Abstract
Purpose
Studies investigating the impact of liposarcoma from the patient’s perspective—including diagnosis, disease presentation, treatment challenges, and quality of life (QOL)—are limited. We report findings from a survey of patients with liposarcoma evaluating the diagnostic/treatment journey, biomarker testing, and QOL.
Methods
Patients with liposarcoma in the United States completed an online survey assessing demographics, diagnostic journey, disease burden, treatment, QOL, and sources of support during diagnosis and treatment. Descriptive statistical analyses were performed, and qualitative written responses from participants were assessed.
Results
Seventy-seven patients with liposarcoma participated in the survey. The proportion of participants with stage I/II/III/IV/unknown liposarcoma at diagnosis was 22%/16%/26%/13%/23%, respectively. Within 12 months of symptom onset, 44/60 respondents (73%) saw a medical professional, and 39/77 (51%) first consulted an internal medicine specialist. Fifty-one of 77 participants (66%) were aware of biomarker testing, and 32/51 (63%) reported receiving it. The most common initial treatments were surgery (47/77, 61%), radiotherapy (15/77, 19%), and chemotherapy (9/77, 12%). Health insurance provided coverage for biomarker testing for 27/31 respondents (87%). Of 77 participants, 38 (49%) traveled ≥1 hour to reach their treatment site, and 27 (35%) relied on care partners such as family members for transportation. Upon treatment initiation, QOL improved for 30/77 participants (39%), did not improve for 29/77 (38%), and 10/77 (13%) were unsure. Forty participants (52%) used social media to obtain disease-specific information and support.
Conclusion
Our survey identified gaps in patients’ awareness and understanding of key aspects of liposarcoma diagnosis and treatment, highlighting a need for better patient education, more effective communication between medical professionals and patients, and a deeper understanding of how treatment impacts patients’ QOL. A fuller picture of the patient burden due to liposarcoma underscores the need for research aimed at improving symptom and disease management, including the development of more effective therapeutics.
Introduction
Liposarcomas constitute ≈15%–20% of soft tissue sarcomas, 1 which are a group of rare heterogeneous tumors that account for ≈1% of adulthood malignancies.1,2 These neoplasms arise from fat cells and are further classified into four subtypes based on their histology: well-differentiated liposarcomas (or atypical lipomatous tumors), dedifferentiated liposarcomas, myxoid liposarcomas, and pleomorphic liposarcomas.3,4 The most common subtypes are well-differentiated liposarcomas and dedifferentiated liposarcomas, accounting for 40%–50% and 15%–20%, respectively, of all liposarcomas cases.4,5 These subtypes differ in their disease course, molecular pathology, and treatment sensitivity. 1
Diagnosis of liposarcomas can be challenging, given the rare incidence and prevalence of this disease and the fact that symptoms vary.6,7 Liposarcomas are categorized according to subtype, stage, and location; all of which inform treatment selection,1,6,8 as well as grade, which is an indicator of prognosis. 9
The current standard of care for liposarcomas—surgery for localized tumors and chemotherapy for patients with advanced, unresectable, or metastatic disease—is guided by tumor stage.1,10,11 Although chemosensitivity varies across liposarcoma subtypes, response to first-line chemotherapy is generally poor, and local recurrence and metastasis are common. 1 A higher recurrence rate (45.5%) has been reported among patients with high-grade (grade 3) liposarcomas than in patients with grade 2 (15.8%) or grade 1 (12.5%) disease. 9 The combination of diagnostic challenges and poor responses to existing therapies contributes to a high patient burden. Additionally, there has been little progress in developing new, effective therapies. Research aimed at improving diagnostic methods, developing more effective therapies, and expanding access to specialized care is needed.
The low incidence and prevalence of liposarcoma contribute to a lack of awareness among medical professionals, which can delay diagnosis and impact timely and appropriate patient care. Many patients with liposarcoma also face logistical or financial barriers to specialized care, such as the need to travel long distances or lack of insurance coverage. Therefore, there is an urgent need for multidisciplinary education on liposarcoma, targeting not only oncologists but also the broader healthcare community, including physicians, nurses, physical therapists, physician assistants, pharmacists, nurse/patient navigators, and others involved in the patient journey. A better understanding of the disease burden by those providing care can, in turn, lead to more effective patient education and shared decision-making conversations.
