Abstract
Lenalidomide, an immunomodulatory drug that the US Food and Drug Administration (FDA) approved for the treatment of multiple myeloma, 5q- myelodysplasia and mantle-cell lymphoma (MCL), has encouraging efficacy in other B-cell malignancies. Its unique mechanism of action is in part due to altering the tumor microenvironment and potentiating the activity of T and natural-killer (NK) cells. Impressive clinical activity and excellent tolerability allows broad applicability. Lenalidomide has been used in a wide range of B-cell malignancies for years, but in 2013, the FDA marked its approval as a single agent only in relapsed/refractory mantle-cell lymphoma. Perhaps most impressive is the efficacy of lenalidomide when combined with monoclonal antibodies. Impressive efficacy and toxicity profiles with the combination of lenalidomide and rituximab in B-cell lymphomas in both the upfront and relapsed/refractory setting may allow a shift in our current treatment paradigm in both indolent and aggressive non-Hodgkin lymphoma (NHL). This review will summarize the current data in the relapsed/refractory and front-line setting of NHL with single-agent lenalidomide as well as its use in combination with other agents.
Introduction
Non-Hodgkin lymphoma (NHL) represents a heterogeneous group of diseases, the majority of which are of B-cell origin. B-cell NHL is generally classified within two broad subdivisions: aggressive and indolent lymphoma, of which the most common subtypes include diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL), respectively. Historically, these cancers are treated with chemoimmunotherapy using rituximab, a monoclonal antibody directed against CD20, in addition to cytotoxic agents such as doxorubicin, cyclophosphamide, vincristine, and bendamustine [Shankland et al. 2012]. NHL is highly treatable and curable, particularly in the aggressive setting. However, efficacy is often limited by toxicity, and many patients eventually relapse or cannot tolerate current cytotoxic approaches necessitating the need for alternative treatment strategies.
Lenalidomide, an analogue of thalidomide, is FDA approved for the treatment of multiple myeloma, myelodysplastic syndrome associated with deletion 5q, and most recently in relapsed/refractory mantle-cell lymphoma (MCL). Moreover, it has a more favorable side-effect profile than its parent drug, thalidomide. Its mechanism of action (MOA) is complex and includes immunomodulation, antiangiogenic effects, and direct cytotoxic activity [Kotla et al. 2009]. Much of our understanding of lenalidomide is derived from data in multiple myeloma; however, recent developments that further elucidate the MOA have broadened its potential application. Specifically, its ability to enhance natural-killer (NK) cell activity, direct cytotoxic effects; and its inhibitory effects of the NF-κb pathway likely mediate dramatic responses in the difficult-to-treat subset of DLBCL, nongerminal center B-cell (non-GCB), or activated B-cell (ABC) subtype [Gribben et al. 2015]. Furthermore, the synergistic effects of lenalidomide in combination with monoclonal antibodies, particularly rituximab, have led to impressive activity in both first line and relapsed/refractory indolent NHL, MCL and DLBCL, setting a precedent for the use and further evaluation of ‘non-chemotherapeutic,’ lenalidomide-containing options for patients with lymphoma.
Here, we review previous and ongoing studies evaluating lenalidomide’s efficacy, both as a single agent and in combinations that may eventually change the standard of care for B-cell NHL.
What is known about the mechanism?
Studies spanning multiple cancer types have demonstrated that the therapeutic effects of lenalidomide are mediated through a number of different mechanisms. Specifically, these include modulation of the immune response, direct cytotoxic effects, reduction of cellular proliferation and inhibition of angiogenesis [Kotla et al. 2009; Gribben et al. 2015].
