Abstract
Background:
5-azacytidine (5-AZA) improves survival of patients with higher-risk myelodysplastic syndromes (MDSs) and oligoblastic acute myeloid leukemia (AML); however, predictive factors for response and outcome have not been consistently studied.
Methods:
This study of the Hellenic MDS Study Group included 687 consecutive patients with higher-risk MDS and oligoblastic AML treated with 5-AZA.
Results:
The International Prognostic Scoring System (IPSS) revised version (IPSS-R), Eastern Cooperative Oncology Group Performance Status (ECOG PS) (0 or 1 versus ⩾2) and baseline serum ferritin (SF) levels > 520 ng/ml were shown to independently predict response to 5-AZA. In the survival analysis, the IPSS and IPSS-R risk classification systems along with the ECOG PS and SF levels > 520 ng/ml proved to be independent prognosticators for overall survival (OS), as well as for leukemia-free survival (LFS). Next, we built new multivariate models for OS and LFS, incorporating only ECOG PS and SF levels besides IPSS or IPSS-R risk classification systems. Thereby, the new modified IPSS and IPSS-R risk classification systems (H-PSS, H-PSS-R) could each discriminate a low, an intermediate and a high-risk patient group regarding OS and LFS. The H-PSS and H-PSS-R proved to be better predictors of OS than their previous counterparts as well as the French prognostic score, while the most powerful OS predictor was the new, H-PSS-R system.
Conclusions:
ECOG PS and SF levels > 520 ng/ml independently predict response to 5-AZA, OS and LFS. Their incorporation in the IPSS and IPSS-R scores enhances these scores’ predictive power in 5-AZA-treated higher-risk MDS and oligoblastic AML patients.
Introduction
Myelodysplastic syndromes (MDSs) are a heterogeneous group of hematopoietic stem cell clonal disorders characterized by bone marrow dysplasia and ineffective hematopoiesis, leading to peripheral cytopenias and increased risk of progression to acute myeloid leukemia (AML). 1
The prognosis of patients with MDSs regarding survival and risk of transformation to AML is mainly determined by the International Prognostic Scoring System (IPSS) and its revised version (IPSS-R), which are based on the number of cytopenias, percentage of bone marrow (BM) blasts and cytogenetic abnormalities.2,3 In general, these systems separate MDS patients into two broad prognostic groups: those with lower-risk disease and those with higher-risk (HR) MDSs. HR-MDS patients are defined as those with intermediate-2 or high-risk score by the IPSS or intermediate (with >3.5 points), high or very-high-risk score by IPSS-R. The median overall survival (OS) for HR-MDS patients ranges from a few months to 1.2 years. Although the IPSS-R showed an improved prognostic accuracy over the IPSS, both the IPSS and the IPSS-R were built using cohorts of untreated MDS patients. IPSS-R has subsequently been shown to have prognostic value among treated MDS patients in retrospective analyses.4–7
In the last decade, 5-azacitidine (5-AZA) has been the standard of care for HR-MDS patients, as well as for AML patients who are ineligible for intensive chemotherapy and allogeneic hematopoietic stem cell transplantation (allo-HSCT). Treatment with 5-AZA prolongs time to transformation to AML and has shown a significant advantage in OS of HR-MDS patients compared with conventional care (median OS, 24.5 versus 15 months) in the randomized AZA001 trial. 8 However, it should be noted that in real life, the OS benefit from 5-AZA is considerably lower, ranging from 12 to 18 months in several studies.9–12 Although the recognition of predictive factors of response and outcome after treatment with 5-AZA is extremely important, the available data are very limited. In the largest series to date, Itzykson et al. evaluated 282 HR-MDS patients treated with 5-AZA and found that previous low-dose cytarabine treatment, BM blasts > 15% and abnormal karyotype independently predicted poor response to 5-AZA, whereas ECOG performance status (PS) ⩾ 2, presence of circulating blasts, red blood cell (RBC) transfusion dependency ⩾4 units in 8 weeks, and intermediate- or high-risk cytogenetics independently predicted inferior OS. Based on these factors Itzykson et al. developed the French prognostic score (FPSS), which separates 5-AZA-treated patients with HR-MDS into three prognostic groups with significantly different OS. 9 The FPSS was validated by the same group in 161 HR-MDS patients treated in the AZA001 trial, who represented an independent but highly selected patient cohort, and by two other groups in two small single-institution cohorts of 60 patients 13 and 90 patients, 12 respectively.
In this study from the Hellenic National Registry of MDS and Hypoplastic Syndromes, we sought to identify predictive factors for response and outcome in a large cohort of HR-MDS and oligoblastic AML patients who received 5-AZA as frontline treatment in the participating centers and we propose a simple modified prognostic model including IPSS or IPSS-R, along with ECOG PS and serum ferritin (SF) levels for leukemia-free survival (LFS) and OS.
