Abstract
Background:
Primary central nervous system lymphoma (PCNSL) has received more attention because of an inferior prognosis. Less is known about the incidence rate, histological type, and survival rate of PCNSL, especially in the 2010s.
Methods:
Data of PCNSL from the Surveillance, Epidemiology, and End Results (SEER) registry database (SEER 9 registries and SEER 18 registries) were used. Incidence was estimated by age, gender, race, site, and histological type. Trends were analyzed using joinpoint regression and described as annual percent change (APC) and average annual percent change (AAPC). Five-year overall survival estimates were compared using log-rank tests.
Results:
Most PCNSL occurred in the brain, followed by the spinal cord. The most frequent histological type of PCNSL was diffuse large B-cell lymphoma, followed by marginal zone lymphoma. Incidence rate increased from 0.1/100,000 to 0.5/100,000 with an AAPC of 5.3% from 1975 to 2017. Incidence rates varied greatly between the younger and older age population. The 5-year overall survival rates in SEER 9 registries and SEER 18 registries were 30.5% and 37.4%, respectively. Even though the 5-year overall survival rate significantly increased from 27.9% for the 1975–1979 time period to 44.8% for the 2010–2017 time period, survival benefit could not be expected for patients ⩾60 years. The 5-year survival rate for elderly patients was about 30% in the 2010s.
Conclusion:
With aging, the incidence of PCNSL in the elderly is increased. Over the past decade, no advances have been made in the treatment of elderly PCNSL. Prospective trials with PCNSL are warranted to improve the survival of elderly patients.
Introduction
Primary central nervous system lymphoma (PCNSL) is an aggressive extranodal non-Hodgkin lymphoma that emerges directly from the central nervous system (CNS), including the eye, leptomeninges, brain, and spinal cord. Like other lymphomas, it is initially sensitive to chemo- and radiation therapy. Unfortunately, the prognosis for PCNSL remains poor in comparison with systemic lymphomas outside the CNS. PCNSL is a rare disease comprising about 4% of newly diagnosed CNS tumors and only 4–6% of all extranodal lymphomas, 1 with an annual incidence rate of 0.5 per 100,000 in the United States. 2 The incidence of PCNSL is steadily increasing as the population ages. 3 A study 4 based on the Surveillance, Epidemiology, and End Results (SEER) database showed a peak incidence rate of PCNSL in the 1990s because of the HIV epidemic.5,6 Risk of PCNSL increases with age and the incidence in older people is rising over time. In the past decade, a rising incidence rate of 4.3 per 100,000 has been observed in patients aged 70–79 years. 7 However, primary intraocular lymphoma (PIOL), a subset of PCNSL, was not included in several recent studies.3,7,8 Hence, we performed a retrospective cohort study of patients with histologically confirmed PCNSL using the SEER database to update the incidence rate, trends, and survival of PCNSL, and to characterize its pathological features.
