Abstract
The management of acute lymphoblastic leukemia (ALL) during pregnancy requires treatment with high-dose chemotherapy that can pose risks to both the mother and fetus. Special consideration to chemotherapy regimen and its doses and to fetal gestational age at the time of chemotherapy administration should be taken in order to limit fetal exposure while still providing optimal therapy to the mother. Here we describe a 22-year-old patient who was diagnosed at 26 weeks gestation with ALL and was treated in the third trimester with HyperCVAD (cytoxan, vincristine, adriamycin, dexamethasone) combination chemotherapy giving birth via Caesarean section to a healthy baby girl 4 weeks after induction chemotherapy.
Introduction
The occurrence of cancer and pregnancy is relatively rare, occurring in about one in 1000 pregnancies [Donegan, 1983]. The most common tumors diagnosed during pregnancy are breast and cervical cancer followed by melanoma, leukemia and lymphoma.
The management of a pregnant patient with malignancy is very challenging and requires a multidisciplinary approach. It is best to avoid chemotherapy during the first trimester as harmful effects to the fetus are highest during the period of organogenesis. Giving chemotherapy during the second and third trimesters of pregnancy has been widely reported, however, there are case reports of growth restriction, intrauterine and neonatal death, prematurity and myelosuppression.
In this paper we will present our patient together with a concise literature review of acute lymphoblastic leukemia (ALL) treatment during pregnancy and a review of long-term outcomes of children exposed to chemotherapy in utero.
Case report
A 22-year-old female primigravida at 26 weeks gestation presented to the emergency department with a 5-day history of fever, fatigue, bruising along with nose and gum bleeding. Laboratory investigation revealed a white blood cell count of 76.2 × 103/mm3, hemoglobin 3.6 (g/dl) and platelets 6 × 103/µl. A bone marrow biopsy revealed precursor B-cell ALL, 95% blasts with BCR-ABL rearrangement (Philadelphia chromosome) by fluorescent in situ hybridization (FISH) in 82% of cells and reverse transcriptase-polymerase chain reaction (RT-PCR) detecting BCR-ABL breakpoint fusion.
Cytogenetics revealed 46,XX,t(9;22)(q34;q11.2) and immunohistochemistry revealed the following: moderately positive for CD10, CD19, CD22, CD34, CD45, HLA-DR and cytoplasmic Tdt while negative for CD3, CD5, CD11b, CD15, CD20, CD33, CD38, CD56, CD71, CD117, kappa and lambda light chain surface antigen.
Ultrasound revealed a single living fetus at 26 weeks with weight corresponding to the 39th percentile and normal amniotic fluid. The patient received red blood cell and platelet transfusions.
The decision was made to wait until 30 weeks to deliver the baby via Caesarean section due to an operative risk of hemorrhage and sepsis to the mother if delivered while pancytopenic and to give more time for fetal maturity. The patient was started on intravenous HyperCVAD (cytoxan, vincristine, adriamycin, dexamethasone) combination chemotherapy (cyclophosphamide 300 mg/m2 q12h days 1–3, mesna 450 mg/m2 days 1–3, doxorubicin 50 mg/m2 over 24 hours on day 4, vincristine 2 mg on day 4 and day 11, and oral dexamethasone 40 mg days 1–4 and days 11–14) without any dose reduction. The patient was started on diflucan and acyclovir for initial prophylaxis. No prophylactic bactrim was given as delivery was planned in 4 weeks.
At 30 weeks gestation (4 weeks after initiation of induction therapy), a Caesarean section was performed with the delivery of a baby girl with a weight of 1346 grams corresponding to the 26th to 50th percentile for the gestational age. The baby required transient respiratory assistance for 12 hours post delivery due to cyanosis and poor respiratory effort.
On postpartum day 10 the mother was started on HyperCVAD combination chemotherapy and dasatinib at 50 mg twice a day for 14 days given with each cycle of chemotherapy. She was also given prophylactic intrathecal chemotherapy. Cerebrospinal fluid analysis done after delivery was negative for central nervous system involvement.
Each cycle of HyperCVAD chemotherapy was alternated with a cycle of intravenous chemotherapy with high-dose methotrexate 200 mg/m2 on day 1 and cytarabine 3 g/m2 every 12 hours on days 2–3 along with leucovorin 800 mg/m2. A bone marrow biopsy performed after completion of cycle one of high-dose cytarabine and methotrexate revealed normal cytogenetic. BCR-ABL was negative by FISH and PCR detected 0.005% residual BCR-ABL. No blasts in bone marrow biopsy were detected and flow cytometry was negative.