Methods
Patients
This study included 687 consecutive patients with intermediate-2 or high-risk MDS and AML with BM blasts 20–30% who were treated frontline with 5-AZA in the participating centers, between July 2004 and February 2019. The data cutoff date for the analysis was July 21, 2019. Diagnosis of MDS was made according to the WHO 2016 diagnostic criteria. 14 BM aspiration, biopsy, and cytogenetic analysis had been performed in all patients before 5-AZA treatment. Risk stratification was made based on the IPSS and IPSS-R.2,3 Assessment of demographic, clinical and laboratory data was performed retrospectively. The values of the various biochemical parameters studied were at the time of diagnosis and before initiation of 5-AZA treatment. For C-reactive protein (CRP), α-2 globulin, and gamma globulin levels, increased values were considered those above the upper normal limit for each laboratory. Regarding SF levels, their categorization into two groups (i.e. high versus low SF levels) was performed using the X-tile software, which allowed the selection of the optimal cutoff for prognostic purposes; this cutoff value for SF levels was equal to 520 ng/ml.
The current study was performed in accordance with the ethical standards of the World Medical Association Declaration of Helsinki (version 2008) and it was approved by the “Attikon” University Hospital Institutional Review Board (approval number: 17-04-2018 Γ.8); all patients provided written informed consent for participation in the study.
Treatment and response categories
All patients received a single dose of 5-AZA at 75 mg/m2/day for seven consecutive days, or for 5 days/weekend off/2 days (5/2/2) subcutaneously on 28-day cycles. All patients were re-evaluated with BM aspiration after six cycles of 5-AZA and at the time of best response. Dose reductions of and/or treatment delays were considered for severe myelotoxicity or myelosuppression-related complications. Granulocyte colony-stimulating factors and erythropoiesis stimulating agents were used at the discretion of the treating physician. Response to treatment was evaluated using the International Working Group 2006 criteria. 15 Response categories were overall response (OR) defined as complete response (CR), partial response (PR), or any hematological improvement (HI); nonresponse defined as progressive disease or treatment failure; and stable disease (SD) as none of the above. OS and time to transformation to AML were calculated from the onset of treatment.
Biostatistical analysis
Univariate Cox regression models were built in order to assess the association between each categorical factor (or age at treatment onset) and the relative risk for transformation to AML and/or death of patients. The examined variables included patient sex, age at treatment onset, CRP, need for platelet and RBC transfusion, SF levels, ECOG PS, as well as IPSS, IPSS-R, and FPSS risks. The proportional hazard assumptions were tested by graphic illustration of Schoenfeld residuals. 16 The multivariate Cox regression analysis included only those variables that proved to be statistically significant in the univariate Cox regression analysis. The reliability of the developed prognostic models was strengthened by performing bootstrapping using 1000 samples as well as bias-corrected and accelerated (BCa) 95% confidence intervals (CI). Moreover, logistic regression was performed to assess the predictive value of CRP, SF level, and ECOG PS, with regard to response to 5-AZA treatment. Finally, the prognostic value of categorical factors regarding LFS and OS was also checked by Kaplan–Meier survival analysis; differences between Kaplan–Meier survival curves were evaluated by the log-rank (Mantel–Cox) test. Moreover, we examined the prognostic power of IPSS, IPSS-R, FPSS and the H-PSS and H-PSS-R risk classification systems, using Kaplan–Meier survival analysis. The new prognostic systems were compared with IPSS, IPSS-R, and FPSS, using Akaike’s information criterion [(AIC) a measure indicating the relative quality of a statistical model based on a given set of data] and the C-statistic (a measure indicating overall adequacy of prediction models with censored survival data). A smaller AIC value is considered as an indicator of a better fit of a prediction model to the data. A higher C-statistic indicates a better measure of goodness of fit for binary outcomes in a logistic regression model. The level of significance for all statistical tests was set at a probability value lower than 0.050 (p < 0.050).
Results
Biological and clinical characteristics of MDS patients
In this study, 687 patients (468 men and 219 women) with HR-MDS and oligoblastic AML were included, with a median age of 75 years (range: 33–95 years). Their categorization according to CRP and SF levels, need for RBC or platelet transfusions, ECOG PS, WHO 2016 classification, as well as IPSS, IPSS-R, and FPSS risk classification are summarized in Table 1. Moreover, biological characteristics of the patients as well as time intervals are presented in Supplemental Table 1. WHO 2016 diagnosis included 26 (3.7%) cases of MDS with single lineage dysplasia (MDS-SLD)/MDS with multilineage dysplasia (MDS-MLD)/MDS with ringed sideroblasts and MLD (MDS-RS-MLD), 112 (16.3%) cases of MDS with excess blasts (MD-EB-1), 371 (54%) cases of MD-EB-2, one case with MDS unclassifiable (MDS-U), one case with MDS with isolated del(5q) and 176 (25.6%) cases of AML (with BM blasts 20–30%). The IPSS and IPSS-R scores were determined for all patients included in the current study, while the FPSS score could be determined for 596 out of 687 patients. According to IPSS, 408 (59.4%) patients were classified in the intermediate-2 risk group and 279 (40.6%) patients in the high-risk group. In addition, according to IPSS-R, 86 (12.5%) patients were categorized in the intermediate risk group, 303 (44.1%) patients in the high-risk group, and 298 (43.4%) patients in the very-high-risk group. Finally, according to FPSS, 53 (8.9%) patients categorized in the low-risk group, 494 (82.9%) patients in the intermediate risk group and 49 (8.2%) in the high-risk group. Patients were treated with a median of seven cycles of 5-AZA administration (interquartile range: 4–15 cycles), with a median time of treatment onset with 5-AZA after diagnosis of 2 months (interquartile range: 1–10 months). The median follow-up time was 28 months.
