Abstract
With a median age of 72 years at first diagnosis, chronic lymphocytic leukaemia (CLL) is a disease of the elderly. At this age, many patients cannot bear an intensive chemoimmunotherapy like fludarabine, cyclophosphamide and rituximab (FCR), and therapeutic decisions are commonly complicated by a high burden of accompanying comorbidities. Clinical trials, on the other hand, are mostly designed to include a far healthier and younger trial population, with a median age in most studies well below 70 years, leading to an insufficient reflection of clinical reality. With the introduction of new targeted therapies, treatment of CLL is currently undergoing a profound change. New compounds like ibrutinib or idelalisib have enlarged the therapeutic options in treating CLL. However, so far, these oral medications imply continuous intake by the patient, which will at some point lead to the issue of adherence in most patients. In addition, long-term experiences are largely missing. In this setting, one of the oldest chemoactive substances remains a viable option for many CLL patients and their treating physicians: bendamustine, a nitrogen–mustard derivative, has proven to be a safe and efficient agent for treatment of CLL in the first- and second-line setting. In particular, there is some evidence that the substance is relatively well tolerated in elderly and unfit patients. In this review, we summarize the current data on bendamustine in the treatment of elderly and unfit patients with CLL and aim to provide a concise analysis and outlook on the current and future role of this substance in the era of new targeted agents.
Introduction
Many patients with chronic lymphocytic leukaemia (CLL) experience an indolent course of their disease for many years and do not require therapy at initial diagnosis. However, with a median age of 72 years at diagnosis, this also means that most patients have reached an advanced age and mostly have relevant comorbidities when they ultimately require therapy. This affects their eligibility for treatment and cytostatic regimens. Hence, treatment decisions for CLL should be individualized depending on the patient’s characteristics, especially comorbidities and medical fitness, as well as on the individual situation. Validated scoring systems, like the Cumulative Illness Rating Scale (CIRS) [Salvi et al. 2008; Eichhorst et al. 2009] can be helpful tools, to objectify the patient’s burden of comorbidity.
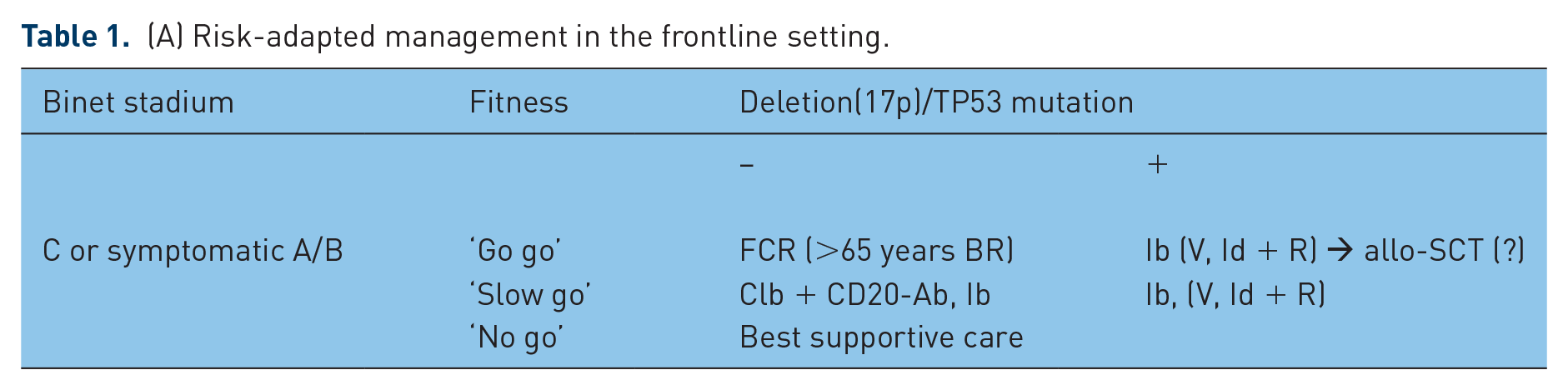
An intensive chemoimmunotherapy with fludarabine, cyclophosphamide and rituximab (FCR) achieving long-term remissions in a large proportion of patients is currently considered the standard for medically fit CLL patients without deletion 17p or TP53 mutation [Hallek et al. 2010; Fischer et al. 2016 (see Table 1)].
