Abstract
The water-based combination of two inorganic chemical compounds such as sodium tungstate dihydrate-Na2WO4 × 2H2O and Aluminum sulfate octadecahydrate-Al2 (SO4) 3 × 18H2O that we have conditionally named ‘Vomifal’ has a broad antiviral activity in various DNA and RNA viruses, including Human Herpes Virus (HHV), African Swine Fever Virus (ASFV), Vaccinia Virus (VV), Hepatitis C Virus (HCV), Foot and Mouth Disease Virus (FMDV), Influenza A virus (A/Aichi/2/68 (H3N2)). In vitro and In vivo assays in several tissue cultures as well as in laboratory animals, conformed ‘Vomifal’ has a very low toxicity and the antiviral properties partially are due to its ability to induce gamma-IFN. Based on the results obtained, we can assume the presence of at least two mechanisms of the antiviral action of the studied drug. First or early stage - an unknown mechanism, possibly related to the effect on cellular receptors. Second or late stage – main antiviral properties probably associated with an interferonogenic effect.
Introduction
According to arising of several new danger viruses epidemics (and at least one pandemic) became very actual development of several clinically effective antiviral compounds. However, majority of modern and/or historically known antivirals have negative features, such as poor bioavailability, limited antiviral activity, toxic side effects or early development of drug resistance. Also the majority of existing antivirals are still directed at few members from more than 15 virus families that can affect humans. This leads to vulnerability to new viruses that have the ability to quickly spread in the human population. So the current epidemic status in world has demonstrated that our preparedness for emerging respiratory viral infections is far too inadequate. 1 In the last years, many antiviral compounds have been identified but only few have been further evaluated in pre-clinical or clinical trials. The main reasons for this are problems in such parameters of new drugs as low efficiency, narrow specificity and partially low stability. 2 To search for new antiviral drugs, devoid of these disadvantages, we studied several representatives from the group of polyoxymetalates (POMs). Our study is devoted to the description of the antiviral characteristics of one of them, containing tungsten.
POMs are negatively charged aggregates of transition metals, mainly Vanadium, Molybdenum and Tungsten with oxygen. 3 Literature suggests that POMs, which contain tungstate, have been proven to have broad spectrum antiviral, antibacterial, and antitumor properties.4,5 Antiviral activity of POMs containing tungstate was reported first by Raynaud et al., as early as 1971. 6 Authors noted that polytungstosilicate heteropoly compounds inhibited murine leukemia sarcoma virus in vitro. It is important to note that under in vitro and in vivo conditions, antiviral activity of POMs was proven experimentally, in both DNA and RNA viruses. Tungsten heteropoly compounds have been described to have a significant inhibitory effect against many RNA viruses such as VSV, MLSV, rubella, RLV and polioviruses. Most of these POMs have shown to have a good antiviral activity with a low cytotoxicity effect to the host. 6 Several studies have described the antiviral mechanisms of tungstate as the cell surface localization of chemical as physical barrier, therefore blockage of viral adsorption and penetration into the cells.3,7 It is also explained by Polyanionic nature of POMs, the state that is similar to other polyanionic species such as heparin and dextran sulfate that have long been known to act at the cell surface to block virus entry to the host. 6 However, despite a huge number of researches the final mechanisms of antiviral properties of POMs are still unknown.
This is the new study demonstrating that a novel preparation including tungsten heteropoly compound can be used as broad spectrum antiviral drug.
Materials and methods
Preparation
To obtain the initial preparation, it is necessary to prepare a mixture of two inorganic compounds. To sodium tungstate dihydrate Na2WO4 × 2H2O (Sigma Adrich No: 72069) with a concentration of 6 × 10−5–6 × 10−2 mol/L, add Aluminum sulfate octadecahydrate Al2 (SO4) 3 × 18H2O (Sigma Aldrich No: 227617) with a concentration of 5 × 10−6–5 × 10−3 mol/L. In a dilute aqueous solution in an acidic medium (Ph 3.35–3.8), a stable hexa-tungsten-aluminum heteropoly acid of polyoxometalates (POMs) is synthesized. As an intermediate name of the drug was proposed “Vomifal”.
