Abstract
Background
High-dose chemotherapy with melphalan (HD-Mel) followed by autologous hematopoietic stem-cell transplantation (autoSCT) has become the standard treatment for younger and/or medically fit patients with multiple myeloma (MM). This treatment approach is based on trials that demonstrated improved overall survival (OS) in comparison with conventional chemotherapy [Child et al. 2003; Harousseau and Moreau, 2009]. In addition, data from the IFM group favoured a tandem transplant approach in comparison with a single course of HD-Mel/autoSCT [Attal et al. 2003]. Although high-dose therapy and autoSCT improved OS in comparison with standard dose chemotherapy, for most MM patients this treatment is not curative and a high portion of patients relapse and finally die from the disease. A curative option, allogeneic SCT (alloSCT), is restricted to young and medically fit patients having a human leukocyte antigen (HLA)-identical donor. The routine use is still a matter of debate because of high transplantation-related morbidity and mortality as well as high relapse rates [Bruno et al. 2007].
Novel therapies with the immunomodulatory drugs thalidomide [Dimopoulos and Anagnostopoulos, 2003] and lenalidomide [Dimopoulos et al. 2007], and the proteasome inhibitor bortezomib [Richardson et al. 2005] have been studied extensively in patients with relapsed or refractory disease. However, the median time to progression (TTP) is less than 1 year with these new agents for relapsed or refractory MM [Palumbo and Anderson, 2011].
For patients who have relapsed late after initial transplantation retreatment followed by late high-dose therapy and autoSCT can be applied, and the procedure is safe with regard to toxicity and reasonably effective with reported TTP of more than 12 month for patients with a long interval between the first and later HD-Mel and autoSCT [Elice et al. 2006; Kumar et al. 2008; Olin et al. 2009]. The term ‘long interval’ is however not clearly defined and some authors state that a long interval is >12 months, others suggest >18 months from completion of first intensive treatment [van de Donk et al. 2011]. In addition, the role of the novel substances in the re-induction before late high-dose therapy and autoSCT is unclear.
Here we report efficacy and survival data of a series of 24 consecutive patients who were treated with a late HD-Mel chemotherapy followed by autoSCT after initial intensive treatment. Importantly, only three of the patients received a novel drug in the first-line treatment and all but one patient received a novel drug for remission induction before late HD-Mel and autoSCT.
Patients and methods
Study design
All consecutive patients with relapsed MM, who had received late repeat myeloablative chemotherapy after initial myeloablative chemotherapy and autoSCT were included into this retrospective study. All patients were treated at the University Hospital of the Technische Universität München. Patients were treated within clinical trials or following local standard (at the time) treatment protocols. All patients gave their informed consent. Follow-up data from medical records were collected until February 2011 with a median follow up of 99 months (range 21–156). Patient characteristics are given in Table 1.
Patient characteristics.

Conditioning regimen, CD34+ dose and supportive care
The HD-Mel regimen consisted of either infusional melphalan 100 mg/m2 day 1 + day 2 (Mel200) for patients with an Eastern Cooperative Oncology Group Performance Status (ECOG PS) ≤1 and age ≤60 years or melphalan 70 mg/m2 day 1 + day 2 (Mel140) for patients older than 60 years. The initial tandem treatment included a second course of HD-Mel treatment if patients did not achieve a very good partial remission (VGPR) after the first melphalan treatment or refused the second treatment. In case of a tandem transplant the interval between the first and second HD-Mel was less than 12 weeks. All patients received antibiotic prophylaxis with ciprofloxacin 500 mg twice daily from start of melphalan treatment until change of antibiotics due to fever or until recovery to absolute neutrophil counts >500/µl. We recommended post-HD-Mel/autoSCT prophylaxis with valaciclovir (500mg twice daily) for 3 months after CD34+ reinfusion. Bisphosphonate treatment was continued monthly and extended to every 3 months for patients in stable remission.
Clinical outcome and response assessment
Data were collected for the occurrence of progression defined as evidence of a new monoclonal gammopathy for patients in complete remission (negative immunofixation) or an increase in paraprotein levels of ≥25% above the lowest value achieved after HD-Mel/autoSCT for patients in VGPR, partial remission (PR) or stable disease (SD) according to the international uniform response criteria for MM [Durie et al. 2006]. Progression-free survival (PFS) was defined as the time interval from the date of transplantation to the date of progression. Time to next treatment (TTNT) was defined as the interval from the date of transplantation (first or second if performed) until the next MM-specific treatment (drug treatment or irradiation) was initiated. OS was defined as the time from the beginning of initial myeloma-specific treatment until death due to any cause or last follow up.