(A) Risk-adapted management in the frontline setting.
(B) Risk-adaptive management in relapsed patients.

FCR, fludarabine, cyclophosphamide, rituximab; Clb, chlorambucil; BR, bendamustine, rituximab; Ib, ibrutinib; Id+R, idelalisib + rituximab; V, venetoclax; Ofa, ofatumumab; allo-SCT, allogenic stem-cell transplantation; ICT, immunechemotherapy; BKI, B-cell kinase inhibitor.
Patients with a high comorbidity burden (i.e. a CIRS score higher than 6) or relevant organ impairment should receive a therapy adapted to their physical status. This includes agents like chlorambucil in combination with a CD20 antibody, that is, rituximab, obinutuzumab or ofatumumab, bendamustine, and as of late, the bruton–tyrosine kinase inhibitor ibrutinib. For patients with del17p and possibly TP53 mutations, frontline treatment should be initiated with ibrutinib. In cases of contraindications or ibrutinib failure, a combination of the PI3K inhibitor idelalisib and rituximab or monotherapy with the Bcl2 inhibitor venetoclax are valid options (see Table 1). However, so far, therapy with small molecules implies continuous administration of medication. In addition, long-term experiences are largely missing.
In this setting, one of the oldest chemoactive substances remains a cornerstone in modern CLL management. Bendamustine, a nitrogen–mustard derivative, has proven to be a safe and efficient agent for treatment of CLL in the first- and second-line setting. In particular, in elderly and unfit patients it has been shown to be well tolerated, as well as comparatively efficient.
This review summarizes the current and future status of bendamustine in the era of antibodies and small-molecule therapies.
Characteristics
Bendamustine is one of the oldest approved drugs for CLL treatment. It was first synthesized in the former German Democratic Republic (GDR) by Werner Ozegowski and Dietrich Krebs in 1963 [Ozegowski, 1971]. First, it was used solely in the GDR for the treatment of CLL, indolent non-Hodgkin’s lymphoma and multiple myeloma. It was not until the 1990s that the first clinical studies were established with bendamustine. In 2008, the Food and Drug Administration approved bendamustine for the treatment of CLL in combination with rituximab or rituximab-containing regimens.
The bendamustine molecule is a nitrogen–mustard derivate and consists of a gamma-[1-methyl-5-bis(β-chloroethyl)-amino-benzimidazolyl-2]-butyric acid hydrochloride. It contains a meclorethamine group, butyric acid and a benzimidazole ring (Figure 1). The alkylating properties, comparable with those of cyclophosphamide, chlorambucil or melphalan, are delivered by the meclorethamine group. Like other alkylating agents, bendamustine induces deoxyribonucleic acid (DNA) breaks. However, under bendamustine, the DNA damage seems to be more widespread and more sustained, as DNA repair mechanisms are slower when compared with chlorambucil treatment [Hartmann et al. 1972; Leoni et al. 2008]. The butyric acid adds to bendamustine’s water solubility, while the benzimidazole ring, which replaces chlorambucil’s benzene ring, is suggested to act as a purine analogue and to thereby provide antimetabolic characteristics [Tageja, 2011]. However, there are no final data on this assumption.

Molecular structure of bendamustine.

Molecular structure of chlorambucil.
Apart from the alkylating effect, bendamustine also stimulates apoptosis, inhibits mitotic checkpoints, and induces mitotic catastrophe. In contrast to other alkylating agents, chemoresistance via alkylguanyl transferase expression does not affect bendamustine’s efficacy, which underlines its unique characteristics as a therapeutic compound [Cheson et al. 2011].