Cytotoxicity in cell cultures
The cytotoxicity of “Vomifal” in in vitro conditions was evaluated in (SK-N- MC, Hela, BHK21, Vero, BSC40, Huh7-Lunet cell lines – Figure 1) by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Confluent cells in 96-well cell culture plate (2 × 104 cell/well) were treated with increasing concentrations of the “Vomifal” ranging from 0.06% to 4%. Treated cells were incubated for 96 h at 37 °C in 5% CO2.
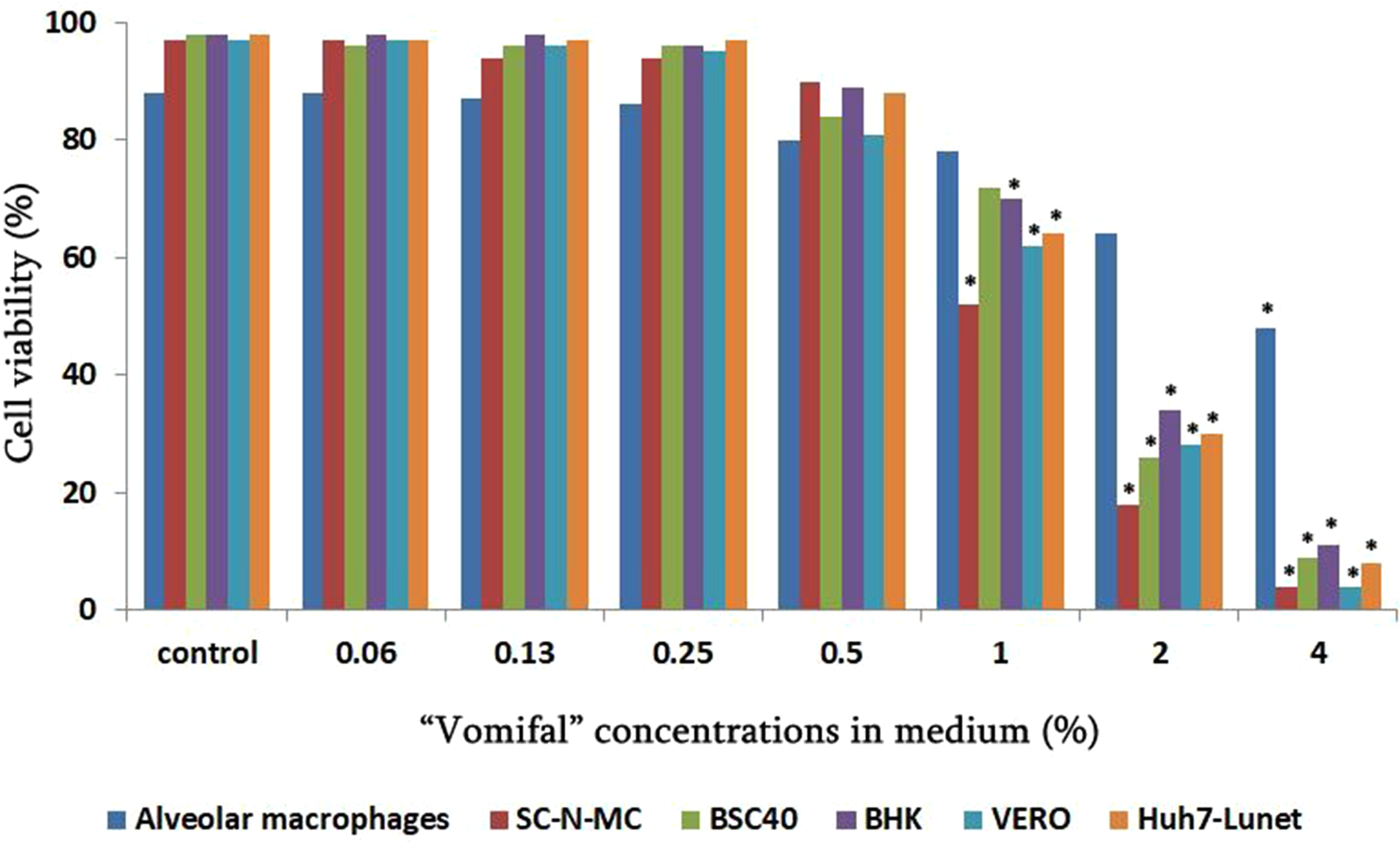
Cytotoxicity of different concentrations of “Vomifal” in cultural medium. Values represent mean from three independent experiments. Data have been analyzed by Student's t-test. Significant differences compared to control are denoted by * p < 0.05–0.01.
