Abstract
Extranodal natural killer/T-cell lymphoma is a rare and aggressive malignancy with a predilection for Asian and Latin American populations. While typically presenting in the nasal cavity, extranasal forms—particularly primary pulmonary extranodal natural killer/T-cell lymphoma —are exceedingly rare and diagnostically challenging. We report the first documented case of primary pulmonary extranodal natural killer/T-cell lymphoma arising after chemotherapy for classical Hodgkin lymphoma (cHL), presenting concurrently with a nodal relapse of cHL in a 26-year-old male. Histopathological analysis confirmed synchronous diagnoses of Ebstein Barr virus-positive extranodal natural killer/T-cell lymphoma in lung tissue and Hodgkin lymphoma in lymph node biopsy. The patient responded favorably to DDGP chemotherapy followed by haploidentical stem cell transplantation. This case highlights the need for heightened clinical suspicion and histological confirmation in atypical lymphoma relapses, and underscores the complex interplay between Ebstein Barr virus infection, prior chemotherapy, and lymphomagenesis. Further research is warranted to clarify the pathogenesis and optimize treatment of metachronous extranodal natural killer/T-cell lymphoma following Hodgkin lymphoma.
Introduction
Extranodal natural killer/T-cell lymphoma (ENKTL) is a rare malignancy, accounting for less than 2% of all lymphomas and approximately 28.6% of T/NK-cell lymphomas. 1 It is more prevalent in Asian and Latin American populations, where it constitutes 40% to 50% of T/NK-cell lymphomas, compared to only 8% in Europe. 2 The median age at diagnosis ranges from 40 to 50 years, with a male predominance, reflected by a male-to-female ratio of 2 to 3:1. 3 ENKTL presents a significant challenge due to its clinical and biological heterogeneity. Clinically, ENKTL is classified based on the site of origin. More than 80% arise in the nose, nasopharynx, oropharynx, Waldeyer’s ring, and upper digestive tract, collectively termed nasal NKTL. In contrast, approximately 10% to 20% of cases occur in other anatomical sites, including the skin, bones, gastrointestinal tract, lungs, liver, and reproductive organs, and are classified as extranasal ENKTL. 4 Extranasal ENKTL typically exhibits a more aggressive clinical course and poorer prognosis.
Advances in therapeutic strategies have significantly improved the prognosis of ENKTL in recent years.5,6 For patients with Ann Arbor stage I/II disease, the 5-years overall survival has increased from less than 50% in the early 2000s to more than 80% with the introduction of early radiotherapy. In advanced-stage disease, the median OS has improved from a few months to over 3 years, largely due to the incorporation of asparaginase-based chemotherapy and PD-1-targeted immunotherapy. 7 Despite these advances, significant gaps in knowledge remain. The rarity of ENKTL limits the availability of large-scale clinical trials, and the biological mechanisms underlying its pathogenesis are not fully understood. Furthermore, cases of ENKTL arising after chemotherapy for Hodgkin lymphoma (cHL) are exceedingly rare, and the mechanisms driving their development remain unclear.
We present a case of a patient with extranasal ENKTL presenting as isolated pulmonary disease and a synchronous nodal relapse of cHL. To our knowledge, this is the first reported case of primary pulmonary ENKTL arising after chemotherapy for cHL and occurring concurrently with a synchronous cHL relapse. This case contributes to the expanding body of knowledge surrounding this diagnostically challenging malignancy.
Case report
A 26-year-old male presented with a 2-month history of a painful, indurated mass on the left side of the neck. This was accompanied by unintentional weight loss of 12 kg, nocturnal diaphoresis, and fever. His medical history was notable for stage IA nodular sclerosis Hodgkin lymphoma diagnosed at age of 16, for which he received two cycles of chemotherapy with the OEPA protocol (vincristine, etoposide, prednisone, doxorubicin) and 12 fractions of radiotherapy (2400 cGy), achieving complete remission confirmed by subsequent PET/CT follow-up imaging in 2015. Notably, he reported frequent outdoor activities, including spelunking and rafting in tropical waters.
On admission, he was febrile (39°C) and mildly tachycardic (109 bpm). Physical examination revealed a palpable cervical mass with adjacent lymphadenopathy, while the remainder of the examination was unremarkable. Laboratory results showed mild pancytopenia with lymphopenia and mild microcytic anemia (leukocytes: 2800/μL, neutrophils: 1700/μL, lymphocytes: 600/μL, hemoglobin: 12.4 g/dL, hematocrit: 37%, mean corpuscular volume: 77.9 fL, mean corpuscular hemoglobin: 33.4 pg, platelets: 133,000/μL) and elevated lactate dehydrogenase (LDH) at 498 U/L (reference range: 120–246 U/L). Renal function, liver function, electrolytes, and coagulation tests were within normal limits.
