Abstract
BAY 81-8973 (Kovaltry®) is an unmodified, full-length recombinant factor VIII (rFVIII) approved for the prevention and treatment of bleeding episodes in patients with hemophilia A. The amino acid sequence for BAY 81-8973 is identical to that of sucrose-formulated rFVIII (rFVIII-FS; Kogenate® FS/KOGENATE®, Bayer), but the two products differ in their manufacturing approaches. The manufacture of BAY 81-8973 includes several modifications and enhancements, such as the introduction of the gene for human heat shock protein 70, a molecular chaperone protein that facilitates folding of proteins; no addition of human- or animal-derived proteins in the cell culture, purification process, or final formulation; and use of a 20-nm filter to remove any potential aggregates and pathogens. BAY 81-8973 was extensively studied in the LEOPOLD clinical development program, which enrolled participants of all age groups (children, adolescents, and adults) with severe hemophilia A. The pharmacokinetic profile of BAY 81-8973 was shown to be noninferior to, and for some variables more favorable than, rFVIII-FS and another commercial full-length rFVIII product. BAY 81-8973 was shown to be efficacious when used for prophylaxis, on-demand treatment, and perioperative hemostasis. The efficacious prophylaxis dose of BAY 81-8973 was approximately 20–40 IU/kg given two or three times per week, which achieved low annualized bleeding rates. Either the one-stage or the chromogenic assay provides accurate measurements for postinfusion monitoring of BAY 81-8973 levels, with no product-specific calibration standard needed. The incidence of treatment-related adverse events was ⩽7% across all LEOPOLD studies, and no previously treated patient developed anti-BAY 81-8973 inhibitors in the completed primary studies.
Introduction
Hemophilia A is a congenital bleeding disorder resulting from deficiency of clotting factor VIII (FVIII). The clinical severity of hemophilia A correlates with plasma FVIII concentration (FVIII:C). In phenotypically mild hemophilia A, FVIII:C is >5 to <40 IU/dl; in moderate hemophilia A, FVIII:C is 1–5 IU/dl; and in severe hemophilia A, FVIII:C is <1 IU/dl. 1 Patients with severe hemophilia A bleed spontaneously, and joints are the most common site of bleeding. 1 Bleeding also occurs into other sites, including muscles and subcutaneous tissues. Recurrent bleeding into joints can lead to progressive and irreversible hemophilic arthropathy. 1
Prophylaxis is the recommended standard of care, is effective in preventing bleeding episodes in patients of all ages,2,3 and can prevent joint damage if started at a young age (usually before the first joint bleeding episode). 3 The FVIII half-life of 8–12 h requires infusions typically three times per week for effective prophylaxis treatment, 1 a dosing frequency that may have a negative effect on patient adherence.4,5 Newer modified FVIII products with an extended half-life may allow less frequent prophylaxis dosing and increase acceptance of and adherence to prophylaxis.
Replacement FVIII products are either plasma-derived or recombinant. 1 Plasma-derived FVIII products are derived from pooled plasma donations; recombinant FVIII (rFVIII) products are created using recombinant DNA techniques. 6 rFVIII products became available in the early 1990s; the first rFVIII products used human serum albumin as a stabilizer, raising concerns about possible pathogen transmission. 6 Advances in rFVIII development led to replacement of albumin with sucrose as a stabilizer. 6 The new-generation rFVIII products are devoid of human- or animal-derived proteins in all stages of manufacture.7–13 In the formulation of rFVIII, the FVIII moiety can be either full length9,11 or a B-domain-deleted FVIII protein.7,8,10–13
BAY 81-8973 (Kovaltry®, Bayer, Berkeley, CA, USA) is an unmodified full-length recombinant human FVIII approved in 2016 for on-demand treatment, prophylaxis, and perioperative management of patients with hemophilia A.9,14 BAY 81-8973 builds on the history of Bayer’s rFVIII products for hemophilia A, which have demonstrated long-term efficacy and safety in clinical trials and real-world settings.2,3,15–20 BAY 81-8973 has an amino acid sequence identical to that of sucrose-formulated rFVIII (rFVIII-FS; Kogenate® FS, Bayer, Whippany, NJ, USA), 15 but is manufactured using innovative techniques.
BAY 81-8973 pharmacokinetics (PK) are noninferior to, and for some PK variables significantly better than, those for rFVIII-FS 15 and another commercial unmodified rFVIII [antihemophilic factor (recombinant) plasma/albumin-free method (rAHF-PFM; Advate®, Baxalta, Westlake Village, CA, USA)] 21 in terms of higher area under the curve (AUC), prolonged half-life and mean residence time after intravenous injection, and slower clearance (all exploratory p values <0.02). In clinical trials, BAY 81-8973 effectively prevented and treated bleeding episodes in patients of all ages22–24 with both a twice-weekly and three-times-weekly dosing regimen. 24 This review describes advances made in the BAY 81-8973 manufacturing process and provides a comprehensive overview of results from the BAY 81-8973 Long-Term Open-Label Program in Severe Hemophilia A Disease (LEOPOLD) clinical development program.
