Abstract
Background:
Inhibitor development is the most serious treatment-related complication of replacement coagulation factor VIII (FVIII) therapy for patients with haemophilia A. Immune tolerance induction (ITI), which involves intensive and prolonged treatment with plasma-derived or recombinant FVIII, is the only clinically proven strategy for eradication of inhibitors. The bispecific antibody emicizumab is approved for use in patients with and without inhibitors to prevent bleeding but does not eliminate inhibitors. MOTIVATE (www.motivate-study.com) aims to capture different approaches to the treatment and management of patients with haemophilia A and inhibitors, document current ITI approaches from real-world clinical experience, and evaluate the efficacy and safety of ITI, emicizumab prophylaxis and ITI with emicizumab prophylaxis.
Methods:
The investigator-initiated MOTIVATE study [ClinicalTrials.gov identifier: NCT04023019; EudraCT 2019-003427-38] will investigate in real-life clinical practice the management of patients with haemophilia A of any severity who have developed inhibitors to FVIII. All treatment is at the investigator’s discretion. The following treatment approaches will be evaluated: Group 1 – ITI with Nuwiq®, octanate® or wilate® and aPCC/rFVIIa if needed to treat bleeding episodes (BEs) or during surgery or for prophylaxis; Group 2 – ITI with Nuwiq®, octanate® or wilate® and emicizumab prophylaxis and aPCC/rFVIIa if needed to treat BEs or during surgery; Group 3 – routine prophylaxis with emicizumab, aPCC or rFVIIa without ITI and aPCC/rFVIIa if needed to treat BEs or during surgery. Patients will not be randomised to a treatment group and may change groups during the study.
Conclusions:
It is planned to enrol 120 patients who will be followed for up to 5 years. Optional sub-studies will explore factors that may influence ITI results as well as the impact of different treatment approaches on important aspects of patient health, including joint and bone health and the risk of thrombotic events.
Keywords
Introduction
Haemophilia A is a blood coagulation disorder caused by deficient or dysfunctional clotting factor VIII (FVIII) leading to incomplete haemostasis.1,2 Patients with severe haemophilia A are predisposed to recurrent bleeding episodes (BEs) in joints and soft tissues that culminate in debilitating arthropathy and long-term morbidity.3,4 Prophylaxis with plasma-derived or recombinant FVIII concentrates effectively restores FVIII levels in patients with haemophilia A, and significantly reduces the risk of bleeding.2,5,6 A critical concern for patients receiving FVIII replacement therapy is the development of neutralising antibodies (inhibitors) against the treatment.7,8 Inhibitors develop in up to 40% of patients with severe haemophilia A when first exposed to FVIII treatment, 9 typically within the first 20–30 exposure days (EDs) although a residual risk remains until after 75 EDs.10,11 Inhibitors preclude the use of FVIII replacement therapy for prevention and treatment of bleeding. 2
Emicizumab is a bispecific monoclonal antibody that binds to factor IX and factor X, mimicking FVIII but without sharing structural homology, and is effective in the prevention of bleeding in haemophilia A patients with and without inhibitors.12,13 Whereas the benefits of FVIII prophylaxis are well established, 14 the impact of long-term emicizumab use, including on bone and joint health, is unknown. Importantly, emicizumab does not eradicate inhibitors, and patients on emicizumab may still experience BEs or need to undergo surgery, requiring the use of another haemostatic agent.9,15 Patients with FVIII inhibitors therefore rely on bypassing agents, including recombinant activated factor VII (rFVIIa) and activated prothrombin complex concentrates (aPCC).13,16 However, bypassing agents are expensive and provide incomplete haemostatic correction in some patients, and the risk of thrombotic events associated with bypassing agent use is a concern.13,16 Serious thrombotic events and fatalities have been reported in patients receiving emicizumab and have been postulated to be associated with concomitant aPCC treatment.17 –19 Patients with inhibitors may also be unable to access potential future therapies, such as gene therapy. 13
Eradication of inhibitors therefore remains an important objective for haemophilia A patients with inhibitors. 16 Immune tolerance induction (ITI) therapy is the only clinically proven strategy for inhibitor eradication, and at least one attempt should be offered to patients with inhibitors.9,16 However, while ITI is well-studied and has a 60–80% success rate, 20 treatment regimens can be expensive and burdensome to patients. 9
There are limited data on the use of emicizumab prophylaxis together with FVIII ITI.21 –23 MOTIVATE (www.motivate-study.com) was designed to explore strategies for managing patients with haemophilia A and inhibitors with a focus on evaluating the safety and efficacy of ITI and/or emicizumab prophylaxis. MOTIVATE includes multiple optional sub-studies to investigate factors that may influence ITI results as well as the impact of different treatment approaches on important aspects of patient health, including joint and bone health and the risk for thrombosis.