Neck computed tomography (CT) confirmed lymphadenopathy in the left IIb station. Chest CT revealed well-defined pulmonary solid nodules with surrounding ground-glass opacities and a random distribution, the largest measuring 20 mm in the right superior lower lobe, with one nodule showing adjacent cavitation. Additional findings included areas of ground-glass opacities and features suggestive of infectious bronchiolitis (Figure 1). Abdominal CT demonstrated splenomegaly without other lymphatic involvement. Extensive workup excluded bacterial endocarditis, tuberculosis, systemic embolism, pulmonary aspergillosis, and histoplasmosis. Serial blood cultures and bronchoalveolar lavage were unremarkable. Initial tomographic assessment. Upper images show a transversal view (a) and a coronal view of a neck CT with a left cervical lymphadenopathy (b). Lower images show a transversal view (c) and coronal view (d) of a chest CT showing multiple solid pulmonary nodules with ground-glass opacities.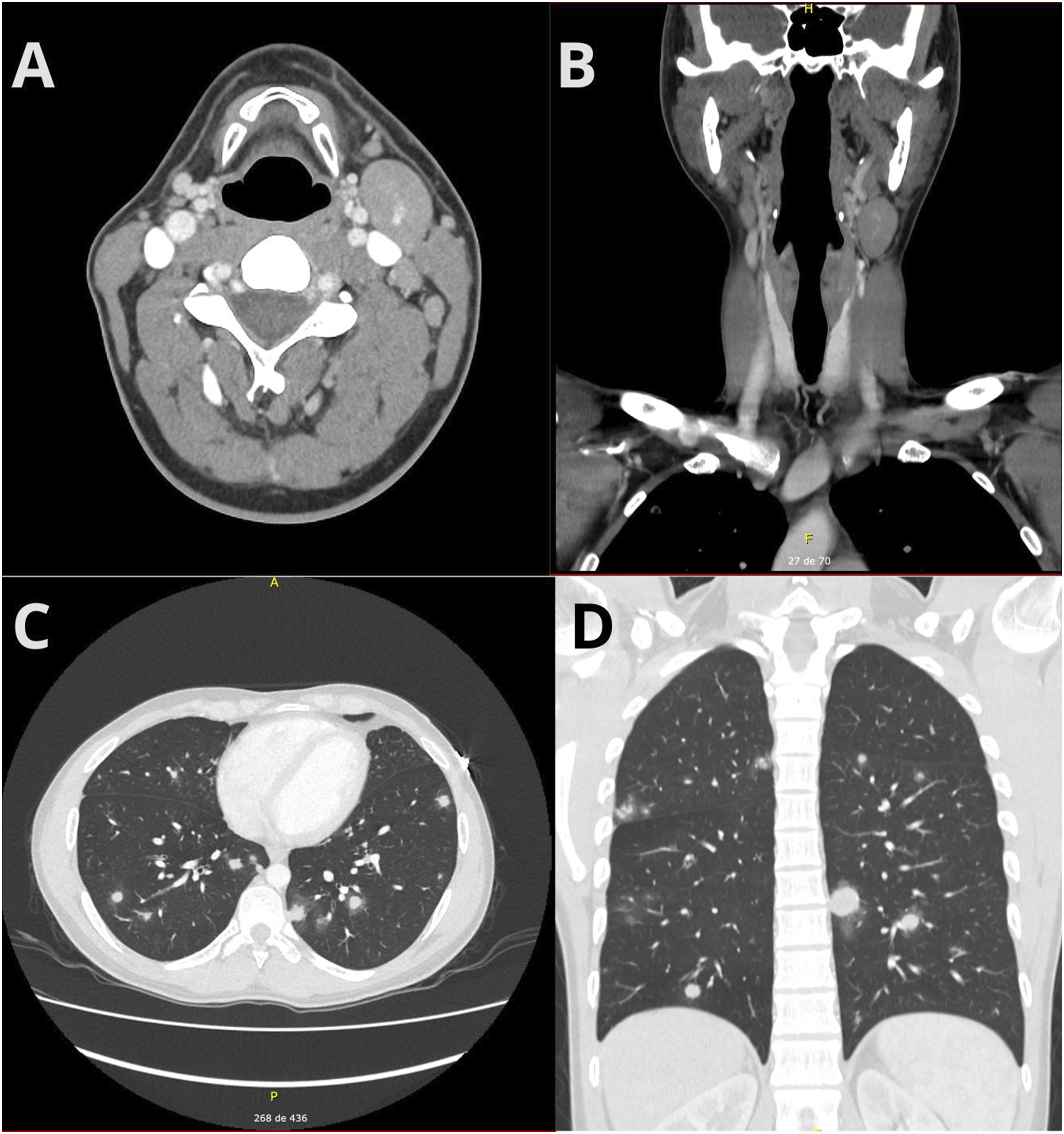
A cervical lymph node biopsy revealed disrupted nodal architecture with Reed-Sternberg cells and areas of interstitial hemorrhage. Immunohistochemistry was positive for CD30, CD15, MUM1, OCT2, BOB1, and BCL6, with a Ki-67 index of 70%. Negative markers included CD3, CD5, CD4, CD8, CD7, CD20, CD79a, and PAX5. In situ hybridization for Epstein-Barr encoding RNA (EBER-ISH) was negative, confirming a late recurrence of Hodgkin lymphoma. Despite antimicrobial therapy, the patient remained febrile and developed shock, requiring intensive care unit admission and broad-spectrum antibiotics, including liposomal amphotericin B.
