Abstract
Bortezomib is a first in class proteasome inhibitor, initially approved by the US Food and Drug Administration for the treatment of plasma cell myeloma. Bortezomib has been approved for the treatment of relapsed and refractory mantle cell lymphoma (MCL) and, more recently, in the upfront setting as well. Treatment algorithms for MCL have rapidly evolved over the past two decades, and the optimal regimen remains to be defined. The choice of treatment regimen is based on disease risk stratification models, the expected toxicity of antineoplastic agents, the perceived patient ability to tolerate the planned treatments and the availability of novel agents. As new drugs with novel mechanisms of action and variable toxicity profiles come into use, treatment decisions for a given patient have become increasingly complex. This article provides an overview of the evolving use of bortezomib in the rapidly changing management landscape of MCL
Keywords
Mantle cell lymphoma: overview
Mantle cell lymphoma (MCL) comprises 5–6% of non-Hodgkin lymphomas (NHLs). Median age at diagnosis is 65 years of age, with a male to female ratio of 3:1 and frequent involvement of extranodal sites, particularly bone marrow, blood, and gastrointestinal tract [Armitage and Weisenburger, 1998; Avivi and Goy, 2015]. MCL is derived from antigen-naïve cells that proliferate in the mantle zone around germinal centers, with morphologic (diffuse, nodular, mantle zone) as well as cytologic variants (small cell, pleomorphic, blastoid). The specific immunophenotype of MCL cells is CD20+, CD5+, CD23−, and FMC7+ with overexpression of cyclin D1 as a result of the translocation t(11;14). This genetic abnormality, usually identified by fluorescent in situ hybridization (FISH), is required to make the diagnosis [Avivi and Goy, 2015]. Rare cases of cyclin D1-negative MCL show cyclin D2 or D3 overexpression (through rearrangements of other immunoglobulin loci) and have similar clinical behavior as typical, cyclin D1-positive MCL cases [Rosenwald et al. 2003]. Since its recognition as a separate disease entity in 1994, the median overall survival (OS) of MCL has improved from less than 2.5 to over 5 years, based on a recent Surveillance, Epidemiology, and End-Results database analysis [Chandran et al. 2012].
Treatment approaches to MCL
The treatment algorithms for MCL have changed significantly over the past three decades. Several treatment approaches have been developed, with the choice based upon age, disease risk, expected treatment tolerance, and other clinician-dependent factors.
Older patients and those with poor functional status with low-risk disease, based on MCL International Prognostic Index (MIPI) [Hoster et al. 2008, 2014a] and Ki-67 staining less than 30% [Determann et al. 2008] have frequently been treated conservatively or with watchful observation [Vose, 2013; Rajabi and Sweetenham, 2015]. Younger patients with good functional status and high-risk disease are often treated aggressively due to the high mortality of intermediate- to high-risk MCL [Vose, 2013; Rajabi and Sweetenham, 2015]. Standard upfront treatment consists of a cytarabine-based induction regimen that is usually followed by an autologous stem cell transplant (ASCT) [Geisler et al. 2012; Hermine et al. 2012; Lacasce et al. 2012; Delarue et al. 2013]. Patients with moderate- or high-risk disease with decreased ability to tolerate toxic regimens have been treated initially with a variety of regimens, including the combination of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) or, more recently, BR (bendamustine/rituximab) [Rummel et al. 2013; Vose, 2013; Rajabi and Sweetenham, 2015]. Rituximab maintenance therapy, after R-CHOP induction, has shown improved survival (4-year survival 87% with rituximab maintenance versus 63% with interferon α) and improved chance of being in remission at 4 years (58% in remission with rituximab maintenance versus 29% in the interferon α group) [Kluin-Nelemans et al. 2012]. The R-BAC regimen, popular in Europe, combined moderate doses of cytarabine (up to 800 mg/m2) BR with no increased toxicity, except for transient grade 3 or 4 thrombocytopenia, and was highly active in older patients with MCL [Visco et al. 2013].