After the second cycle of high-dose cytarabine and methotrexate combination the patient was admitted with septic shock, pancytopenia and Clostridium difficile colitis. She required mechanical ventilation with vasopressor support along with broad spectrum antibiotics.
It was decided after she recovered from this life-threatening episode to stop her chemotherapy with HyperCVAD and start maintenance chemotherapy with vincristine 2 mg on day 1 with oral prednisone 200 mg on days 1–5, repeated every 28 days, along with continuous dasatinib given at a dose of 150 mg orally every day.
A bone marrow biopsy performed after the first month of maintenance therapy was normal with negative FISH, PCR and normal cytogenetics.
The treatment plan formulated was to repeat HyperCVAD at months 7 and 13 of maintenance treatment with continued maintenance with vincristine/prednisone for 2 years and to continue dasatinib indefinitely [Ravandi et al. 2010]. She developed recurrence of Ph+ ALL after 16 months of initial remission induction and attaining a complete molecular response. Testing for ABL Kinase Domain Mutation analysis by sequencing was positive for F317L which is a common mutation leading to dasatinib resistance [Khorashad et al. 2013]. She was started on ponatinib at the time of recurrence and after 1 month attained a hematological complete response on flow cytometry. Currently she is awaiting allogenic bone marrow transplant [Cortes et al. 2012].
The baby continues to do well and has reached normal developmental milestones at 16 months of age.
ALL in pregnancy
Acute leukemia is uncommon during pregnancy. It affects approximately 1 in 75,000 pregnancies [Terek et al. 2003]. A total of 28% of leukemia cases diagnosed during pregnancy are ALL while acute myeloid leukemia (AML) and chronic myeloid leukemia (CML) represent the remainder [Terek et al. 2003]. A literature review of case reports of ALL during pregnancy is shown in Table 1. A total of 17 patients received high-dose chemotherapy between gestational age of 15 and 33 weeks. Chemotherapy was mainly given during the second and third trimester. Chemotherapy regimens varied, with most regimens including anthracyclines, vincristine and steroids. While most patients’ long-term outcomes are unclear, about half of the women in this review achieved remission and the other half either relapsed or died from progression of the disease. Some of the infants were born with few adverse outcomes including transient pancytopenia, respiratory distress and preterm delivery.
Case reports of acute lymphoblastic leukemia treated during pregnancy.
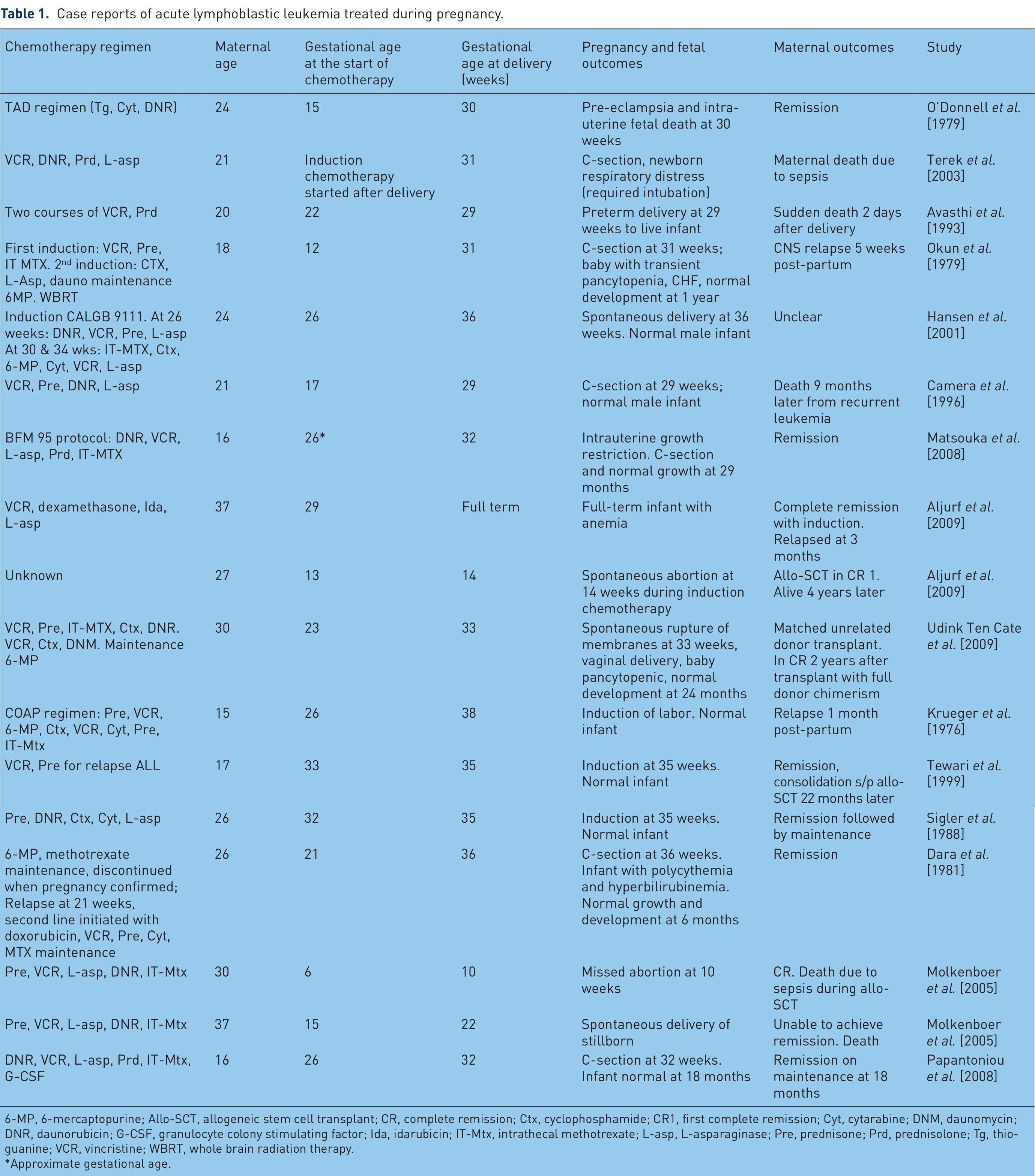
6-MP, 6-mercaptopurine; Allo-SCT, allogeneic stem cell transplant; CR, complete remission; Ctx, cyclophosphamide; CR1, first complete remission; Cyt, cytarabine; DNM, daunomycin; DNR, daunorubicin; G-CSF, granulocyte colony stimulating factor; Ida, idarubicin; IT-Mtx, intrathecal methotrexate; L-asp, L-asparaginase; Pre, prednisone; Prd, prednisolone; Tg, thioguanine; VCR, vincristine; WBRT, whole brain radiation therapy.