Studies in follicular lymphoma have shown that tumor infiltrating CD4- and CD8-positive T cells can have defective synapse formation that results in impaired antigen presentation. Consequently,
Cereblon, an E3 ubiquitin ligase, was identified as the primary target for thalidomide-induced teratogenesis [Ito et al. 2010]. Unsurprisingly, lenalidomide also targets cereblon and it has been shown that this leads to the reduction of c-Myc and IRF4 expression, while increasing expression of p21 that leads to G1 cell-cycle arrest [Lopez-Girona et al. 2012]. In DLBCL, a screen for synthetic lethality also showed that lenalidomide decreased IRF4 expression as well as reduction in Spi-B transcription factor in a cereblon-dependent manner [Yang et al. 2012]. This, in turn, impacts NF-kappa B signaling and the type I interferon response which is likely the mechanism that is responsible for the improved efficacy seen in the ABC subtype of DLBCL. The lenalidomide-mediated effects on cereblon may also play a role in the immune-modulating effects by mediating T-cell activation [Gandhi et al. 2014].
In terms of antiangiogenic activity, it has been shown that lenalidomide mediates inhibition of lymphangiogenesis in murine models of MCL as well as restraining the angiogenic potential of endothelial cells in multiple myeloma patients [De Luisi et al. 2011; Song et al. 2013]. A more detailed overview of the MOA has recently been published by Gribben and colleagues [Gribben et al. 2015].
Mantle-cell lymphoma
MCL represents approximately 6–8% of all NHL; clinically, patients have a distinctive but highly variable phenotype [Weisenburger and Armitage, 1996]. MCL patients are often elderly males, and because comorbidities are often present in this population, treatment can be challenging and limited by toxicity particularly when considering dose-intensive regimens [Bosch et al. 1998]. Younger, fit patients are generally treated with an anthracycline-containing regimen alternating with high-dose cytarabine followed by autologous stem-cell transplantation. Transplant-ineligible patients are often treated with R-CHOP, or bendamustine and rituximab [Geisler et al. 2008]. There are many options for relapsed MCL, including bortezomib, ibrutinib, temsirolimus and lenalidomide.
Relapsed/refractory mantle-cell lymphoma
Lenalidomide has been studied extensively in the relapsed/refractory population and was FDA approved in June 2013. The MCL-001/EMERGE study evaluated 134 patients with relapsed/refractory MCL who received single-agent lenalidomide [Goy et al. 2013]. The overall response rate (ORR) was 28% with a complete response (CR) in 8%, and a median duration of response (mDOR) of 16.6 months (Table 1). Prior to this study, multiple trials examined lenalidomide in combination with a variety of biologic agents and cytotoxic therapies. In 2008, a phase II trial evaluated lenalidomide alternating with thalidomide and rituximab in patients with relapsed/refractory NHL- three of whom had MCL [Coleman, 2008]. ORR was 100% with two patients achieving a complete remission. Subsequently, in 2009, a subset analysis was performed in the NHL-002 trial with 15 patients with relapsed/refractory MCL [Habermann et al. 2009]. The ORR was 53%, with 20% CR. Most common significant AEs for both these trials were neutropenia and rash.A phase II trial by Ahmadi and colleagues published in 2014, studying the combination of lenalidomide with dexamethasone, and eventually incorporating rituximab in patients with relapsed/refractory NHL, including MCL, was performed, and demonstrated that with the addition of rituximab, the ORR was 60%, with AEs consistent with those reported in previous studies [Ahmadi et al. 2014]. These results corroborated similar findings from a trial undertaken in 2012 [Zaja et al. 2012], (Table 1), but the former trial was the first that showed efficacy with the combination regimen in patients who had been resistant to rituximab in the past.
Lenalidomide for mantle-cell lymphoma.

MCL, mantle-cell lymphoma; R2, lenalidomide and rituximab; Len, lenalidomide; Dex, dexamethasone; Ritux, rituximab; Thal, thalidomide; Benda, bendamustine; Bortez, bortezomib; mFUT, median follow-up time; ORR, overall response rate; CLL,; CR, complete response; CRu, unconfirmed complete response; PR, partial response; PFS, progression-free survival; mPFS, median progression-free survival; OS, overall survival; mOS, median overall survival; DOR, duration of response; mDOR, median duration of response; NR, not reached; PD, disease progression; pts, patients; MRD, minimal residual disease; DLT, dose-limiting toxicity; TL, transformed lymphoma; FL-III, follicular lymphoma grade III; R-DHAP, rituximab, dexamethasone, cytarabine and cisplatin; R-CHOP, rituximab, doxorubicin, vincristine, cyclosphamide and prednisone; SLL, small lymphocytic lymphoma; MTD, maxium tolerated dose; CLL, chronic lymphocytic leukemia.
