 with CR, 2 with GPR, and 10 died due to GVHD or severe infections during the HSCT), including three patients within 6 months and 20 patients within 12 months (one patient died due to graft-versus-host disease (GVHD) and 19 patients had a CR after allo-HSCT). The mortality rate associated with HSCT was 16.7% (10/60). The other nonresponse patients at 6 months were not administered HSCT as rescue treatment due to unavailability of suitable donors or failure to afford the high cost of HSCT.
Six hundred and ninety-seven patients had an HR during the treatment period. The median follow-up time was 64.0 (10.6–137.5) months. By the follow-up time point, CsA had been discontinued in 45.4% of those who had a response. The median duration of CsA use was 39.3 (18.6–120.8) months. Moreover, 9.2% (64/697) of the patients had experienced a relapse. The percentage of patients with relapse who had a response again after reuse or adjustment of the dose of the immunosuppressive agent was 48.4% (31/64), including six with CR after HSCT, two with CR after the second course of IST, and three with CR after moderate dose cyclophosphamide. Thirty-three patients were in a constant state of NR, even with the use of CsA, and 12 (36.4%, 12/33) died due to hemorrhage and infection. The 5-year cumulative incidence of relapse was 9.0% [95% confidence interval (CI) = 4.2–16.0%] (Figure 3(a)).

Cumulative incidence of relapse, clonal evolution, and hPNH. The shaded areas indicate 95% confidence intervals. Graph (a) showed that the 5-year cumulative incidence of relapse was 12.2% (95% CI = 7.1–18.9%). Graph (b) showed that the 5-year cumulative incidence of clonal evolution was 4.5% (95% CI = 1.4–10.6%). Graph (c) showed that the 5-year cumulative incidence of hPNH in all patients was 4.7% (95% CI = 1.2–12.4%). The red line indicates the cumulative incidence of hPNH in patients with PNH clones at diagnosis. The 5-year cumulative incidence of hPNH in PNH clone-positive patients at diagnosis was 22.5% (95% CI = 10.1–37.8%).
Karyotypic abnormalities, myeloid cancers
The clinical and chromosomal data were available for 959 patients at the final follow-up. Therefore, these patients were enrolled for clonal evolution. The median follow-up time was 57.3 (6.2–137.5) months, the clonal evolution rate was 4.5% (43/959), and the median evolution time was 19.6 (2.9–78.8) months. Of the 43 patients, 20 had confirmed karyotypic abnormalities that met the definition of karyotypic evolution without myeloid cancers, 19 progressed to myelodysplastic syndrome (MDS) (17 cases had karyotypic abnormalities), and 4 had acute myeloid leukemia (AML). The 5-year cumulative incidence of clonal evolution was 4.5% (95% CI = 1.4–10.6%) (Figure 3(b)).
Hemolytic paroxysmal nocturnal hemoglobinuria
The incidence of hemolytic paroxysmal nocturnal hemoglobinuria (hPNH) was 3.5% (34/959), and 82.4% of cases occurred in PNH clone (+) patients prior to the IST. The 5-year cumulative incidence of hPNH was 4.7% (95% CI = 1.2–12.4%). In patients with PNH clones at diagnosis, the 5-year cumulative incidence of hPNH was 22.5% (95% CI = 10.1–37.8%) (Figure 3(c)).
Long-term outcomes
The 5-year OS rate was 83.7% (95% CI = 81.1–86.0%) (Figure 4(a)), while the 5-year EFS rate was 50.4% (95% CI = 47.1–53.5%) (Figure 4(b)).

The 5-year overall survival and event-free survival. Graph (a) showed that the 5-year OS was 83.7% (95% CI = 81.1–86.0%). Graph (b) showed that the 5-year EFS was 50.4% (95% CI = 47.1–53.5%).
Compared with the OS according to AA severity, the 5-year OS was similar regardless of AA severity [90.8% (95% CI = 84.6–94.6%) in TD-NSAA, 86.9% (95% CI = 83.3–89.9%) in SAA, and 77.0% (95% CI = 72.2–81.0%) in VSAA, Figure 5(a)]. In the ⩾65-year-old group, the 5-year OS of 68.4% (95% CI = 42.8–84.4%, p < 0.001, Supplementary Figure S1) was significantly lower than that of the other age groups [86.2% (95% CI = 71.7–93.6%) in those aged <10 years, 89.8% (95% CI = 84.8–93.3%) in those aged 10–18 years, 84.1% (95% CI = 80.1–87.4%) in those aged 18–40 years, and 78.5% (95% CI = 72.5–83.3%) in the 40–65 years group].