Characteristics of HR-MDS and oligoblastic AML patients at the treatment onset.

AML, acute myeloid leukemia; CRP, C-reactive protein; EB, excess blasts; ECOG PS, Eastern Cooperative Oncology Group Performance Status; FPSS, French prognostic score; HR-MDS, higher-risk myelodysplastic syndromes; IPSS, International Prognostic Scoring System; IPSS-R, International Prognostic Scoring System (revised); MDS, myelodysplastic syndrome; MLD, multilineage dysplasia; n/a, not available; RBC, red blood cell; RS, ringed sideroblasts; SLD, single lineage dysplasia; U, unclassifiable; WHO, World Health Organization.
Response analysis by the prognostic model risk groups and other baseline characteristics
To determine baseline characteristics that could predict OR, the 625 patients were categorized into overall responders (CR, PR, HI), SD, and nonresponders. Univariate logistic regression was used to assess the predictive power of patient sex, α-2 globulin, gamma globulin, CRP, SF levels, need for platelet transfusion, need for RBC transfusion, ECOG PS, IPSS risk, IPSS-R risk, and FPSS risk. Interestingly, besides IPSS-R (p = 0.022), ECOG PS and SF levels were also shown to predict response to 5-AZA (p = 3.5 × 10–5 and p = 0.003, respectively; Table 2); their predictive power was independent of that one of the IPSS-R risk classification system, as shown in the multivariate logistic regression analysis (Table 2).
Univariate and multivariate logistic regression analyses, for the prediction of response of HR-MDS and oligoblastic AML patients to the treatment with 5-AZA.

Multivariate models were adjusted for ferritin, ECOS PS, and IPSS-R risk at the treatment onset.
OR, estimated from logistic regression model.
CI of the estimated OR.
5-AZA, 5-azacytidine; AML, acute myeloid leukemia; CI, confidence interval; CRP, C-reactive protein; ECOG PS, Eastern Cooperative Oncology Group Performance Status; FPSS, French prognostic score; HR-MDS, higher-risk myelodysplastic syndromes; IPSS, International Prognostic Scoring System; IPSS-R, International Prognostic Scoring System (revised); OR, odds ratio; RBC, red blood cell.
The number of cycles of treatment with 5-AZA was negatively correlated with ECOG PS, as shown by Spearman correlation (rs × = × –0.16, p × = × 3.0 × 10–4). After having excluded those patients that had failed to complete four cycles of treatment with 5-AZA, we performed again logistic regression analysis. In this group of patients, ECOG PS was the unique significant predictor of response to 5-AZA (p = 4.0 × 10–6; Supplemental Table 2).
Survival analysis
As shown in the univariate Cox regression analysis for OS (Table 3), besides IPSS (p = 4.0 × 10–7), IPSS-R (p = 6.3 × 10–14), and FPSS (p = 2.4 × 10–15) risk classification systems, the need for RBC transfusion (p = 3.3 × 10–6), ECOG PS (p = 1.2 × 10–7), and SF levels (p = 1.7 × 10–5) proved to be significant prognosticators. Kaplan–Meier survival analysis confirmed the strong prognostic significance of these variables, particularly with regard to OS [Figure 1(a and b), and Supplemental Figure 1]. Figure 1(c and d), and Supplemental Figure 2 depict the prognostic significance of the same factors for LFS. Interestingly, the SF levels are also a strong prognosticator among patients without any need for RBC transfusion, strongly predicting LFS (p = 6.0 × 10–6), as shown by stratified Kaplan–Meier survival analysis [Supplemental Figure 3(a)]; moreover, they show a trend of predicting LFS in patients needing RBC transfusion, as well [p = 0.060; Supplemental Figure 3(b)]. Regarding OS, the SF levels possess significant prognostic value in both subgroups of patients (i.e. patients without any need for RBC transfusion and those needing for RBC transfusion), as shown by stratified Kaplan–Meier survival analysis [p = 0.011 and p = 0.005; Supplemental Figure 3(c and d)].