While R-CHOP is commonly used in certain clinical situations, its use as initial treatment without subsequent consolidation with ASCT or maintenance rituximab has lost favor [Rajabi and Sweetenham, 2015]. A retrospective review conducted by LaCasce and colleagues showed R-CHOP resulted in inferior outcomes than the more intense R-HyperCVAD (rituximab, cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with methotrexate and cytarabine) regimen, regardless of the use of consolidation with ASCT. Based on these results, it remains unclear if patients who receive R-HyperCVAD benefit further from ASCT. The retrospective nature of this study and the relatively small number of patients who received ASCT after R-HyperCVAD (n = 34) impede a definitive conclusion regarding the use of transplant after this intensive chemotherapy regimen [Lacasce et al. 2012]. An earlier study of 25 patients with MCL treated upfront with HyperCVAD followed by ASCT or allogenic stem cell transplantation showed an impressive 3-year survival of 92% [Khouri et al. 1998]. However, multiples studies have shown that R-HyperCVAD is associated with significant toxicity, with up to 39% of patients being unable to complete the planned course due to complications from therapy [Bernstein et al. 2013]. As new agents have been shown to have activity against relapsed or refractory MCL, the possibility of achieving equal long-term clinical outcomes without the toxicity of ASCT or cytarabine-based regimens has become a key clinical question in the initial management of MCL.
Different treatment approaches are taken for relapsed or refractory MCL compared with initial treatment of newly diagnosed disease. Relapsed and refractory disease have been managed with bortezomib, lenalidamide, ibrutinib, bendamustine, temsirolimus, R-CHOP, various combination regimens, as well as ASCT and allogeneic stem cell transplantation in well selected patients [Vose, 2013; Avivi and Goy, 2015; Rajabi and Sweetenham, 2015]. Many studies evaluating the treatment of relapsed or refractory MCL are small in size, single arm, with sometimes limited follow up to evaluate survival [Friedberg et al. 2011; Kouroukis et al. 2011; Morrison et al. 2015].
Proteasome inhibition in MCL: mechanism of action of bortezomib
The intact 26S proteasome is the major site of protein degradation in eukaryotic cells, responsible primarily for degrading intracellular proteins. Bortezomib (Velcade; Millennium Pharma-ceuticals, Cambridge, MA, USA) is a first-in-class compound that reversibly interacts with a threonine residue on the β subunit of the 20S proteasome, responsible for chymotrypsin proteolytic activity. Proteins destined for proteasomal degradation become polyubiquitinated and are recognized by the proteasome by their polyubiquitin ‘tag’. The many proteasomal substrates include key cell-cycle regulatory proteins, such as cyclins, the endogenous CDK inhibitors p21 and p27 and the CDC25 family of phosphatases, the tumor suppressor p53, several proapoptotic and antiapoptotic proteins of the Bcl-2 family, oncoproteins such as c-fos, c-jun and N-myc, and IκB, an inhibitory protein that maintains the transcription factor nuclear factor κB (NFκB) in an inactivated state in the cytoplasm under normal conditions [Adams, 2004; Bose et al. 2014]. Bortezomib induces tumor cell apoptosis in multiple lymphomas [Pham et al. 2003; Nasr et al. 2004; Sors, 2006; Bonvini et al. 2007; Olejniczak et al. 2010; Juvekar et al. 2011], primarily through NFκB inhibition achieved by decreased proteasome degradation of IκB. In addition, bortezomib is capable of killing B-cell NHL cells via caspase-independent mechanisms [Olejniczak et al. 2010].
Bortezomib induces cell-cycle arrest and apoptosis in MCL cells [Pham et al. 2003] and increased proteasomal degradation of p27 is associated with decreased OS in MCL [Chiarle et al. 2000]. MCL is characterized by constitutive activation of the NFκB pathway [Jares et al. 2012; Bose et al. 2014]. The ability of IκB kinase inhibitors to induce apoptosis in MCL cells in vitro validated NFκB as a therapeutic target in MCL [Pham et al. 2003; Roue et al. 2007]. Independent of inhibition of the NFκB pathway, it has been demonstrated that bortezomib induces apoptosis in MCL cells through reactive oxygen species generation and upregulation of the BH3-only proapoptotic protein Noxa, independent of p53 status [Perez-Galan, 2006; Rizzatti et al. 2008].