As the patient’s clinical condition continued to deteriorate, a percutaneous and surgical lung biopsy was performed. Histopathological examination of the lung biopsy revealed fragments of pulmonary parenchyma infiltrated by a malignant neoplastic process characterized by large, atypical cells with irregular, cleaved, hyperchromatic nuclei, absent nucleoli, and moderate eosinophilic cytoplasm. Frequent apoptosis, mitosis, and areas of necrosis were observed. Periodic acid-Schiff (PAS) and Grocott staining showed no fungal structures. Immunohistochemistry was positive for CD3, CD56, CD5, CD4, CD7, MUM1, BCL2, BCL6, CD30 (<10%), CD2, granzyme B, perforin, and TIA-1, with a Ki-67 index of 60%. CD15, TTF1, ALK, and C-MYC were negative, and EBER-ISH was positive. These findings were consistent with ENKTL (Figure 2). The pulmonary ENKTL was classified as stage IE by the modified Ann Arbor system and stage II by the Chinese Southwest Oncology Group and Asia Lymphoma Study Group (CA) system, consistent with an extranasal primary without nodal involvement. According to the PINK model, the patient scored 1 point (extranodal site), corresponding to an intermediate-risk category. Histopathological features of extranodal NK/T-cell lymphoma, nasal type. Low-power view showing angiocentric and angiodestructive lymphoid infiltrate with extensive necrosis (H&E, left panel), and high-power