Toxicity in animal models
Before carrying out virus infection experiments and addressing antiviral properties we first studied the toxicity of compound in animals (mice, rats and guinea pigs). Outbred mice rats and guinea pigs of both sexes were assigned to groups of at least four animals each. Various doses of “Vomifal” were administered to determine minimal toxic doses of the individual agents. Parameters evaluated included mortality, clinical observations, body weights, and gross necropsy. Acute toxicity studies in vivo have indicated that oral administration to mice, rats and guinea pigs of both sexes does not cause death and does not affect their overall health and behavior. Based on these data, was suggested maximal nontoxic dose 500 µg/kg.
Viruses
For investigations was used ASFV (genotype II) isolate Arm07. Infectious titer of ASFV was calculated by the method of haemadsorption. 8
Hepatitis C (HCV) - 1b isolate 274933RU. Doses of the virus genome were evaluated by rtPCR.
Foot and mouse disease virus - A (FMDV-A, isolate A22); Infectious titer of FMDV-A was calculated by the method of biological titration on BHK-21 cells. For in vivo experiments doses were calculated by tenfold dilutions of ulcers on guinea pigs.
Influenza A virus (A/Aichi/2/68 (H3N2)). The infectious titers of the virus were determined by the method of Reed and Mench when infecting mice with tenfold dilutions of the lung suspension.
Wild-type herpes simplex virus HHV-1. Infectious doses of the virus were evaluated by the method of Reed and Mench when mice were infected with tenfold dilutions of the brain suspension.
Wild type Western Reserve Vaccinia virus (WT-WR VV). Infectious doses of the virus were evaluated by the plaque assay. 9
EMCV (Columbia-SK strain) was used at multiplicity of infection 105 TCD50/mL on SK-N-MC. Viral titers were calculated by the method of Reed and Mench. Parallel with viral titration passages of non-infected culture were used as a control.
Animal model human herpes virus
For in vivo studies, was used HHV-1, previously adapted for reproduction in the brain of mice at a dose of 100 LD50. The infectious activity of the virus was determined by the method of Reed and Mench when mice were infected with tenfold dilutions of the brain suspension. To investigate the effect of ‘Vomifal’ in vivo study we use Human Herpes Virus (HHV-1), for this purpose the white outbred mice with the weight of 10–12 g (n = 35) were used. Mice were fed with none or different concentration of ‘Vomifal’ by oral administration (Figure 2(a)), followed by intracerebral inoculation of virus with the dose of 7.0 lethal doses (LD) 50/mice, survivals ratio were monitored up to 16 days

Antiviral action of “Vomifal” in vitro. (a) ‘Vomifal’ reduced HCV viral RNA level in Huh7-Lunet cells. Cells were pretreated at control or indicated the concentration of “Vomifal” subsequently infected with 4 × 103-HCV particles. At 5 dpi viral RNA level was measured by rtPCR. IFN was used as inhibitory control. (b) Effect of ‘Vomifal’ on FMDV-A. BHK-21 cells were infected with FMDV-A virus in the absence or presence of “Vomifal” at 0.2% of stack solution in culture medium, at the indicated time post infection cell lysate was harvested and virus titer was determined in BHK-21 cells. (c) ASFV viral titer in primary porcine alveolar macrophages. Control or ‘Vomifal’ pretreated AMs were infected with ASFV at conc. of 104 HAD50/mL at indicated time post infection cell lysates were harvested and titer was determined in primary porcine AM. (d) Vaccinia virus plaque formation was reduced with ‘Vomifal’. BSC40 cells were pretreated with medium alone (a, b) or with medium containing 1:20 dilution of ‘Vomifal’ (c, d) for 1hr at 37 °C cells were then infected with wild type WR-VV of 100 PFU (a, c) or 50 PFU (b, d) cultured in medium containing ‘Vomifal’ fixed and stained with crystal violet at 2 day pi. * Data were analyzed by Student's t-test Significant comp’red to control (p < 0.05–p < 0.01); ** tendency (p < 0.1).
Murine serum IFN-gamma measurements
Five healthy, control outbred white mice were sacrificed at the time of infection (n = 5). In first group was used single per os administration of “Vomifal” in dose 430 µg/kg. In second group was used single per os administration of “Vomifal” in dose 250 µg/kg. In third group was used every day per os administration of “Vomifal” in single dose 150 µg/kg (total in five days 750 µg/kg). Five outbred mice in the each treated groups were bled under ether anesthesia on each of the days 1, 2, 3, 4 and 5 (n = 75). Each blood sample was collected in and centrifuged. The resultant serum was stored at-20 °C until assay. The levels of IFN-γ were determined by ELISA (Mybiosource; MBS175777) according to protocol.
