Abstract
Background:
Letermovir prophylaxis is currently the standard of care for the prevention of cytomegalovirus (CMV) infections in allogeneic hematopoietic stem-cell transplantation (allo-HSCT). However, drug–drug interactions between letermovir and azoles or calcineurin inhibitors and the high financial burden of letermovir remain problematic, especially in resource-limited countries. It has not been clarified whether a lower dose of valganciclovir would constitute an effective strategy for CMV prevention in patients with acute leukemia undergoing allo-HSCT.
Methods:
We retrospectively assessed 84 consecutive adult patients with acute leukemia who underwent allo-HSCT. These 84 patients were stratified into a valganciclovir prophylaxis group (
Results:
Patients in the valganciclovir prophylaxis group had a lower possibility of CMV DNAemia at week 14 after allo-HSCT than those in the non-valganciclovir prophylaxis group (15.0%
Conclusion:
Low-dose valganciclovir prophylaxis could be an alternative to letermovir to prevent CMV infection in allo-HSCT, especially in resource-limited countries.
Introduction
Allogeneic hematopoietic stem-cell transplantation (allo-HSCT) is a necessary treatment for various hematological diseases. For patients with acute myeloid leukemia with intermediate- and poor-risk cytogenetic and molecular features, allo-HSCT is considered the preferred post-remission therapy. 1 In terms of acute lymphoblastic leukemia, complex, Philadelphia, or other high-risk chromosome abnormalities at diagnosis, and persistent minimal residual disease detected with multicolor flow cytometry, polymerase chain reaction assay for B cell immunoglobulin and T cell receptor gene rearrangements, or next-generation sequencing after remission induction chemotherapy are indications for allo-HSCT. 2 Although allo-HSCT provides a survival benefit to patients with acute leukemia, infection remains a profound complication and may result in transplant-related mortality. After allo-HSCT, cytomegalovirus (CMV) is notorious among the numerous types of infectious agents because CMV infection may cause multiorgan diseases in allo-HSCT recipients. 3
Currently, preemptive therapies and prophylaxis are the two primary strategies to prevent CMV diseases in allo-HSCT. A European conference on infections in leukemia has proposed preemptive intravenous ganciclovir or oral valganciclovir as the drug of choice for asymptomatic allo-HSCT recipients with detected CMV DNAemia greater than 1000 copies/mL.
4
Regarding preventive strategies, CMV prophylaxis in allo-HSCT started with acyclovir in 1981. With delayed CMV reactivation, acyclovir prophylaxis did not effectively prevent CMV diseases in allo-HSCT.
5
The second CMV preventive agent studied was ganciclovir. Unlike acyclovir, intravenous ganciclovir prophylaxis significantly reduced the risk of CMV infection in allo-HSCT. However, ganciclovir prophylaxis was not associated with reduction in all-cause mortality in allo-HSCT.
6
Additionally, ganciclovir-induced myelosuppression remains an unresolved issue. Long-term intravenous ganciclovir administration is also impractical. In the past decade, maribavir and brincidofovir have emerged as potential candidates for CMV prophylaxis in allo-HSCT. However, a phase III trial by Marty
Letermovir is currently the standard of care for CMV prophylaxis in allo-HSCT because a 14-week prophylactic course of letermovir has been shown to significantly decrease CMV infection by 23.5% at week 24. 9 However, drug–drug interactions between letermovir and azoles or calcineurin inhibitors require more attention. Moreover, the associated high financial burden remains a concern, especially in resource-limited countries. To avoid the inconvenience of intravenous ganciclovir administration and the drug–drug interactions between letermovir and azoles or calcineurin inhibitors, oral valganciclovir could be an alternative to prevent CMV infection in allo-HSCT. A randomized controlled study showed that compared with preemptive CMV treatment, late prevention with daily 900 mg valganciclovir did not improve CMV disease-free survival. In addition, a greater degree of treatment-related neutropenia was observed in patients undergoing late valganciclovir prophylaxis. 10
Whether early intervention with a lower dose of valganciclovir would be an effective and safe strategy for CMV prophylaxis in patients with acute leukemia undergoing allo-HSCT remains unclear. Accordingly, we conducted this retrospective study to compare the incidence of CMV DNAemia at week 14 after allo-HSCT among patients with acute leukemia with and without low-dose valganciclovir prophylaxis to address the possible application of low-dose valganciclovir in this setting. We also analyzed the overall survival (OS) difference at week 48 between these two groups of patients.