Currently, data on quality of life (QOL) for patients with liposarcoma are limited and do not capture the full impact of liposarcoma on patients’ well-being throughout diagnosis and treatment. Patient surveys that include open-ended questions allow patients to provide descriptive responses in their own words. These responses can offer valuable qualitative insights and a more detailed understanding of key factors that influence treatment choices and prognosis. For example, patients should be aware of the growing importance of biomarker testing in liposarcoma diagnosis and management. Biomarker testing can identify unique genetic signatures, such as aberrant expression of the MDM2 and CPM genes, which are frequently upregulated in liposarcomas.12–15 As targeted therapies emerge in the treatment landscape, biomarkers are expected to gain greater prominence, yet many patients with sarcoma or liposarcoma are unaware of their existence or future value.
Knowledge of the patient perspective can identify areas where medical professionals can optimize the diagnostic and treatment journey, improve access to specialized care, and help ease the burden of disease on both patients and their caregivers. To better understand the patient experience, an online survey was conducted among adults living with liposarcoma residing in the United States to assess disease diagnosis, treatment patterns, and the impact of liposarcoma on participants’ QOL.
Methods
Participants and study design
Participants were eligible to take part in the survey (Data Supplement 1) if they were aged 18 years or older, resided in the United States, and had received a diagnosis of liposarcoma. An online survey collected responses to questions about patient demographics, the diagnostic journey, burden of disease and treatment, QOL, and sources of support during diagnosis and therapy. The survey was designed and facilitated by Savvy Cooperative in cooperation with Boehringer Ingelheim Pharmaceuticals, Inc., the Sarcoma Foundation of America, and a patient advocate living with liposarcoma.
Ethics approval and informed consent
This survey received an exemption from institutional review board oversight by Advarra (Pro00072826). Participants received an email with an informed consent document that they read, signed, and returned. Patients were prompted to provide informed consent again before beginning the online survey by clicking on an “I agree and consent to participate” button. They were informed about the purpose of the survey, the information we were seeking to collect, how their information would be captured and used, and how their privacy would be ensured.
Data analysis
Descriptive statistical analyses were performed, and participants’ qualitative responses were assessed to identify trends.
Results
Patient demographics
Patient demographics and clinical characteristics.

aMore than one selection permitted for race/ethnicity.
bItalian/American.
cOther types of health insurance were as follows: employer-provided insurance through spouse’s insurance plan; Medicare and TRICARE For Life; Samaritan Ministries group sharing program; TRICARE; and Medi-Cal.
dExplanations provided by participants who marked “unsure” were as follows: intermediate; PT3PN0 myxoid/round cell liposarcoma; “I had two instances of liposarcoma, I had one grade 3 and one grade 1”; very large retroperitoneal liposarcoma (40 cm); was labeled well-differentiated; pending biopsy results; “My type was not ‘staged.’ My surgical oncologist has called it ‘low-grade’ and has said it is not ‘graded’ when I asked him. My type of liposarcoma is retroperitoneal liposarcoma, well-differentiated lipoma-like and sclerosing. This qualifies as ‘low-grade.’”; no explanation given (n = 12).
eOther locations: “In the scrotum next to testicles”; “right groin/pelvic area and down to the top of right thigh”; left hip/glute; left buttock.
