Abstract
Patients with haemophilia A (HA) undergoing neurosurgical procedures have a high risk of haemorrhage with potential fatal outcome. Here, we present a successful perioperative haemostatic concept applying an extended half-life factor VIII (EHL FVIII), Efmoroctocog alfa, in two patients with HA undergoing neurosurgery for paramedian right-sided disc herniation (case 1) and astrocytoma (case 2). After adequate EHL FVIII treatment the surgical procedures were performed without any bleeding complications despite the high-risk interventions. Laboratory measurements confirmed stable FVIII levels throughout the hospital stay. We suggest close interdisciplinary collaboration between involved clinicians as mandatory prerequisite for an optimized perioperative management in patients with HA. The presented cases indicate, that the increased stability, safety and fewer injections provide a rationale to use EHL FVIII products in HA patients undergoing surgical interventions with a very high bleeding risk.
Introduction
Perioperative management of patients with haemophilia A (HA) is complex and requires a multidisciplinary team of the clinicians involved (i.e. surgeon, anaesthesiologist, haematologist). Collaboration with haemophilia centres is strongly advised to ensure an optimal treatment plan and to prevent morbidity and mortality of perioperative bleeding. 1 Neurosurgical procedures confer a very high risk of haemorrhage with potential fatal outcome. We here report successful perioperative management using an extended half-life recombinant factor VIII (EHL FVIII) product in two HA patients undergoing neurosurgical interventions.
Case presentation
Case 1
The 40-year-old patient had been diagnosed with severe HA in his childhood and prophylactic FVIII treatment was started at young age. Genetic work-up confirmed HA diagnosis by showing a deletion at exome 20 (c.6185-6186delAT). Despite FVIII prophylaxis, the patient underwent various orthopaedic surgeries due to haemophilia arthropathy. In recent years, the patient received prophylactic therapy with a standard FVIII product only twice weekly (50 IU/kg bodyweight) for personal reasons to reduce injection frequency. In February 2018, the patient suffered another joint bleed approximately 60 h after the last prophylactic application of a standard FVIII product. Consequently, treatment was switched to Efmoroctocog alfa, a recombinant FVIII-Fc fusion protein (rFVIIIFc) with an extended half-life of approximately 1.5-times that of standard FVIII products. 2 Prophylactic treatment was scheduled twice a week at a dosage of 50 IU/kg bodyweight. At the first follow-up visit, FVIII activity of 25% was observed 72 h after the last application. Further FVIII measurements at the next visits revealed FVIII activities of 7% and 100% at 90 h and 30 min after EHL FVIII application (incremental recovery: 1.98 IU/dl per IU/kg), respectively. The patient experienced a good quality of life without relevant major bleeding episodes.
In July 2019, the patient presented with radicular hypaesthesia and pain in the right arm refractory to prolonged conservative measures. Imaging revealed a paramedian right-sided disc herniation at C5/C6 and a clear indication for surgical anterior cervical discectomy and fusion. Preoperatively, the patient was referred to our department to fix an optimal concept for perioperative haemostaseological management. Prophylactic therapy with rFVIIIFc at a dosage of 25 IU/kg the day before surgery, 50 IU/kg directly prior to surgery (with a target FVIII activity ⩾80) and 37.5 IU/kg 12 h after surgery was suggested. Thereafter, 37.5 IU/kg rFVIIIFc was applied daily (targeting trough levels ⩾60%) for 6 consecutive days and thereafter every second day until day 10 using the same dose.
No bleeding complications during surgery were observed. Of note, despite the advice of the haemophilia treatment centre, a higher dosage of rFVIIIFc (62.5 IU/kg) was administered the day prior to surgery leading to FVIII levels above the target range (FVIII activity: 225%) prior to surgery. No bleeding episodes occurred after the surgical intervention during the daily administration of 37.5 IU/kg rFVIIIFc. FVIII measurements confirmed stable FVIII activity >80% during all timepoints. Figure 1a displays the timepoints of rFVIIIFc administration and the corresponding FVIII activity levels. Comparison of the FVIII levels, as measured by one-stage and chromogenic assays, showed only marginal differences. As a consequence of successful perioperative haemostaseological management, the patient was discharged already on day 4 after surgery. The patient continued FVIII treatment as mentioned above, and no further bleeding episodes were noted during follow up.