The proteasome inhibitor bortezomib has potent activity in MCL, thus its combination with lenalidomide has been studied in the first- and second-line setting. In 2012, lenalidomide combined with bortezomib was evaluated in the phase II CALGB 50501 study [Morrison, 2011]. Of the 53 patients with relapsed/refractory MCL, eight of them (15.1%) achieved a CR, and 13 (24.5%) achieved a PR. The 1-year event-free survival (EFS) was 25%. Consistent with previous studies, common AEs included myelosuppression and rash, however, a 6% incidence of venous thromboemobolism (VTE) was also reported.
Previously untreated mantle-cell lymphoma
A landmark study published by Ruan et al. (2015) used lenalidomide and rituximab in 38 treatment-naïve MCL patients, yielding an impressive ORR of 92%, CR of 64%, with a median progression-free survival (mPFS) that was estimated to be 85% at 2 years, with an overall survival (OS) of 97% [Zaja, 2015]. The combination therapy was well tolerated and may prove to be an effective and well tolerated approach for previously untreated MCL.
In another study, lenalidomide/bortezomib was combined with rituximab in the first- or second-line setting in 22 patients with MCL [Flinn, 2012]. The ORR was reported at 86%, with a CR of 36% with notable AEs primarily being neutropenia and rash.
Bendamustine, a cytotoxic agent with a favorable toxicity profile that has shown much promise in lymphoid malignancies including MCL, was studied in combination with lenalidomide and rituximab in a phase I/II trial [Albertsson-Lindblad, 2015]. In 51 elderly patients with newly diagnosed MCL, the reported ORR was 91%, with a CR of 78% and a median OS of 53 months. AEs were most notable for infections, particularly opportunistic infections.
Ongoing clinical trials are evaluating the use of lenalidomide in a variety of combinations in both the relapsed/refractory and untreated MCL.
Indolent lymphoma
Indolent lymphomas comprise approximately 40% of all NHL and include a number of histologies including follicular, small lymphocytic, marginal zone and lymphoplasmacytic lymphoma [Lymphoma Research Foundation, 2016]. Indolent lymphomas typically have a long natural history and initially respond well to a variety of chemotherapeutic and biologic agents; however, they invariably recur and are considered mostly incurable. Follicular lymphoma (FL) is the most common indolent subtype of NHL.
FL typically presents with slowly progressive, waxing and waning adenopathy, often delaying diagnosis; thus most patients with FL are diagnosed at an advanced stage. Since FL, like other low-grade lymphomas, tends to recur, alternative therapeutic options are indicated. One such combination studied extensively in recent clinical trials is lenalidomide and rituximab, otherwise known as R2. Evidence of lenalidomide’s ability to synergistically work with rituximab to increase antitumor activity has been reported in multiple trials evaluating R2 in both untreated, relapsed/refractory and rituximab-refractory patient populations [Gribben et al. 2015].
Relapsed/refractory indolent lymphoma
In 2009, a phase II multicenter study (NHL-001) investigated lenalidomide monotherapy using 25 mg/day for 21 days of a 28-day cycle in 43 patients with previously treated indolent NHL [Witzig et al. 2009]. The ORR for the entire study population was 23%, with a CR or unconfirmed complete response (CRu) of 7%. The ORR for the 22 patients with FL was 27%. The most recent report on this study noted that the DOR was greater than 16.5 months, with ongoing responses up to 28 months. The most common AEs were primarily hematologic. Grade 1 or 2 tumor-flare response occurred in a total of four patients [Witzig et al. 2009].