Cox regression analyses regarding the overall survival of HR-MDS and oligoblastic AML patients.
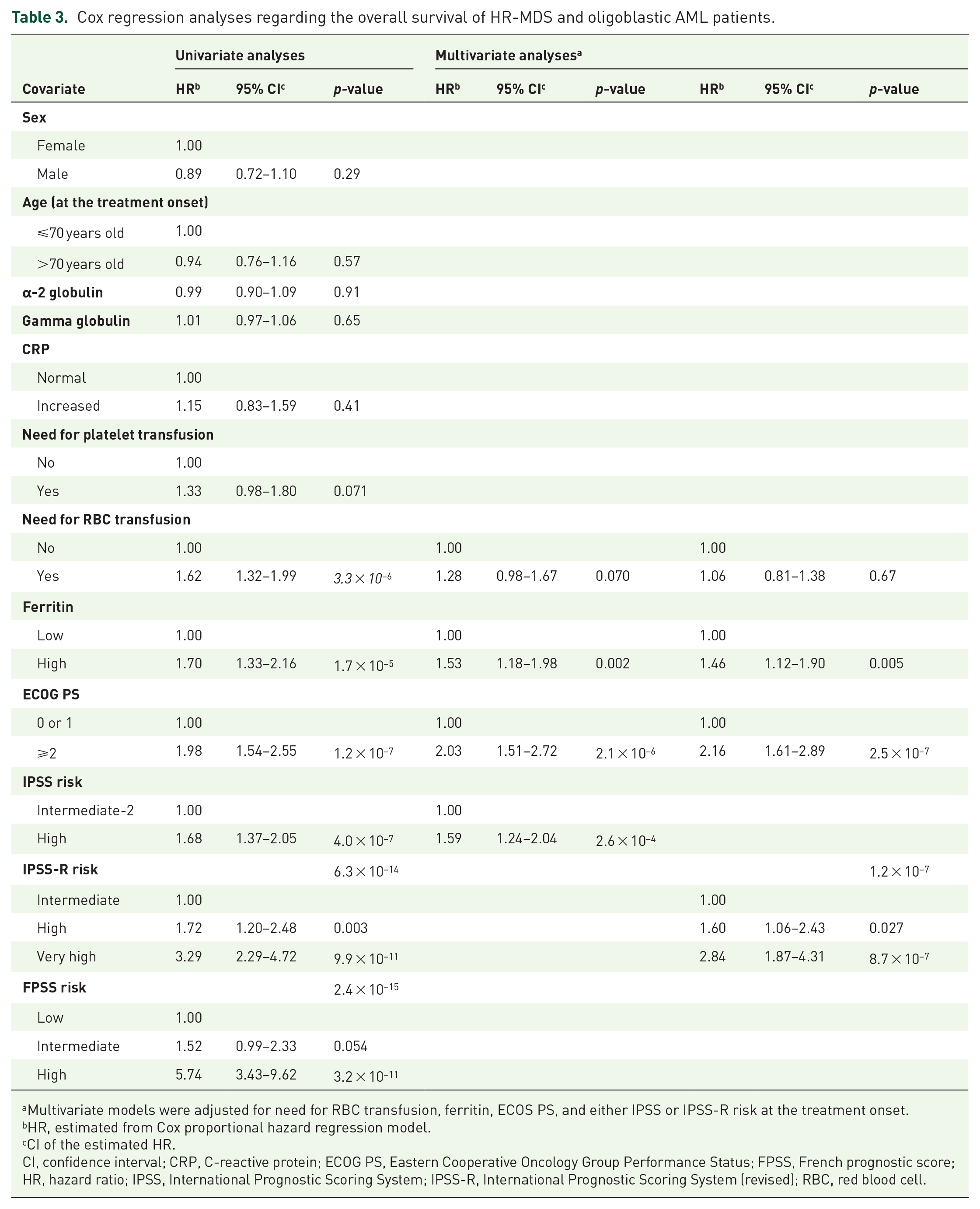
Multivariate models were adjusted for need for RBC transfusion, ferritin, ECOS PS, and either IPSS or IPSS-R risk at the treatment onset.
HR, estimated from Cox proportional hazard regression model.
CI of the estimated HR.
CI, confidence interval; CRP, C-reactive protein; ECOG PS, Eastern Cooperative Oncology Group Performance Status; FPSS, French prognostic score; HR, hazard ratio; IPSS, International Prognostic Scoring System; IPSS-R, International Prognostic Scoring System (revised); RBC, red blood cell.

Kaplan–Meier OS and LFS curves, showing the prognostic significance of ECOG PS and SF levels in patients with higher-risk myelodysplastic syndromes and oligoblastic AML, treated with 5-azacytidine. Poor ECOG PS (⩾2) and high SF levels (⩾520 ng/ml) predict unfavorable [(a) and (b), respectively] and inferior LFS [(c) and (d), respectively]. Total number of patients of each cohort and number of events are shown.