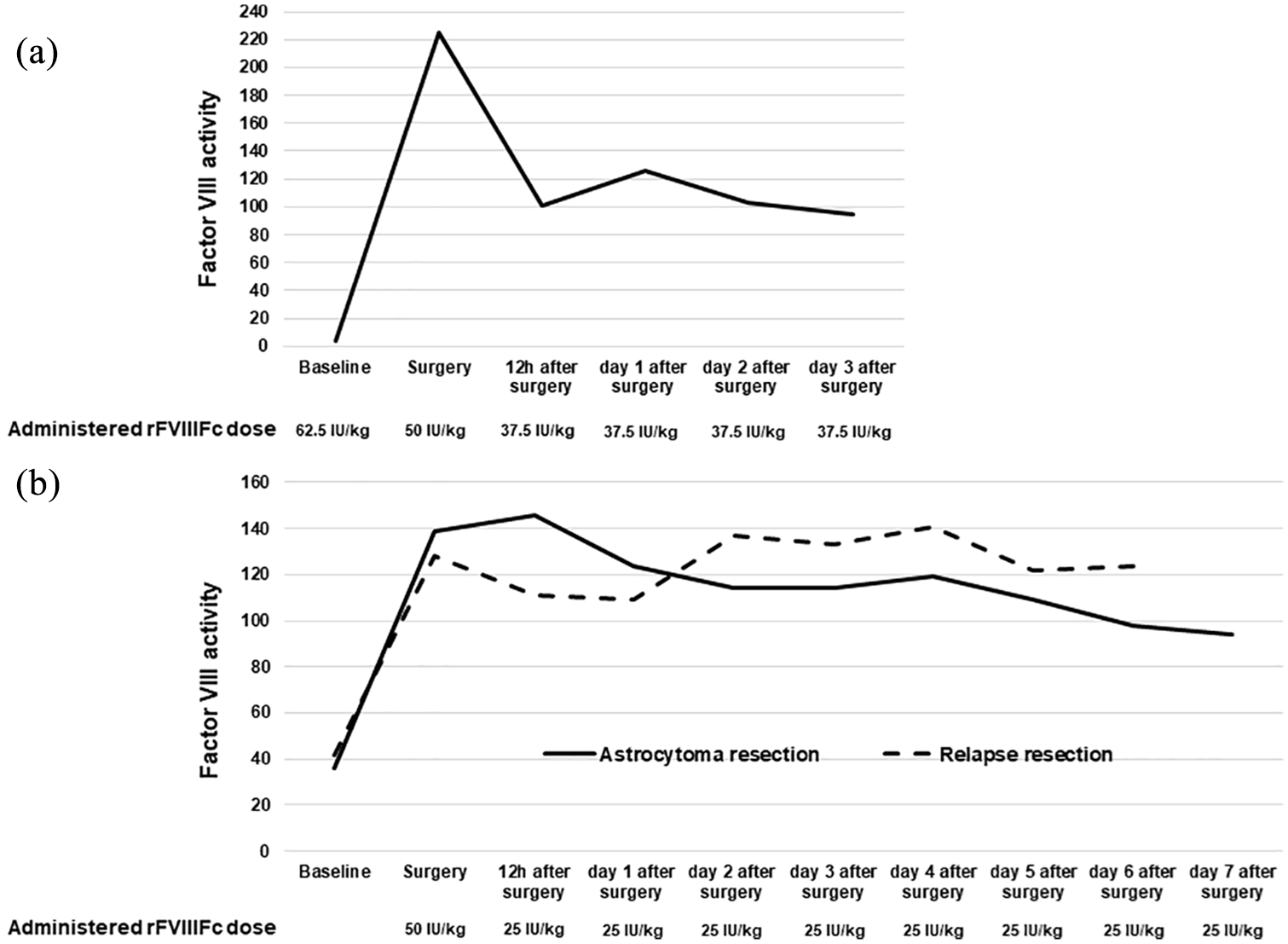
a,b Intra- and post-operative FVIII trough activity levels measured by one-stage assay during the presented neurosurgical procedures in case 1 (a) and case 2 (b). Administered EHL FVIII doses are provided as IU per kilogram bodyweight.
Case 2
This patient had been diagnosed with mild HA as an adolescent, with a FVIII activity of 25%. No prophylactic or on-demand FVIII substitutions had been performed. Medical history revealed an increased susceptibility for haematomas during exercise and one major bleeding event after tonsillectomy in childhood. In April 2018, at the age of 58 years, the patient suffered from fatigue and discomfort, finally prompting central nervous system (CNS) imaging, which revealed a left temporal lesion. Subsequently, his complaints were interpreted as mild epileptic seizures. A navigation-guided cerebral parenchyma biopsy was performed in another tertiary care hospital leading to the histological diagnosis of a grade II astrocytoma [isocitrate dehydrogenase (IDH)-wildtype, O(6)-methylguanine-DNA methyltransferase (MGMT) unmethylated]. Despite adequate substitution according to World Federation of Haemophilia (WFH)-guidelines with conventional recombinant FVIII, 3 the patient experienced postoperative intracerebral haemorrhage, which led to the decision not to surgically resect the tumor, but to start radio- and chemotherapy (54 Gy and five cycles of temozolomide). In July 2019, however, follow-up cerebral magnetic resonance imaging (MRI) showed progression of astrocytoma, the patient was referred to our centre for further treatment evaluation and resection of the lesion was planned. Due to the history of intracranial haemorrhage and the known bleeding diathesis, a perioperative haemostatic concept was made in collaboration with the haemophilia treatment centre. Laboratory analysis revealed FVIII activity of 36%. Perioperative treatment with rFVIIIFc was conducted. Prior to surgery, the patient received 50 IU/kg and 25 IU/kg 12 h after start of surgery. On days 1–7 after surgery, 25/IU/kg were administered daily with the aim of achieving constant FVIII activity >60%. Following the haemostatic management plan, no bleeding occurred during or after complete resection of the temporal astrocytoma and FVIII activity remained >90% at all measurements (shown in Figure 1b). In November 2019, the patient suffered a generalized epileptic seizure. At brain imaging, a relapse of the astrocytoma was suspected and a second resection was performed. A similar perioperative rFVIIIFc substitution concept was followed and again no relevant bleeding complications occurred, and FVIII activity remained >100% during the hospital stay (shown in Figure 1b).