In addition, bortezomib has been shown to result in sensitization of malignant lymphoid cells to the cytotoxic effects of chemotherapy [Vega et al. 2008; Mozos et al. 2011] and glucocorticoids [Chandra et al. 1998]. This is mostly mediated by blocking the effects of NFκB activation, a physiological response to cellular stress, cell adhesion molecules and apoptosis inhibitors [Wang et al. 1996; Wang, 1998].
Peripheral neuropathy is a well recognized major adverse effect of bortezomib, with an incidence ranging between 31% and 45%, according to the results of major phase II/III clinical trials. Pretreatment with neurotoxic chemotherapeutic agents appears to increase the incidence neuropathy [Argyriou et al. 2014]. Subcutaneous administration of bortezomib has been shown to have similar efficacy but with a lower incidence of toxicity in patients with myeloma [Moreau et al. 2011], but the equivalency of subcutaneous bortezomib has not yet been widely demonstrated in the treatment of lymphoid malignancies.
The clinical pharmacokinetics of bortezomib are poorly understood, partly due to analytical challenges. Overall, the kinetic profile is characterized by a large volume of distribution and a high systemic clearance. The plasma elimination rate supports the current dosage schedule (twice weekly for 2 of every 3 weeks), which has been based on toxicity. The excretion routes remain to be determined, as well as the contribution of biotransformation to the plasma half life of bortezomib. No dose reduction is typically needed for renal dysfunction. Dose reduction is recommended for moderate or severe hepatic dysfunction (serum bilirubin >1.5 upper limit of normal), although dose can be escalated based on tolerance. No drug–drug interactions have been reported, but some might be expected given the metabolic profile of bortezomib [Leveque et al. 2007].
Upfront treatment of MCL in older patients: evolving role of bortezomib
Howard and colleagues published the results of a phase II study of R-CHOP for upfront treatment of MCL. This early study included 40 patients and suggested R-CHOP resulted in peripheral blood or bone marrow molecular response. However, no advantage in progression-free survival (PFS) was identified for patients who experienced molecular response [Howard et al. 2002]. The study did not report OS data, and was done before the currently used MIPI score was introduced as a mechanism of risk stratification for patients with MCL. A subsequent study by Lenz and colleagues compared CHOP with R-CHOP as first-line therapy for MCL. This larger study included 120 patients and demonstrated a significant improvement in overall (94% versus 75%) and complete response rates (34 versus 7%) in favor of R-CHOP. However, at the initial time of publication this study also failed to demonstrate an improvement in significant difference in PFS [Lenz et al. 2005]. A pooled analysis from the German Lymphoma Study Group (GLSG) comparing 386 patients assigned to either R-CHOP or CHOP found an OS benefit of R-CHOP, with median OS of 5.9 years versus 4.8 years in patients who received CHOP. In this analysis, baseline age and MIPI scores were similar between the two treatment groups [Hoster et al. 2014b].
As evidence gathered that in older patients, who were not deemed eligible for aggressive cytarabine-based immunochemotherapy with or without ASCT, R-CHOP was an appropriate option yet rate of progression remained high with poor long-term survival. Given the efficacy of bortezomib in relapsed or refractory MCL (detailed below), the LYM-3002 study was conducted to evaluate the use of bortezomib in the upfront setting [Robak et al. 2015]. Four hundred and eighty-seven previously untreated patients with MCL were randomized to either frontline R-CHOP or VR-CAP (substituting bortezomib, 1.3 mg/m2 intravenously on days 1, 4, 8, and 11, for vincristine; Table 1). An earlier study [Ruan et al. 2011] that combined bortezomib with R-CHOP in patients with untreated MCL or diffuse large B-cell lymphomas has shown a 64% incidence of neuropathy (8% grade 2 and 4% grade 3) which made the combination of vincristine and bortezomib less favored for future studies despite high-response rates observed (Table 1). Notably, rituximab maintenance was not employed in either group, as the benefit of rituximab maintenance after R-CHOP had not yet been demonstrated at the time of trial design [Kluin-Nelemans et al. 2012]. In addition, only patients considered unable to tolerate ASCT were eligible for the LYM-3002 study, based on advanced age and medical comorbidities. The majority (73%) of patients accrued were over 60 years old. Exclusion criteria were consistent with contemporary standards of care for ASCT in that setting [Vose, 2013; Rajabi and Sweetenham, 2015]. The VR-CAP arm had a significantly longer median PFS (24.7 versus 14.4 months) (Robak et al. 2015). Moreover, there was a nonsignificant trend towards improved OS after VR-CAP, with a 4-year OS of 64% versus 54% in the R-CHOP group [Robak et al. 2015]. It is worth noting that the PFS in the R-CHOP group was inferior to what has been reported in earlier trials (40.9 months) of similar patient population [Rummel et al. 2013]. In the LYM-3002 trial, disease progression was assessed by frequent computed tomography scanning, and the scans were interpreted through stringent, central radiographic review in which the interpreters of the scans were ‘blinded’ of group assignment which can explain the difference in PFS reported in other trials which relied on unblinded investigator assessment of progression. As observed in the landmark Nordic MCL network trials [Geisler et al. 2012], it is likely that an OS benefit will be demonstrated with longer follow up.