BAY 81-8973 manufacturing
The BAY 81-8973 manufacturing process is similar to that of rFVIII-FS, with several modifications and enhancements. These changes were made to take advantage of the latest manufacturing technologies, eliminate use of human- and animal-derived raw materials in the manufacturing process, further enhance pathogen safety, and optimize production by reducing the number of manufacturing sttif. 25 Mammalian cell lines are used for expression of rFVIII because of the large size and high degree of glycosylation of the FVIII molecule. 26 BAY 81-8973 and rFVIII-FS use the same cell expression system [baby hamster kidney (BHK) cells]. An important distinction in manufacture of BAY 81-8973 is the introduction of the human heat shock protein 70 (HSP70) gene in the production cell line, a novel, patented (US Patent No: US 2005/0048608 AI) enhancement step. The HSP70 is an intracellular molecular chaperone expressed in response to cellular stress 27 that plays an essential role in ensuring proper folding of newly formed proteins 28 and refolding of misfolded or aggregated proteins. 29 In BAY 81-8973 manufacturing, HSP70 increases FVIII expression and improves the productivity of the cell bank, 25 provides quality control, and inhibits apoptosis, which might enhance rFVIII expression and yield a product with high and consistent purity. 27 No HSP70 is detectable in the cell culture harvest or final product. 27 The BAY 81-8973 manufacturing process was further enhanced through use of continuous cell separation and membrane-based ion exchange chromatography that rapidly capture and concentrate rFVIII from the cell culture harvest. 25
No human- or animal-derived raw materials are added to the cell culture medium used for BAY 81-8973, 25 reducing the theoretical risk and potential of BAY 81-8973 to transmit pathogens through such additives. Nanofiltration is an additional measure added in the manufacturing process that reduces the theoretical risk of pathogen transmission. BAY 81-8973 is passed through a 20-nm pore-size viral filter, which is capable of removing viruses, including parvovirus, and transmissible spongiform encephalopathy agents. 25 The 20-nm filter also captures and removes any potential protein aggregates. 25
The BAY 81-8973 purification process includes immunoaffinity, metal affinity, and ion exchange chromatography sttif. 25 The BAY 81-8973 manufacturing process results in a rFVIII product of enhanced purity, with a consistently high degree of sialylation of N-terminal glycans on the molecular surface. 25 Compared with rFVIII-FS, BAY 81-8973 has glycans shifted to higher branched structures and somewhat higher sialylation (Figure 1); these changes may contribute to the improved PK, including slower clearance, higher AUC, and longer half-life, seen with BAY 81-8973.15,23,30 Potency labeling is based on the chromogenic assay.

Glycan structures in BAY 81-8973 and rFVIII-FS.
BAY 81-8973 clinical evaluation
BAY 81-8973 has been extensively studied in the LEOPOLD clinical development program, comprising three open-label trials in patients with severe hemophilia A. These were multicenter, multinational studies conducted in compliance with International Conference on Harmonisation Good Clinical Practice guidelines, regulatory requirements, and study-specific protocols. The studies were LEOPOLD I parts A, B, C, and extension; LEOPOLD II; and LEOPOLD Kids parts A, B, and extension (Figure 2).22–24 In addition to the LEOPOLD studies, a comparative PK study was conducted. 21

Patients eligible for LEOPOLD I and II were males aged 12–65 years with severe hemophilia A (endogenous FVIII <1%), ⩾150 exposure days (EDs) to any FVIII product, and no history of or current FVIII inhibitors. 23 LEOPOLD I part A was a phase I trial to assess and compare BAY 81-8973 PK with that of rFVIII-FS. 23 LEOPOLD I part B was a phase III efficacy and safety study of BAY 81-8973 prophylaxis. 23 LEOPOLD I part C evaluated efficacy and safety in patients undergoing major surgery. 23 The LEOPOLD I extension assessed the long-term efficacy and safety of BAY 81-8973 over a treatment period of up to 2 years.
LEOPOLD II was a randomized phase II/III study designed to demonstrate the superiority of BAY 81-8973 prophylaxis over on-demand treatment. 24 Patients eligible for LEOPOLD II were currently being treated on demand and had not received regular prophylaxis for >6 months in the previous 5 years.