Alveolar macrophage (Am) culture
Pigs 3 month aged were euthanizated and the lungs removed. Cells obtained during bronchoalveolar lavage (BAL) were resuspended in sterile Hank's balanced salt solution. They were centrifuged at 600 g for 10 min and resuspended in RPMI 1640 with 5% fetal bovine serum (FBS) at a cell concentration of 1 × 106 mL. After 3 h at 37 °C in a humidified, CO2 incubator, the adhered cells were washed three times with RPMI to remove contaminating non-adherent cells and then reincubated in RPMI 1640 with 10% FBS. 10
Infection of porcine alveolar macrophage in vitro
AM were washed twice with RPMI 1640 and inoculated with virus at 104 haemadsorption units HADU50/mL. After 1 h at 37 °C in a humidified, CO2 incubator, the monolayers were again washed twice, cell maintenance medium was added and they were reincubated. The titer of African Swine Fever Virus (ASFV) used for infection was 104 HADU/mL. Virus doses were measured by Enjuanes et al. 8
Animal model FMDV
Guinea pigs weighing about 300 g, with no antibodies to Foot and Mouth Disease Virus (FMDV), were maintained under pathogen-free conditions with free access to pathogen-free food and water.
After the “Vomifal” introduction (0.2 µl/g of 10% solution), anesthetized guinea pigs were challenged subcutaneously and intradermal in the left rear leg with 0.2 mL 200 infection dose (ID50) of FMDV A. After 2, 3, 5 days after infection, protection against FMDV was examined by clinical signs as follows: guinea pigs showing FMDV lesions only at the original injection site were judged to be protected, and those showing any FMDV clinical signs in the other three feet were judged to be unprotected. 11
Animal model A/Aichi/2/68 (H3N2)
The influenza A/Aichi/2/68 (H3N2) virus previously adapted for reproduction in the lungs of mice at a dose of 100 LD50 was used for research in vivo. The infectious activity of the virus was determined by the method of Reed and Mench when infecting mice with tenfold dilutions of the lung suspension. Outbred white mice weighing 12–14 g were infected with the virus intranasally under light ether anesthesia at a dose of 0.05 mL / mouse.
The drug was administered orally at a dose of 430 μg/kg once a day according to the following groups: 1st groups (20 animals): once 24 h before infection of mice with influenza virus (prophylactic scheme). 2nd groups (20 animals): simultaneously with infection with the virus (one-time treatment and prophylactic scheme). 3rd groups (20 animals): multiple times - immediately after infection and every day for 3 days after infection of mice with the virus. Control virus infection (20 animals).
In vitro HCV rtPCR
According to Perin et al., 12 prior to infection with Hepatitis C (HCV), Huh7-Lunet cell cells were pretreated for one hour with culture medium or with medium containing “Vomifal” at different concentration. Subsequently cells were infected with 4 × 103-HCV particles. At 5 dpi viral RNA level was measured by rtPCR using specific primer sequences that were chosen according early description. 13
JFH1-Forward/TGGGTTCGCATGGTCCTAATGACA, Reverse/TGGAAGGTCCAAAGGATTCACGGA (AB237837);
OPN-Forward/TGAAATTCATGGCTATGGAA,
Reverse/TGAAACGAGTCAGCTGGATG (NM_000582);
CD44-Forward/AGCAACCAAGAGGCAAGAAA,
Reverse/GTGTGGTTGAAATGGTGCTG (NM_000610);
β-Actin-Forward/TGGCATCCACGAAACTACCT,
Reverse/ACGGAGTACTTGCGCTCAG. (NM_001101)
Plaque assay for Wild Type Western Reserve Vaccinia Virus
For plaque assay Wild Type Western Reserve Vaccinia Virus (WT-WR-2 VV) strain was used. 14 The confluent monolayer of BSC40 cells in 6 well plates, were pretreated with medium alone or with medium containing 0.2% of ‘Vomifal’ for 1 h at 37 °C subsequently cells were infected with 100 or 50 VV plaque forming unit per plate (PFU/plate) in the presence or absence of ‘Vomifal’, after 1 h of infection viral inoculums was removed and infected cells were overlaid with 1% agar in growth media, incubated for 3 days, fixed, stained with crystal violet and photographed. 9
Statistics
Statistical analyses were performed using the Student's t- test, and by non-parametric Mann-Whitney U test.