Bortezomib: upfront non-cytarabine based regimens.

In evaluable patients with MCL (n = 32).
For patients with MCL only.
CR, complete response rate; DLBCL, diffuse large B-cell lymphoma; Int, intermediate; IV, intravenous; MCL, mantle cell lymphoma; MIPI, Mantle cell International Prognostic Index; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PO, orally.
The VR-CAP group had significantly more hematologic toxicity, particularly thrombocytopenia, with 57% of patients experiencing grade 3 or higher thrombocytopenia. While incidence of infection was higher in the VR-CAP group (60% versus 46% in the R-CHOP group), the rate of events of febrile neutropenia grade 3 or higher was essentially the same in both groups (15% in patients with VR-CAP, 14% in patients with R-CHOP). Of note, peripheral neuropathy rates were 29% in the R-CHOP group and 30% in the VR-CAP group (grade ⩾ 3, 4% versus 8%). Peripheral neuropathy was reversible in the majority of patients, with a complete resolution rate of 75% in the R-CHOP group and 81% in the VR-CAP group, in a median of 5.5 months and 3.0 months, respectively. This study showed VR-CAP resulted in an impressive improvement in PFS, although it must be noted that the largest affect was in patients with low- and intermediate-risk MCL [Robak et al. 2015]. Given the positive results of the LYM-3002, both the US Food and Drug Administration and the European Commission have approved the use of bortezomib in upfront treatment of MCL.
Prior to the LYM-3002 study, Houot and colleagues studied the use of rituximab, bortezomib, doxorubicin, dexamethasone and chlorambucil (RiPAD+C) in 39 older patients with MCL (Table 1). The median age of patients was 72 years and most had intermediate or high MIPI scores. The overall response rate (ORR) was 79% with a complete response rate (CR) of 51% [Houot et al. 2012]. After a median follow up of 64 months, median PFS was 22 months, median OS has not been reached and the 3-year OS rate was 63.5% [Dubreuil et al. 2014]. The RiPAD+C treatment regimen had significant toxicities, with 45% of subjects experiencing some degree of neuropathy and 18% having grade 3 neuropathy. Further, two patients died of severe sepsis, and at the time of interim follow up, four deaths had been attributed to treatment toxicities [Houot et al. 2012; Dubreuil et al. 2014].
E1411 [ClinicalTrials.gov identifier: NCT01415752] is an ongoing phase II study by ECOG (Eastern Cooperative Oncology Group) comparing the efficacy of four different upfront treatment approaches in patients with MCL. Patients are randomized to bendamustine or rituximab (BR) induction with or without bortezomib. Patients will also be randomized, upfront, to a 2-year continuation with rituximab, with or without lenalidomide. In light of the favorable LYM-3002 results, E1411 may be helpful in determining the optimal maintenance regimen after bortezomib-based induction. However, the differences in induction regimens used for LYM-3002 and E1411 will make the choice of initial induction regimen for older patients with MCL more complicated.