In the years following NHL-001, a number of studies evaluated the safety and efficacy of R2, sometimes in comparison with lenalidomide monotherapy, (Table 2). In previously treated patients, R2 produced ORRs ranging from 33% to 74%, and a CR/CRu that ranged from 18% to 44% (Table 2) [Witzig et al. 2009; Ahmadi et al. 2014; Smith, 2014; Tuscano et al. 2014; Leonard et al. 2015]. Common AEs were similar to those observed in NHL-001, including moderate myelosuppression.In 2014, another phase II study further evaluated the efficacy of R2 in patients with relapsed/refractory indolent NHL [Tuscano et al. 2014]. A total of 30 patients were enrolled and all patients had previously received rituximab, 15 of whom were refractory to rituximab. Lenalidomide 25 mg/day was given for the first 21 days of a 28-day cycle and continued until intolerance or progression. Subsequently, two of the first four patients developed grade 3 tumor-lysis syndrome (TLS), prompting a dose reduction to 20 mg/day and administration of TLS prophylaxis. Rituximab 375 mg/m2 was administered as 4-weekly infusions beginning on day 15 of cycle 1. Out of 27 evaluable patients, ORR was 74.1%, with 44% CR/CRu. At a median follow up of 43 months, the mDOR was 15.4 months for all responders and not reached for complete responders, and mPFS was 12.4 months. Median time to response was 1.8 months, and 34% of patients achieved the best response during the maintenance phase. Among the 22 patients with FL, ORR was 77.3%, consisting of nine CR/Cru patients.
Lenalidomide for indolent lymphoma.
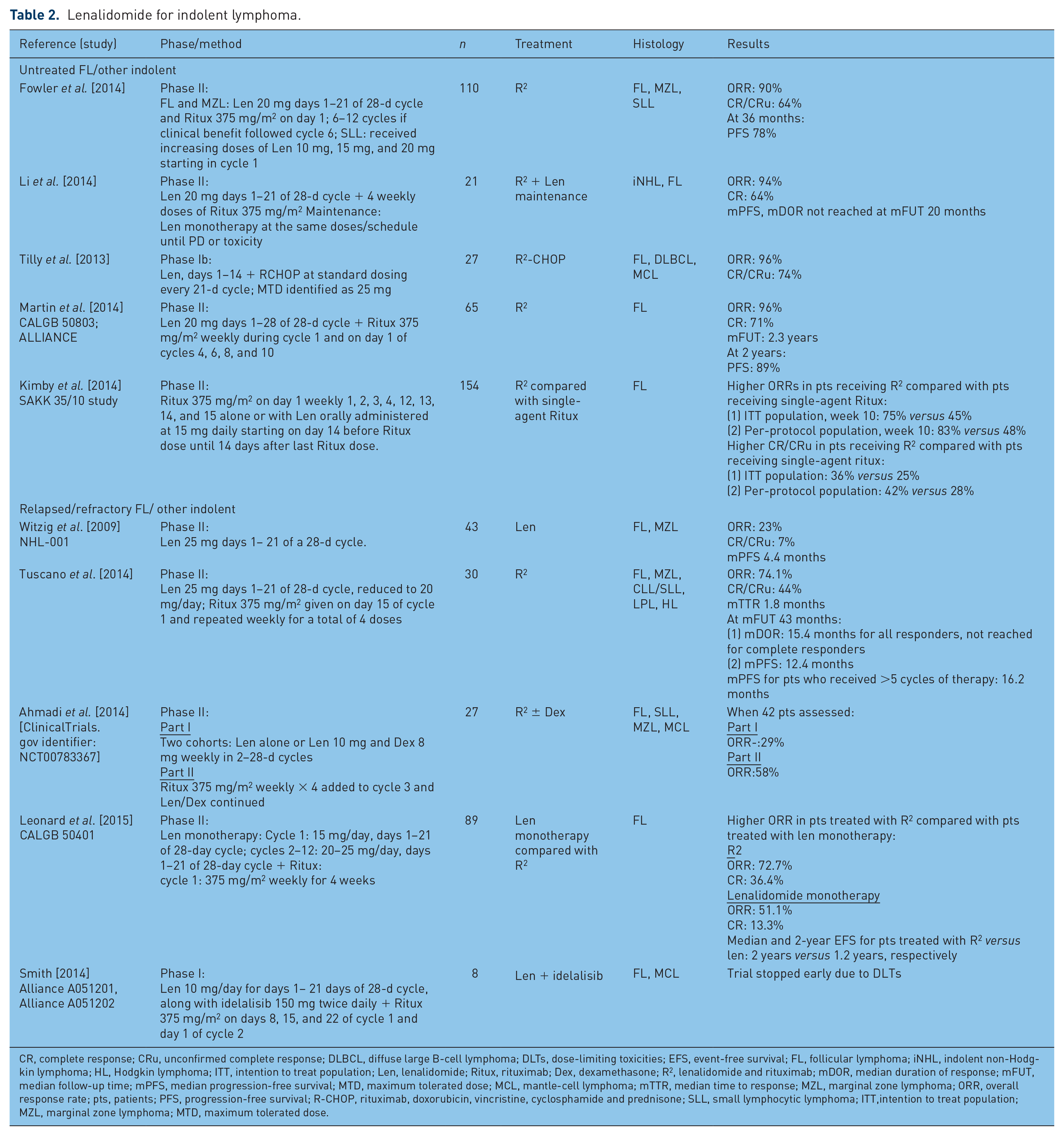
CR, complete response; CRu, unconfirmed complete response; DLBCL, diffuse large B-cell lymphoma; DLTs, dose-limiting toxicities; EFS, event-free survival; FL, follicular lymphoma; iNHL, indolent non-Hodgkin lymphoma; HL, Hodgkin lymphoma; ITT, intention to treat population; Len, lenalidomide; Ritux, rituximab; Dex, dexamethasone; R2, lenalidomide and rituximab; mDOR, median duration of response; mFUT, median follow-up time; mPFS, median progression-free survival; MTD, maximum tolerated dose; MCL, mantle-cell lymphoma; mTTR, median time to response; MZL, marginal zone lymphoma; ORR, overall response rate; pts, patients; PFS, progression-free survival; R-CHOP, rituximab, doxorubicin, vincristine, cyclosphamide and prednisone; SLL, small lymphocytic lymphoma; ITT,intention to treat population; MZL, marginal zone lymphoma; MTD, maximum tolerated dose.