Methods
Patients
We retrospectively reviewed the medical records of 84 consecutive adult patients with acute leukemia in complete remission undergoing allo-HSCT at our hospital between February 2010 and December 2019. To investigate the impact of CMV prophylaxis with a low dose of valganciclovir in patients with acute leukemia receiving allo-HSCT, these 84 patients were further stratified into the valganciclovir prophylaxis group (
Clinical characteristic comparison between patients with and without valganciclovir prophylaxis.
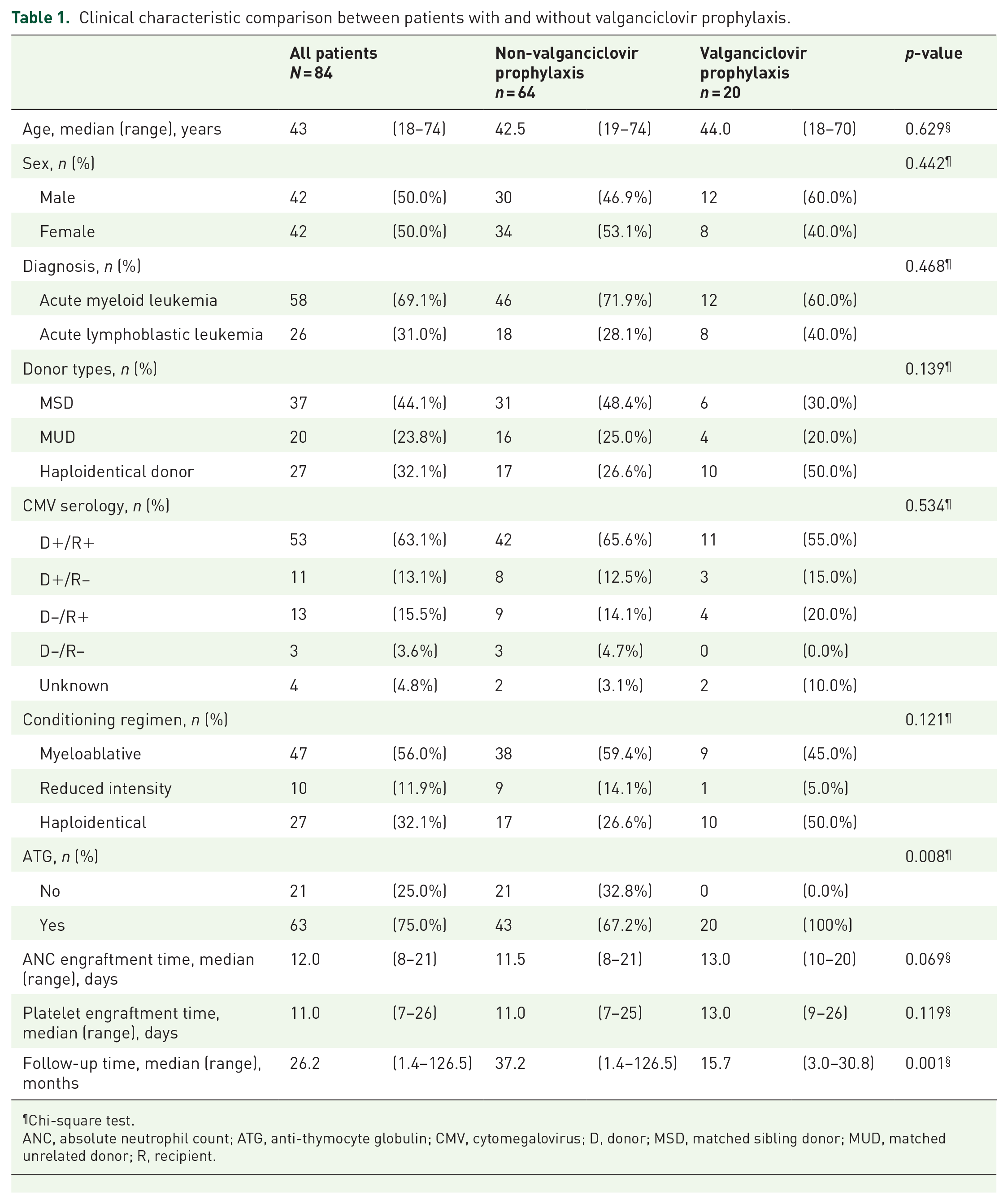
Chi-square test.