Upfront treatment of MCL in younger patients: bortezomib plus cytarabine based chemoimmunotherapy and ASCT
Given the demonstrated single-agent activity of bortezomib in relapsed and refractory MCL, William and colleagues conducted a trial of the feasibility and efficacy of adding bortezomib to high-dose chemotherapy with BEAM (BCNU, etoposide, cytarabine, and melphalan) followed by ASCT (Table 2). The phase I portion of the study identified the maximum tolerated dose for bortezomib of 1 mg/m2 when given in combination with BEAM conditioning. In the ASCT context, the dose-limiting toxicities included gastrointestinal complications and peripheral neuropathy (21% with grade 3 or greater anorexia and 19% with grade 3 or greater peripheral neuropathy). The study accrued 42 patients with median age of 58 years: 52% had MCL in first complete remission (other NHL histologies were included). After a median follow up of 32 months, the estimated 5-year PFS and OS was 57% and 72%, respectively, for the 23 patients with MCL treated with ‘V-BEAM’ on the trial. Comparison with 26 matched historic controls who received BEAM followed by ASCT did not demonstrate statistically significant outcome differences [William et al. 2014].
Bortezomib: upfront cytarabine-based, transplant, and maintenance post-transplant regimens.
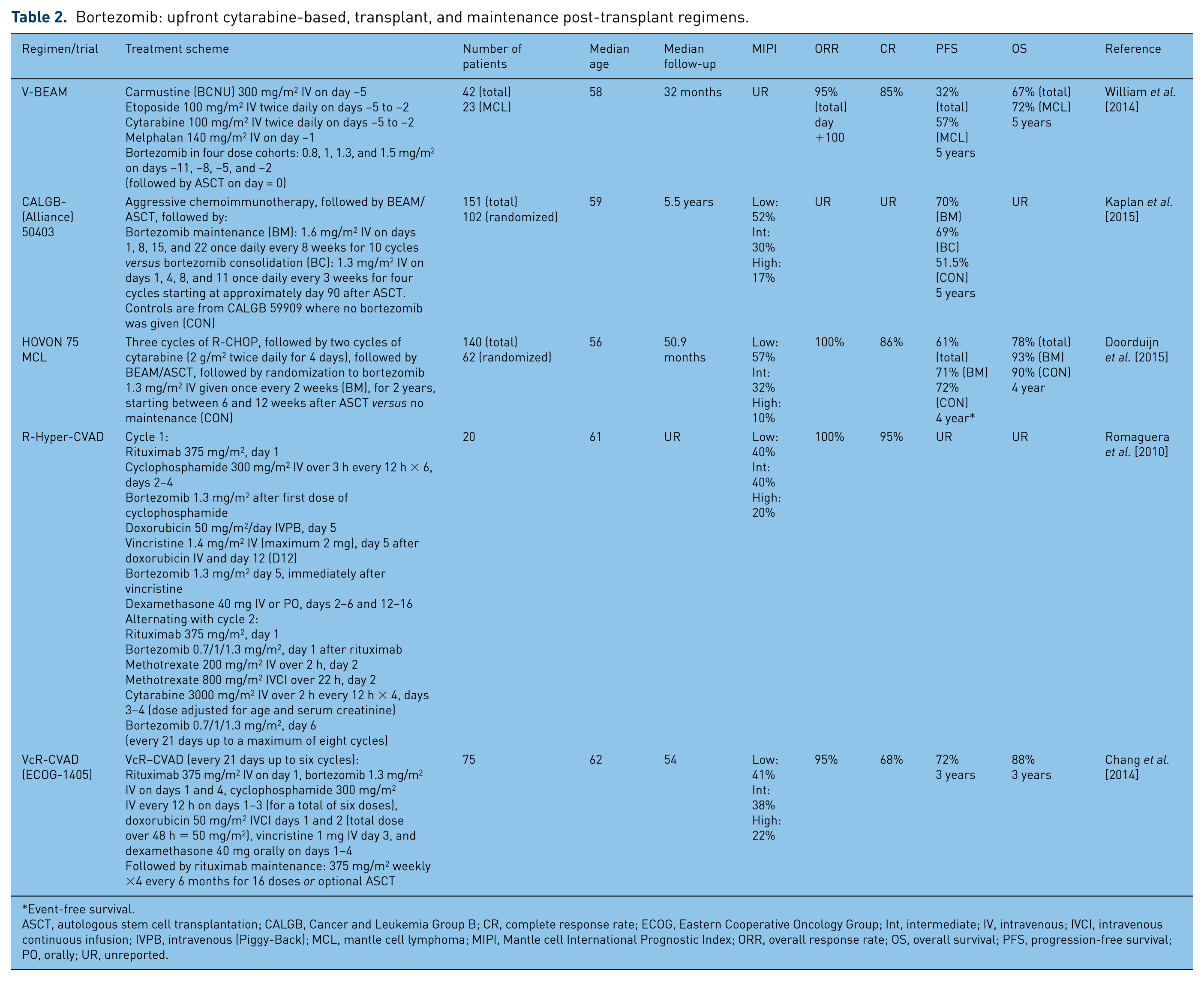
Event-free survival.