LEOPOLD Kids part A, a phase III trial, was conducted to evaluate the efficacy and safety of BAY 81-8973 for prophylaxis and treatment of bleeds in pediatric previously treated patients (PTPs). 22 Boys aged <12 years with severe hemophilia A could enroll in LEOPOLD Kids part A if they had ⩾50 EDs to any FVIII product and no history of or current FVIII inhibitors.
The additional study was conducted to compare the PK of BAY 81-8973 with those of another commercial rFVIII product (rAHF-PFM). 21
Potency labeling for BAY 81-8973 is based on the chromogenic substrate assay per European Pharmacopoeia (CS/EP).9,14 To demonstrate the efficacy of BAY 81-8973 regardless of the assay used to assign potency (either the one-stage or chromogenic assay), LEOPOLD I part B and LEOPOLD II incorporated a direct intraindividual comparison of annualized bleeding rate (ABR) during treatment periods with dosing based on the CS/EP assay and dosing based on a chromogenic substrate assay with the BAY 81-8973 vial label adjusted to mimic potency results that would be obtained with the one-stage assay (CS/ADJ).23,24 This comparison was made to determine whether dosing based on the assay used for potency labeling affected BAY 81-8973 efficacy, given the estimated differences of approximately 20% in FVIII potency measured using the one-stage assay versus the chromogenic assay. 31 The ABR was analyzed for each of the randomized, intraindividual, crossover, 6-month treatment periods. No difference in efficacy was demonstrated in this comparison, showing that the difference in potency labeling had no effect on clinical efficacy.23,24,32
Pharmacokinetics
Results from the 26 patients [mean (range) age, 29.5 (12–61) years] in LEOPOLD I with evaluable PK data demonstrated that the PK of BAY 81-8973 was noninferior to, and for some variables superior to, those for rFVIII-FS (Table 1).15,23,33 Compared with rFVIII-FS, BAY 81-8973 had a significantly longer half-life (geometric mean, 12.2 versus 13.4 h; p = 0.011), higher AUC (1175.7 versus 1397.5 IU·h/dl; p < 0.0001), and slower clearance (0.043 versus 0.036 dl/h/kg; p < 0.0001) based on the one-stage assay; similar differences were seen for PK parameters based on the chromogenic assay.15,23
AUC, area under the curve; CL, clearance; MRTIV, mean residence time after intravenous injection; rFVIII-FS, sucrose-formulated recombinant factor VIII; Vss, volume of distribution at steady state.
Data are geometric mean (%CV).
In a subset of 19 pediatric patients aged <12 years enrolled in LEOPOLD Kids part A, BAY 81-8973 values for maximum concentration and AUC in children tended to be lower than those for adults; 15 half-life was consistently similar for younger and older patients (11.8 h for ages 0 to <6 years and 11.0 h for ages 6 to <12 years). 22
Additional PK data were derived from a population PK (popPK) model based on all available FVIII measurements from all LEOPOLD trials, including several preinfusion and postinfusion FVIII levels obtained for determining recoveries during the study. 14 Results for the predicted PK parameters for all patients included in the studies from this PK model are shown in Table 2. 14 PK parameters determined using noncompartmental analysis (NCA), which is calculated from a subset of patients with dense PK sampling, and those estimated based on the popPK model for the total population (Table 2) were largely comparable. Higher clearance per kilogram was observed in children (n = 51) versus adults (n = 132) using the popPK approach; similar results were observed using NCA (children, n = 18; adults, n = 26). 34 A key difference between the two analyses was that the geometric mean half-life was higher based on popPK compared with NCA for patients aged <6 years (13.3 versus 11.8 h) and 6 to <12 years (14.1 versus 11.0 h). The shorter half-life observed in children using NCA might reflect limitations of the NCA method with a limited number of sample time points. Although NCA may be sufficient for compounds with monophasic PK profiles, it may have a bias for compounds exhibiting biphasic PK profiles. In children, a limited number of plasma samples were available (before infusion and 30 min, 4 h, and 24 h after infusion); therefore, NCA may not sufficiently differentiate between the short distribution half-life and the terminal (long) half-life. Pharmacokinetic values predicted for a total population based on a robust popPK model may provide a more robust PK description. The popPK model can use structural information of a biphasic concentration–time profile to better describe the terminal half-life and variability in PK within a population. 14
Pharmacokinetic parameters based on population pharmacokinetic modeling in previously treated children, adolescents, and adults with severe hemophilia A.