Metabolism
Bendamustine is administered intravenously and approximately 95% of the drug is bound to plasma proteins like albumin. However, only free bendamustine is pharmacologically active. Hepatic phase I metabolization via cytochrome P460 1A2 leads to dealkylation and gamma hydroxylation of bendamustine, which in turn produces the two main metabolites gamma hydroxyl-bendamustine and N-desmethyl-bendamustine [Gandhi, 2002]. These are less active and appear in much lower concentrations than the original substance. Hence, the main therapeutic effect is mediated by bendamustine rather than its metabolites [Teichert et al. 2007].
Around 90% of bendamustine is eliminated via the faeces and only 10% via the kidneys. Thus, bendamustine can be safely applied even in patients with moderate-to-severe renal insufficiency [Cephalon, 2008; Shoji et al. 2013]. Hepatic insufficiency might limit the applicable maximum bendamustine dosage, however, there are only few reports on the use of bendamustine in patients with moderate hepatic insufficiency [Mccloskey et al. 2013]. Likewise, older age and sex do not seem to influence bendamustine’s pharmacokinetics or metabolization.
Compared with other nitrogen mustards, bendamustine has moderate adverse effects. Several studies report fatigue, fever and anticholinergic symptoms like dry mouth as dose-limiting toxicities. Moreover, infusion-related reactions like fever and rash are more likely to appear under bendamustine than other alkylating agents [Knauf et al. 2015; Cephalon, 2008]. Like all alkylating compounds, bendamustine suppresses the bone marrow leading to neutropaenia, thrombocytopenia and anaemia. Nausea can occur up to one week after drug administration and should be treated prophylactically with 5HT3-antagonists like ondansetron [Tombleson et al. 2012].
First-line therapy with bendamustine
Monotherapy
Bendamustine is still a backbone in the treatment of CLL in the frontline setting. In 2009, Knauf and colleagues published a phase III trial comparing bendamustine with chlorambucil in previously untreated CLL patients with Binet stage B or C and up to 75 years of age [Knauf et al. 2009]. Bendamustine was administered at a dose of 100 mg/m2 on days 1 and 2 every 4 weeks for six cycles, while chlorambucil was given at a dose of 0.8 mg/kg on days 1 and 15 every 4 weeks for six cycles. Chlorambucil was chosen as a comparator because it was approved for first-line treatment of CLL at the time of trial initiation in 2001.
The trial showed that bendamustine monotherapy is superior to single-agent chlorambucil treatment with regards to overall response rate (ORR) (67% versus 30%) and progression-free survival (PFS) (21.5 months versus 8.3 months). The rate of adverse events was higher with bendamustine treatment (89%) than with chlorambucil therapy (79%). The most frequent events were neutropaenia (28%), pyrexia (24%) and thrombocytopenia (23%). Serious adverse events (SAEs) also occurred more often during bendamustine treatment (18% versus 11%). Updated results showed no difference with regards to overall survival (OS) between the two groups [Knauf et al. 2012]. However, median age of patients at study entry was 63 years and thus comparatively young for the CLL population, and patients were not stratified for comorbidities or impaired organ function. Based on this trial, it is therefore difficult to draw specific conclusions for elderly CLL patients or CLL patients with substantial comorbidities.
Combination therapy
In a phase II trial, Fischer and colleagues demonstrated that the combination of bendamustine and rituximab is a safe and efficient regimen in CLL. In the first-line setting, up to six cycles of 90 mg/m2 bendamustine on days 1 and 2 of each cycle, in combination with 375 mg/m2 rituximab on day 1 in the first course and 500 mg/m2 in all subsequent cycles, achieved an ORR of 88%, including a complete response (CR) rate of 23.1%; the median PFS was 33.8 months [Fischer et al. 2012].
Based on these satisfactory data, a large phase III trial testing bendamustine and rituximab (BR) versus FCR for noninferiority in young, fit patients with CLL was initiated [Eichhorst et al. 2016]. Patients were randomized to either receive FCR (fludarabine 25 mg/m2 d1–3, cyclophosphamide 250 mg/m2 d1–3, rituximab 375 mg/m2 d0 at first cycle and 500 mg/m2 d1 all subsequent cycles; every 28 days up to six cycles) or BR with a dosage of bendamustine of 90 mg/m2 on day 1 and 2, respectively. BR was inferior to FCR with regard to the primary endpoint PFS (43.2 versus 53.7 months) and CR (31.5% versus 40.7%). There was no significant difference between ORR (95.7% versus 95.4%) and OS at 36 months (92.2% versus 90.6%). However, in a subset analysis in 194 patients older than 65 years, no significant difference in terms of PFS was detected. Instead, the significantly higher toxicity and infection rates during FCR led to more frequent dose reduction in fit patients older than 65 years. Although the study was not powered for this subgroup analysis, this result suggests that BR should be considered for elderly fit patients or patients with a higher risk of infections.