In 2012, the CALGB 50401 study compared lenalidomide monotherapy with R2 in 89 patients who remained sensitive to rituximab, with relapsed/refractory FL after at least one rituximab-based regimen [Leonard et al. 2015]. Patients treated with R2 had a higher ORR compared with patients treated with lenalidomide alone (72.7%
It is important to note that not all drug combinations have produced such convincing evidence of synergy. The combination of R2 with idelalisib in relapsed/refractory NHL has produced serious AEs including unexpected immune-mediated dose-limiting toxicities and led to cessation of rituximab. Other dose-limiting toxicities following rituximab removal included culture-negative sepsis, lung infection, and grade 4 rash that led to termination of the trial [Smith, 2014]. However, the majority of clinical results highlight the promising potential for combination therapies that incorporate lenalidomide or R2 in patients with relapsed/refractory indolent lymphoma.
Previously untreated indolent lymphoma
Clinical studies evaluating previously untreated patients with indolent NHL treated with R2 reported ORRs ranging from 75% to 96%, and CR/CRu ranging from 36% to 71%, (Table 2).
In 2012, a phase II, single-arm study evaluated lenalidomide in combination with rituximab in 110 previously untreated patients with indolent NHL [Fowler et al. 2014]. The ORR was a notable 90% in 103 evaluable patients, including a 64% CR/CRu. The PFS at 36 months was 78%. For the FL subset, the ORR was 98% and the CR/CRu was 87%, with a 36-month PFS of 81%.
Lenalidomide has also been combined with other cytotoxic regimens with excellent efficacy in the upfront setting. In 2013, a study evaluating R2-CHOP in previously untreated high-burden FL yielded an ORR of 96%, with 74% CR/CRu [Tilly et al. 2013], prompting ongoing clinical trials to further evaluate this combination.
