Abstract
Background
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm driven by the Philadelphia chromosome, typically progressing through chronic, accelerated, and blast phases. Blast crisis may be myeloid or lymphoid. Isolated central nervous system (CNS) involvement is rare and represents a diagnostic and therapeutic challenge.
Case presentation
We report a 40-year-old man with no prior hematologic history who presented with four months of progressive lower-limb paresthesias, neuropathic pain, and subacute weakness impairing ambulation. Neurologic examination revealed asymmetric paraparesis, hyporeflexia, and distal hypoesthesia in a stocking distribution, consistent with a subacute asymmetric sensorimotor polyradiculoneuropathy. Complete blood count demonstrated marked leukocytosis with neutrophilia, left shift, and circulating blasts. Contrast-enhanced spinal magnetic resonance imaging showed extensive leptomeningeal enhancement. Cerebrospinal fluid (CSF) analysis revealed pleocytosis, hypoglycorrhachia, and elevated protein levels; flow cytometry identified 14% B-lymphoid blasts (CD34+, CD19+, CD20+, CD22+). Bone marrow aspirate and biopsy showed hypercellularity with trilineage hematopoiesis, basophilia, and small hypolobated megakaryocytes, without a significant increase in blasts (<5%). Cytogenetic analysis identified t (9; 22), and molecular testing confirmed the BCR::ABL1 p210 transcript, consistent with chronic-phase CML. An isolated CNS lymphoid blast crisis was diagnosed. Treatment with HyperCVAD (Block B only) plus dasatinib 140 mg daily resulted in neurologic improvement and hematologic normalization. After five cycles, CSF clearance was achieved, followed by consolidation with haploidentical allogeneic hematopoietic stem cell transplantation. The patient remains in complete remission.
Conclusions
Isolated CNS blast crisis may represent the initial manifestation of CML. Prompt recognition through integrated neurologic, hematologic, and molecular evaluation is critical to enable timely, targeted therapy.
Introduction
Chronic myeloid leukemia (CML) is a myeloproliferative neoplasm characterized by the balanced translocation t(9;22) (q34; q11.2), also known as the Philadelphia chromosome. 1 This cytogenetic alteration generates the BCR::ABL1 fusion protein, which constitutively activates tyrosine kinase signaling and promotes uncontrolled cell proliferation. 2 The annual incidence is approximately 2 cases per 100,000 population, and CML-related mortality has decreased to about 0.5–1% in the tyrosine kinase inhibitor (TKI) era. 1 The median age at diagnosis is 64 years, and the disease is more common in men (male-to-female ratio ∼1.4:1). 3
The prognosis of CML has changed radically with the advent of TKIs, which inhibit the BCR::ABL1 tyrosine kinase domain. 4 First-generation TKIs include imatinib; second-generation TKIs include dasatinib, nilotinib, and bosutinib; and the third-generation TKI ponatinib is also available. 4 Since their introduction into routine practice, 10-year survival for patients with CML has improved from approximately 20% to 80–90%. 2
Three phases are described in the natural history of CML: chronic, accelerated, and blast phases. Blast phase is the most aggressive, occurs in 1–4% of patients per year in the absence of adequate treatment, and resembles acute leukemia with increased morbidity and mortality. 5 Blast crises may be myeloid (65–75% of cases) or lymphoid (20–30%), which influences both diagnostic approach and treatment. 5
Approximately 50% of patients with CML are diagnosed incidentally after leukocytosis with neutrophilia is detected on a complete blood count, while they remain asymptomatic. 1 In blast phase, symptoms may include bone pain, headache, fever, or infections. 1 CNS involvement in CML has been reported in about 5% of cases and is usually associated with advanced disease or post-transplant relapse. 6 Here, we describe a patient with B-lymphoid blasts in CSF as the initial manifestation of CML, associated with leptomeningeal infiltration and axonal sensorimotor polyneuropathy. The reporting of this case conforms to the CARE guidelines. 7
Case presentation
A 40-year-old man, an industrial engineer, with past medical history significant only for dyslipidemia treated with statins, presented to the emergency department with four months of lower-limb paresthesias with ascending progression, accompanied by burning pain in the lumbar region and hips. Three weeks before admission, he developed progressive lower-limb weakness with limitation of gait, without sphincter dysfunction or constitutional symptoms. Neurologic examination showed an asymmetric paraparesis (right-predominant), hyporeflexia, and distal hypoesthesia in a “stocking” distribution, consistent with a subacute asymmetric sensorimotor polyradiculoneuropathy of the lower limbs.
Baseline laboratory findings.


Contrast-enhanced magnetic resonance imaging of the lumbosacral spine (T1-weighted sequence after gadolinium) showing extensive leptomeningeal enhancement.