Management of chronic lymphocytic leukaemia in elderly or unfit patients
Even though not designed to test for these subgroup analyses, the results of the CLL 8 trial conducted by the The German CLL Study Group (GCLLSG) suggest that chemoimmunotherapy with FCR improves response rates and time to progression, but does not increase survival time in elderly patients compared with FC [Hallek et al. 2010]. In a retrospective analysis, the German CLL study group pointed out that CLL was the leading cause of death in patients with two or more comorbidities, which implies that disease control is crucial for overall survival [Goede et al. 2014; Ysebaert et al. 2015]. Hence, management of CLL in this group of patients is still a challenge. To this day, the number of clinical studies with a considerable number of unfit or elderly patients is very limited (see Table 2, last column). Recommendations have been based on subgroup analyses of older study participants rather than on dedicated studies with elderly or unfit cohorts.
A selection of bendamustine-containing trials in chronic lymphocytic leukaemia.

Notably, only three trials had an elderly patient population. All other trials included much younger patients.
PFS did not differ significantly between FCR and BR in patients older than 65 years.
No disease progression in either arm of the study was reported during follow up.
ORR, overall response rate; CR, complete response; PFS, progression-free survival; B, bendamustine; CLB, chlorambucil; F, fludarabine; C, cyclophosphamide; R, rituximab; Ob, obinutuzumab; Of, ofatumumab; L, lenalidomide; A, alemtuzumab; Cyt, cytarabine; M, mitoxantrone; P, placebo; r, retrospective; o, observational.
Until recently, a monotherapy with chlorambucil was considered standard of care for elderly or unfit patients with CLL. However, response rates (31–72%) and complete remission (CR) rates (0–7%) are relatively low with this treatment (reviewed by Goede et al. 2015). A landmark study for the use of chemoimmunotherapy in older and unfit patients was the CLL11 trial of the GCLLSG which included 781 patients with a higher comorbidity burden, defined by a CIRS score of >6 points with or without an impaired renal function. The trial demonstrated that the addition of the CD20 antibodies rituximab or obinutuzumab to chlorambucil was superior to single-agent chlorambucil with regards to PFS, OS, ORR and MRD negativity rate [Goede et al. 2014]. Likewise, the COMPLEMENT-1 trial showed that the CD20-antibody ofatumumab in combination with chlorambucil is also superior to chlorambucil alone in patients who were elderly or had comorbidities [Hillmen et al. 2015a]. These two randomized controlled studies showed that addition of an anti-CD20 antibody to chlorambucil improves the outcome as compared with a monotherapy with chlorambucil [Goede et al. 2015].
Laurenti and colleagues performed a retrospective analysis of the use of bendamustine in elderly or unfit patients in Italy [Laurenti et al. 2015]. They showed that in a cohort of 70 elderly or unfit CLL patients, a combination of BR lead to an ORR of 88.6% and a CR rate of 31.4%; median PFS was 35 months, despite the fact that more than half of the patients required a dose reduction from 90 mg/m2 down to 70 mg/m2 due to increased toxicity. The most frequent adverse events were grade 3 to 4 neutropaenia and infections. The Italian CLL guidelines therefore recommend that elderly and comorbid patients should receive a reduced bendamustine dose of 70 mg/m2 for up to six cycles. The minimum effective dose of bendamustine is approximately 50 mg/m2 [Cheson, 2014, Cuneo et al. 2014].