Multivariate Cox regression models for OS, adjusted for the need for RBC transfusion, ECOG PS, SF levels, and either IPSS or IPSS-R risk classification showed the independent prognostic value of ECOG PS and SF levels besides that of the two risk classification systems (Table 3). Similar results were found in the univariate Cox regression analysis for LFS (Table 4). More specifically, the need for RBC transfusion (p = 0.006), ECOG PS (p = 0.012), and SF levels (p = 2.2 × 10–5) were shown to strongly predict LFS. Multivariate Cox regression models for LFS, adjusted for the need for RBC transfusion, ECOG PS, SF levels and either IPSS or IPSS-R risk classification showed the marginal, independent prognostic significance of ECOG PS and SF levels besides that of the two risk classification systems (Table 4).
Cox regression analyses regarding the leukemia-free survival of HR-MDS and oligoblastic AML patients.
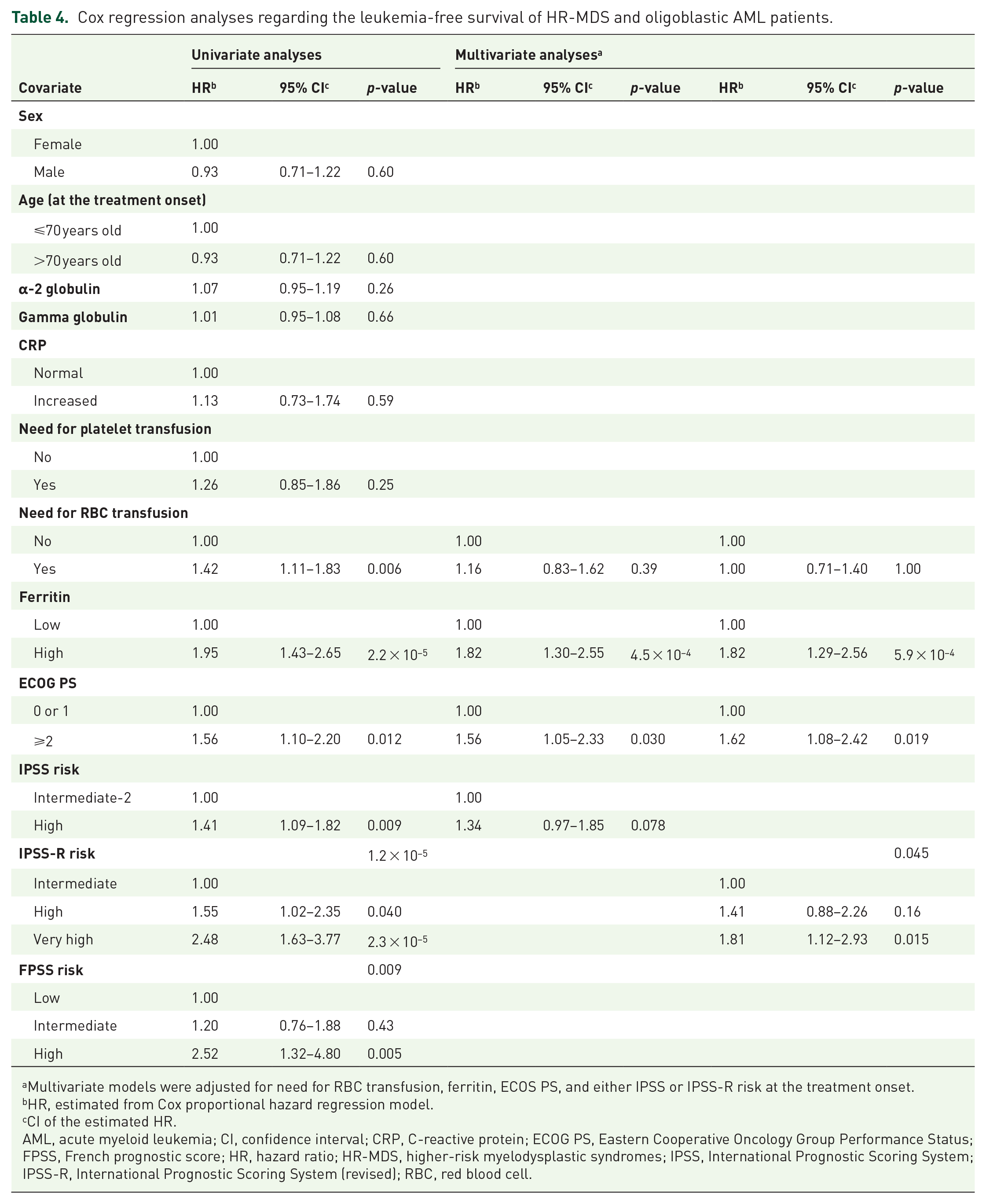
Multivariate models were adjusted for need for RBC transfusion, ferritin, ECOS PS, and either IPSS or IPSS-R risk at the treatment onset.