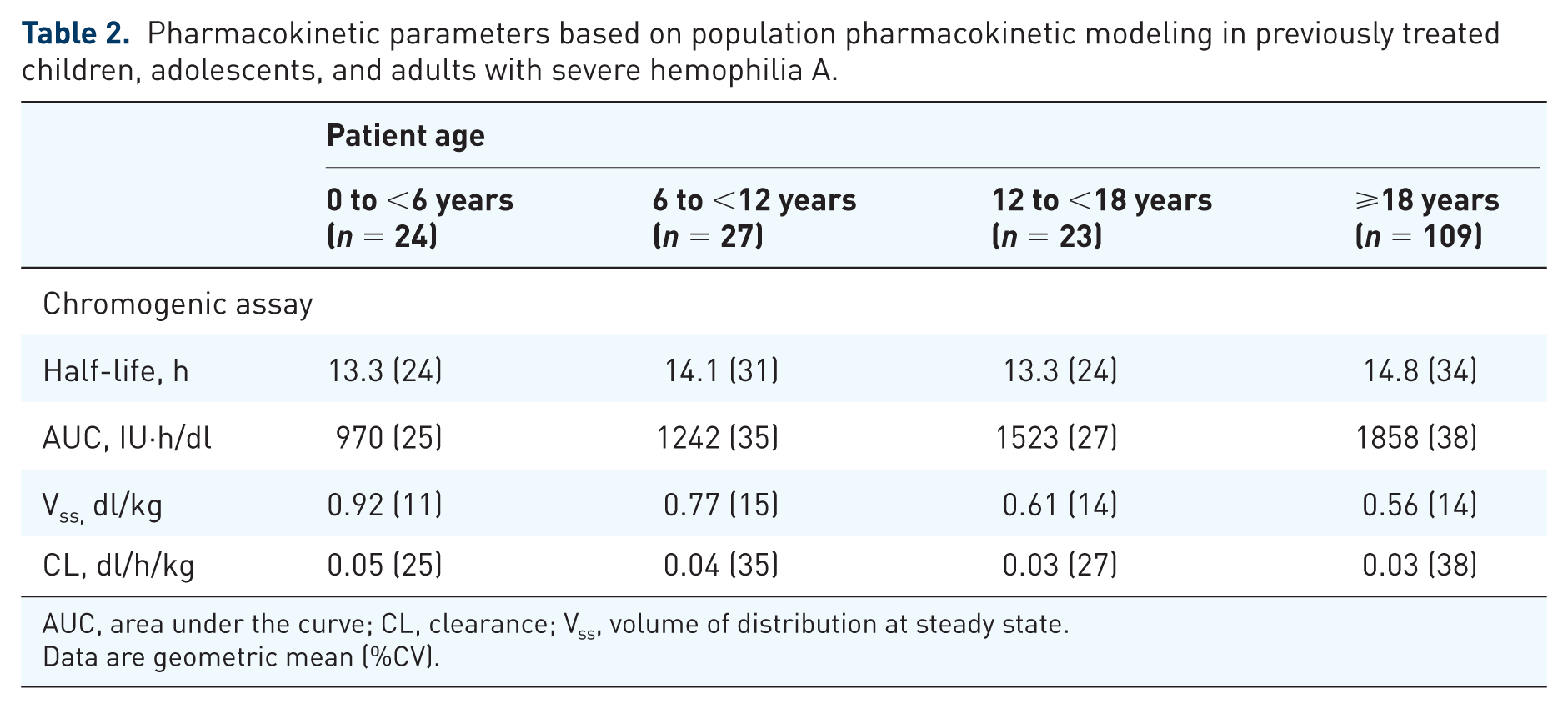
AUC, area under the curve; CL, clearance; Vss, volume of distribution at steady state.
Data are geometric mean (%CV).
In a phase I study separate from the LEOPOLD trials, the PK of BAY 81-8973 was superior to that of rAHF-PFM. 21 In this open-label, randomized, crossover study, 18 male patients aged 18–65 years with severe hemophilia A received single 50 IU/kg infusions of BAY 81-8973 and rAHF-PFM. Compared with rAHF-PFM, BAY 81-8973 had a significantly longer half-life, higher AUC, and slower clearance, whether measured using the one-stage or chromogenic assay (all p < 0.0001; Table 3). 21
Pharmacokinetic results following single-dose administration of 50 IU/kg BAY 81-8973 and rAHF-PFM in previously treated adults with severe hemophilia A.

AUC0–last, area under the curve from time 0 to the last data point; CI, confidence interval; CL, clearance; Cmax, maximum concentration; CV, coefficient of variation; MRT, mean residence time; NA, not applicable; rAHF-PFM, antihemophilic factor (recombinant) plasma/albumin-free method.
Ratio of BAY 81-8973:rAHF-PFM.