Results and discussion
Toxicity evaluations
Firstable we have screened the cytotoxicity of different concentrations of “Vomifal” on (AM, SK-N- MC, BHK-21, Vero, BSC40 and Huh7-Lunet cells) in vitro, by using the MTT assay. As shown on Figure 1, “Vomifal” concentration in cell medium less than 1% is nontoxic. It can be also indicated that only porcine alveolar macrophages survive in high concentrations of investigated preparations.
“Vomifal” treatment to animals was performed by feeding them 3 times a day for 14 days (500 µg/kg), euthanized and organs were investigated for their comparison with control samples. Almost no difference was observed in size, weight, or pathomorphological characteristic (data not shown).
Antiviral activity of “Vomifal” against HCV in vitro
In order to see whether prophylactic action of “Vomifal” on could inhibit HCV infection Huh7-Lunet cells were left untreated-control or pretreated with different concentrations of “Vomifal”, for one hour prior to virus inoculation; on the fifth day of post infection rtPCR was performed. As we can see from (Figure 2(a)) in the presence of “Vomifal” HCV infection was reduced at a dose-dependent manner. Interestingly, the highest concentration inhibitory effectiveness was similar to IFN inhibition (see below).
Antiviral activity of “Vomifal” against FMDV-A in vitro
Another RNA virus that has been tested in vitro for its sensitivity to ‘Vomifal’ is FMDV (Figure 2(b)). FMDV-A virus titer was measured in cells using one-step growth analysis, control or ‘Vomifal’ pretreated BHK-21 were infected with FMDV-A at various time post infection cell lysates were collected and the virus titer was determined in BHK-21 cells as shown in Figure 2(b) ‘Vomifal’ reduced FMDV-A virus titer at approximately 2 fold. This effect was measured at 24 and 48 h after infection.
Antiviral activity of “Vomifal” against EMCV in vitro
Another picornavirus that has been tested in vitro for its sensitivity to ‘Vomifal’ is EMCV (Figure 2(c)). EMCV virus titer was measured in cells using one-step growth analysis, control or ‘Vomifal’ pretreated SK-N-MC were infected with EMCV at various time post infection cell lysates were collected and the virus titer was determined in SK-N-MC cells as shown in Figure 2(c) ‘Vomifal’ reduced EMCV titer at approximately 2.5 fold. This effect was measured at 6, 12, 24 and 48 h after infection.
Antiviral activity of “Vomifal” against ASFV in vitro
Next was evaluated the effect of ‘Vomifal’ on DNA viruses, first, control or ‘Vomifal’ pretreated swine AMs were infected with ASFV virus at the concentration of 104 HAD50/mL at 0, 24 and 48 hpi., cells were harvested and virus titer was determined by haemadsorption assay on AMs. As shown in (Figure 2(d)) ‘Vomifal’ reduced ASFV infection, though to a low extent.
Antiviral activity of “Vomifal” against Vaccinia Virus in vitro
Another DNA virus that tested for sensitivity to “Vomifal” is VV (Figure 2(e)). BSC40 cells were pretreated with medium alone (Figure 2(e), (a), and (b)) or with medium containing 1:20 dilution of ‘Vomifal’ (Figure 2(e), (c), and (d)) for 1 h at 37 °C cells were then infected with wild type VV of 100 PFU (Figure 2(e), (a), and (c)) or 50 PFU (Figure 2(e), (b), and (d)). As shown in Figure 2(e) “Vomifal” had a great inhibitory effect (∼80%) on (both VV doses) plaque formation in BSC40 cells.
Antiviral activity against HHV in vivo
The antiviral activities of “Vomifal” against HHV were measured by the evaluation of average life expectancy and the percentage of survived mice (Figure 3(a)). From the figure follows that at the highest concentration with ‘Vomifal’ up to 60% of animals were protected by 14 dpi, while without ‘Vomifal’ all animals died on the second day. To understand if the high virus replication in the mice brain is the reason for animal to be killed and that ‘Vomifal’ acts to reduce virus replication we tend to evaluate the amount of virus in the brain. Therefore, tissue samples from infected with the virus of control were collected at 1st, 3rd, 5st and 7st dpi were titrated as we can see from Figure 3(b) while in control samples virus titer reached to ∼5l g TCD50/mL, with ‘Vomifal’ it has ∼2 lg TCD50/mL (Figure 3(b)). Thus, we can conclude that ‘Vomifal’ provide a satisfactory level of security for animals from HHV1 infection in vivo.