Approximate gestational age.
Tyrosine kinase inhibitor use during pregnancy
There are few case reports of women being treated with tyrosine kinase inhibitors (TKI) during pregnancy. Owing to the longer history of patients treated with imatinib, there are more reports on pregnancy outcome after gestational exposure to imatinib compared with the newer TKIs. A review of pregnancy outcomes of 180 females exposed to imatinib during pregnancy was performed [Pye et al. 2008]. The review found 50% of the patients with known outcomes (125 out of 180) delivered normal infants, and 28% underwent elective terminations, 3 following identification of abnormalities. Abnormalities were identified in 12 infants with 9 of those defects occurring in the infants with known first trimester exposure to imatinib. The review concluded that most pregnancies exposed to imatinib are likely to have successful outcome but fetal malformations remain a risk.
Cases of exposure to newer TKI’s during pregnancy have also been reported. One case of exposure to nilotinib during the first trimester was described in a patient who unexpectedly became pregnant while taking nilotinib 200 mg orally twice a day until 7.4 weeks of pregnancy [Conchon et al. 2009]. She delivered a baby with no congenital malformation identified. The baby was shown to have normal development when followed longitudinally [Conchon et al. 2009].
A case of fetal exposure to dasatinib during pregnancy was also reported in a female patient with CML intolerant to imatinib who was treated with dasatinib 70 mg orally twice a day when she became pregnant unexpectedly [Conchon et al. 2010]. It was estimated the patient had been on dasatinib for 4 weeks while pregnant. The patient gave birth to a healthy male baby at 33 weeks with no congenital malformation [Conchon et al. 2010].
Long-term outcomes of children after exposure to chemotherapy in utero
The risks of in utero exposure to chemotherapy are expected to include abnormalities in growth, organogenesis, intellect and reproduction [Garber, 1989]. Overall, long-term follow-up reports show few side effects. Most of these reports come from observation following the treatment of pregnant patients with breast cancer, lymphomas, leukemia and other solid tumors [Gulati et al. 1986; Partridge et al. 2000; Aviles et al. 2001; Peres et al. 2001; Hahn et al. 2006; Ali et al. 2009; Cardonick et al. 2010]. Many of the studies reported that these children have normal height and weight compared with controls, have no abnormal behavior or learning deficit, and have normal psychological and neurological evaluations [Peres et al. 2001; Hahn et al. 2006; Cardonick et al. 2010].