Methods and analysis
Design
MOTIVATE is a multicentre, investigator-initiated study registered as a non-interventional study in the US [ClinicalTrials.gov identifier: NCT04023019] and a low-interventional, pragmatic trial in Europe (EudraCT No. 2019-003427-38). A total of 120 male patients of any age with haemophilia A of any severity who have developed inhibitors to any FVIII product will be enrolled at haemophilia treatment centres worldwide. All treatments are defined by the treating physicians prior to study inclusion and take into consideration the participants’ clinical condition and local prescribing information. For ITI, patients can receive either Nuwiq®, octanate® or wilate® for ITI, all of which have proven effective for ITI in inhibitor patients.24 –26 The FVIII product used may or may not be the same product to which the patient had developed an inhibitor.
The study will evaluate the following approaches (Figure 1):

MOTIVATE design.
Group 1 – ITI with either Nuwiq®, octanate® or wilate® and aPCC/rFVIIa if needed to treat BEs or during surgery or for prophylaxis
Group 2 – ITI with either Nuwiq®, octanate® or wilate® and emicizumab prophylaxis and aPCC/rFVIIa if needed to treat BEs or during surgery
Group 3 – Routine prophylaxis with emicizumab, aPCC or rFVIIa without ITI and aPCC/rFVIIa if needed to treat BEs or during surgery
Patients are not randomised to a particular treatment group and may switch to another group if their treatment is changed. Patients may also switch FVIII product during the study at the discretion of the treating physician. Concomitant medications can be administered at the discretion of the treating physician.
Compliance will be monitored by the treating physician as part of routine clinical practice. Participants may withdraw from the trial or be withdrawn by their parent(s)/legal guardian(s) at any time for any reason without the need to justify their decision. Participants may also be withdrawn at any time at the discretion of the treating physician for safety, behavioural, compliance or administrative reasons.
Objectives and endpoints
The overall objectives are to capture different approaches to the treatment and management of participants with haemophilia A and inhibitors, document current ITI approaches from real-world clinical experience and evaluate the efficacy and safety of ITI, emicizumab prophylaxis and ITI with emicizumab prophylaxis (Box 1). The primary endpoint for Groups 1 and 2 is ITI outcome, as determined by achievement of internationally recognised ITI criteria: inhibitor titre <0.6 Bethesda units (BU)/ml, FVIII recovery ⩾66% of the predefined reference value of 1.5% IU/kg body weight, and FVIII half-life ⩾6 h (Box 2). 27 The primary endpoint for Group 3 is the annualised bleeding rate (ABR) compared with the ABR in Groups 1 and 2 (Box 3).
MOTIVATE objectives.

Definitions of ITI outcome.

MOTIVATE endpoints.

Secondary endpoints for Groups 1 and 2 include the time to achieve ITI outcomes, use of FVIII, emicizumab, aPCC and rFVIIa during ITI and rate of FVIII inhibitor relapse in participants who have achieved complete ITI success. The secondary endpoints for all three groups include the frequency and severity of BEs (all BEs, treated BEs, spontaneous BEs, joint BEs and target joint BEs), number of infusions required to control BEs, frequency and severity of bleeding during and after surgical procedures, proportion of participants experiencing adverse drug reactions (ADRs), thrombotic events (location, treatment, outcome) and treatment costs. The Haemophilia Joint Health Score (HJHS) is used to assess joint health in all three groups.