Methods
Data source
Data of PCNSL were collected from the SEER program of the National Cancer Institute (NCI). Non-Hodgkin’s lymphoma (NHL) was defined using the SEER site recoding based on the World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues (2008). 9 PIOL was included in our study, and primary ocular adnexal lymphoma, originating from conjunctiva, lacrimal gland, orbit, eyelid, and extraocular muscle, was excluded from this study. All NHLs with topography codes C69.1–C69.4, C69.9, C70.0, C70.1, C70.9–C72.5, C72.8, and C72.9 were classified as PCNSL. We included data from two cohorts of patients: SEER 9 registries (1975–2017, Connecticut, Hawaii, Iowa, New Mexico, and Utah, and the metropolitan areas of Atlanta, Detroit, San Francisco–Oakland, and Puget Sound) and SEER 18 registries (2000–2017, SEER 9 plus Los Angeles, San Jose–Monterey, rural Georgia, Alaska, Greater California, Greater Georgia, Kentucky, Louisiana, and New Jersey). The SEER 9 registries were selected for this analysis in order to compare historical trends starting from 1970s. This study followed the guidelines of the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement. 10
Statistical analyses
To maximize the representativeness of our study, we calculated the 1975–2017 incidences using SEER 9 registries, and the 2000–2017 incidences using SEER 18 registries. All incidences were age-adjusted to the 2000 US standard population and were calculated using SEER∗Stat (version 8.3.8). The Joinpoint Regression Program (version 4.8.0.1) was used to compare temporal trends and to estimate the average estimated annual percent change (APC). Time from PCNSL diagnosis to death from any cause was evaluated using SEER∗Stat (version 8.3.8). We provided overall survival (OS) by gender, race, age, site, time of diagnosis, and pathological type with a maximum follow-up time of 40 years using data from the SEER 9 registries. To evaluate the recent trends in survival, we also conducted survival analyses from the SEER 18 registries. OS curves were constructed using the Kaplan–Meier method and compared with the log-rank test. Univariable and multivariable Cox regression analyses were performed using GraphPad Prism (version 8.0), and only variables with p ⩽ 0.2 in univariate analysis were included in multivariate analysis. Two-sided tests were used, and a p value of less than 0.05 was considered significant.
Results
Clinical character
In total, we identified 3841 cases of PCNSL in SEER 9 registries and 6647 cases in SEER 18 registries. Table 1 shows patient characteristics. The median age at diagnosis in SEER 9 registries was 63.0 years, and the median age in SEER 18 registries was 65.0 years. PCNSL in males was found to be slightly more common than in females. More than 50% of PCNSL patients were White or elderly who were >60 years of age. Most PCNSLs occurred in the brain, followed by the spinal cord. The most common histological type of PCNSL was diffuse large B-cell lymphoma (DLBCL), followed by marginal zone lymphoma (MZL) and rarely mantle-cell lymphoma (MCL) or peripheral T-cell lymphoma (PTL). The distribution of histological type of PCNSL varied greatly according to the diseased parts (Figure 1).

Histology distribution of PCNSL by sites in SEER 18 registries.
Characteristics of PCNSL in SEER 18 registries (2000–2017) and SEER 9 registries (1975–2017).

IQR, interquartile range; PCNSL, primary central nervous system lymphoma; SEER, Surveillance, Epidemiology, and End Results.
Incidence and trend of PCNSL
The age-adjusted incidence rate of PCNSL increased from 0.1/100,000 in 1975 to 0.5/100,000 in 2017, a fivefold increase (Figure 2). In the 2010s, the incidence rate of PCNSL ranged from 0.4 to 0.5 per 100,000 in the SEER database. The incidence rates of PCNSL varied greatly between age groups, ranging from 0.2 in patients under 60 years to around 2.0 in patients above 60 years over the last decade (Supplemental Appendix). Particularly, in 2016, an incidence rate of 3.2 per 100,000 was observed in patients aged 70–79 years (Figure 3), and incidence rates between other groups did not differ much. PCNSL incidence significantly increased with an average annual percent change (AAPC) of 14.2% from 1975 to 1991 and significantly decreased with an AAPC of −3.3% from 1991 to 2002 (Figure 2). PCNSL APCs in SEER 18 cohort were higher than the APCs in SEER 9 cohort after 2005. Statistical significance was reached in SEER 18 cohort (Figure 2). The incidence of PCNSL had been increasing steadily after 2000. By comparison, a statistically significantly increased trend was observed in patients aged 70–79 years with an AAPC of 2.4% from 2000 to 2017 (Figure 3).

Incidence of PCNSL and annual percent change (APC) trends in SEER 9 registries and SEER 18 registries (*p < 0.05).