Statistical analysis
Median OS, PFS and TTNT were calculated using the Kaplan–Meier method. Cox-proportional hazard models were employed to estimate hazard ratios with 95% confidence intervals (CIs). Owing to the limited sample size and therefore to avoid over fitting of regression models, no multivariable analysis was performed. A two-sided p-value of < 0.05 was considered statistically significant.
Results
Patient characteristics
We identified 24 consecutive patients who underwent a late HD therapy supported with autoSCT. The median age was 64 years (range 45–73 years). The median follow up of patients was 99 months (range 21–156 months). Data on cytogenetic analysis are available for 14 patients (Table 1). Treatment was initiated for stage II and III disease according to Salmon and Durie criteria. Importantly, only 3 patients (12.5%) had bortezomib (n = 1) or thalidomide (n = 2) before initial HD-Mel and autoSCT, while 23 patients (95.8%) had novel drugs before the late HD-Mel and autoSCT treatment. Patients received a median of one treatment line before the initial HD-Mel/autoSCT. A total of 17 patients (70.8%) received an initial tandem transplant (Table 2). All 24 patients received salvage treatment before late HD-Mel/autoSCT. The median interval between the end of first intensive treatment and the start of re-induction upon relapse or progression was 24 months (range 7–119 months; 95% CI 14.4–33.6 months). The median TTNT after late autoSCT was 21 months (range 1–25 months, 95% CI 10–32 months). Prerequisites for proceeding to allogeneic SCT were: (1) Achieving at least a PR after late autoSCT; (2) availability of an HLA-identical donor; (3) ECOG performance status of 0 or 1; (4) written informed consent. Nine of the 24 (37.5%) patients met all criteria and received an alloSCT after reduced intensity conditioning (RIC). All details regarding patient characteristics are given in Table 1, all treatments are listed in Table 2.
Treatment

Ida/Dex (Idarubicin, Dexamethasone), HD-Mel 200 (140) Melphalan 100mg (70mg) day1+day2+autoSCT, Vel/Dex (Bortezomib, Dexamethasone), Thal/Dex (Thalidomide, Dexamethasone), Len/Dex (Lenalidomide, Dexamethasone), IEV (Ifosfamide, Epirubicin, Etoposide), EID (Epirubicin, Ifosfamide, Dexamethasone), VAD (Vincristin, Adriamycin, Dexamethasone), Thal (Thalidomide), BAD (Bortezomib, Doxorubicin, Dexamethasone), EDAP (Etoposide, Cytarabine, Cisplatin), Benda/Thal (Bendamustine, Dexamethasone, Thalidomide), HyperCDT (Cyclophosphamide, Dexamethasone, Thalidomide), Cyclo (Cyclophosphamide), Mel/Pred (Melphalan, Prednisone), Dex (Dexamethasone), Benda (Bendamustine), Len (Lenalidomide), IFN (Interferon), TBI (total body irradiation)
Toxicity and response to HD-Mel/autoSCT, survival and TTNT
The toxicity of initial versus late HD-Mel/autoSCT was not significantly different with regard to nonhematological toxicity grade III/IV (67% and 65% versus 79%, for first and second versus late HD-Mel/autoSCT; see Table 3). After late HD-Mel and autoSCT one patient died early (before engraftment) due to H1N1 infection with acute hemorrhagic pneumonia and subsequent multi-organ failure before H1N1 vaccination was available.
Toxicity and response.

Stem-cell mobilization was chemotherapy plus granulocyte colony stimulating factor (G-CSF) in all patients. In 20 patients chemotherapy for stem-cell mobilization was the IEV regimen (ifosfamide, epirubicin and VP-16) [Straka et al. 2003]. Two patients received cyclophosphamide, one patient idarubicin and one patient epirubicin and ifosfamide. We generally aimed to collect two autologous grafts and one back-up graft. In all but two patients we used the back-up graft for the late autoSCT. In two cases we remobilized with cyclophosphamide for the late HD-Mel/autoSCT. The number of CD34+ cells reinfused was not significantly different with median 4.2 × 106, 4.6 × 106 and 3.65 × 106 CD34+ cells/kg body weight (BW) for first, second and late autoSCT. There was no significant difference in the time to platelet recovery to > 20 G/l and/or transfusion independency (median 11, 10.5 and 12 days) or absolute neutrophil count (ANC) recovery to > 1 G/l (median 12, 10.5 and 11 days) with regard to initial first, second versus late autoSCT (Table 3).