Patient eligibility and recruitment
Patients eligible for study participation must meet all inclusion criteria and none of the exclusion criteria (Table 1). Patients start treatment after study inclusion according to the discretion of the physician and data are recorded prospectively. Patients who have already started treatment (after November 2017 in the US, after February 2018 in Europe, i.e., the date of emicizumab approval in the US and Europe, respectively) can also enter the study if detailed retrospective documentation on treatment, BEs, inhibitor titres and FVIII levels is available. Patients are allocated to a treatment group according to their initial treatment strategy and may change groups during the study. The maximum observation period for each patient is 5 years.
Patient criteria.
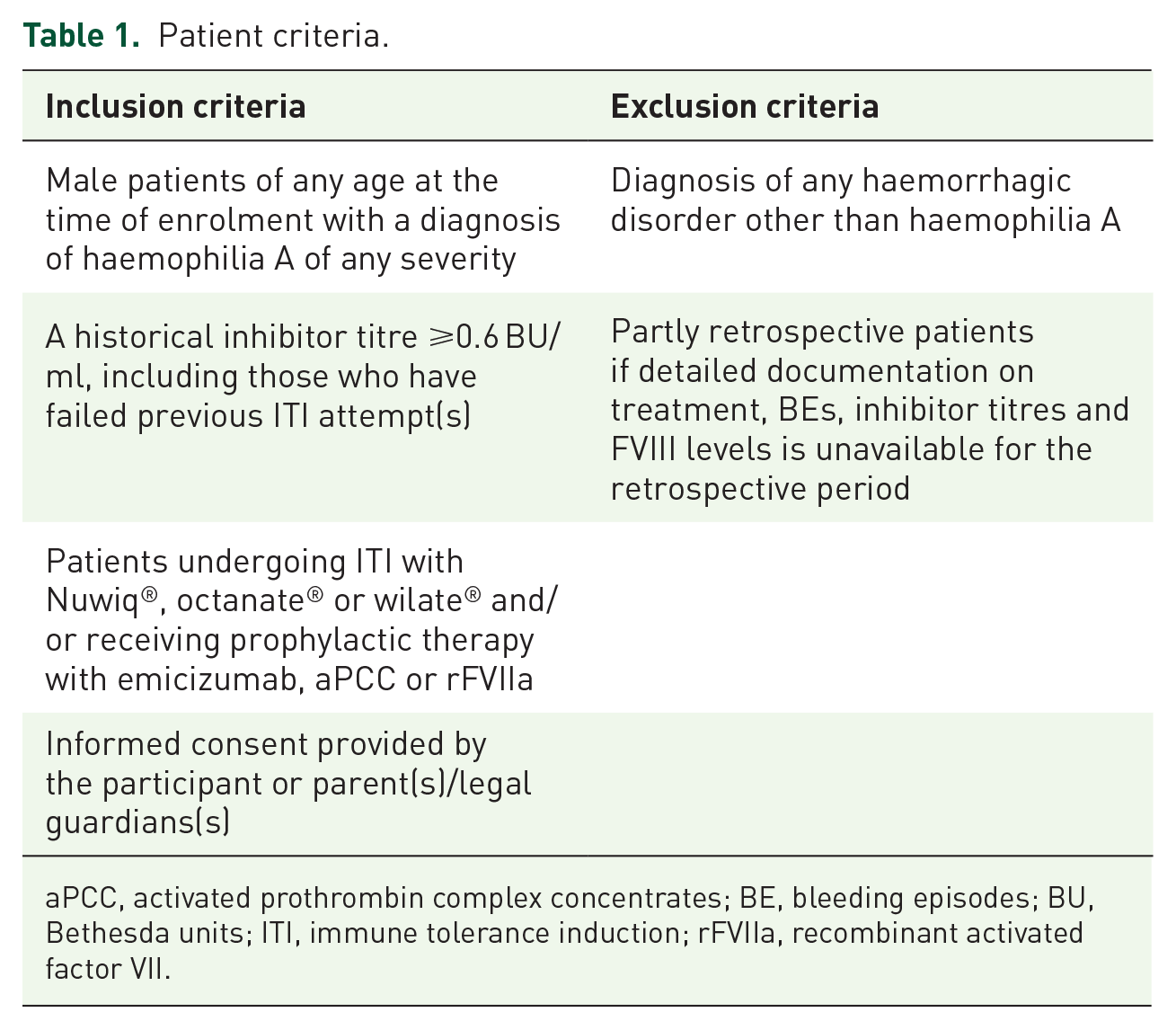
aPCC, activated prothrombin complex concentrates; BE, bleeding episodes; BU, Bethesda units; ITI, immune tolerance induction; rFVIIa, recombinant activated factor VII.