Study population
A total of 204 PTPs were enrolled in the LEOPOLD clinical program, which included 73 PTPs in LEOPOLD I, 23 80 PTPs in LEOPOLD II, 24 and 51 PTPs in LEOPOLD Kids part A trials; 22 LEOPOLD Kids part B in previously untreated patients (PUPs) is ongoing. Of the 204 patients, 153 were aged ⩾12 years and 51 were aged <12 years; 140 patients were treated for ⩾12 months, and 43 of these patients were treated for 24 months in the completed trials. 9 Currently, 76 children have been enrolled in the LEOPOLD Kids extension study (including patients from LEOPOLD Kids parts A and B), which started after 6 months of treatment in the main study. This extension study is ongoing.
Efficacy
Prophylaxis
A total of 62 patients [mean (range) age, 31.5 (12–61) years] were enrolled in LEOPOLD I part B. 23 Prophylaxis dosing was at the investigator’s discretion based on the patient’s individual requirements. The proposed regimen per protocol was two or three times per week in a dose range of 20–50 IU/kg; 44 patients (71%) were treated three times per week, and 18 (29%) were treated twice weekly. 23
During 12 months of BAY 81-8973 prophylaxis treatment, no clinically relevant differences in median ABR for total bleeds were seen between the CS/EP and CS/ADJ dosing periods (median ABR, 1.0 for combined CS/EP and CS/ADJ periods). 23 Median total bleeds were 1.0 for twice-weekly dosing (median nominal dose/infusion, 35 IU/kg) and 2.0 for three-times-weekly dosing (median nominal dose/infusion, 31 IU/kg; Table 49,23,24,33). 23 The median prestudy joint bleed ABR of 3.5 decreased to 1.0 during 12 months of BAY 81-8973 prophylaxis (Table 4).23,33 In total, 16 of the 62 patients (26%) in LEOPOLD I had 0 bleeds during the 1-year treatment period, including 6 (33%) in the twice-weekly group and 10 (23%) in the three-times-weekly group.9,35 A similar bleed rate was maintained during the 1-year extension study [median ABR, 2.0 (twice-weekly group) and 1.9 (three-times-weekly group)] with a decrease in median dose per prophylaxis infusion to 29 IU/kg in the twice-weekly dosing group (median weekly dose, 65 IU/kg) and to 31 IU/kg in the three-times-weekly dosing group (median weekly dose, 91 IU/kg).
NA, not applicable; Q1, quartile 1; Q3, quartile 3.
Data in LEOPOLD I are total bleeds (defined as spontaneous and trauma-related bleeds, untreated bleeds, and unspecified bleeds); data in LEOPOLD II are all bleeds (defined as spontaneous bleeds, trauma-related bleeds, untreated bleeds, and unspecified events for which treatment was administered).
LEOPOLD II enrolled 80 patients [mean (range) age, 29.6 (14–59) years]; patients were randomized to one of the three treatment arms: on demand (n = 21); twice-weekly lower-dose prophylaxis (20–30 IU/kg; n = 28); or three-times-weekly higher-dose prophylaxis (30–40 IU/kg; n = 31). 24
During the 12 months of treatment, median ABR for all bleeds was lower in both prophylaxis groups combined compared with on-demand treatment (2.0 versus 60.0; Table 4), confirming the superiority of prophylaxis over on-demand treatment for both prophylaxis regimens. 24 As in LEOPOLD I part B, no clinically meaningful differences in bleeding frequency were seen between the CS/EP and CS/ADJ potency periods for both prophylaxis regimens. 24 During treatment months 1 through 6, median ABR for all bleeds was 4.1 for twice-weekly dosing and 2.0 for three-times-weekly dosing; during treatment months 7 through 12, median ABR for twice-weekly dosing considerably decreased and was similar to that for three-times-weekly dosing (median, 1.1 versus 2.0, respectively). 24 Median number of joint bleeds, spontaneous bleeds, and trauma-related bleeds were all considerably lower with prophylaxis than with on-demand treatment (Table 4). 24 Overall, during the 1-year treatment period, 16 of the 59 prophylaxis patients (27%) in LEOPOLD II had 0 bleeds, including 8 (29%) in the twice-weekly group and 8 (26%) in the three-times-weekly group. 9
In a post hoc analysis of pooled data from LEOPOLD I and LEOPOLD II, no significant differences were seen between twice-weekly and three-times-weekly dosing in the percentage of patients considered good or moderate responders (defined as having ⩽4 joint bleeds per year; 72% and 84%, respectively, for LEOPOLD I and 61% and 77%, respectively, for LEOPOLD II). 35
The noninferiority of CS/EP versus CS/ADJ dosing was confirmed in a pooled analysis of data from the per-protocol prophylaxis groups in LEOPOLD I and II (n = 118). 32 The Hodges–Lehmann estimate for median intraindividual difference of ABR between the CS/EP and CS/ADJ potency periods (CS/ADJ minus CS/EP) was −0.012 bleeds per year, with a lower limit of the one-sided 95% CI of −1.038 bleeds per year. 32 The noninferiority of CS/EP versus CS/ADJ dosing was statistically demonstrated because the lower limit was above the predefined noninferiority margin of −1.5 bleeds per year, 32 which was considered a clinically meaningful difference in a yearly bleeding rate. Based on results from LEOPOLD I and II and the pooled analysis, use of the chromogenic assay for potency labeling does not affect the clinical efficacy of BAY 81-8973.23,24,32