Diagnostic journey
Participants were asked about the length of time from symptom onset to their initial visit with a medical professional. Of 60 participants who answered this question, 44 (73%) saw a medical professional within 12 months of symptom onset (Figure 1(a)). The first specialist to be consulted was most often an internal medicine physician (internist; 51% of participants) (Figure 1(b)). Across provider types, participants most commonly visited an oncologist (Figure 1(b)). The diagnosing physician for the liposarcoma was most frequently a surgeon or an oncologist (61%; 47 of 77 respondents) (Appendix Fig S1). Prior to their diagnosis of liposarcoma, 55 of 77 participants (71%) had not previously received other diagnoses related to their symptoms of liposarcoma. Other diagnoses reported by more than one participant were lipomas (n = 8), gastrointestinal conditions (n = 3), hernias (n = 2), and cysts (n = 2). Visits to medical professionals in the initial phase of the patient treatment journey.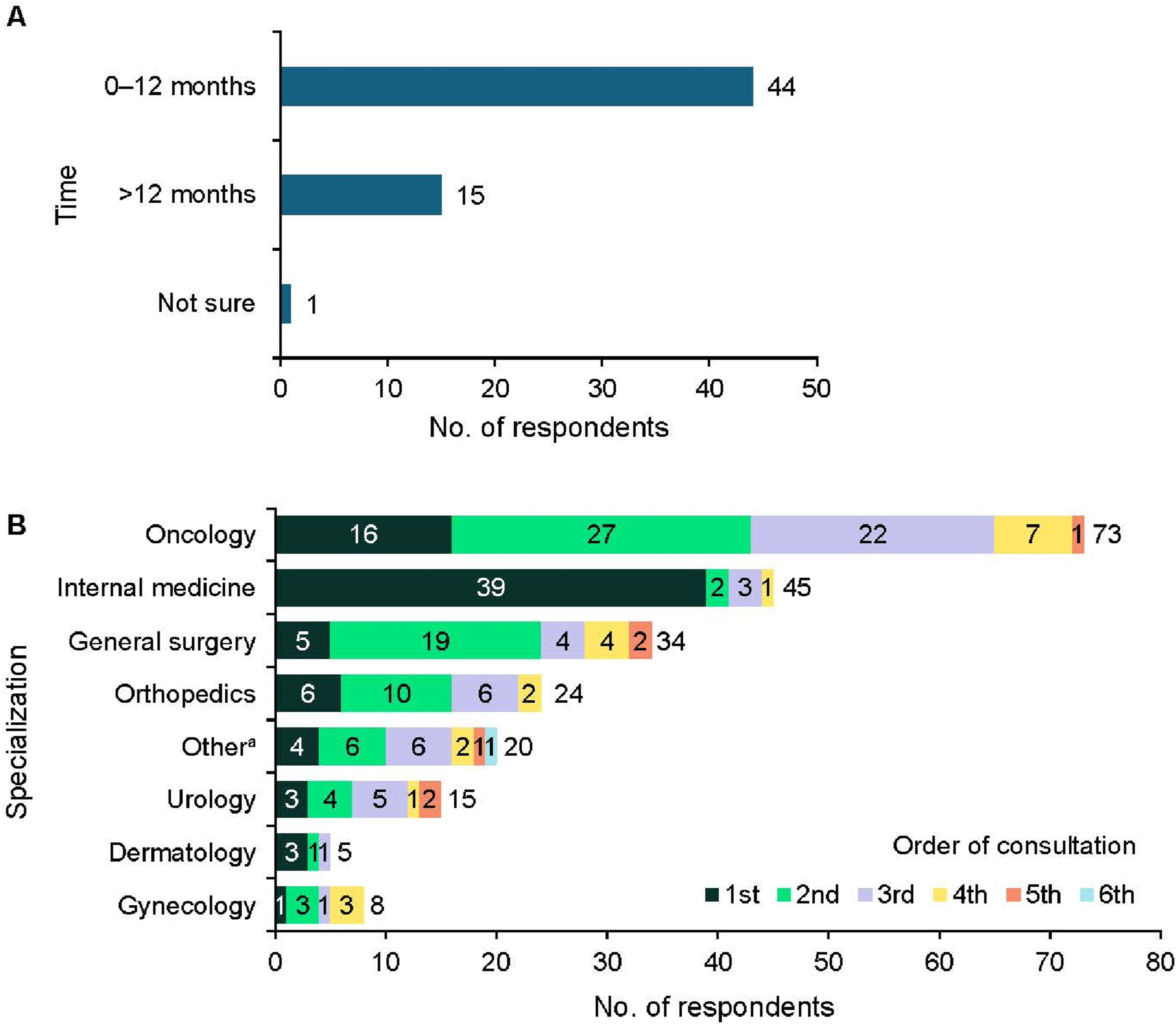
Biomarker testing
Among the 77 participants, 50 reported that they had heard of biomarker testing (Figure 2(a)); however, 51 participants (66%) answered the follow-up question asking if they had received biomarker testing. Of those 51 respondents, 32 (63%) reported having had biomarker testing (Figure 2(b)). Among 31 participants who reported their source of funding for biomarker testing, 27 (87%) cited health insurance, and 2 (6%) cited financial support from a pharmaceutical company; other sources included self-funding (n = 2; 6%), crowdsourcing (n = 1; 3%), and support from family, friends, and/or community (n = 1; 3%) (Figure 2(c)). Biomarker testing awareness, receipt of testing, and funding for testing.