Antiviral effect of “Vomifal” in vivo. (a) HHV1 infection of mice. 8 mice/group were fed by water or the indicated concentration of “Vomifal” followed by intra-brain virus inoculation Average life expectancy white bars and the percentage of survivals black line is shown. Data were analyzed by Student's t-test. (b) HHV1 viral t'ter in brain tissue. On the 1st, 3rd, 5th and 7th day post infected animals with or without ‘Vomifal’ were sacrificed and virus titer in the brain was determined. Data were analyzed by Student's t-test. (c), (d) General’zation of FMDV-A virus on Guinea pigs, with/without ‘Vomifal’. Control (black bar), ‘Vomifal’ (white bar), 1 h prior to subcutaneous inoculation animals were fed (8 animals per group) by 0.2 mL/g (c) or 1 mL/g (d) of ‘Vomifal’. Data were analyzed by Mann-Whitney U-test. (e) Influenza A A/Aichi/2/68 (H3N2)) virus infection of mice. 20 mice/group were fed by water orally the indicated concentration of “Vomifal” followed by injections virus. Average life expectancy dark gray and percentage of lethality (light gray) is shown. Data were analyzed by Mann-Whitney U-test. * Significance (p < 0.05–p < 0.01); ** tendency (p < 0.1).
Antiviral activity against FMDV-A in vivo
The FMDV-A inhibiting potential of “Vomifal” in vivo was assessed by using a standard guinea pig model. After the oral administration of ‘Vomifal’ (single doses: 0.2 mL/g (Figure 3(c)) and 1.0 mL/g (Figure 3(d)) for 1 h, anesthetized animals were challenged with 100ID50 of FMDV-A subcutaneously in the left rear leg. At 2, 3, 5 days of infection, protection was examined by clinical signs as described earlier (materials and methods), as we can see ‘Vomifal’ protected animals to observe lesions in dosage dependent manner (Figure 3(c) and (d)). However, at the 5 dpi even with ‘Vomifal’ virus took over and animals gain lesions similar to control treated mice level (Figure 3(c) and (d)). We can thus conclude ‘Vomifal’ protection of FMDV-A generalization is a transient phenomenon, which needs further examination to understand.
Antiviral activity against influenza A/Aichi/2/68 (H3N2) virus in vivo
The antivirus potential of “Vomifal” in vivo was assessed by using a mouse model for influenza as described earlier (materials and methods).
The relative average life expectancy in the virus control group was 8.5 days with 70.0% mortality. Research results (Figure 3(e)) show that the drug “Vomifal” did not have a protective effect against experimental influenza infection with a single injection of the drug and the virus. However, multiple (immediately after infection and every day for 3 days) administration of the drug was very effective: it increased the survival rate of infected mice to 90.0%. The relative average lifespan of mice in comparison with controls increased accordingly to 58.8 days.
Abilities to induce interferon-gamma in vitro and in vivo
To investigate the abilities of “Vomifal” to induce IFN-gamma production we performed an induction experiment in Primary human mononuclear cells (Figure 4(a)). Our data revealed a pronounced interferon-inducing activity at in vitro conditions on stage 72 h post administration. Also, the result showed that indeed “Vomifal” was able to induce gamma-IFN production similar to PHA level at 24 h and subsequently increased at 48 h, however with the combination of both at 72 h it did not give any greater result (Figure 4(a)), indicating that both were acting through different mechanisms.

Gamma-IFN induction by “Vomifal”. (a) Gamma-IFN induction by “Vomifal” at in vitro conditions. Primary human mononuclear cells were control treated (black) or treated with PHA (white), (0.2% solution in cell medium) ‘Vomifal (gray) or both (light gray). At the indicated time of post treatment, gamma-IFN induction of supernatant was measured using classical ELISA assay. (b) Gamma-IFN induction by “Vomifal” in in vivo conditions on murine model. 1st group - single per os administration of “Vomifal” in dose 430 µg/kg. 2nd group - single per os administration of “Vomifal” in dose 250 µg/kg. 3rd group - every day per os administration of “Vomifal” in single dose 150 µg/kg (total in five days 750 µg/kg). Data were analyzed by Student's t-test. * Significance (p < 0.05–p < 0.01).