During the meeting of the international workshop on CLL (iwCLL) 2015, Michallet and colleagues presented data of the MaBLe trial, in which efficacy and safety of 90 mg/m2 bendamustine intravenously on day 1 and 2 plus rituximab on day 1 every 4 weeks for six cycles were compared with 10 mg/m2 chlorambucil orally on days 1–7 plus rituximab on day 1 every 4 weeks for six cycles (Clb-R) [Michallet et al. 2015]. The median age of the trial population was 72 years and the median number of comorbidities was three. After a median follow up of 24 months, first-line BR patients had a higher CR rate (24 % versus 9%), and longer median PFS (39.6 months versus 29.9 months) than patients treated with Clb-R. OS did not differ significantly between both treatment arms (median 43.8 months versus not reached for Clb-R). However, incidence of grade ⩾ 3 adverse events was greater in the BR group, in particular, due to blood and lymphatic disorders, infections and infestations. According to these findings, next to chlorambucil plus CD20 antibody, BR might be an option for CLL patients who are ineligible for FCR therapy due to advanced age and possibly comorbidities.
Recently, an observational trial in Italy and Austria with 100 patients confirmed that BR is an effective therapy with manageable toxicity for elderly and unfit patients [Ninkovic et al. 2015]. Notably, the BR regimen seems to dominate among first-line treatments in elderly CLL patients in Germany in the recent years, as shown by the evaluation of a tumour registry for lymphatic neoplasms in Germany [Knauf et al. 2015]. In a cohort of 860 CLL patients with a median age of 71 years, 56% were treated with BR and 22% with FCR.
Outlook: combination of bendamustine with novel agents
In recent years, a number of new agents have been introduced for the treatment of CLL (reviewed in Al-Sawaf et al. 2016). Ibrutinib is an inhibitor of bruton’s tyrosine kinase (BTK) and disrupts B-cell receptor (BCR) signalling in lymphocytes. It has shown high efficacy in treatment-naïve as well as heavily pretreated CLL patients; main adverse events reported in clinical trials so far were transient lymphocytosis, diarrhoea, fatigue, as well as an increased risk of atrial fibrillation and bleeding. Idelalisib is an inhibitor of the kinase PI3K via which it also disrupts BCR signalling. Due to an increased risk of cytomegalovirus (CMV) and pneumocystis jiruvecii (PJP) infections under idelalisib, particularly in treatment-naïve patients, it is recommended that idelalisib should only be used in previously treated patients or patients who are not eligible for ibrutinib therapy. Moreover, a PJP prophylaxis, for example, with co-trimoxazole, as well as CMV monitoring should be performed during therapy.
Recently, the Bcl2-inhibtor venetoclax was approved in the US and Europe for treatment of patients with relapsed CLL or patients with deletion 17p/TP53 mutations who are not eligible for ibrutinib therapy. Studies showed that tumour lysis events, which were observed in the first clinical trials, can effectively be avoided by a dose ramp-up and close monitoring of electrolytes and renal function. Other frequent toxicities include cytopenias, pyrexia and respiratory infections.
Several combination regimes with bendamustine have already been investigated. In 2014, Brown and colleagues evaluated the combination of obinutuzumab with bendamustine or FC in a phase I study and documented a high rate of toxicities, especially infusion-related reactions and neutropaenias [Brown et al. 2015]. Recently, a phase III trial showed that a combination of BR with ibrutinib in relapsed fit CLL patients further improved outcome with a manageable safety profile. At a median follow up of 25 months, the 24-month PFS rate was 74.8% in patients receiving BR + ibrutinib as compared with 20.9% in patients receiving BR + placebo [Chanan-Khan et al. 2016; Fraser, 2016].