Liposarcoma treatment journey and outcomes
Participants were asked about the type and order in which treatments were given. Of the 77 participants, 67 (87%) had undergone some form of surgery, 39 (51%) had received radiotherapy, and 24 (31%) had received chemotherapy; 2 (3%) had not received any treatment at the time of survey completion (Figure 3). Among the initial treatments received by participants, surgery was most common (n = 47; 61%), followed by radiotherapy (n = 15; 19%), and chemotherapy (n = 9; 12%). Liposarcoma treatments taken. Data are based on responses from 77 participants.
Clinical outcomes.

aTwenty participants indicated experiencing a recurrence in response to one question, while 21 participants indicated experiencing at least one recurrence in a follow-up question.
Impact of liposarcoma on patient QOL
In response to a question about travel time to their treatment site, 54 of 77 participants (70%) reported traveling at least 30 minutes, and 38 participants (49%) reported 1 hour or more of travel time (Appendix Fig S2). While 43 participants (56%) could drive themselves to the treatment site, 27 participants (35%) relied on commuting assistance from care partners (Appendix Fig S3A). Seventy participants (91%) were able to obtain treatment from a treatment site in their home location; however, 5 participants (6%) reported relocating closer to the treatment site (Appendix Fig S3B).
When asked about changes in their QOL upon initiating treatment, similar numbers of participants reported QOL had either improved (n = 30; 39%) or had not improved (n = 29; 38%) (Figure 4(a)). Of the 30 participants who experienced an initial improvement, 20 (67%) reported a continued benefit at the time of survey completion. Of the 29 participants whose QOL did not improve upon starting treatment, 12 (41%) reported experiencing a benefit at the time of survey completion. Ten participants were unsure of the impact that treatment had on their QOL. Among the eight participants who selected “other” in response to the impact of liposarcoma treatment on their QOL, several cited both benefits of treatment in terms of disease management and survival, as well as negative physical and psychological impacts associated with the disease, its treatment, or follow-up. These sentiments are captured by the representative comment: “The surgery and removal of the tumor greatly improved my quality of life. Living with the possibility of recurrence is stressful.” QOL in patients with liposarcoma.
Multiple aspects of participants’ daily lives were affected by liposarcoma (Figure 4(b)). “Physical condition” was the concern cited most (n = 56; 73%), followed by finances (n = 37; 48%) and ability to partake in recreational and leisure activities (n = 34; 44%). Participants’ personal relationships were also impacted, including those with family, friends, romantic partners, and care partners. The type of support that participants found most useful was peer-to-peer connections (n = 45; 58%), followed by support provided by counselors or therapists (n = 23; 30%), financial assistance, and mental health resources (both n = 21; 27%) (Appendix Fig S4). Over half of participants (n = 40; 52%) looked to Facebook groups specifically when seeking support, and 34 participants (44%) mentioned they had tried to access patient support groups and networks.
Discussion
This survey evaluated the impact of liposarcoma and its treatment on the lives of patients from their own perspectives. We found that patients face diverse challenges throughout their journey with liposarcoma, including difficulties obtaining a diagnosis; physical, financial, and psychological burdens as a result of treatment and disease symptoms; and negative effects of therapy on QOL. Some survey responses indicated conflicting sentiments, where participants expressed an appreciation of disease control and continued survival but lamented the physical and psychological stress associated with liposarcoma symptoms and treatment. The impact of liposarcoma on QOL and financial status were key concerns expressed by patients, illustrating the need not only for greater awareness among medical professionals of the patient burden but also for new strategies to help mitigate this burden. The survey results—reflecting current clinical practice—offer valuable real-world insights into the liposarcoma patient journey and may be used to inform individuals and organizations aiming to improve care and provide education to support these patients.