LEOPOLD Kids part A enrolled 51 PTPs aged <12 years (0 to <6 years, n = 25; 6 to <12 years, n = 26). 22 Prophylaxis dosing was determined by the investigator based on the patient’s individual requirements. 22 At study start, 22 patients were treated twice weekly and 29 were treated three times per week or every other day. 22
Median ABR for total bleeds occurring within 48 h after a prophylaxis infusion (primary endpoint) was 0 for all patients, 1.9 for patients aged 0 to <6 years, and 0 for patients aged 6 to <12 years (Table 59,33). 22 Median ABR for total bleeds independent of time since infusion was 1.9 for all patients, 2.0 for patients aged 0 to <6 years, and 0.9 for patients aged 6 to <12 years (Table 5). 22 Median ABR for joint bleeds was 0 for all patients (Table 5). 22 In total, 23 of the 51 patients (45.1%) in LEOPOLD Kids part A had 0 bleeds during the approximately 6-month treatment period, including 10 (40.0%) in the 0 to <6 years age group and 13 (50.0%) in the 6 to <12 years age group, demonstrating the efficacy of BAY 81-8973 in the previously treated pediatric population. 9
Q1, quartile 1; Q3, quartile 3.
Treatment of bleeds
In LEOPOLD I, 45 of the 62 patients experienced 236 bleeding episodes during the 1-year treatment period. 23 Response to treatment, available for 235 bleeds, was rated as good or excellent for 81% of the bleeding episodes; 9 86% of treated bleeds were controlled with ⩽2 infusions. 23
In LEOPOLD II, 293 bleeds occurred in the combined prophylaxis group and 1204 in the on-demand group. 24 Response to treatment, available for 279 bleeds in the prophylaxis groups and 1196 bleeds in the on-demand group, was rated as good or excellent by 62% of patients in the prophylaxis groups and 70% of those in the on-demand group; 9 96% of bleeds in the prophylaxis groups and 95% of bleeds in the on-demand group were controlled with ⩽2 infusions. 24
In LEOPOLD Kids part A, 97 bleeding episodes occurred. 22 Response to treatment, available for 81 treated bleeds, was rated as good or excellent in 90% of cases (98% for patients aged 0 to <6 years; 81% for patients aged 6 to <12 years), and 90% of bleeding episodes were successfully treated with ⩽2 infusions. 22 Part B in PUPs is ongoing.
Perioperative management
In LEOPOLD I, 10 patients underwent 12 major surgeries and 18 patients underwent 26 minor surgeries. 36 In LEOPOLD II, 1 patient underwent a single major surgery and 14 patients underwent 20 minor surgeries. 36 A total of 7 of the 13 major surgeries were orthopedic procedures; 28 of the 46 minor surgeries were dental procedures. 36 Perioperative blood loss during major surgeries was within expected ranges; hemostasis was assessed as good or excellent in all major and minor surgeries. 36
In LEOPOLD Kids part A, 1 patient underwent major surgery (multiple tooth extractions); hemostasis during this procedure was assessed as good. 36 No minor surgeries were performed during LEOPOLD Kids part A. 36
Influence of patient characteristics on BAY 81-8973 efficacy
A post hoc analysis of LEOPOLD I data was conducted to evaluate the effect of baseline patient characteristics on efficacy outcomes. 37 Median ABR was lower in patients aged ⩾18 years versus those aged 12–17 years, those previously treated with prophylaxis rather than on demand, those aged ⩽1 year when starting treatment, those with ⩽6 total bleeding episodes or ⩽3 joint bleeding episodes in the preceding 12 months, and those with a baseline Gilbert total score ⩽17. 37 A low bleeding rate before study entry was a good predictor of low ABR during the study. 37
In analysis of data from patients treated twice weekly in LEOPOLD I, compared with patients who had ⩾3 bleeding episodes during the study, those with 0 bleeds were significantly older (mean age, 27.8 versus 49.2 years; p = 0.04) and had significantly fewer bleeds in the 12 months before study entry (mean, 11.0 versus 0.2; p = 0.02). 35
A post hoc analysis of pooled LEOPOLD I and LEOPOLD II data found that baseline von Willebrand factor antigen (VWF:Ag) levels considerably influenced BAY 81-8973 PK and clinical outcomes.33,38 Baseline VWF:Ag levels were higher in patients with infrequent bleeding episodes (defined as ⩽1 joint bleed per year) during the LEOPOLD studies versus those who frequently had bleeding episodes (⩾5 joint bleeds per year; mean VWF:Ag level, 127.4 IU/dl versus 99.0 IU/dl; p = 0.049). 33 In patients treated with twice-weekly prophylaxis, high VWF:Ag levels (⩾120 IU/dl) were significantly correlated with low joint bleeding rates (correlation coefficient, r = −0.689; p < 0.0001); patients with VWF:Ag levels <120 IU/dl had a mean joint ABR of 6.2 compared with 0.3 for those with a VWF:Ag >120 IU/dl. This association was not seen in the three-times-weekly prophylaxis group. 33 In general, BAY 81-8973 PK, using the one-stage assay, was significantly better in patients with baseline VWF:Ag levels ⩾120 IU/dl versus those with levels <120 IU/dl (half-life, 17.9 versus 11.8 h; AUC, 2137 versus 1231 U·h/dl; clearance, 0.024 versus 0.041 dl/h/kg; all p < 0.0001). 38 Higher baseline VWF:Ag levels were associated with older patient age. 33 Factor VIII trough levels at 48 h postinfusion were significantly correlated with VWF:Ag levels (correlation coefficient, r = 0.814; p < 0.0001).