Of the 24 patients included into this analysis, 17 (70.8%) initially received a double autoSCT (Table 1). Three patients achieved a CR after the first autoSCT, and two additional patients after the second initial autoSCT (resulting in a total CR rate of 20.8%). Seven patients achieved a VGPR after the first initial autoSCT and 11 patients after first plus second initial autoSCT (total 45.8% VGPR). The overall response rate (ORR) to initial treatment was 100%. A total of 23 of 24 patients were evaluable for response after late autoSCT, one patient was not evaluable due to early death. One (4.2%) patient achieved a CR, 9 (37.5%) a VGPR and 11 (45.8) a PR. One patient achieved SD. The ORR (PR + VGPR + CR) after salvage therapy and late autoSCT was 87.5% (Table 3).
The median OS of the whole cohort was 90 months (95% CI 52.2–127.8, Figure 1). The median PFS after initial HD-Mel/autoSCT was 19 months (95% CI 13–25) compared with 13 months (95% CI 5.7–20.3) after late HD-Mel/autoSCT (Figure 2A). The median TTNT was 25.5 months after initial HD-Mel/autoSCT and 21 months after late HD-Mel/autoSCT (Figure 2B). At the time of final analysis 13 patients were alive without evidence of progression, which includes five of nine patients who underwent alloSCT after the late autoSCT (Figure 2, Table 2 lower part).
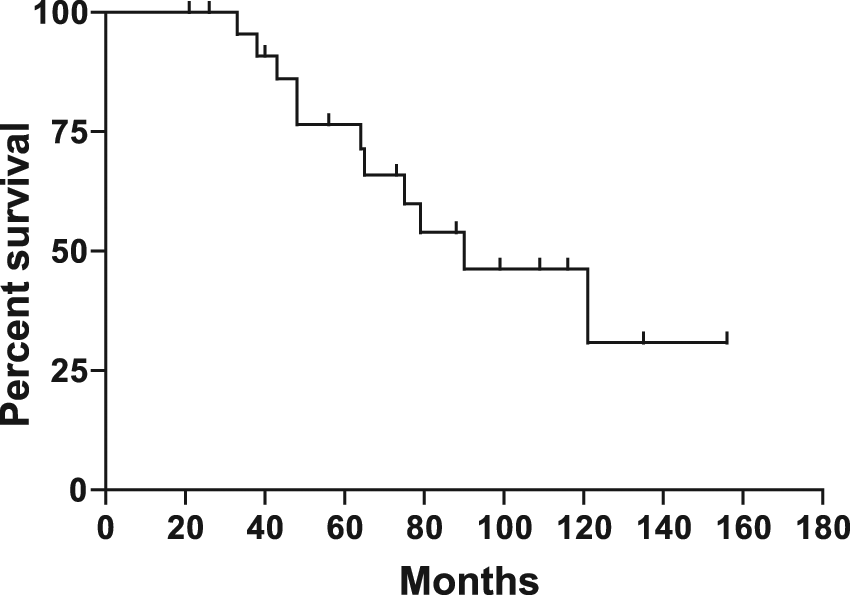
Overall survival.

(A) Progression-free survival after initial (upper panel) versus late (lower panel) autologous hematopoietic stem-cell support (autoSCT). (B) Time to next treatment after initial (upper panel) versus late (lower panel) autoSCT.
Determinants of outcome
In the univariate analysis patient age showed no significant impact on OS (hazard ratio [HR] 0.99, 95% CI 0.89–1.09, p = 0.82). With regard to the risk of progression after late autoSCT, we found time to initial progression (before start of late autoSCT) and response to first autoSCT as significant prognostic factors. According to these analyses there was a 10% risk-decrement per each month of prolonged progression free time (HR 0.90, 95% CI 0.80 to <1.00, p = 0.048) and patients having achieved only partial response after initial autoSCT had an at least 1.6-fold higher risk of relapse compared to patients with VGPR or CR (HR 14.7, 95% CI 1.60–135, p = 0.017).