Chemotherapy regimens during pregnancy for breast cancer, lymphoma and leukemia included anthracyclines, antimetabolites and alkylating agents. Doxorubicin has been the most common drug used during pregnancy with most reports showing no associated cardiac abnormalities, although one study reported on a trend towards a reduction in both wall thickness and left ventricular mass index [Aviles et al. 2006; Van Calsteren et al. 2006]. Some studies reported on children exposed to chemotherapy in utero developing reactive airway disease and recurrent otitis media [Garber, 1989; Aviles et al. 2001; Hahn et al. 2006; Cardonick et al. 2010].
Discussion
Cancer and especially ALL is not a common occurrence during pregnancy. Treatment of any malignancy during pregnancy represents a great challenge to the physician, patient and fetus. Oncologists over the last two decades have started to treat pregnant patients with combination chemotherapy during the second and third trimesters with frequent safe outcomes to the mother and infant. Also, long-term follow up for children exposed to chemotherapy in utero has shown few minor long-term side effects.
Treatment of acute leukemia during pregnancy remains an even greater challenge than treating solid tumors or lymphoma due to the requirement of using much higher doses of induction chemotherapy. There are few reports of using such high-dose chemotherapy in treating leukemia or high-grade lymphoma with good outcomes.
Our patient represents the great challenge in treating leukemia during pregnancy. When she presented at 26 weeks gestation she was extremely pancytopenic with signs of bleeding predicting only supportive treatment with blood product transfusions and antibiotics would not be sufficient to support the pregnancy for an extended period of time. Also delivering the baby with Caesarean section at that stage with such degree of pancytopenia would have been accompanied by a high risk of bleeding, infection and possible death to the mother and risks of immaturity to the infant.
A decision to proceed with induction chemotherapy was reached to avoid the risks of Caesarean section and to provide more time for fetal maturity. In addition, we predicted there would be a good chance the leukemia will attain morphological remission after first induction chemotherapy and it will be optimal to deliver the baby with Caesarean section at 30 weeks of gestation.
We elected HyperCVAD combination therapy for induction treatment because doxorubicin, cyclophosphamide and vincristine were frequently used during pregnancy mainly in treating breast cancer and lymphoma with good outcomes and due to the effectiveness of the regimen in treating adult ALL.
We were aware that the dose of each agent in the HyperCVAD combination is nearly three times higher than the regular doses used to treat breast cancer and lymphoma during pregnancy. The main worry was that the baby would be born with pancytopenia although we felt 4 weeks after induction chemotherapy would be enough time for hematopoietic recovery for the mother and the baby.
Other ALL remission induction regimens are frequently used. These regimens include three drugs (vincristine, steroids and asparaginase) in standard-risk ALL and four drugs (vincristine, steroids, asparaginase and anthracyclines) in high-risk ALL [Pui et al. 2006, 2010; Moricke et al. 2010; Silverman et al. 2010]. Common side effects of asparaginase are pancreatitis, hypofibrinoginemia and bleeding. Any of these side effects of asparaginase occurring during pregnancy will have deleterious effects on outcomes. In addition, these induction regimens may take a longer time to achieve complete response compared with HyperCVAD.
We did not use dasatinib during pregnancy as there is no long-term record of safety. The first-generation TKI, imatinib, has been reported in 180 pregnant women yet there were fetal abnormalities in 12 infants which raises concern when considering the use of imatinib during pregnancy [Pye et al. 2008]. In cases of chronic myeloid leukemia, the benefits of continuing imatinib versus the risks should be discussed with the patient.
The leukemia was in morphologic remission at the time of the Caesarean section and the baby had normal blood counts upon delivery. After delivery we elected to repeat another cycle of HyperCVAD rather than starting the alternate combination of high-dose cytarabine and methotrexate as it is less myelosuppressive leading to reduced risk of infection and bleeding while the patient is recovering from the Caesarean section.
Other issues related to treatment of Philadelphia chromosome positive ALL are related to requirement of allogenic bone marrow transplant although with the addition of TKIs to high-dose chemotherapy there are reports of long-term remission without proceeding to transplant [Foa et al. 2011].
Finally, each pregnant patient with malignancy represents a peculiar challenge requiring the treating oncologist to take into consideration the gestational age at presentation, the type of malignancy and the chemotherapy regimen and its doses while focusing on optimal outcome for both the mother and the infant.
Our case represents the feasibility of treating ALL with HyperCVAD during the third trimester of pregnancy with optimal outcomes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors
Conflict of interest statement
The authors declare that there are no conflicts of interest to disclose.