Graph (a) showed that the 5-year overall survivals were similar in different severity of AA groups (p > 0.05); 90.8% (95% CI = 84.6–94.6%) in TD-NSAA, 86.9% (95% CI = 83.3–89.9%) in SAA, and 77.0% (95% CI = 72.2–81.0%) in VSAA. Graph (b) showed that the 5-year OS was significantly lower in no response at 6 months group [77.3% (95% CI = 72.4–81.5%)] than that in hematologic response group [95.9% (95% CI = 93.5–97.4%), p < 0.001].
According to the HR at 6 months, the 5-year OS was 95.9% (95% CI = 93.5–97.4%) and 77.3% (95% CI = 72.4–81.5%) in the HR and NR groups, respectively (p < 0.001, Figure 5(b)). The OS was comparable between the 2010–2014 and 2015–2019 groups [84% (95% CI = 79.5–87.6%) and 83.4% (95% CI = 80–86.3%, p = 0.685), respectively] (Supplementary Figure S2).
Discussion
Over the past five decades, from the introduction of ATG/ALG in the 1970s to the addition of CsA to ATG in the 1980s, a standard intensive immunosuppressive regimen of ATG combined with CsA was established. There are different series of ATG preparations available, with h-ATG recommended as the first-line preparation for treating AA. 24 Unfortunately, h-ATG is unavailable in China, and some previous studies have suggested that rabbit ATG is inferior to h-ATG.11–15 For Chinese patients with SAA, p-ATG has been the most widely used ATG preparation for the IST regimen in this decade, mainly owing to its recent efficacy, safety, and relatively low financial burden. Our previous study 25 on the pharmacokinetic characteristics of p-ATG showed that the blood concentration of p-ATG increased rapidly after administration, with a mean t1/2 of (10.479 ± 8.242) days. During the administration of p-ATG, the absolute lymphocyte count (ALC) decreased rapidly, with a median ALC of 0.31 × 109/l at the time of peak concentration of p-ATG and the lowest value being only 16.8% of the baseline value of ALC. Besides, the Tmax of p-ATG was similar to that of h-ATG reported in China and abroad, with T1/2β of 12.353 days, which is shorter than that of r-ATG (21.56–29.67 days). The residual drug of p-ATG was detected in vivo at 60 days after administration, which was shorter than that of r-ATG (90 days). The patients’ lymphocytes increased quickly with the clearance of p-ATG and returned to baseline level with the stabilized concentration of p-ATG, which differs from reports that r-ATG has a longer period of lymphocyte inhibition. The immunosuppression characteristics of p-ATG were similar to those of h-ATG. 25 This study aimed to evaluate the efficacy of the IST regimen based on p-ATG for patients with aplastic anemia, and the results showed that p-ATG had not only good short-term efficacy but also favorable long-term outcomes in patients with AA.
Our results showed that the overall response rate was 53.2% at 6 months, with an early death rate of 2.8%. A prospective randomized trial from the National Institutes of Health (NIH) showed that the overall response rate was 68% at 6 months and indicated that h-ATG was superior to r-ATG in AA. 13 Results from other studies, such as that of the European Bone Marrow Transplant Society (EBMT), 14 were in accordance with those of the NIH. Recently, EBMT findings indicated that the overall response rate of h-ATG (ATGAM, Pfizer, New York, NY, USA) plus CsA was 41% and 66% by the EBMT and the NIH criteria for HR, respectively. 26 Some studies, however, have suggested that it is comparable between r-ATG (approximately 65% of OR at 6 months) and h-ATG. Our previous results also showed an overall response rate of 60.3% at 6 months.27–30 The results of the study from Peking Union Medical College Hospital showed that the overall response rate could be as high as 83% after p-ATG combined with CsA treatment, which may be related to bias associated with the small sample size of 48 patients. 16 In addition, our and other research centers have demonstrated that the efficacy of p-ATG is comparable or superior to that of r-ATG in adults and children.17–19,31 The comparison of the outcomes of different preparations of ATG is listed in Table 3.11–18,29,30,32–38 Hence, although h-ATG is not available in China, the early efficacy of p-ATG is comparable to that of r-ATG or h-ATG, and the early death rate is low.
Comparison of the overall response of different preparation of ATG.

ATG, antihuman thymocyte globulin; h-ATG, horse antihuman thymocyte globulin; ORR, overall hematologic response rate; OS, overall survival; p-ATG, antihuman T lymphocyte porcine immunoglobulin; r-ATG, rabbit antihuman thymocyte globulin.