Discussion
One reasonable treatment strategy for newly diagnosed patients with symptomatic MM under the age of 65 years without substantial organ dysfunction is induction therapy including one of the new substances followed by HD chemotherapy and autoSCT, or alloSCT in patients with an HLA-identical donor [Bruno et al. 2007; Kroger, 2010; Kumar et al. 2011]. In older patients and in those with coexisting conditions a reduced melphalan dose should be considered for autoSCT [Bruno et al. 2007; Palumbo and Anderson, 2011]. Before the era of new substances many patients received induction chemotherapy and high-dose chemotherapy followed by autoSCT. A high portion of these patients relapse and the best treatment strategy for this group of patients is unknown. The new substances are safe and effective in patients relapsing after high-dose chemotherapy and autoSCT, but response duration is less than 1 year in most reported studies. Lenalidomide in combination with adriamycin and dexamethasone resulted in a TTP of 10.4 months in relapsed/refractory MM patients [Knop et al. 2009]. Bortezomib alone resulted in a median TTP of 6.5 months and the addition of liposomal doxorubicin resulted in a TTP of 9.3 months [Orlowski et al. 2007]. The combination of bortezomib, cyclophosphamide and dexamethasone resulted in a median PFS of 12 months [Kropff et al. 2007]. Thalidomide in combination with dexamethasone resulted in a PFS of 20.3 month in relapsed MM after high-dose therapy [Palumbo et al. 2005]. A late high-dose treatment/autoSCT is safe and reasonably effective as a salvage treatment for late relapse after initial HD-Mel/autoSCT when used without the new substances with an event-free survival (EFS) of 15–16 months [Alvares et al. 2006; Elice et al. 2006], or a median PFS of 6.8 months [Qazilbash et al. 2006]. In our cohort, the combination of new drugs for remission induction (23 of 24 patients) followed by HD-Mel/autoSCT resulted in a median PFS of 13 months, compared with 8.5 months PFS in a study that used various HD regimens during late autoSCT [Olin et al. 2009]. Of note, the median TTP reported by us is the interval between the date of transplantation and the date of documented progression. In contrast, in the studies of new substances alone [Knop et al. 2009; Kropff et al. 2007; Orlowski et al. 2007; Palumbo et al. 2005] the median TTP is defined as the time interval between the start of the treatment and the date of documented progression.
The major goal in MM is to maximize the duration of response, which in turn will result in delay of MM complications. The depth of response predicts the duration of the time to treatment failure irrespective of the treatment regimen used [Gay et al. 2011]. The cohort we report was selected for a rather late HD-Mel therapy and autoSCT based on a relatively long TTP after the first HD-autoSCT. We anticipated that at a deep response to retreatment with new substances and late HD-autoSCT would subsequently result in a long treatment-free interval. The overall response rate (CR, VGPR, and PR) was 87.5% with only 4.2% CR and 37.5% VGPR rate. Thus, although the depth of second remission was not as good as after initial treatment, the median TTNT was notable at 21 months and, in an indirect comparison, might be superior to new substances alone or the combination of new substances with standard-dose chemotherapy [Knop et al. 2009; Kropff et al. 2007; Orlowski et al. 2007; Palumbo et al. 2005]. Taken together, our results indicate that retreatment of selected patients with new substances followed by HD-Mel/autoSCT can achieve a similar treatment-free period as after initial treatment, despite the significantly lower CR/VGPR rate.
Late alloSCT after failure of autoSCT is not supported by data from a retrospective analysis [Crawley et al. 2005]. RIC was feasible, but heavily pretreated patients (i.e. with >1 prior HD-therapy and autoSCT) had no benefit with regard to OS. In this MM cohort no new agents were used before alloSCT. In the era of new substances, we found it reasonable to proceed to alloSCT in patients with an HLA-identical donor who responded to the sequence of new drug-HD-MEL/autoSCT. Nine of 24 patients in our analysis received alloSCT after RIC-conditioning. Of interest, five of these nine patients are alive at the time of last follow up. Six of the 9 patients that proceeded to late alloSCT had a TTP after the initial HD-therapy of <24 months. Thus, re-induction with new drugs and autoSCT can serve as a bridge to late alloSCT in selected patients.
Conclusions
In summary, our data confirm that depth and duration of the first remission after HD-Mel and autoSCT are predictors for the response duration after late HD-Mel and autoSCT. The use of new substances for remission induction leads to a high ORR. Based on our results and the existing literature we recommend considering relapse treatment with new agents and late HD-Mel/autoSCT for selected patients, potentially as bridge to alloSCT. Thus, collecting three stem-cell grafts should be considered. In the near future many relapsed MM patients will have received a new agent during first-line therapy. It is unclear whether a second remission induction with an alternative new agent with or without chemotherapy followed by HD-Mel/autoSCT will result in a comparable treatment-free interval.
Footnotes
Acknowledgements
The authors would like to thank Dagmar Wäscher and Robert Peter for support with data collection.
The authors declare no conflicts of interest.