HR, estimated from Cox proportional hazard regression model.
CI of the estimated HR.
AML, acute myeloid leukemia; CI, confidence interval; CRP, C-reactive protein; ECOG PS, Eastern Cooperative Oncology Group Performance Status; FPSS, French prognostic score; HR, hazard ratio; HR-MDS, higher-risk myelodysplastic syndromes; IPSS, International Prognostic Scoring System; IPSS-R, International Prognostic Scoring System (revised); RBC, red blood cell.
Next, we built new multivariate models for OS and LFS, incorporating only ECOG PS and SF levels besides IPSS or IPSS-R risk classification systems (Table 5). Since all variables included in these models possess an independent prognostic value, we built two modified risk classifications systems, based on either IPSS or IPSS-R.
Multivariate Cox regression analyses for selected variables, with regard to the leukemia-free survival and overall survival of HR-MDS and oligoblastic AML patients.

HR, estimated from Cox proportional hazard regression model.
CI of the estimated HR.
Multivariate models were adjusted for need for ferritin, ECOS PS, and IPSS risk at the treatment onset.
Multivariate models were adjusted for need for ferritin, ECOS PS, and IPSS-R risk at the treatment onset.
AML, acute myeloid leukemia; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; HR-MDS, higher-risk myelodysplastic syndromes; IPSS, International Prognostic Scoring System; IPSS-R, International Prognostic Scoring System (revised).
New, H-IPSS and H-PSS-R risk classification systems
Patients with either intermediate-2 or high-IPSS-risk MDS (or oligoblastic AML) were classified into three new categories of the H-PSS risk, namely low, intermediate, and high H-PSS risk. Thus, patients with intermediate-2 IPSS risk, normal SF levels and ECOG PS = 0 or 1 were classified into the low-H-PSS-risk group; patients with high IPSS risk, increased SF levels and ECOG PS ⩾ 2 were classified into the high-H-PSS-risk group; all other intermediate-2 and/or high-IPSS-risk patients were categorized in the intermediate H-PSS risk group.
In addition, patients with either intermediate, high, or very-high IPSS-R risk were classified into three new categories of the H-PSS-R risk, namely low, intermediate, and high H-PSS-R risk. Thus, patients with intermediate IPSS-R risk along with ECOG PS = 0 or 1, as well as those with high IPSS-R risk with normal SF level and ECOG PS = 0 or 1 were classified into the low H-PSS-R risk group; patients with very high IPSS-R risk along with ECOG PS ⩾ 2, as well as those with high IPSS-R risk with increased SF levels and ECOG PS ⩾ 2 were classified into the high-H-PSS-R-risk group; all other intermediate-2, high and/or very-high-IPSS-R-risk patients were categorized in the intermediate H-PSS-R-risk group.
Prognostic significance of the new, H-PSS and H-PSS-R risk classification systems
The estimated median OS was significantly different among the three distinct prognostic groups that were generated [Figure 2(a)], based on the H-PSS risk classification system (p = 5.8 × 10–14). Thus, the low H-PSS group of patients had an estimated median OS time of 31 months (95% CI = 23–39), whereas the intermediate H-PSS group had an estimated median OS time of 16 months (95% CI = 13–19) and the high H-PSS-risk group had an estimated median OS time of 7 months (95% CI = 3–11). Similarly, Kaplan–Meier survival analysis [Figure 2(b)] revealed that the estimated OS differed among the three distinct prognostic groups, generated based on the H-PSS-R risk classification system (p = 1.3 × 10–21). Thus, the low-H-PSS-R group of patients had an estimated median OS time of 33 months (95% CI = 27–39), whereas the intermediate-H-PSS-R group had an estimated median OS time of 16 months (95% CI = 13–19) and the high-H-PSS-R-risk group had an estimated median OS time of 8 months (95% CI = 5–11).

Kaplan–Meier survival curves, showing the prognostic significance of H-PSS and H-PSS-R risk classification systems in patients with higher-risk myelodysplastic syndromes and oligoblastic AML, treated with 5-azacytidine. Both H-PSS and H-PSS-R have very high prognostic significance, with regard to OS [(a) and (b), respectively] and LFS [(c) and (d), respectively]. Total number of patients of each cohort and number of events are shown.
With regard to LFS [Figure 2(c)], the estimated median was significantly different among the three distinct prognostic groups that were generated based on the H-PSS risk classification system (p = 2.2 × 10–6). Thus, the low H-PSS group of patients had an estimated median LFS time of 30 months (95% CI = 24–36), whereas the intermediate H-PSS group had an estimated median LFS time of 20 months (95% CI = 17–23) and the high H-PSS risk group had an estimated median LFS time of 7 months (95% CI = 5–9). Similarly, Kaplan–Meier survival analysis [Figure 2(d)] revealed that the estimated LFS differed among the three distinct prognostic groups, generated based on the H-PSS-R risk classification system (p = 7.6 × 10–7). Thus, the low H-PSS-R group of patients had an estimated median LFS time of 31 months (95% CI = 23–39), whereas the intermediate H-PSS-R group had an estimated median LFS time of 17 months (95% CI = 14–20) and the high H-PSS-R risk group had an estimated median LFS time of 11 months (95% CI = 8–14).