Bone marrow aspirate and biopsy showed hypercellularity with trilineage hematopoiesis, basophilia, and small hypolobated megakaryocytes, without a significant increase in blasts (<5%) (Figure 2). Flow cytometric immunophenotyping of bone marrow blasts demonstrated a myeloid phenotype (aberrant CD56 and CD15 expression and heterogeneous cyMPO expression) (Table 2). Conventional karyotyping identified t (9; 22) in 10 metaphases, and molecular testing confirmed the BCR::ABL1 p210 transcript. Despite chronic-phase bone marrow findings, the presence of B-lymphoid blasts in cerebrospinal fluid with radiologic leptomeningeal involvement supported the diagnosis of isolated CNS lymphoid blast crisis, consistent with extramedullary blast-phase CML. Bone marrow aspirate showing hypercellularity with trilineage hematopoiesis, basophilia, small hypolobated megakaryocyte (left arrow) and <5% blasts (right arrow). Bone marrow studies.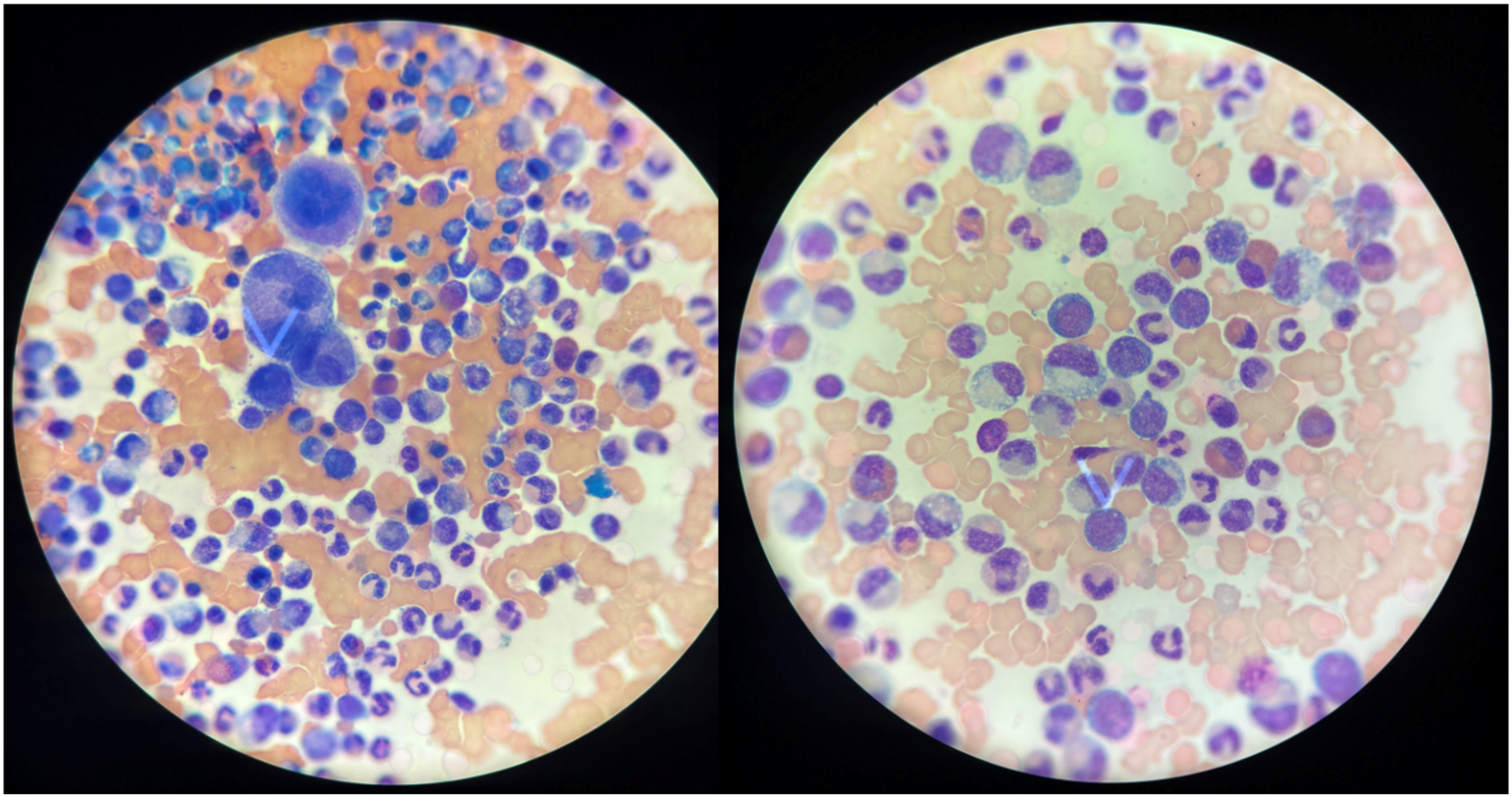

Treatment with HyperCVAD regimen (Block B only, to maximize CNS penetration; Block A was omitted) was initiated, and dasatinib 140 mg orally once daily was added. Intrathecal chemotherapy with methotrexate, cytarabine and dexamethasone was administered twice weekly until CSF clearance. The patient experienced progressive neurologic improvement and normalization of blood counts. After five cycles, CSF became negative by both cytology and flow cytometry, and he underwent consolidation with a haploidentical allogeneic hematopoietic stem cell transplant, with stem cell infusion on May 14th, 2025. To date, no complications have been documented and he remains in complete remission.