Data from LEOPOLD I were also analyzed to identify patient and treatment characteristics that could be associated with assignment to twice-weekly prophylaxis dosing of BAY 81-8973. 39 Compared with patients assigned by the investigator to three-times-weekly dosing, those assigned to twice-weekly dosing were older (median age, 29 versus 40 years), were more likely to have previously been treated with prophylaxis (77% versus 89% of patients) or had previously been treated twice weekly (5% versus 61%; p < 0.0001), had fewer total bleeding episodes in the previous 12 months (median, 4.5 versus 3.5 for patients previously treated with prophylaxis and 41.5 versus 18 for those previously treated on demand), had fewer joint bleeding episodes in the previous 12 months (median, 4.0 versus 2.0), were less likely to have had recurrent bleeding into the same joint (77% versus 56% of patients), and had a lower baseline Gilbert bleeding score (mean ± SD, 4.2 ± 3.7 versus 1.1 ± 1.3; p = 0.0007).39–41 Patients previously treated only on demand were less likely to be assigned to twice-weekly dosing and more likely to be assigned to three-times-weekly dosing. 40
In a post hoc analysis of data from LEOPOLD Kids, patients were more likely to be assigned to twice-weekly prophylaxis dosing if they had been treated on demand rather than with prophylaxis. 42 No other patient characteristics differed significantly between patients treated twice weekly and those treated more frequently. 42
Recovery and trough levels
In adolescents and adults, median incremental recovery was >2.0 IU/dl per IU/kg, measured using either the one-stage or chromogenic assay.23,24 For pediatric patients, median incremental recovery using the chromogenic assay was 1.6 IU/dl per IU/kg for patients aged 0 to <6 years and 1.8 IU/dl per IU/kg for those aged 6 to <12 years. 22
Because BAY 81-8973 dose and dosing frequency varied within specific ranges in the LEOPOLD trials, FVIII trough levels also varied. Factor VIII trough levels were ⩾1% in ⩾70% of patients at 2.5–3.5 days postinfusion during the LEOPOLD I trial. 23 Data from a popPK model showed that median time to reach FVIII levels of 1% and 3% after a 30 IU/kg infusion of BAY 81-8973 was 81 and 58 h, respectively. 33
Safety
No inhibitors developed in any PTPs in the completed LEOPOLD I, LEOPOLD II, or LEOPOLD Kids part A trials.22–24 In an ongoing extension study, a 13-year-old PTP had a titer of 0.6 Bethesda units after 550 EDs concurrent with an acute infection and positive immunoglobulin G anticardiolipin antibodies. Factor VIII recovery was 2.2 IU/dl per IU/kg, ABR was 0, and no change in treatment was required. Follow-up inhibitor test results were negative in this patient. Inhibitor data in PUPs will be available when the ongoing LEOPOLD Kids part B trial concludes.