Comparison between survival prediction power of the new, H-PSS and H-PSS-R classification systems, and the IPSS, IPSS-R, and FPSS
According to the AIC test results, the H-PSS is a better survival discriminator compared to the original IPSS risk classification system. The IPSS-R and FPSS seem to be even better OS discriminators, while the most powerful OS predictor is the new, modified IPSS-R system, since it possesses the lowest AIC value among all these prognostic models. The C-statistic results further supported the conclusion that the new, modified IPSS-R system is superior to IPSS, IPSS-R, and FPSS, in terms of predicting OS, being higher in the first case (Concordance Index C for H-PSS-R: 0.620; for H-PSS: 0.570; for IPSS: 0.550; for IPSS-R: 0.584; and for FPSS: 0.564).
Discussion
In this large cohort of HR-MDS and oligoblastic AML treated with 5-AZA, we have demonstrated that SF levels < 520 ng/mL, ECOG PS 0 or 1 and IPSS-R independently predicted better response to 5-AZA. In addition, poor ECOG PS (⩾2), SF levels > 520 ng/ml and IPSS or IPSS-R independently predicted shortened LFS and OS. Those factors could be combined in a simple prognostic score defining three patient subsets with significantly different LFS and OS. Moreover, the modified scores proved to be better predictors of OS than their previous counterparts as well as the FPSS in our patient cohort.
Increased SF levels although usually associated with transfusion overload, can also be observed in non-transfused MDS patients. This phenomenon has been attributed mainly to ineffective erythropoiesis and increased gut absorption.17,18 Furthermore, iron overload leads to increased oxidative stress causing genomic instability. 19 Increased SF levels at diagnosis have been associated with worse prognosis in MDS patients.20–22 The same finding has also been observed in a small retrospective study of 48 patients with HR-MDS and oligoblastic AML treated with 5-AZA. They found that patients with SF level < 725 ng/mL had significantly better OS (57% at 2 years) than those with SF level ⩾ 725 ng/mL (28% at 2 years). 23 Moreover, our group recently identified the adverse prognostic value of the high SF levels in patients with chronic myelomonocytic leukemia. 24 Of note, ferritin cutoff levels have been inconsistent throughout these studies. Even though retrospective in nature, our trial is the largest to date to explore the effects of SF levels in patients with HR-MDS and oligoblastic AML treated with 5-AZA. We demonstrated that elevated SF levels (>520 ng/ml), along with ECOG PS ⩾ 2 and IPSS-R independently predict poor response to 5-AZA treatment and dismal LFS and OS. More importantly, we showed that the prognostic value of SF levels was independent of RBC transfusion need as well as of inflammatory markers such as CRP, α-2 globulin, and gamma globulin values. Response to 5-AZA treatment, which is observed in a considerable proportion of HR-MDS patients can be, at least partly, attributed to the mitigation of ineffective erythropoiesis in this patient group. Since elevated SF is a consequence of ineffective erythropoiesis,18,19 it would be reasonable to speculate that patients with more ineffective erythropoiesis, (as expressed by high SF levels) have inferior response to 5-AZA treatment. However, more studies are needed to confirm this hypothesis. The assumption that high SF levels represent a surrogate marker of inflammation cannot be supported by our findings since other markers like CRP and the α-2 and gamma globulin values were not of prognostic significance. Moreover, there was no correlation between the RBC transfusion overload and SF levels. One alternative explanation about the negative prognostic role of increased SF in patients treated with 5-AZA could be the increased toxic effect of intracellular iron associated with genetic instability and increased risk of leukemic evolution. 19
To date, few studies have investigated the impact of ECOG PS on the prognosis of HR-MDS patients treated with 5-AZA.9,25 In the noninterventional prospective study of 149 patients with HR-MDS, chronic myelomonocytic leukemia and oligoblastic AML by Wehmeyer et al., patients with ECOG PS 0 had significantly longer progression-free survival than patients with ECOG PS 1 (18.4 versus 9.8 months). 25 In the study by Itzykson et al., ECOG PS ⩾ 2, presence of circulating blasts, RBC transfusion dependency ⩾4 units in 8 weeks, and intermediate- or high-risk cytogenetics independently predicted inferior OS and they were consequently incorporated in the FPSS score. 9 However, ECOG PS did not predict response to 5-AZA treatment. In our study, we showed that ECOG PS was a predictor for response as well as for outcome and it was proven to be the only predictor for survival for patients who managed to receive at least four cycles of 5-AZA, which was the mean time to first response in our patient cohort. The incorporation of ECOG PS in the modified H-PSS and H-PSS-R further underlines its prognostic value.