Recommended data collection and primary analyses
The study schedule is summarised in Table 2. Baseline data and retrospective data are collected at the time of enrolment. Throughout the study, the participants or participants’ parent(s)/legal guardian(s) will document clinical data in a treatment eDiary, and the treating physician will review the content of the patient eDiary.
Study schedule.
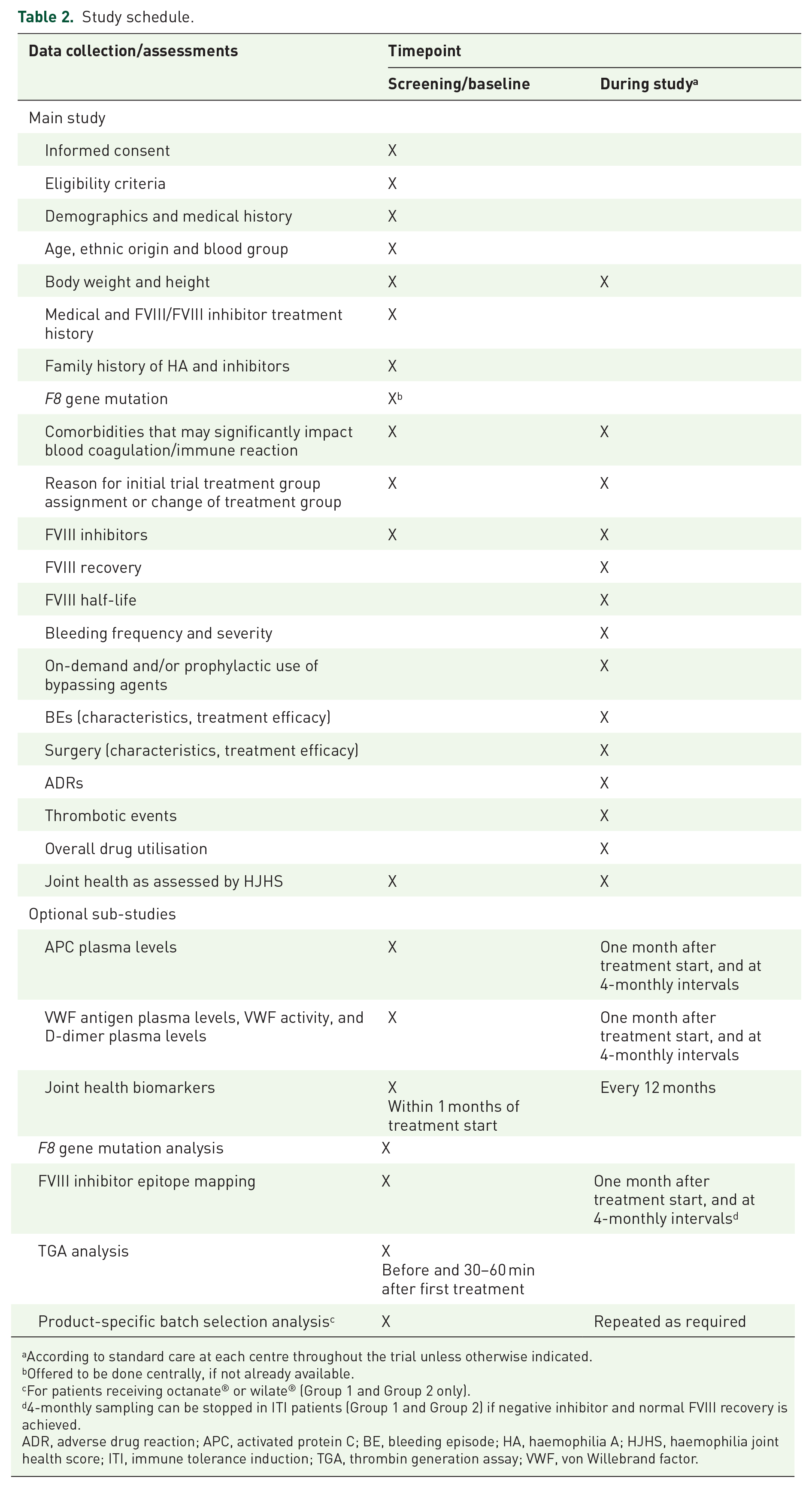
According to standard care at each centre throughout the trial unless otherwise indicated.