The most commonly reported adverse events during BAY 81-8973 treatment were headache, pyrexia, and pruritus, 9 which are similar to those reported in other FVIII replacement studies. The incidence of treatment-related adverse events was ⩽7% across all completed LEOPOLD studies.22–24 The frequency, type, and severity of adverse events in previously treated children was similar to that in previously treated adolescents and adults. 9 One patient in the LEOPOLD Kids trial withdrew from the study because of a central venous access device infection; this event was not considered related to BAY 81-8973 treatment. 22
During the LEOPOLD I extension, a myocardial infarction occurred in a 62-year-old man with cardiovascular risk factors, including hypertension, smoking, hyperlipidemia, and coronary arteriosclerosis, who had been treated with BAY 81-8973 for 13 months. 23 Symptoms of myocardial infarction occurred approximately 4 h after a BAY 81-8973 prophylaxis infusion. 23 The patient recovered after placement of a metal stent in the occluded artery. 23 The investigator considered the event to be related to FVIII treatment leading to a temporary normalization of clotting, but not specifically related to BAY 81-8973. 23
Anti-BHK antibodies were detected in four patients in LEOPOLD I and II at baseline; no patient had any hypersensitivity reactions.24,33 Anti-BHK antibodies were not assessed in LEOPOLD Kids.
Anti-HSP70 antibodies were measured in clinical studies every 3 months in adults (LEOPOLD I and II) and at the start and end of LEOPOLD Kids in children; no immune responses to HSP70 or safety concerns related to HSP70 were detected. 27 Anti-HSP70 antibodies were detectable in most patients at baseline but below the 95th percentile of a normal population (the predefined cutoff for positivity). 27 Positive anti-HSP70 antibody levels were detected during BAY 81-8973 treatment in 10 of the 189 patients (5.3%) who tested negative for anti-HSP70 antibodies pretreatment; 27 5 of these patients had transient positivity at one time point, and the other 5 were positive until study end. Anti-HSP70 antibody development was not associated with any clinical effect and was most likely related to concomitant infections or normal fluctuations. 27
Assays for postinfusion monitoring of factor VIII levels
Analysis of data from LEOPOLD I and II demonstrated that either the one-stage or chromogenic assay can be used for postinfusion monitoring of FVIII levels in patients treated with BAY 81-8973. 32 Accurate measurements were obtained with either assay using plasma standards for calibration; a product-specific calibration standard is not needed or recommended. 32 Mean incremental recovery was similar when measured with either assay. 32 Further support for use of either assay was provided in an international comparative laboratory field study, in which 41 laboratories in 11 countries measured BAY 81-8973 levels in spiked plasma samples with the assays routinely used in their laboratory; no clinically relevant differences were found in the ability of laboratories to accurately measure BAY 81-8973 levels using either the one-stage or chromogenic assay. 43
Conclusions
BAY 81-8973 is an unmodified, full-length recombinant human FVIII with the same amino acid sequence as rFVIII-FS but with an innovative manufacturing process. The enhancements and modifications made to the BAY 81-8973 manufacturing process include use of a 20-nm filter that removes potential protein aggregates; in addition, no human- or animal-derived proteins are added to the cell culture, purification process, or final formulation. These changes result in a product of enhanced purity with a consistently high degree of sialylation of N-linked glycans on the molecular surface. The increased N-glycan branching and higher sialylation resulting from the manufacturing process changes translate into a PK profile for BAY 81-8973 that is more favorable than that for rFVIII-FS and another commercial unmodified rFVIII. 30 Among the youngest patient group (those aged <6 years), BAY 81-8973 half-life was comparable to that of patients aged 6–12 years (11.8 and 11.0 h, respectively). 22
BAY 81-8973 is approved for the prevention and treatment of bleeding episodes in children, adolescents, and adults with hemophilia A. BAY 81-8973 has been extensively studied in the LEOPOLD clinical trial program. Results from these studies demonstrated the efficacy of BAY 81-8973 for twice-weekly and three-times-weekly prophylaxis, on-demand treatment, and hemostasis during surgery in patients of all ages. BAY 81-8973 was generally well tolerated, with no FVIII inhibitors developing in PTPs in the completed primary studies.
Footnotes
Acknowledgements
Medical writing assistance was provided by Karen L. Zimmermann from Complete Healthcare Communications, LLC (West Chester, PA, USA) and was funded by Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bayer.
Conflict of interest statement
Dr. Mahlangu has received research grants from Bayer, Biogen, CSL Behring, Novo Nordisk, and Roche; is a member of the scientific advisory committees of Amgen, Baxalta, Biogen, CSL Behring, Novo Nordisk, and Roche; and is on the speakers bureaus of Amgen, Bayer, Biogen, Biotest, and CSL Behring.
Dr. Ahuja is a member of the advisory boards of Bayer, Baxalta, and Biogen, and has been on the speakers bureaus of Biogen, Grifols, and Novo Nordisk.
Dr. Windyga received honoraria or research grants from Bayer, Baxalta, CSL Behring, Novo Nordisk, Octapharma, Pfizer, and Sobi.
Dr. Church and Dr. Shah are employees of Bayer. Mr. Schwartz was a Bayer employee at the time this manuscript was originally submitted.