Also, overall and CR rates, as well as the minimal residual disease (MRD) negativity rate were improved. Thus far, due to a short follow-up time and a cross-over of the patients relapsing after BR to receive ibrutinib, only a trend towards improvement of OS could be demonstrated [hazard ratio (HR) 0.670; 95% confidence interval (CI), 0.44–1.02; p = 0.0587]. Notably, no transient lymphocytosis due to ibrutinib was observed when combined with BR, and the regime seems to bring patients faster into deeper remission with more CRs and MRD negativity. Therefore, this strategy might be useful in patients with a higher tumour load and the necessity to achieve a quick response. However, a comparison of the single-agent ibrutinib and BR plus ibrutinib was not yet performed. It is thus not clear whether chemoimmunotherapy is necessary or whether there is any long-term benefit in comparison with ibrutinib monotherapy. Based on an adjusted indirect comparison of the HELIOS and RESONATE trials, Hillmen and colleagues recently suggested that addition of BR to ibrutinib does not improve PFS or OS compared with single-agent ibrutinib. Long-term follow up is needed to elucidate whether ibrutinib monotherapy is sufficiently effective for patients with relapsed/refractory CLL [Hillmen et al. 2015b].
A phase III trial showed that addition of idelalisib to BR is superior to BR alone in relapsed CLL patients. Median PFS in the BR + idelalisib arm was 23.1 months versus 11.1 months in the group receiving BR + placebo. The study also found a significant OS benefit, with a 45% reduction in the risk of death for patients additionally treated with idelalisib (HR 0.55; 95% CI, 0.36–0.86; p = 0.008) [Zelenetz, 2015].
Addition of the modified CD20-antibody ofatumumab to bendamustine might be beneficial in patients with relapsed or refractory CLL, as was shown by Cortelezzi and colleagues in a phase II trial, where patients receiving bendamustine and ofatumumab had an ORR of 72% and a median PFS of 23.6 months [Cortelezzi et al. 2014]. Other regimes are currently under investigation, for instance a combination of bendamustine with the PI3K inhibitor idelalisib [ClinicalTrials.gov identifier: NCT01569295] or addition of the Bcl2-inibitor venetoclax to a BR regimen [ClinicalTrials.gov identifier: NCT01671904]. First data of the latter trial suggests that the combination of venetoclax with BR has an acceptable safety profile, regardless of whether treatment is initiated with venetoclax or BR first [Stilgenbauer et al. 2016].
Taken together, there are promising approaches for combination regimes of bendamustine with new compounds; however, none of the quoted trials was or is primarily designed for elderly or unfit patients. Moreover, the Resonate-2 trial has recently established the superiority of ibrutinib monotherapy in elderly patients as compared to chlorambucil monotherapy, which poses the question whether addition of chemotherapy is necessary for the positive outcome in elderly patients [Burger et al. 2015].
The ability of bendamustine to quickly reduce tumour load, that is, lymphocyte count and lymphadenopathy, suggests a role as a debulking agent in the era of targeted therapies. The GCLLSG proposed a ‘triple T’ concept, meaning a tailored and targeted treatment aiming at a total eradication of MRD [Hallek, 2013]. This includes an optional debulking with bendamustine in patients with high lymphocyte count with or without substantial lymphadenopathy in order to reduce tumour load, prior to continuing with an induction and MRD-tailored maintenance treatment with an antibody plus a kinase inhibitor or bcl-2-antagonist (BXX trials of the GCLLSG [ClinicalTrials.gov identifiers: NCT02689141, NCT02445131, NCT02401503, NCT02689141]).
Conclusions
Although the current data on bendamustine-containing regimes is mostly based on younger patients, the data presented in this review suggest that bendamustine can be a viable option in the treatment of elderly or unfit CLL patients. However, in light of new therapeutic options with targeted agents showing promising efficacy and tolerability across several trials in elderly patients, the role of bendamustine likely needs to be redefined in the years to come and potentially will be limited to selected cases.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
MH received fees from Roche, Glaxo Smith Kline, Mundipharma, Janssen, Gilead and Celgene.
NP received travel grants and other compensations from Celgene and Roche.
OA had no relevant conflict of interest to declare.
PC received research funding from Gilead Sciences (Inst), Roche (Inst), Janssen-Cilag (Inst), Novartis (Inst), GlaxoSmithKline (Inst); travelgrants and other compensations from Astellas Pharma, Gilead Sciences, Janssen-Cilag, Roche and Mundipharma.
VG received fees from Roche, Glaxo Smith Kline, Mundipharma and Bristol Myers Squibb.