ASCT, autologous stem cell transplantation; CALGB, Cancer and Leukemia Group B; CR, complete response rate; ECOG, Eastern Cooperative Oncology Group; Int, intermediate; IV, intravenous; IVCI, intravenous continuous infusion; IVPB, intravenous (Piggy-Back); MCL, mantle cell lymphoma; MIPI, Mantle cell International Prognostic Index; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PO, orally; UR, unreported.
The CALGB (Alliance) 50403 trial randomized patients with MCL to maintenance (BM) versus consolidation bortezomib (BC) after cytarabine-based immunochemotherapy and ASCT (Table 2). After a median follow up of 5.5 years, the 5-year PFS estimates from study entry in the BM and BC arms were 70% and 69%, respectively. There was a nonsignificant trend toward more sensory neuropathy in the consolidation arm. Compared with nonrandomized controls from the CALGB 59909 trial, both the bortezomib arms on the 50403 trial (BM and BC) significantly improved PFS in an intention-to-treat analysis (72.7% versus 51.5%), favoring the 50403 trial which differed from 59909 only by the addition of post-transplant bortezomib. Minimal residual disease (MRD) results were available in 47 patients.Five-year PFS from study entry was 93% if MRD negative (n = 15) and 51% if MRD positive (n = 32) following induction chemoimmunotherapy [Kaplan et al. 2015]. Also, results from the similar European HOVON 75 MCL trial were recently reported in which patients with MCL were randomized to bortezomib maintenance or no maintenance after induction with R-CHOP and cytarabine followed by ASCT (Table 2). Peripheral neuropathy was observed in the maintenance arm (14% grade 2 and 4% grade 3). Event-free survival (EFS) at 4 years for all patients is 61%, and the OS 78%. The median follow up of the randomized patients still alive was 42.9 months. The EFS at 4 years is 72% without maintenance versus 71% with bortezomib maintenance. The OS at 4 years also shows no significant difference between the groups, 90% versus 93% respectively [Doorduijn et al. 2015]. Based on these conflicting results, the role of maintenance bortezomib remains undefined in MCL after ASCT.
Khouri and colleagues reported the outcomes of 25 previously untreated patients who received Hyper-CVAD alternating with high-dose methotrexate and cytarabine (M/A), followed by ASCT or allogeneic stem cell transplantation. At 3 years median follow up, OS was 92% and EFS was 72% [Khouri et al. 1998]. Romaguera and colleagues added bortezomib to the standard R-Hyper-CVAD regimen at a dose of 1.3 mg/m2 intravenously on days 2 and 5, with escalation of bortezomib after the alternating R-MA regimen, in doses of 0.7, 1, and 1.3 mg/m2 (Table 2). The principal toxicity was haematologic and did not differ from that observed with R-HyperCVAD-MA without bortezomib [Romaguera et al. 2010].