The survey responses indicate a high level of variability in the diagnostic journey, consistent with the variability in patient characteristics and circumstances encountered in a real-world population, as well as with the challenges inherent in diagnosing liposarcoma. For example, although the majority of the respondents had symptoms or concerns that led them to consult a medical professional, 17/77 (22%) reported having tests performed for a different purpose that led to the discovery of their liposarcoma. In addition, although survey participants were most frequently diagnosed by a surgeon or an oncologist, 30/77 (39%) were diagnosed by other specialists, such as orthopedists, urologists, or internists. One patient reported being diagnosed by a dermatologist.
Many participants in our survey faced logistical barriers that could hinder timely diagnosis, as well as access to specialized care. These included the need to consult multiple specialists and the time required for travel to the treatment site. Nearly half of the participants required at least 1 hour of travel time to their treatment site, with 5 participants relocating to be closer to the treatment site. One-third of those surveyed relied on a care partner for transportation to the treatment site, illustrating that the burden of disease is often shared with patients’ friends or family members. These challenges can make it harder for patients to routinely access the treatment and care they require, potentially having a negative impact on management and follow-up—which are critical for optimizing patient outcomes.
The survey results also indicate a need for more effective communication and patient education by medical professionals, particularly regarding tumor stage, grade, and biomarker testing. More than one in five participants were unsure of their tumor stage at diagnosis, and survey responses from some participants indicate a confusion between tumor stage versus histologic grade and/or subtype. Because tumor stage is a key factor driving treatment decisions, 10 the significance of tumor stage should be accurately communicated and reinforced to help patients understand their disease status and treatment options. Furthermore, histologic grade is the most important prognostic factor for all soft tissue sarcomas, 16 and morphologic subtypes of liposarcoma differ in their clinical course and optimal treatment. 1 Because patients may be distressed when they are first diagnosed—due to feeling overwhelmed and experiencing “information overload,” for example—effective, stepwise education and frequent reinforcement of key concepts such as stage and grade may help address patients’ key information needs to assist them with shared decision-making regarding their treatment at different points in their liposarcoma management journeys. In addition to more traditional resources, such as members of their care team, many participants listed peer support and social media as key resources for support and assistance. Taken together, these findings indicate that raising medical professionals’ awareness of patients’ knowledge gaps and providing potential sources of information and support could facilitate more effective conversations with patients, thereby improving shared decision-making and meeting patients’ information and education needs during their liposarcoma management journey.
Approximately one-third of participants had no awareness of biomarker testing for liposarcoma. Of those who were aware, many were uncertain if they had been tested or not. This uncertainty suggests a need for better communication and/or understanding of biomarkers, how they can inform diagnosis and prognosis of rare cancers such as liposarcoma, the different types of biomarker testing, and what results mean for the patient. Biomarker testing for participants was most commonly funded through health insurance or financial support from a pharmaceutical company. Greater advocacy and communication may help improve patient awareness of the importance of biomarker testing for liposarcoma.
Recent studies providing a deeper understanding of liposarcoma tumor biology highlight the diagnostic and prognostic value of molecular markers for liposarcoma 17 and underscore the potential of biomarker testing. For example, evaluation of MDM2 status is considered to be a crucial part of the diagnostic workup for liposarcoma, 14 and MDM2 amplification/overexpression has also been associated with a worse prognosis. 18 Determining biomarker status may help identify patients for clinical trials of targeted therapies and also prospectively identify patients likely to benefit from new therapies as they become available. While there are currently no targeted therapies approved for liposarcoma, therapies targeting MDM2, CDK4/6, or both have been investigated. Knowing their biomarker status could also empower patients to initiate conversations with medical professionals about emerging treatment options or opportunities to enroll in clinical trials. Unfortunately, the lack of clear guidance regarding biomarker testing in liposarcoma affects not only clinical practice but also access to testing. Many insurance plans will not pay for testing if guidelines do not call for it, and medical professionals are unlikely to order testing if they think it will not be covered.
This study was limited by its small size and the qualitative and descriptive nature of survey data, particularly the freeform responses from participants. Because the aim of the survey was to gain insights into the overall patient experience that are not typically captured in medical records, detailed data on each patient’s diagnostic workup and course of treatment were not collected. However, the qualitative methodology was also a strength as the survey responses provided a more comprehensive narrative of the burden of liposarcoma from the patient’s perspective, often in the patient’s own words. This produced new insights beyond those gained solely from medical records and outcomes data. To our knowledge, this is the first study to evaluate the patient journey for liposarcoma based on findings from a survey of patients in the United States.