Offered to be done centrally, if not already available.
For patients receiving octanate® or wilate® (Group 1 and Group 2 only).
4-monthly sampling can be stopped in ITI patients (Group 1 and Group 2) if negative inhibitor and normal FVIII recovery is achieved.
ADR, adverse drug reaction; APC, activated protein C; BE, bleeding episode; HA, haemophilia A; HJHS, haemophilia joint health score; ITI, immune tolerance induction; TGA, thrombin generation assay; VWF, von Willebrand factor.
The primary analysis for Groups 1 and 2 will be ITI success as determined using internationally recognised criteria, as shown in Box 2. To evaluate FVIII recovery, it is recommended that FVIII plasma levels are measured prior to and approximately 15–30 min after FVIII treatment. To evaluate FVIII half-life, it is recommended that FVIII plasma levels are measured prior to and 15–30 min, 2 h, 4 h, 8–12 h and 24 h after administration of FVIII. When FVIII trough levels are >1% during regular prophylaxis, normal half-life can be expected and half-life can be evaluated from fewer samples (4, 24 and 48 h post injection) using a population pharmacokinetic model (e.g. WAPPS-Hemo).28,29
The primary analysis for Group 3 will be ABR, calculated as the number of BEs divided by the time on the treatment regimen, in years, compared with the ABRs in Groups 1 and 2. ABRs will be calculated for all BEs, treated BEs, spontaneous BEs, joint BEs and target joint BEs.
The following parameters will be used to assess the efficacy of BE treatment: BE start; BE end; BE site; BE severity; BE classification; haemostatic interventions; duration of hospitalisation; and the assessment of the effectiveness of treatment using a four-point scale (excellent, good, moderate or none). Information on product consumption will be collected where available.
For surgical procedures, the following data are collected where available: type of surgery; location of surgery; minor or major surgery; details on dose(s) of treatments given pre-, intra-, or post-operatively; pre-, intra- and post-operative FVIII plasma levels; estimated and actual peri-operative and post-operative bleeding volumes; relevant concomitant medications; and overall effectiveness assessment at the end of the surgical prophylaxis.
It is recommended that joint health is assessed using the HJHS at screening, every 12 months and at study completion.
Participating centres have the opportunity to send samples to a central laboratory service [Guy Young (US and Canada) and CEE (Europe and rest of world)] for FVIII and inhibitor measurements. Both one-stage and chromogenic assays can be used to determine FVIII activity levels.
Safety
The safety of all treatments will be assessed based on the incidence of ADRs. Thrombotic events (including location, treatment and outcome) are considered to be of special interest and will be monitored carefully. Serious ADRs and other significant safety findings will be reported to the Institutional Review Board/Independent Ethics Committee (IRB/IEC) in accordance with IRB/IEC procedures.
Optional sub-studies
MOTIVATE includes a number of optional sub-studies (Table 3).
Optional sub-studies.

APC, activated protein C; BALP, bone-specific alkaline phosphatase; CTX, C-terminal telopeptide of type I collagen; ETH, Eidgenössische Technische Hochschule; ITI, immune tolerance induction; NTX, N-terminal telopeptide of type I collagen; US, United States; VWF, von Willebrand factor.