Subsequently, Chang and colleagues reported the outcomes of the ECOG-1405 trial combining bortezomib with rituximab, cyclophosphamide, doxorubicin, vincristine and dexamethasone (VcR-CVAD) (Table 2) followed by either 2-year rituximab maintenance (MR) or ASCT in patients who achieved a CR or partial remission. Choice of consolidation was based on patient or physician preference. A total of 75 eligible patients with a median age of 62 (range 40–76) years were enrolled. The ORR was 95%, with 68% of patients achieving a CR. After a median follow up of 4.5 years, 3-year PFS and OS were 72% and 88%, respectively. No substantial difference in PFS or OS was observed between patients treated with MR (n = 44) versus ASCT (n = 22). There were no unexpected toxicities with this regimen [Chang et al. 2014]. Table 2 summarizes the trials investigating the addition of bortezomib to cytarabine-based immunochemotherapy regimens
The LYM-3002 clinical trial specifically excluded younger patients eligible for cytarabine-based immunochemotherapy regimens typically followed by consolidation with ASCT. Therefore, this strategy remains a standard of care for younger patients with MCL with intermediate- to high-risk disease. The results of a 6-year follow up of the Nordic MCL2 trial of upfront treatment with cytarabine-based immunochemotherapy followed by consolidation with ASCT showed that more than 70% of patients with low to intermediate MIPI were alive at 10 years, but only 23% of the patients with high MIPI remained alive at the same time point [Geisler et al. 2012].
Historically, the higher nonrelapse mortality rates observed with allogeneic stem cell transplant has been a deterrent from using this treatment modality in the upfront setting [Chaudhary et al. 2013; Fenske et al. 2014]. However, nonmyeloablative allogeneic stem cell transplantation (NMA SCT) was shown to achieve durable remissions in 20–25% of patients with chemorefractory disease [Hamadani et al. 2013]. Given the increasing safety of NMA SCT in patients with relapsed or refractory MCL, this strategy has become an increasingly attractive alternative, as it capitalizes on the immunologic graft versus lymphoma effect.
Bortezomib for relapsed and refractory MCL
The landmark multicenter PINNACLE trial enrolled 155 patients with MCL who experienced relapse or progression after one to three lines of therapy. Median age was 65 years, 44% had an IPI score of at least 3 (the study was conducted prior to widespread use of MIPI), 55% presented bone marrow involvement, and 96% of patients had received a prior regimen including rituximab. Patients received bortezomib, 1.3 mg/m2 on days 1, 4, 8, and 11 in 21-day cycles. Treatment continued until progressive disease or toxicity limitations were met [Fisher et al. 2006]. The ORR was 32% with 8% CR/unconfirmed CR [Fisher et al. 2006; Goy et al. 2009]. In an updated analysis, the median time to progression was 6.7 months, and median OS was 23.5 months [Goy et al. 2009]. Toxicities associated with single-agent bortezomib were consistent with prior clinical trials conducted in myeloma. Of note, 13% of patients experienced grade 3 or higher peripheral neuropathy. Grade 3 or greater lymphopenia was seen in 34%, as could be expected in such a heavily pretreated patient population. It is of interest that similar response rates were observed in patients with relapsed and refractory MCL, suggesting that there may be little cross resistance with other conventional cytotoxic agents and that patients with MCL with refractory or poorly responsive disease may still derive meaningful clinical benefit from treatment with bortezomib [O’Connor et al. 2009].
Given the significant activity and tolerability of BR in patients with relapsed MCL, and the potential synergy between chemotherapy agents and bortezomib, the combination of bendamustine, bortezomib, and rituximab (BVR) was investigated in 30 patients with relapsed indolent lymphoma and MCL (seven had MCL). The ORR was 71% in patients with MCL. After a median follow up of 29 months, 2-year PFS for the entire cohort of 29 evaluable patients was 47%. Most toxicities observed were hematologic and nonhematologic toxicities were rare [Friedberg et al. 2011]. Given the high efficacy and tolerability of the BVR regimen, it has quickly gained popularity as a first-line salvage regimen in patients with relapsed MCL treated initially with either R-CHOP or cytarabine-based chemoimmunotherapy. BVR is currently being examined in the upfront treatment of MCL in the E1411 trial (as detailed above).