In our study, OR rate was comparable with that observed in the AZA001 study; 8 however, the OS in our patient cohort appeared to be shorter than the OS of the AZA001 analysis (16.0 versus 24.5 months), in line with other real-life 5-AZA studies.9,10,12,26 For instance, similar results were reported by the Spanish group. 10 In 200 MDS patients of all risk groups according to IPSS treated with 5-AZA an estimated median OS of 16.5 months (95% CI = 12.4–19.1 months). The French group reported a median OS of 13.5 months for 282 HR-MDS patients treated with 5-AZA in the French compassionate, patient-named program. 9 This difference in survival outcomes between the randomized trial and real-life studies may reflect the unselected nature of the patients included in real-life analyses compared to the strictly selected patients enrolled in randomized trials. Therefore, a median survival of 16–17 months seems to be more realistic when treating HR-MDS patients with 5-AZA, than the median survival of 24.5 months reported in the AZA-001 trial.
In addition, in the current study we confirmed the prognostic utility of IPSS, IPSS-R and FPSS in our 5-AZA-treated patient population. All the above prognostic systems discriminated patients with different LFS and OS. Our findings agree with the results of other series in terms of OS4,27 and further demonstrate their importance for LFS. Moreover, with the incorporation of ECOG PS and SF levels in the IPSS and the IPSS-R, we developed two new prognostic scores, namely the H-PSS and the H-PSS-R. The two modified prognostic scores could classify patients in three distinct groups with markedly different LFS and OS depending on risk category. In fact, they were proven to be better survival discriminators than their traditional counterparts and the FPSS, while the H-PSS-R was the most powerful among all of them. Both of these new prognostic scores distinguish a subset of patients with very good outcomes after 5-AZA treatment, with low-risk patients achieving an estimated median LFS of 30 months (95% CI: 24–36 months) and 31 months (95% CI: 23–39 months), and estimated median OS of 31 months (95% CI: 23–39 months) and 33 months (95% CI: 27–39 months) for H-PSS and H-PSS-R respectively. More importantly, these two scores identified a group of high-risk patients with a very poor outcome after 5-AZA treatment with an estimated median LFS of 7 months (95% CI: 5–9 months) and 11 months (95% CI: 8–14 months) and estimated OS of 7 months (95% CI: 3–11 months) and 8 months (95% CI: 5–11 months) for H-PSS and H-PSS-R, respectively. These high-risk patients should be strongly considered for allogeneic stem cell transplant, 28 or alternative aggressive approaches and experimental treatments such as 5-AZA-based combinations.29–31
Our study has two main limitations: Firstly, it is retrospective and thus susceptible to selection bias. Secondly, even though the bootstrap analysis performed supports our findings, the current study does not include a validation cohort. Therefore, additional prospective studies are needed to evaluate our findings.
In conclusion, our results suggest that the IPSS-R, ECOG PS and SF levels at baseline can predict response and survival in HR-MDS and oligoblastic AML patients treated with 5-AZA. Moreover, with the incorporation of ECOG PS and SF levels into the IPSS or IPSS-R, we developed two new prognostic scores that recognize a subset of patients with favorable prognosis and long-term survival and a subset of very-high-risk patients who may not benefit from treatment with 5-AZA and may be candidates for alternative therapeutic approaches.
Supplemental Material
sj-pdf-1-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-pdf-1-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-2-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-pdf-2-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-3-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-pdf-3-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-1-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-tif-1-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-2-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-tif-2-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Supplemental Material
sj-tif-3-tah-10.1177_2040620720966121 – Supplemental material for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes
Supplemental material, sj-tif-3-tah-10.1177_2040620720966121 for Serum ferritin and ECOG performance status predict the response and improve the prognostic value of IPSS or IPSS-R in patients with high-risk myelodysplastic syndromes and oligoblastic acute myeloid leukemia treated with 5-azacytidine: a retrospective analysis of the Hellenic national registry of myelodysplastic and hypoplastic syndromes by Sotirios G. Papageorgiou, Ioannis Kotsianidis, Anthi Bouchla, Argyris Symeonidis, Athanasios Galanopoulos, Nora-Athina Viniou, Eleftheria Hatzimichael, Theodoros P. Vassilakopoulos, Dimitrios Gogos, Aikaterini Megalakaki, Panagiotis Zikos, Panagiotis Diamantopoulos, Alexandra Kourakli, Panagiota Giannoulia, Menelaos Papoutselis, Elias Poulakidas, Maria Arapaki, Anna Vardi, Achilles Anagnostopoulos, Despoina Mparmparousi, Maria Papaioannou, Eleni Bouronikou, Maria Dimou, Helen Papadaki, Panayiotis Panayiotidis and Vasiliki Pappa in Therapeutic Advances in Hematology
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