Aggressive lymphoma
Aggressive lymphomas fall into a broad category with a variety of molecularly and phenotypically defined subtypes, the most common of which is diffuse large B-cell lymphoma (DLBCL) which comprise approximately 30% of all NHL [Alizadeh et al. 2000]. Although aggressive, these lymphomas are potentially curable, with an OS rate of 50–75% at 5 years, depending on prognosis [Shipp, 1994]. Most relapses occur within the initial 2 years after completion of therapy, and once relapsed, can be effectively salvaged and in eligible patients, consolidative autologous stem-cell transplant (SCT) will produce long-term durable remissions in approximately 20–40% of patients [Coiffier et al. 1998].
Relapsed/refractory aggressive lymphoma
In 2011, Witzig and colleagues studied patients with aggressive lymphoma who received single-agent lenalidomide at 25 mg daily, days 1–21 of a 28-day cycle. The study reported an ORR of 35%, with a CR/CRu of 13%. Notably, the ORR in patients who had undergone a prior autologous SCT was 37% and the median DOR 10.6 months [Witzig et al. 2011]. In another study, using single-agent lenalidomide at a similar dosing schedule in various subsets of lymphoma, including 14 relapsed/refractory DLBCL patients, lenalidomide was administered for 52 weeks or until progressive disease or intolerance was observed [Hitz et al. 2013]. Of the 17 patients evaluable for analysis, the ORR for the entire cohort was 35%, with three patients achieving a CR/CRu. For both of these studies, the major AEs were hematologic, including neutropenia, anemia, and thrombocytopenia. In a study using R2 in 45 patients with relapsed aggressive lymphomas, treatment yielded an ORR of 33%, of which 22% achieved a CR, with a median DOR of 10.2 months [Wang et al. 2013]. The majority of the grade 3 and 4 AEs were, again, hematologic.
Lenalidomide has also been combined with a variety of other cytotoxic agents. From the phase I/II portion of the SAKK 38/08 trial, where lenalidomide was combined with bendamustine in 13 patients with recurrent high-grade lymphomas, four patients had unacceptable toxicities leading to treatment termination; one patient had an unconfirmed CR and remained in remission after 9 months of follow up; and two patients achieved PR [Hitz et al. 2013]. The grade 3 and 4 AEs included myelosuppression, cardiac events, rash, and fatigue.
Lenalidomide has been combined with other agents including rituximab, dexamethasone, cytarabine and cisplatin (R-DHAP), mammalian target of rapamycin (m-TOR) inhibitors, and ibrutinib with varying degrees of success. When combined with R-DHAP, the observed ORR was 55%, with 12 CR/CRu patients [Glass, 2014]. When combined with temsirolimus, an ORR of 47% was reported, while everolimus yielded an ORR of 20%, with one CR and eight PR patients [Smith, 2012; Ernst, 2014]. Finally, ibrutinib combined with lenalidomide showed an ORR of 36% with two patients achieving a CR [Christian, 2014]. These combination trials resulted in a higher incidence of hematologic and infectious toxicity when compared with single-agent lenalidomide or R2. See Table 2 for more details.
Untreated aggressive lymphoma
Of intense interest are subsets of DLBCL that are considered more resistant to chemotherapy in which promising responses to lenalidomide have been reported. Molecular profiling has allowed us to identify distinct subtypes of DLBCL that include germinal center B-cell (GCB) and ABC subtypes that have significantly different outcomes when treated with R-CHOP [Rosenwald et al. 2002]. Treatment with R-CHOP produced an OS at 5 years in the GCB group of 60%, as compared with 35% in the ABC subtype [Rosenwald et al. 2002]. The ABC subtype responds poorly to R-CHOP with a significantly inferior 5-year OS (76%
Lenalidomide for aggressive lymphomas.