Other combinations of bortezomib and cytotoxic chemotherapy in relapsed and refractory MCL were proposed (Table 3); BVR remains the most popular in the USA. Furtado and colleagues randomized 46 patients with MCL in first relapse to CHOP chemotherapy with or without bortezomib. Median age was 71 years in the CHOP arm and 69 years in the CHOP–bortezomib arm, and 95% of patients in each arm received fludarabine-based therapy and were not consolidated with ASCT upfront. The trial was terminated early because of significantly superior ORR to CHOP–bortezomib (82.6%) compared with CHOP (47.8%), as well as superior OS (35.6 versus 11.8 months, respectively). More patients in the CHOP–bortezomib arm developed sensory neuropathy than in the CHOP arm (23.9% versus 10.9% respectively), however the majority of this was low grade. The incidence of severe (⩾ grade 3) sensory neuropathy was similar in both arms. The other reason for early termination of the trial was the growing evidence that rituximab improves survival in MCL (rituximab was not part of standard treatment of MCL in the UK), therefore changing the standard of care to a rituximab-containing regimen and decreasing the validity of the comparison [Furtado et al. 2015].
Bortezomib: relapsed or refractory regimens.

ASCT, autologous stem cell transplantation; CALGB, Cancer and Leukemia Group B; CR, complete response rate; IV, intravenous; IVCI, intravenous continuous infusion; MCL, mantle cell lymphoma; NCIC, National Cancer Institute of Canada Clinical Trials Group; ORR, overall response rate; OS, overall survival; PO, orally; PFS, progression-free survival; UR, unreported.
Including all ‘high-intensity regimens’ with or without ASCT (Hyper-CVAD, ICE, ESHAP, or DHAP, all with or without rituximab [Fisher et al. 2006]).
Given the single-agent activity of lenalidomide in relapsed or refractory MCL [Wang et al. 2012; Goy et al. 2013], and the demonstrated tolerability of the bortezomib–lenalidomide combination in plasma cell myeloma [Richardson et al. 2009], the CALGB 50501 trial examined this combination in a phase II study that included 53 patients with relapsed or refractory MCL. Median age was 67 years and 40% had prior ASCT. The ORR of 40% observed for the combination was disappointing and may have been secondary to insufficient lenalidomide dosing and dose reductions in both agents related to toxicity. The limited amount of protocol therapy, administered in only 37% of patients, was related to disease progression and tolerability issues [Morrison et al. 2015].
Weigert and colleagues treated eight patients with relapsed or refractory MCL with a combination of bortezomib and high-dose cytarabine. Median age was 65, all patients had received first-line CHOP and two patients were consolidated with ASCT. As expected, grade 3 or 4 hematologic toxicity was observed in all patients. Overall response was 50%, with 25% CR. Median PFS and OS were 5 and 15.5 months, respectively [Weigert et al. 2009]. Kouroukis and coworkers treated 26 patients with relapsed or refractory MCL with a combination of bortezomib and gemcitabine. Overall response was 60% and median PFS was 11.4 months. Unfortunately, 40% of patients experienced grade 3 or 4 thrombocytopenia and 48% has experienced 3 or 4 granulocytopenia [Kouroukis et al. 2011]. Despite their high activity in relapsed or refractory MCL, the significant hematologic toxicity of these regimens prohibits further development, given the availability of other active and much less toxic regimens. Multiple other bortezomib-based combinations are in clinical trials for the treatment of MCL and other B-cell malignancies.
Conclusion
The efficacy of bortezomib in relapsed and refractory MCL has been supplemented by recent studies showing significant activity and a tolerable side-effect profile in the upfront setting. The high response rates and good tolerability of VR-CAP makes this regimen an attractive alternative for first-line use in older and unfit subjects who cannot tolerate cytarabine-based chemoimmunotherapy regimens. Given the demonstrated survival benefit of cytarabine-based chemoimmunotherapy regimens in younger, medically fit patients (as demonstrated in the Nordic MCL network trials), these aggressive regimens remain the recommended upfront treatment choice for this patient population. The benefit of consolidation with high-dose chemotherapy and ASCT after cytarabine-based chemoimmunotherapy is questionable, but is still offered to medically fit and motivated, patients. The emergence of highly active, less toxic regimens such as BVR may permit similarly good outcomes in younger patients while sparing them the toxicity of cytarabine-based regimens and ASCT. However, this remains to be demonstrated and it is expected that ongoing trials including E1411 can help clarify the role of these less toxic regimens. The emergence of novel, targeted agents, including BTK and phosphoinositide 3-kinase (PI3K) inhibitors suggests the optimal treatment of MCL will remain a rapidly evolving field over the next few years.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