A serious consequence of repeated joint bleeding in patients with haemophilia A is debilitating arthropathy, which can be further exacerbated by the presence of FVIII inhibitors. 30 Biomarkers of joint health will be analysed in a sub-study to evaluate the impact of different treatment approaches on joint and bone outcomes in patients with inhibitors. The sub-study also seeks to identify novel biomarkers of joint and bone health, which could be used to detect degeneration at an early stage.
Reduced response to activated protein C (APC) and higher levels of VWF and D-dimer in the plasma have been associated with risk of thrombosis.31 –33 The impact of the different approaches on the levels of these markers will be assessed in MOTIVATE sub-studies.
There is evidence that clinical outcomes in patients receiving ITI are affected by the type of F8 gene mutation and the target binding epitopes of FVIII.34,35 The F8 gene mutation analysis and FVIII inhibitor epitope mapping sub-studies aim to help identify predictors of treatment outcome.
Other optional sub-studies include thrombin-generation assay (TGA) analysis and product-specific batch selection analysis (for patients receiving ITI with octanate® or wilate®). Variations in the reactivity of inhibitory antibodies against the FVIII molecules in different concentrates, or batches of the same concentrate, have been observed.36 –38 To select an individual batch for ITI, in vitro batch selection is performed according to a modified Oxford method and the process repeated as required during the study.
Statistical methods
All statistical analyses will be performed using SAS® for Windows (version 9.4 or higher) and in accordance with the International Conference on Harmonisation (ICH) guidelines. 39 Descriptive statistics for continuous data will include the mean, standard deviation (SD), minimum value, lower quartile, median, upper quartile and maximum value. These descriptive statistics will be determined for measured values and for differences from baseline. Descriptive statistics for categorical data will be provided as contingency tables of absolute and relative frequencies of the categories. Confidence intervals (CIs) will be calculated where appropriate. When incidences are computed, these will be in relation to all patients at risk in the associated analysis population (e.g. incidences of ADRs).
If a patient switches treatment group, then data for that patient will be analysed as part of the initial group and the group to which the patient switched for the respective time periods in the two groups.
No inferential analysis involving formal testing is planned and therefore no formal sample size estimation was performed. A total of 120 participants will be enrolled in the study. In order to avoid over-enrolling into group 3, a maximum of 30 participants should be enrolled in this group. The goal to enrol a total of 120 patients with haemophilia A and FVIII inhibitors is based on experience of feasibility and not a statistical case number calculation taking into account the statistical power. Nevertheless, it will allow for medically valuable conclusions regarding the outcome of ITI and/or alternative approaches to treatment in patients with haemophilia A and FVIII inhibitors. Interim analyses are planned if data for a sufficient number of participants are available (up to three interim analyses are expected).
Ethics and dissemination
MOTIVATE is conducted in accordance with ethical principles of international guidelines including the Declaration of Helsinki and the Council for International Organizations of Medical Sciences (CIOMS), Good Clinical Practice (GCP) Guidelines and applicable laws and regulations. The protocols (and amended protocols, if applicable), informed consent forms and other relevant documents were submitted to the Ethics Commission of the Medical Association of Hessen, Germany, before the study was initiated (approval number 2019-1399-fAM). Any amended protocols will require IRB/IEC approval before implementation.
The treating physician obtains freely given written consent from each participant or the participant’s parent(s)/legal guardian(s).
The results are planned to be published in a peer-reviewed journal and presented at scientific meetings. Personal trial-related data will be used in accordance with local data protection law.
Patient and public involvement
There has been no public or patient involvement in the design of MOTIVATE. Results will be distributed to participants via the investigators who will receive the study report.
Discussion
MOTIVATE was designed to observe treatment strategies in patients with haemophilia A and inhibitors with a focus on evaluating and comparing the safety and efficacy of FVIII ITI, emicizumab prophylaxis, and FVIII ITI with emicizumab prophylaxis. MOTIVATE will collect data relating to the long-term impact of different treatment strategies on bleeding rates, inhibitor eradication, joint and bone health and risk for thrombosis. The international, multicentre, observational design will allow comparison of data from routine clinical practice in different countries and provide a broad representation of the haemophilia A patient population.