ANC, absolute neutrophil count; ATG, anti-thymocyte globulin; CMV, cytomegalovirus; D, donor; MSD, matched sibling donor; MUD, matched unrelated donor; R, recipient.
Immunosuppressants for GVHD prophylaxis
We used cyclosporine as the primary immunosuppressant for GVHD prophylaxis since day −2 for patients allografted from both matched sibling donors and matched unrelated donors. A trough level between 150 and 250 ng/mL was the target concentration of cyclosporine. We gradually reduced the cyclosporine dose after day 90. Rabbit ATG (2 mg·kg−1day–1, from day –3 to day –2) was routinely administered to patients allografted from matched unrelated donors, but it was not mandatory for patients allografted from matched sibling donors. For haploidentical allo-HSCT, we implemented a GVHD prophylaxis strategy modified from the Johns Hopkins protocol. 11 Briefly, in addition to post-transplantation cyclophosphamide, patients undergoing haploidentical allo-HSCT also received both cyclosporine (since day 5) and rabbit ATG (2 mg·kg–1day–1, from day –3 to day –2) as part of their immunosuppressants’ regimen. To further prevent GVHD, we used myfortic acid at a dose of 720 mg twice daily starting on day –2 for patients allografted from matched sibling donors and matched unrelated donors and on day 5 for patients undergoing haploidentical allo-HSCT. Myfortic acid was discontinued on day 60.
CMV monitoring, prophylaxis, and preemptive treatment
We used the quantitative polymerase chain reaction assay of the COBAS® AmpliPrep/COBAS TaqMan CMV system (Roche Molecular Systems, Inc., Branchburg, NJ, USA) to monitor serum CMV DNAemia. This monitoring was conducted before allo-HSCT and once a week in the first 14 weeks after allo-HSCT. After week 14, we generally monitored for CMV DNAemia every 2 weeks until week 48. Notably, none of the allo-HSCT recipients in the current study were detected to have CMV DNAemia before allo-HSCT.
Regarding CMV prophylaxis, patients in the valganciclovir prophylaxis group received valganciclovir 450 mg per day, from the day of absolute neutrophil engraftment to approximately 100 days after allo-HSCT. The median duration of valganciclovir prophylaxis was 98.5 (range: 21–107) days. In contrast, patients in the non-valganciclovir prophylaxis group did not receive any prophylaxis for CMV. Preemptive antiviral therapy with ganciclovir and CMV-specific intravenous immunoglobulin started when the CMV viral load exceeded 1000 copies/mL in both groups of patients. The preemptive treatment continued until CMV DNAemia was no longer detected.
Outcome measurements and definitions
Comparison of the incidence of CMV DNAemia at week 14 between the valganciclovir prophylaxis group and non-valganciclovir prophylaxis group to identify whether a low dose of valganciclovir prophylaxis could decrease CMV DNAemia in allo-HSCT was the primary outcome measurement in the current study. This study also compared the difference in the OS rate at week 48 between the groups. The incidence of GVHD and relapse rate and causes of death were further analyzed (Figure 1).

Comparison of the cumulative incidence of cytomegalovirus (CMV) DNAemia between the study groups. The cumulative incidence of CMV DNAemia at week 14 after allogeneic hematopoietic stem-cell transplantation (allo-HSCT) in the valganciclovir and non-valganciclovir prophylaxis groups was 15.0% and 50.4%, respectively, with the difference between the groups being significant (
Regarding causes of death, death before week 14 after allo-HSCT was considered to constitute transplant-related mortality. Furthermore, we defined acute leukemia as the cause of death if leukemia cells were still identified from either peripheral blood or bone marrow at the time of death. GVHD was considered the cause of death if GVHD and CMV DNAemia or CMV diseases were simultaneously active at the time of death.