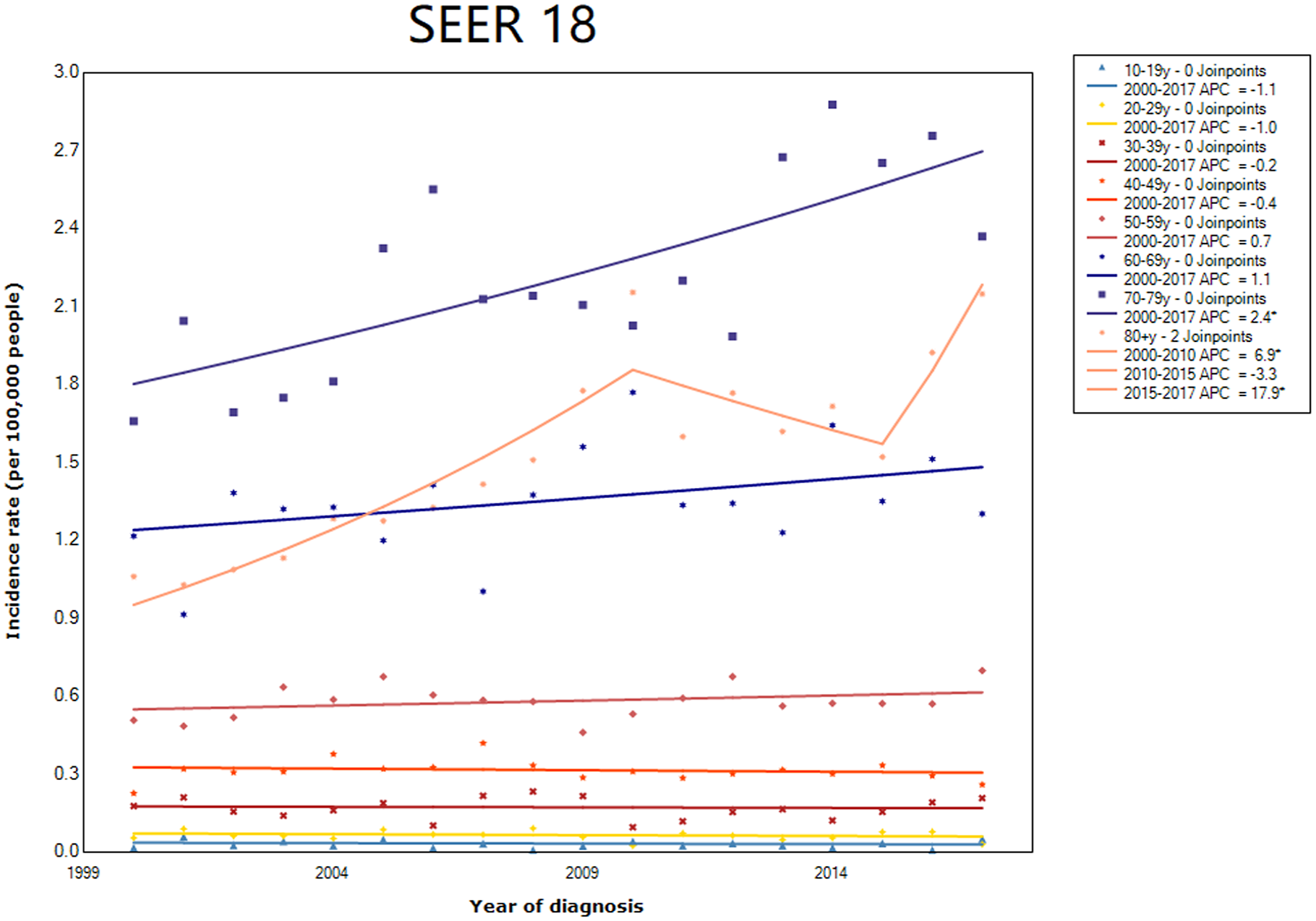
Incidence of PCNSL and annual percent change (APC) trends based on patient age in SEER 18 registries (*p < 0.05).