Discussion
We present two cases of successful neurosurgical treatment of HA patients receiving perioperative treatment with the EHL FVIII product Efmoroctocog alfa (rFVIIIFc). This is noteworthy, as neurosurgical resections and, in particular, intracranial procedures, have a very high postoperative bleedings risk and postoperative bleeding is associated with high morbidity and mortality. 4 Most studies investigating the efficacy of FVIII substitutions do not report on the safety of those products during neurosurgery. Up to now, literature regarding such constellations is scarce, and only a few case reports are available dealing with hemostaseological concepts going alongside intracranial procedures.5,6 As a consequence, surgeons and anaesthesiologist are often reserved when indicating surgical interventions in HA patients. As an example, the standard procedure of resecting the astrocytoma upon diagnosis was not performed in the second patient due to known bleeding disorder. Even though a certain risk of bleeding exists even in mild HA, adequate substitution of EHL FVIII provided stable FVIII activity and successfully prevented relevant peri-procedural bleeding episodes.
The guidelines recommend a preoperative factor level of 80–100 percent during major surgeries. 3 According to a retrospective analysis of 50 surgeries in patients with HA, pre- and post-surgery FVIII levels were below the target range in 6.7% and 12.8% of patients, respectively. 7 In this study conventional FVIII products were administered. The use of rFVIIIFc in our patients resulted in stable activity levels above 80% during in hospital treatment. Therefore, it seems obvious that extended half-life formulations provide a more stable FVIII activity throughout the peri-operative procedures and may prevent perioperative bleeding and facilitate wound healing. 8 Moreover, increased stability and longer half-life result in reduced injections and fewer FVIII units needed per day. There is an ongoing discussion whether activity of EHL FVIII should be measured by one-stage or chromogenic assays. Only subtle differences were detected during the various measurements. Therefore, our data indicate that rFVIIIFc can safely be monitored either by one-stage or by chromogenic assays, as shown previously. 9 Even though continuous perioperative infusion of EHL FVIII has been suggested, 10 analysis of FVIII activity in our patients revealed stable FVIII activity levels during the hospital stay even with bolus treatment. Daily bolus management is certainly more convenient for the patient and might also be beneficial in the perioperative management of those patients in surgical wards.
Especially in mild HA, treating physicians have to be aware of the potential of inhibitor development. Since those patients have rarely been treated or are naive to FVIII products, danger signals during surgery in combination with high-dose FVIII treatment might lead to increased risk of inhibitor development. It has been shown that up to 57% of patients with mild HA develop an inhibitor upon intensive FVIII exposure. 11 Of note, no inhibitor development was observed in either patient upon rFVIIIFc despite intensive treatment and major surgeries.
In times of increasing life expectancy in HA patients, it can be assumed that HA patients will require more and more surgical procedures. 12 Therefore, it is of utmost importance to educate involved clinicians on the best possible haemostatic management of HA by providing optimal perioperative treatment algorithms to prevent bleeding and subsequent morbidity and mortality.
Conclusion
The cases presented show that treatment with EHL FVIII is feasible and represents a valid option even in patients requiring high bleeding risk surgical procedures such as neurosurgery. A prerequisite for optimal patient management is the close collaboration between the clinicians involved. The increased stability, safety and lower injection frequency might be an argument for the routine peri-operative use of EHL FVIII in HA patients requiring surgical procedures.
Footnotes
Author contributions
All authors contributed to the writing and editing of the manuscript and approved the final version of the manuscript.
Conflict of interest statement
Clemens Feistritzer reports grants, fees and research support from Sobi, Shire/Takeda, CSL Behring, Novo Nordisk, Biotest and Bayer; Florian Kocher, Andreas Seeber, Johannes Kerschbaumer, Stefan Schmidt and Dominik Wolf declare that they received no financial support related to this manuscript. All authors declare that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Publication fees were funded by Sobi. However, no financial support was recieved for the research and authorship.
Statement of ethics
The presented work complies with the guidelines for human studies and the research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. Both patients provided written informed consent for publication of the case presentation. According to the local regulations approval by the local ethical committee is not required for publication of case reports.