MCL, mantle-cell lymphoma; NHL, non-Hodgkin lymphoma; R2, lenalidomide and rituximab; Len, lenalidomide; Ritux, rituximab; Thal, thalidomide; Benda, bendamustine; mFUT, median follow-up time; ORR, overall response rate; CR, complete response; CRu, unconfirmed complete response; PR, partial response; PFS, progression-free survival; mPFS, median progression-free survival; OS, overall survival; mOS, median overall survival; DOR, duration of response; mDOR, median duration of response; NR, not reached; PD, disease progression; f/u, follow up; DLT, dose-limiting toxicity; IV, intravenously; TL, transformed lymphoma; FL, follicular lymphoma; FL-III, follicular lymphoma grade III; R-DHAP, rituximab, dexamethasone, cytarabine and cisplatin; R-CHOP, rituximab, doxorubicin, vincristine, cyclosphamide and prednisone; DLBCL, diffuse large B-cell lymphoma; pts, patients; HL, Hodgkin lymphoma; PTCL, peripheral T-cell lymphoma; CLL, chronic lymphocytic leukemia; SLL, small lymphocytic lymphoma; MTD, maximum tolerated dose; TCL, T-cell lymphoma; LPL, lymphoplasmacytic lymphoma; SD, stable disease; BTK, Bruton’s tyrosine kinase; mTTR, median time to response; WM, Waldenstrom’s macroglobulinemia; MZL, marginal zone lymphoma.
While lenalidomide is currently only FDA approved for the treatment of relapsed MCL, its application in NHL is broadening clinically. A plethora of small clinical trials have demonstrated impressive activity in indolent and aggressive NHL particularly in heavily pretreated patients. What is of further interest is the durability of response, even in difficult-to-treat patient populations. As expected, a higher response rate is consistently observed in patients who are treatment naïve, as compared with those who are heavily pretreated. However, even in heavily pretreated FL patients, although the ORR was a modest 27%, those that responded had durable remission lasting approximately 20 months [Witzig et al. 2009].
The synergistic effects of lenalidomide and rituximab allow for enhanced efficacy likely mediated by NK and T-cell-mediated cytotoxicity, making it an obvious combination in NHL. The striking efficacy in untreated FL patients as demonstrated by an ORR of 98%, with a CR rate of 87% with R2 and with median DOR that was not reached at a median follow-up time of 40.6 months, is a testament to the potential of the regimen [Tilly et al. 2013; Fowler et al. 2014] and has provided the basis for other combination studies with lenalidomide.
Although the mechanism of action is complex and incompletely understood, what is known thus far suggests that the effects of lenalidomide are pleotropic, and include direct cytotoxic effects, modulation of cereblon and NF-κB levels and activity, as well as immune modulation. The effects on cereblon and NF-κB likely also contribute to the immunomodulatory activity in ABC DLBCL providing an excellent rationale for therapy in this subtype [Gribben et al. 2015]. R2-CHOP has provided promising data in untreated ABC DLBCL which has not seen advances in the recent past. These data need to be further matured and verified in ongoing phase III trials to further validate these results.
When combined with agents other than rituximab, although response rates are higher, the toxicity is often correspondingly higher. Lenalidomide is generally well tolerated with the most common AEs being hematologic, primarily myelosuppression. Although often asymptomatic, significant neutropenia was the most common AE, however the rate of infections was also notable. Lenalidomide’s risk of VTE is well documented, particularly when combined with dexamethasone and other cytotoxic therapy in patients with myeloma [Khorana et al. 2009], however the incidence of VTE does not appear higher in NHL [Chanan-Khan and Cheson, 2008] but needs to be better defined. Of further interest is the incidence of secondary primary malignancies (SPMs) that were noted initially in a small percentage of patients with multiple myeloma, that was hypothesized to be associated with prior treatment, particularly alkylating agents such as melphalan [Gribben et al. 2015]. The incidence of SPMs in the NHL population is not as well studied and available data is relatively immature.
In summary, lenalidomide is a promising agent with a complex MOA that has shown impressive activity in a variety of lymphoid malignancies. Clinical trials with lenalidomide alone, in combination with rituximab, other antibodies and cytotoxic therapy, as well as the recently developed targeted kinase inhibitors, are currently open to accrual and will further define the role of this promising agent for the treatment of lymphoma.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of Interest statement
Joseph Michael Tuscano receives research funding and honoraria from Celgene.