OS of PCNSL
The median follow-up time was 12 months. The 5-year OS in SEER 9 registries and SEER 18 registries was 30.5% and 37.4%, respectively. Sex-related survival difference was observed in SEER 9 (Supplemental Appendix). No gender-related survival difference was observed on the Kaplan–Meier curve in SEER 18 (Figure 4). There was no difference in survival between White and Black race (Figure 4). Older PCNSL age (⩾60 years old) was associated with worse OS (Figure 4). Brain parenchyma PCNSL had the worst prognosis compared with other sites (Figure 4). Also, significantly improved OS was observed in patients diagnosed in the 2010s in comparison with any other years (Figure 4). However, the prognosis of patients above 60 years remained poor, with a 5-year OS rate of about 30% (Supplemental Appendix). Multivariate analysis showed age, gender, time of diagnosis, and pathological type were independent indicators of prognosis. Race and site were not prognostic indicators. The detailed information is shown in the Supplemental Appendix.

Kaplan–Meier survival curves by subgroup in SEER 18 registries. (a) Gender, (b) Race, (c) Age,(d) Site, (e) Time of diagnosis, (f) Pathological type.
Discussion
Our study described the incidence rate, histology type, and outcomes of PCNSL patients in the United States based on the SEER database. In our analysis, the incidence of PCNSL steadily increased from 1975 to 2017, which could be due to improvement in access to care, diagnostic methods, and awareness. The sharp spike in the early 1990s was triggered by population age and the HIV epidemic.5,6 Regardless of age, the incidence of PCNSL was higher in males. 4 Incidence varied greatly by age group. In a population under age 40, incidence significantly decreased after 1996. The population people over 60 years is growing faster than any other age group, as a result of both longer life expectancy. More cases were observed in the elderly population, with the incidence being five times higher than the general population. Mendez et al. 7 reported that the annual incidence of PCNSL in patients aged 70–79 years was 4.32 per 100,000 population; however, only about 2.8 per 100,000 incidences were observed in our study and 1.90 per 100,000 incidences were observed by the patients aged ⩾75 years by Villano et al. 1 Incidence data of the study of Mendez et al. the Central Brain Tumor Registry of the United States (CBTRUS) were collected from database, which seems to be the main reason for the difference.
Miller et al. 11 reported more than 80% of PCNSL were DLBCL in Massachusetts General Hospital from 1958 to 1989. Sixty-two cases (86%) were classified as DLBCLs in the study of Camilleri-Broët et al. 12 Based on these studies, several reviews about PCNSL stated that approximately 90% of PCNSLs are DLBCL.13–17 Both data from SEER 9 registries and SEER 18 registries identified DLBCL as the most common histological type in approximately 80% of PCNSLs. The possible cause of the difference is that intraocular lymphoma was not included in this research. PIOL is a subtype of PCNSL, and 58% of PIOL carries a 50% risk of secondary involvement in the CNS beyond the eye. 18 The most common PIOL by far is primary vitreoretinal lymphoma (PVRL). 19 DLBCL is the most common PIOL with a prevalence estimated at 30–40%, followed by MZL. The histological type of intraocular lymphoma varies greatly according to whether the neoplasm predominantly involves the retina or the uvea. Primary retinal lymphomas are mostly aggressive, and primary choroidal lymphomas are typically low-grade lymphomas. 20
We investigated that age, sex, time of diagnosis, and pathological type were associated with survival in the multivariate analysis. Age is a strong negative prognostic factor in all lymphoma subtypes. Severity of physiologic dysfunction or comorbidity increasing with age can further affect the tolerability of therapy, which is associated with decreased survival. 21 Many studies have confirmed that women have a longer cancer-specific survival than men.22–24 Male sex was an adverse risk factor in patients with DLBCL, 25 follicular lymphoma (FL), 26 or MCL. 27 programmed cell death-1 (PD-1) Female sex was associated with decreased interferon signaling, transcription, cell cycle, and signaling, 28 which might affect treatment responses. Female patients with MCL are more sensitive to lenalidomide than male patients in a UK study. 29 The observation of improved survival after 2010 suggests current therapeutic approaches have improved survival.