Patients with SAA are in a life-threatening state; therefore, any treatment is based on keeping the patient alive. With the development of bone marrow transplantation or IST, the survival rate of patients with SAA has improved from 20–25% to 60–80%. In our large cohort with long-term follow-up, the 5-year OS was 83.7%, which was a favorable result and comparable to that of a recent EBMT study. 39 The 5-year OS was higher in pediatric patients with AA, which was in accordance with the results of Nishikawa et al. 40 and those of our pediatric therapy center. 41 Although the 5-year EFS was only 50.4% compared with the OS, this result indicated that more patients with NR could be alive, which is attributed to the partial improvement in blood counts after IST and intensive supportive care (including transfusion and broad-spectrum antibiotics).
Surprisingly, about 26% of patients without an HR at 6 months had a response between 6 and 12 months without rescue therapy due to personal reasons, accounting for 10.1% of the total number and 15.9% of those with a response. Whether patients with aplastic anemia acquire an HR after IST depends on the presence of immune pathogenesis, the intensity of the immunosuppressive regimen, and the number of residual hematopoietic cells. Our results showed that there was more VSAA, indicating that the number of residual hematopoietic cells was less in patients with a delayed HR. Thus, these patients may need more time to recover hematopoiesis after IST. To now, however, we do not recommend this therapeutic strategy. TPO-RAs, such as eltrombopag and hetrombopag, which can stimulate the proliferation and differentiation of hematopoietic stem/progenitor cells, have been approved for refractory SAA and have shown good efficacy;42,43 meanwhile, these drugs can be obtained in China. Hence, applying TPO-RAs could be a rescue therapy regimen for patients with refractory aplastic anemia for IST, who were unsuitable for HSCT.
Late disorders are of special concern in IST, including relapse and clonal hematopoietic disorders such as PNH, MDS, and AML. This is an inevitable problem that needs to be addressed. It, however, does not reduce the value of IST in patients with SAA who are ineligible for HSCT. With our long-term follow-up, the 5-year cumulative incidence of relapse was only 9.0%, which was lower than that reported in most Western studies. We believe that the slow tapering of CsA contributed to the low relapse rate. For the other concerning issue, our results showed that the 5-year cumulative incidence of clonal evolution to MDS/AML was 4.5%, which was lower than the previously reported rate of 10–15% 44 but comparable to the transformation rate of our previous r-ATG study. From this point of view, the late clonal evolution of AA after IST may not be related to ATG preparations and is still caused by its own hematopoietic pressure. Therefore, regular monitoring of chromosome karyotypes remains crucial for patients with AA who are treated with IST. The main limitation of this study is being a single-center retrospective study. In future, prospective, randomized controlled clinical trials on h-ATG will be needed in China.
Conclusion
The p-ATG combined with CsA for the treatment of AA was effective and safe, and p-ATG could be used as an alternative ATG preparation for the standard IST regimen in areas in which h-ATG is not available.
Supplemental Material
sj-docx-1-tah-10.1177_20406207221146031 – Supplemental material for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study
Supplemental material, sj-docx-1-tah-10.1177_20406207221146031 for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study by Wenrui Yang, Xu Liu, Xin Zhao, Li Zhang, Guangxin Peng, Lei Ye, Kang Zhou, Yuan Li, Jianping Li, Huihui Fan, Yang Yang, Youzhen Xiong, Liping Jing and Fengkui Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-2-tah-10.1177_20406207221146031 – Supplemental material for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study
Supplemental material, sj-tiff-2-tah-10.1177_20406207221146031 for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study by Wenrui Yang, Xu Liu, Xin Zhao, Li Zhang, Guangxin Peng, Lei Ye, Kang Zhou, Yuan Li, Jianping Li, Huihui Fan, Yang Yang, Youzhen Xiong, Liping Jing and Fengkui Zhang in Therapeutic Advances in Hematology
Supplemental Material
sj-tiff-3-tah-10.1177_20406207221146031 – Supplemental material for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study
Supplemental material, sj-tiff-3-tah-10.1177_20406207221146031 for Antihuman T lymphocyte porcine immunoglobulin combined with cyclosporine as first-line immunosuppressive therapy for severe aplastic anemia in China: a large single-center, 10-year retrospective study by Wenrui Yang, Xu Liu, Xin Zhao, Li Zhang, Guangxin Peng, Lei Ye, Kang Zhou, Yuan Li, Jianping Li, Huihui Fan, Yang Yang, Youzhen Xiong, Liping Jing and Fengkui Zhang in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