Building on the limited amount of previously published data on the patient experience, the results of this study could guide future research aimed at enhancing liposarcoma care and patient QOL. For example, development of new diagnostic tools could facilitate a more accurate and timely initial diagnosis, which is important for selecting appropriate treatment, while identification of new biomarkers that inform prognosis or predict therapeutic response may advance the current understanding of treatment implementation. There is also a need for practical initiatives aimed at transforming the patient experience by increasing the understanding of liposarcoma throughout the medical community, improving the uptake of biomarker testing, enhancing access to sarcoma treatment centers, and providing additional sources of educational information and support to patients. A more comprehensive understanding of the disease’s impact—as illustrated by the qualitative statements on lived experiences collected directly from patients—could strengthen the case made to policy makers responsible for funding these efforts.
Supplemental material
Supplemental material - Burden and unmet needs of liposarcoma in the United States: Patient perspectives on the liposarcoma diagnostic journey
Supplemental material for Burden and unmet needs of liposarcoma in the United States: Patient perspectives on the liposarcoma diagnostic journey by Dan Mulligan, Amy Shaberman, Jennifer Han, Jennifer Horonjeff, Brandi Felser in Rare Tumors
Footnotes
Acknowledgments
The authors meet criteria for authorship as recommended by the International Committee of Medical Journal Editors (ICMJE). The authors did not receive payment related to the development of the manuscript. Medical writing and editorial support were provided by George Hsu, PhD and Amy Volpert, MA, CMPP™ of HCG, and funded by BIPI. BIPI was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations.
Ethical considerations
This survey received an exemption from institutional review board oversight by Advarra (Pro00072826).
Consent to participate
Participants received an email with an informed consent document that they read, signed, and returned. Patients were prompted to provide informed consent again before beginning the online survey by clicking on an “I agree and consent to participate” button. They were informed about the purpose of the survey, the information we were seeking to collect, how their information would be captured and used, and how their privacy would be ensured.
Author Contribution
Conceptualization: DM, AS, JHan, JHoronjeff, BF.
Data curation: JHoronjeff.
Validation: DM, AS, JHan, JHoronjeff, BF.
Writing – original draft: DM, AS, JHan, JHoronjeff, BF.
Writing – reviewing & editing: DM, AS, JHan, JHoronjeff, BF.
All authors conceived the study and wrote, reviewed, and edited the manuscript. Jennifer Horonjeff curated the survey data. All authors approved the final version of the manuscript
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported and funded by Boehringer Ingelheim Pharmaceuticals, Inc. (BIPI). The medical writing and editorial support were funded by BIPI.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dan Mulligan reports employment and consultancy with Corin Group, leadership with Stryker Corporation and 270 Surgical, and travel accommodations from Tyber Medical. Amy Shaberman reports employment at Boehringer Ingelheim Pharmaceuticals, Inc., stock ownership with Abbott Laboratories, Amgen, Bristol Myers Squibb, Johnson & Johnson, Pfizer, and United Healthcare, and travel accommodations from Boehringer Ingelheim Pharmaceuticals, Inc. Jennifer Han reports employment at Boehringer Ingelheim Pharmaceuticals, Inc., stock ownership with Incyte Corporation and Johnson & Johnson, and travel accommodations from Boehringer Ingelheim Pharmaceuticals, Inc. Jen Horonjeff reports employment at Savvy Cooperative and consultancy for AbbVie, Inc. Brandi Felser reports no disclosures.
List of where and when the study has been presented in part elsewhere,if applicable
Interim results were previously presented at the 2024 Connective Tissue Oncology Society Annual Meeting (Mulligan D et al., abstr 206); the 2024 American Society of Clinical Oncology Quality Care (ASCO QC) Symposium (Mulligan D et al., abstr 255), the 2024 The Academy of Oncology Nurse & Patient Navigators (AONN+) Annual Conference (Mulligan D et al., abstr I2; ASCO QC encore), and the 2024 Journal of the Advanced Practitioner in Oncology (JADPRO) Live Meeting (Mulligan D et al.; ASCO QC encore).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