Norden et al. 30 found out that despite treatment advances, the OS of PCNSL patients remains poor based on the SEER database from 1973 to 2004. In our study, the OS of PCNSL significantly improved from the 2000s, mainly because of the administration of high-dose methotrexate-based chemotherapy, immune-checkpoint inhibitors, and targeted molecules like the immunomodulatory drugs (IMiDs) or Bruton tyrosine kinase (BTK) inhibitor. 31 OS of PCNSL in the 1990s was an exception with an OS rate of 17.8% in our analysis. Studies conducted by the Japanese Society for Therapeutic Radiology and Oncology (JASTRO) Lymphoma Study Group or the Chubu Radiation Oncology Group showed that the 5-year survival rate of PCNSL was 18.0% in the 1990s, which was consistent with our analysis. 32 The French oculo-cerebral lymphoma network (LOC) We are unsure as to why survival declined in the 1990s. Study showed that age and sex were independent prognostic factors of PCNSL in the modern era. 33 Patients 60 years and older have an inferior prognosis even though the treatment of PCNSL has changed significantly over the past decades with superior survival. Our data underscore the urgency for clinical trials in elderly PCNSL to unearth mainstay options to evade poor prognosis.
The International Extranodal Lymphoma Study Group (IELSG) score 34 and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic score 35 emphasized the crucial role of age in the evaluation of prognosis in PCNSL. Previous studies had shown that the incidence of PCNSL gradually increases with aging.3,7 Unlike other studies, we chose 60 years as the cutoff value for age to perform subgroup analysis. First, the incidence of PCNSL in patients aged 60–69 years was two times higher than patients who were 50–59 years in our analysis. Second, no difference in OS was observed between patients aged 40–49 years and patients aged 50–59 years. However, a significant difference in OS was observed between patients aged 50–59 years and patients aged 60–69 years. Using 50 years as the age cutoff, the study of Jahr et al. 36 failed to verify the age for prognostication of OS in PCNSL. Patients <60 years displayed a median OS of 53 versus 10 months for those ⩾60 years (p < .05) in our study, which was consistent with the literature. 36
This study has some limitations to consider when discussing the current research. First, we must be aware that SEER 18 APCs are higher than SEER 9 APCs after 2005, because SEER 18 represents 28% of the US population versus 9% in SEER 9. 37 However, the SEER data may not reflect true incidence due to regional variation. Second, we did not evaluate the impact of HIV on the incidence and survival of PCNSL. The HIV epidemic resulted in a sharp spike in PCNSL incidence in the early 1990s. The observation of reduced OS in the 1990s might be a reflection of increased mortality from HIV. Since 2000, remarkable progress has been made in the diagnosis and treatment of persons living with HIV. Mendez et al. 7 reported that the incidence of PCNSL in patients with or without HIV was similar after 2000. Third, due to the nature of the SEER database, only six variables were selected for the Cox model. Survival analysis was unable to evaluate the value of the IELSG score 34 and the MSKCC prognostic score. 35 The MSKCC score may be better than the IELSG score for prognostication of survival in PCNSL. 36 B cell receptor (BCR) Due to the lack of genetic data, we cannot evaluate the role of BTK inhibitor in PCNSL patients with mutations altering the subunit CD79B and MYD88.
In summary, the incidence of PCNSL in the elderly increased. OS of elderly PCNSL was not improved significantly over the past decade. Prospective trials in elderly patients are essential to finding a therapeutic option in clinical routine to improve the outcome.
Supplemental Material
sj-doc-1-tah-10.1177_20406207211066166 – Supplemental material for Primary central nervous system lymphoma in the United States, 1975–2017
Supplemental material, sj-doc-1-tah-10.1177_20406207211066166 for Primary central nervous system lymphoma in the United States, 1975–2017 by Chenglan Lv, Jing Wang, Min Zhou, Jing-Yan Xu, Bing Chen and Yuan Wan in Therapeutic Advances in Hematology
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Binghamton University Faculty Startup Fund 910252-35. The work was partially supported by National Cancer Institute (1R01CA230339 subaward and 1R37CA255948).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