Statistical analysis
Continuous variables were compared between the valganciclovir and non-valganciclovir prophylaxis groups using the Mann–Whitney
Results
The patients in the valganciclovir prophylaxis group had a lower incidence of CMV DNAemia at week 14 than the patients in the non-valganciclovir prophylaxis group
The incidence of CMV DNAemia at week 14 after allo-HSCT was 15.0% and 50.0% in the valganciclovir and non-valganciclovir prophylaxis groups, respectively (
Outcome comparison between patients with and without valganciclovir prophylaxis.

Chi-square test.
Fisher’s exact test.
CMV, cytomegalovirus; GVHD, graft-
Low-dose valganciclovir prophylaxis significantly reduced the risk of CMV DNAemia at week 14
Next, we investigated the risk factors for CMV DNAemia at week 14 in patients with acute leukemia undergoing allo-HSCT. Univariate analysis revealed that older age (HR, 1.02; 95% CI, 1.00–1.05;
The multivariate analysis further validated this result, showing that allografting from matched unrelated donors (HR, 2.90; 95% CI, 1.16–7.29;
Risk factors of CMV DNAemia at week 14.

ATG, anti-thymocyte globulin; CI, confidence interval; CMV, cytomegalovirus; HR, hazard ratio; MSD, matched sibling donor; MUD, matched unrelated donor.
Patients in the valganciclovir and non-valganciclovir prophylaxis groups had similar transplant-related mortality and OS rates at week 48
In addition to the incidence of CMV DNAemia at week 14, we also studied the impact of valganciclovir prophylaxis on survival. The results showed that the transplant-related mortality rates in the valganciclovir and non-valganciclovir prophylaxis groups were 10.0% and 3.1%, respectively (

Comparison of overall survival (OS) between the study groups. The OS rates at week 48 after allogeneic hematopoietic stem-cell transplantation (allo-HSCT) in the valganciclovir and non-valganciclovir prophylaxis groups were 75.0% and 76.6%, respectively. The groups had similar OS rates at week 48 after allo-HSCT (
Cause of death analysis
Although patients in the valganciclovir and non-valganciclovir prophylaxis groups had similar acute GVHD (
Comparison of causes of death at weeks 14 and 48 between patients with and without valganciclovir prophylaxis.

Fisher’s exact test.
Chi-square test.
CMV, cytomegalovirus; GVHD: graft-
Adverse events of low-dose valganciclovir prophylaxis in allo-HSCT
Among the 20 patients receiving low-dose valganciclovir prophylaxis, eight (8/20; 40.0%) did not complete the prophylactic protocol, primarily because of neutropenia (
Discussion
This study found that the patients in the valganciclovir prophylaxis group had a lower possibility of CMV DNAemia at week 14 after allo-HSCT than the patients in the non-valganciclovir prophylaxis group (
Previous studies have shown the efficacy of prophylactic valganciclovir in various types of solid organ transplantations. One randomized controlled trial showed that CMV (+) renal allograft recipients receiving preventive 450 mg valganciclovir twice a day for 100 days had significantly lower rates of CMV infection or diseases up to month 84 than patients treated with preemptive valganciclovir.
12
Moreover, extended prophylaxis with 900 mg valganciclovir per day led to substantially lower CMV pneumonitis in lung transplant recipients. However, more than 50% of patients needed to discontinue valganciclovir prophylaxis either temporally or permanently, primarily because of leukopenia.
13
To minimize the valganciclovir-associated bone marrow suppression in allo-HSCT, our patients in the valganciclovir prophylaxis group received 450 mg/day valganciclovir from the day of absolute neutrophil engraftment to day 100 after allo-HSCT. Among patients receiving a low dose of valganciclovir CMV prophylaxis, we identified only three patients with CMV DNAemia before week 14 (3/20; 15%). This failure rate was similar to the clinically significant CMV infection incidence at week 14 in patients undergoing letermovir prophylaxis (7.7%).