Even with the availability of emicizumab, current expert opinion states that inhibitor eradication remains a key goal and that patients with inhibitors should be offered at least one round of ITI that is personalised according to their risk profile for ITI success.9,16 However, more understanding is needed on how best to manage patients with haemophilia A and inhibitors.
ITI is the only clinically proven strategy for eradication of inhibitors.9,40 While ITI is highly successful and enables the return to effective FVIII therapy, ITI treatment regimens can be expensive and burdensome to patients. Low-dose/low-frequency ITI regimens have been investigated and are less costly than high-dose/high-frequency regimens and may reduce the need for central venous access devices (CVADs), which are associated with mechanical device failure and infections. 9 However, these low-dose/low-frequency regimens may take longer to eradicate inhibitors and have been associated with higher bleeding rates. 41
The use of FVIII ITI together with emicizumab could be an effective strategy to eradicate inhibitors and manage BEs while enabling low-dose, low-frequency regimens to offer a more convenient dose administration protocol to patients. 9 Small investigations have provided preliminary data on ITI together with emicizumab. In an ongoing study of seven patients with haemophilia A and inhibitors treated with low-dose ITI and emicizumab, three patients (43%) had achieved a negative inhibitor titre at the time of the interim report. 42 The four patients with measurable inhibitors had a median inhibitor titre of 1.3 BU/ml (range 0.8–3.2 BU/ml) at the last measured inhibitor titre compared with 2.5 BU/ml (range 0.7–30.7 BU/ml) at the start of ITI and emicizumab. Four of the patients experienced nine BEs, and no thrombotic complications were reported.
In addition to inhibitor eradication, there are other potential long-term benefits of FVIII treatment together with emicizumab in patients with inhibitors. Uncontrolled bleeding into joints may result in debilitating arthropathy in haemophilia A patients. While there are demonstrated benefits of FVIII prophylaxis for the maintenance of joint and bone health,43 –45 there is a lack of long-term clinical data on preservation of joint and bone health with emicizumab. In a primate model of haemophilia A, although emicizumab prevented macroscopic joint impairment subclinical histopathological changes in the joint synovium were found. 46 There is also evidence that FVIII plays a direct role in bone metabolism. 47 The impact of FVIII ITI and/or emicizumab on joint and bone health will be analysed in a sub-study of MOTIVATE, which will monitor levels of various markers of joint and bone health.
As treatment options for haemophilia A patients with inhibitors evolve, there is a need for real-world data on patient outcomes to inform clinical decision-making. MOTIVATE will prospectively collect valuable real-world data on the short- and long-term impact of treatment choice in patients with haemophilia A and inhibitors. These data may help to inform decisions on the standard of care for people with haemophilia A and inhibitors.
Footnotes
Acknowledgements
The authors would like to thank the MOTIVATE Steering Committee: Johannes Oldenburg (Germany; Chair), Cedric Hermans (Belgium), Victor Jiménez-Yuste (Spain), Roseline d’Oiron (France), Claire Berger (France), Maria Elisa Mancuso (Italy), Nadezhda Zozulya (Russia), Jan Astermark (Sweden), Ri Liesner (UK) and Guy Young (US), Sub-study Lead Investigators and Central Laboratory Service Coordinators. The authors also thank in advance the investigators, trial personnel, patients and caregivers for their participation in MOTIVATE. Medical writing assistance was provided by nspm ltd (Meggen, Switzerland) and funded by Octapharma AG.
Author contributions
The initial draft of the manuscript was prepared by CEE and RFS Jr. Both authors reviewed and approved the final version of the manuscript.
Conflict of interest statement
CEE has acted as a consultant, received speaker’s fees and/or received research funding from the following companies: Alnylam, Bayer Healthcare, Biomarin, Biotest, CSL Behring, Freeline, Grifols, Kedrion, LFB, Novo Nordisk, Octapharma, Roche/Chugai, Sobi, Takeda.
RFS Jr. has acted as a consultant and/or received research funding from the following companies: Octapharma, Genentech, Biomarin, Uniqure, Kedrion, Grifols, Bayer, Pfizer, Novo Nordisk, Bioverativ, Hema Biologics, Shire.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MOTIVATE is an investigator-initiated study funded by Octapharma AG, Lachen, Switzerland.