Discussion
We report a patient without prior hematologic disease who presented with isolated CNS blast phase with a lymphoid phenotype as the first manifestation of CML, associated with leptomeningeal infiltration and axonal sensorimotor polyneuropathy leading to paraparesis.
CML presents in blast phase in approximately 2–4% of patients; among these, a lymphoid phenotype accounts for only 20–30%. 5 Although lymphoblasts have greater CNS tropism than myeloblasts, isolated CNS lymphoid blast crisis is exceptionally rare and has mainly been described in isolated case reports of progression or relapse, particularly in the post-transplant setting or after prolonged TKI exposure.8–13 To date, isolated CNS lymphoid blast crisis as the initial presentation of CML has been reported only in two pediatric patients,14,15 whereas adult cases are exceedingly rare and previously limited to myeloid blasts detected in cerebrospinal fluid. 16
This case is also notable for the association between CNS lymphoblastic involvement and sensorimotor polyneuropathy. Proposed pathogenic mechanisms include direct leukemic infiltration of leptomeninges and nerve roots, disruption of the blood–brain barrier facilitating CNS homing of lymphoblasts, microvascular ischemic injury, immune-mediated paraneoplastic processes, and cytokine-driven neurotoxicity. Such neurologic manifestations have been infrequently described in leukemias, lymphomas, and select solid tumors, 17 suggesting a multifactorial pathophysiology rather than a single dominant mechanism.
From a biological perspective, CNS lymphoid blast crisis may reflect clonal evolution within a protected sanctuary site, where subtherapeutic drug exposure permits expansion of BCR::ABL1-positive lymphoid progenitors. 18 Recent WHO and European LeukemiaNet classifications recognize blast phase CML as biologically distinct from chronic phase disease, requiring integrated morphologic, immunophenotypic, cytogenetic, and molecular assessment, even when marrow blast burden remains low. 18
Previous reports of CNS-isolated blast crisis in CML14–16 have described neurologic presentations including headache, cranial neuropathies, radiculopathy, or paraparesis, with imaging evidence of leptomeningeal disease and blast detection in cerebrospinal fluid despite minimal or absent marrow blast involvement. Therapeutic approaches have varied, including intrathecal chemotherapy, CNS-penetrant TKIs, ALL-based systemic regimens, and allogeneic stem cell transplantation, with heterogeneous outcomes ranging from transient responses to durable remissions.
CML blast phase should be treated similarly to acute leukemia. 5 Accordingly, TKIs should be combined with intensive chemotherapy according to blast phenotype.1,2 Because the blood–brain barrier may serve as a sanctuary site for blast clones, the presence of CNS lymphoblasts supports the use of TKIs with good CNS penetration, such as dasatinib, in combination with acute lymphoblastic leukemia (ALL)–based regimens. 19 Current guidelines recommend allogeneic hematopoietic stem cell transplantation for eligible patients who return to chronic phase, as it remains the only curative option. 19 In the present case, combined HyperCVAD and dasatinib resulted in neurologic recovery, cerebrospinal fluid clearance, and hematologic remission.
This report is inherently limited by its single-patient design, precluding generalization of therapeutic success. In addition, long-term neurocognitive outcomes were not systematically assessed, and publication bias toward favorable responses in rare case reports cannot be excluded. Nevertheless, this case contributes meaningful clinical insight into an exceptionally uncommon presentation and emphasizes the importance of early neurologic evaluation, integrated molecular diagnostics, and CNS-penetrant targeted therapy. Further studies aggregating reported cases are needed to better define disease biology, neurologic sequelae, and optimal treatment strategies.
Conclusion
Isolated CNS lymphoid blast crisis as the initial manifestation of CML in adults is exceedingly rare. Prompt recognition through integrated neurologic, hematologic, and molecular evaluation is critical to enable timely, targeted therapy. This case adds to the limited adult literature and highlights the need for heightened clinical suspicion and multidisciplinary management in atypical presentations of CML.
Footnotes
Acknowledgments
The authors would like to thank the staff of the hematology unit at Hospital Universitario San Ignacio for their efforts over the years in caring for patients.
Ethical considerations
Ethics committee approval was not required for a single case report according to institutional guidelines.
Consent to participate
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author contributions
A.Z.V, M.A.Z, A.C.A and A.Z.M contributed to the conceptualization, data collection, supervision, and writing of the article. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data is available upon request.