The results of in vivo studies, presented in Figure 4(b), show that the highest IFN-gamma titers in the blood serum of experimental animals (more than 2000 pg/mL in some animals) are determined 96 h after a single oral administration of the drug. It can also be noted that the induction of interferon synthesis does not depend on the dose of the drug (comparison of groups 1 and 2). Repeated administration of the drug also does not increase the synthesis of interferon (group 3).
Discussion
Tungsten as a component of inorganic antivirials was detected several decades ago e.g., Tungsten in composition with HPA-23 showed antiviral activity against human immunodeficiency virus (HIV). 15 Later it was observed that Tungsten as PM-19 caused a decrease in plaque formation by several strains of herpes simplex virus (HSV) type 1, and also was effective against human cytomegalovirus. 16 In composition with mixed valence K15H2[Ln(BW9W2O39)2].28H2O was determined the highest antiviral activity against influenza A(FluVA) strain (A/H1N1/Jingfang/1/91 and A/H3N2/Jingfang/30/95) and influenza virus B(FluVB) (B/Hufang/1/87) in MDCK cells. 17 As polyoxometalates were reported the latter's anti-HIV activity, 18 anti- Hepatit B, 19 anti-respiratory syncytial agent (RSV), 20 and anti-Hepatit c activities. 21 In the composition of heteropolyoxotungstate was examined antiviral activity against herpes simplex virus type 2 (HSV-2),22,23 hepatitis B (HBV) and SARS virus. 24 Some studies were done for the inhibition of viral DNA polymerases in vitro and in vivo. 25 In the last 10 years as discrete metal-oxide cluster anions with high solubility in water and photochemically and electrochemically active property, was reported it's antiviral activity against DNA and RNA viruses (such as HSV, HIV, influenza, and SARS). 26 Tungsten's powder has shown antiviral activity against bacteriophage Φ6. Inactivation of alkaline phosphatase enzyme proteins was assumed as one mechanism of the antiviral activity of the latter. 27
In this study we demonstrate the role of a new composition of the tungsten in its inhibitory effect in several RNA/DNA viruses. Our experiments showed the good level of inhibitory properties of the compound. However, despite our results or previously described studies for other polyoxometalates3–6 the exact mechanism of the compound still remains to be explored. One possible explanation is that the compound is situating in the cell surface like physical barrier, 5 not allowing effective virus attachment and penetration into the host. So based on the results obtained, we can assume the presence of at least two mechanisms of the antiviral action of the studied drug. First or early stage - an unknown mechanism, possibly related to the effect on cellular receptors. Second or late stage – main antiviral properties probably associated with an interferonogenic effect.
Together, these results offer that the suggested tungsten heteropoly compound (“Vomifal”) can warrant further clinical investigation for complete development of investigations and furthermore for and implementation in clinical practice antiviral drug with a broad spectrum of activities.
Conclusion
In conclusion in this study we provide the compound, which has several important features: The ‘Vomifal’, is not costly, it is easily soluble in water, is not toxic to animals as well as for a variety of cell cultures, it has shown a high inhibitory effect in DNA and RNA viruses in both in vitro and in vivo.
Footnotes
Acknowledgements
The work was partially supported by the Science Committee of MESCS RA and State Committee of Science and Technology of RB in the frames of the joint research project 21SC-RBSCST-1F004. We are thankful to the Directorate of the Institute of Molecular Biology of NAS for all technical supports. We are also thankful to Prof. Wen Chang, ![]() Sinica, Taipei, Taiwan, ROC for supporting with Vaccinia Virus experiments.
Sinica, Taipei, Taiwan, ROC for supporting with Vaccinia Virus experiments.
Declaration of conflicting interests
All authors confirm that they have no conflict interest. All animal care and euthanasia were done according to the AVMA Guidelines on Euthanasia, and local guideline for animal care and use (Institutional Review Board/Independent Ethics Committee of the Institute of Molecular Biology of NAS, IRB00004079).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science Committee of MESCS RA and State Committee of Science and Technology of RB in the frames of the joint research project 21SC-RBSCST-1F004., (grant number 21SC-RBSCST-1F004).