9
Furthermore, the incidence of CMV DNAemia at week 14 among patients without a lower dose of valganciclovir prophylaxis in our study and the incidence of clinically significant CMV infection at week 14 among patients without letermovir prophylaxis in the cohort by Marty
Although the dose of prophylactic valganciclovir in the current study was lower than the dose applied to kidney and lung transplantation, valganciclovir-induced adverse events remain problematic in the allo-HSCT setting. In our study cohort, four patients (three because of neutropenia and one because of diarrhea) in the valganciclovir prophylaxis group needed to prematurely and permanently discontinue valganciclovir use before week 14 (4/20; 20%). Compared with maribavir or brincidofovir, 5 the toxicity of low-dose valganciclovir appears acceptable in allo-HSCT. However, only 10.2% of allo-HSCT recipients with letermovir prophylaxis needed to discontinue treatment before week 14, 9 suggesting that letermovir could be more tolerable than low-dose valganciclovir for CMV prophylaxis in allo-HSCT. Notably, the financial burden of prophylactic letermovir and drug–drug interactions between letermovir and calcineurin inhibitors or azoles require more attention than that paid to valganciclovir.
Identifying patients at high risk of CMV reactivation might be the best strategy to balance the benefits and adverse events associated with low-dose valganciclovir CMV prophylaxis in allo-HSCT. Although numerous factors may increase the possibility of CMV infection during allo-HSCT, the CMV seroprevalence rate in both recipients and donors remains a fundamental one. Unfortunately, the adult Taiwanese population’s CMV seropositivity rate can be as high as 90%, 14 rendering CMV infection a more severe problem in Taiwanese allo-HSCT recipients than in those in Western countries. In addition to CMV seroprevalence, older age, 15 donor types, 16 and ATG-containing conditioning regimens 17 are also associated with CMV infection in allo-HSCT. Our study showed that older age and haploidentical HSCT were significant factors for CMV reactivation. These results suggest that a low dose of valganciclovir may be potentially beneficial for CMV prevention in allo-HSCT, especially among older patients and patients undergoing haploidentical allo-HSCT.
Regarding OS, a low dose of valganciclovir prophylaxis did not result in a superior OS rate at week 48 after allo-HSCT. This result was not surprising because similar data were observed for letermovir prophylaxis, showing that patients in the letermovir prophylaxis and placebo groups had similar cumulative death rates from any cause at week 48. 18 Moreover, in the current study, relapsed acute leukemia remained the primary cause of death in both groups of patients, accounting for 60.0% of deaths in the valganciclovir prophylaxis group and 73.3% of deaths in the non-valganciclovir prophylaxis group. Notably, only two patients in the whole study cohort eventually died of CMV infection, suggesting that intensive CMV surveillance and appropriate preemptive ganciclovir treatment could effectively reduce CMV-related mortality in allo-HSCT. 19 Furthermore, we did not observe CMV-related mortality among patients failing valganciclovir prophylaxis, suggesting that low-dose valganciclovir prophylaxis might not result in ganciclovir therapeutic resistance in allo-HSCT.
Different transplant eras, the retrospective study design, and a limited number of patients in the valganciclovir prophylaxis group were notable limitations of the current study. In addition, we did not analyze the incidence of CMV DNAemia at week 24 because the CMV DNAemia surveillance schedule after week 14 could vary for each patient. Prospective and randomized-controlled studies with large numbers of patients are needed to validate our results. In summary, this study showed that with tolerable toxicity, low-dose valganciclovir prophylaxis significantly reduced CMV DNAemia at week 14 after allo-HSCT. However, this preventive approach did not result in superior OS at week 48 after allo-HSCT. Older patients and patients undergoing haploidentical allo-HSCT could benefit most from this treatment. Low-dose valganciclovir could be an alternative to letermovir to prevent CMV infection in allo-HSCT, especially in resource-limited countries.
Footnotes
Conflict of interest statement
Chieh-Lin Jerry Teng received an honorarium and consulting fees from Novartis, Roche, Takeda, Johnson & Johnson, Amgen, BMS Celgene, Kirin, AbbVie, and MSD. The other authors declare that they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from TCVGH-1093704D.