view revealing atypical lymphoid cells with irregular nuclei and pale cytoplasm (H&E, upper right panel). Immunohistochemistry demonstrates positivity for CD3, CD56, TIA-1, granzyme B, and perforin, with loss of CD5 expression (lower right panel).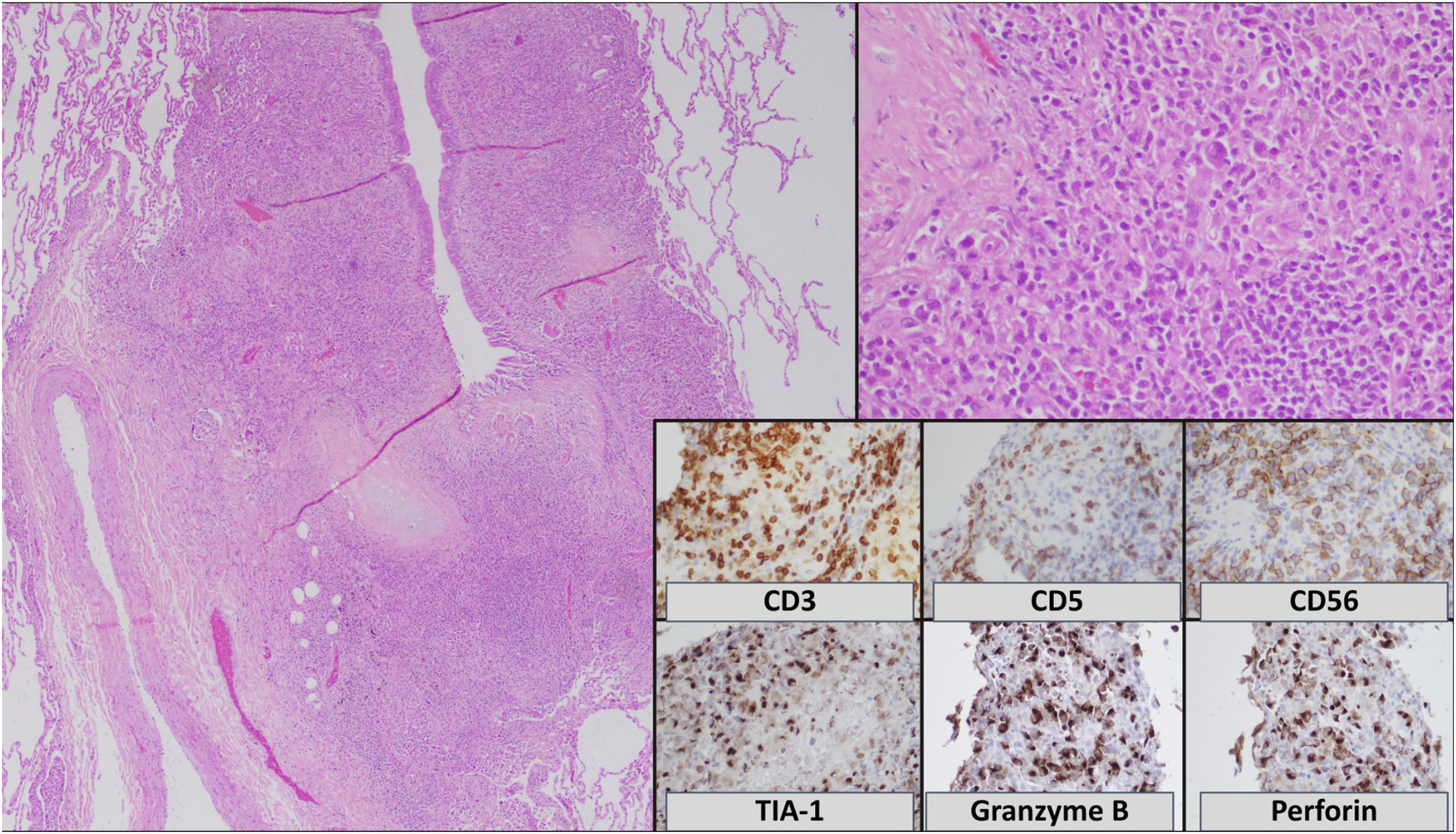
Chemotherapy was initiated with a DDGP protocol (dexamethasone, 15 mg/m2 intravenously on days 1–5; cisplatin, 20 mg/m2 intravenously on days 1–4; gemcitabine, 800 mg/m2 intravenously on days 1 and 8; L-aspargase, 2500 IU/m2 intramuscularly on day 1).
8
The patient showed clinical improvement within days, allowing extubation and transition to outpatient care (Figure 3). Follow-up chest CT demonstrated a significant reduction in pulmonary nodule size. After initiation of DDGP chemotherapy, the patient showed rapid clinical improvement, with resolution of fever and reduction in cervical lymphadenopathy. Post-treatment PET/CT after six cycles confirmed complete metabolic remission of both the relapsed Hodgkin lymphoma and the pulmonary ENKTL. The patient subsequently underwent allogeneic stem cell transplantation from a haploidentical donor without complications reported to date. He has been followed for 1 year with semiannual PET/CT imaging and monthly evaluations by the hospital’s hematology service, maintaining sustained complete remission and good functional status. Serial contrast-enhanced chest CT scans demonstrating the evolution of bilateral pulmonary infiltrates in a patient with extranodal NK/T-cell lymphoma. Progressive increase in ground-glass opacities and consolidations is observed from day 14 to day 40. Chemotherapy was started followed by partial radiological improvement by day 60.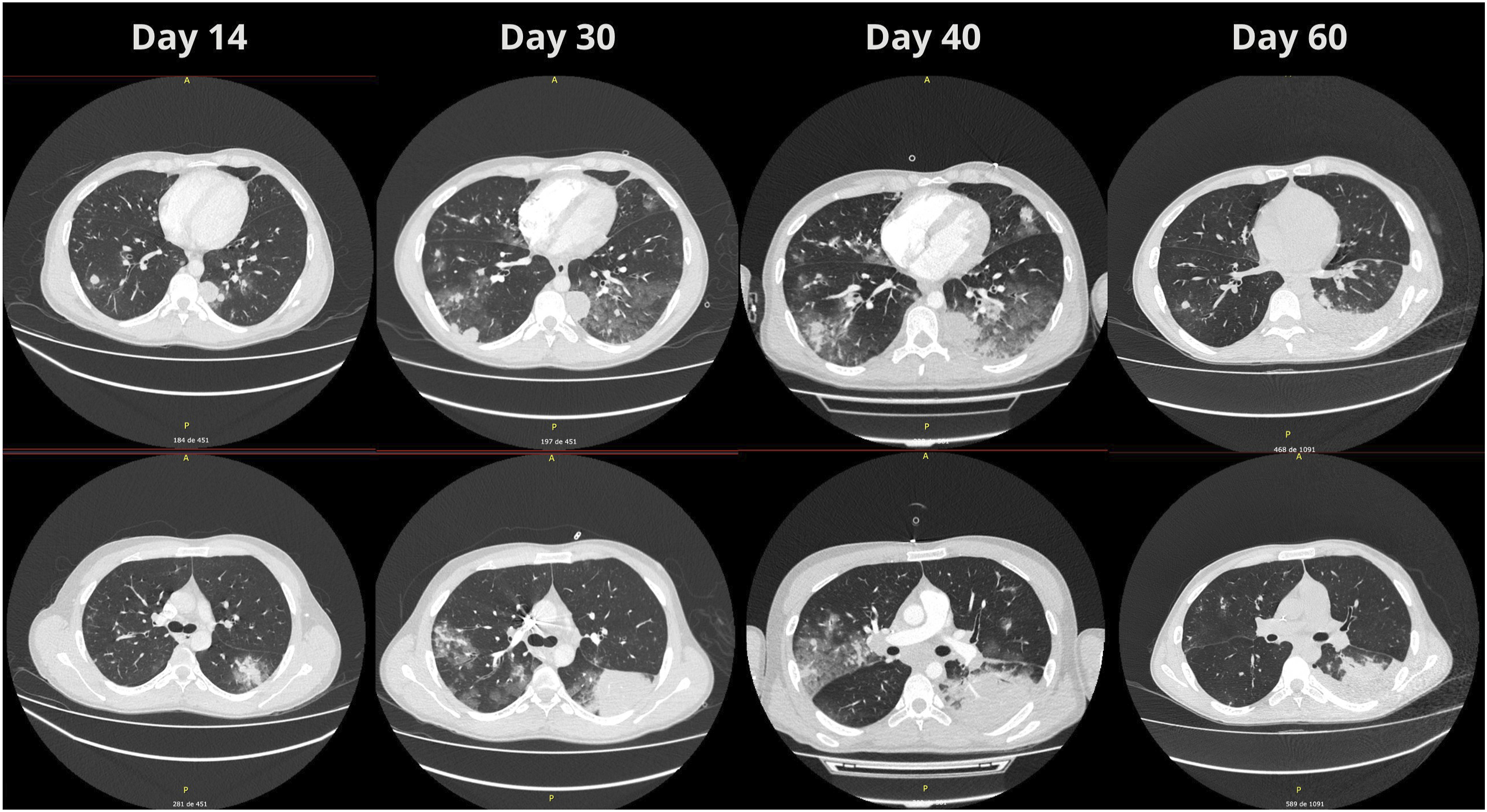
Discussion
Comparison between reported cases of ENKTL since the year 2000.

ASCT: Autologous stem-cell transplantation.
The introduction of asparaginase-based chemotherapy regimens has significantly improved the overall survival (OS) of patients with ENTKL, extending life expectancy from a few months to over 3 years in advanced-stage disease. 7 The SMILE protocol (steroids, methotrexate, ifosfamide, L-asparaginase, and etoposide) is the most widely studied, achieving complete response (CR) in 40-54% of patients with stage III/IV disease and a 5-years OS of 47%. 29 Other asparaginase-based regimens, including AspaMetDex, MEDA, and P-GEMOX, have shown comparable efficacy. The DDGP regimen (dexamethasone, gemcitabine, cisplatin, and pegylated asparaginase) has demonstrated improved CR rates and survival compared to SMILE, although existing studies lack the statistical power to establish definitive superiority. 30 To date, no randomized clinical trials have directly compared these regimens, leaving the optimal treatment approach unresolved.
Data on outcomes for primary pulmonary ENTKL treated with asparaginase-based regimens are limited. Historically, primary pulmonary ENTKL had a dismal prognosis, with a median survival of 2 months despite conventional chemotherapy. 4 More recent case series of 20 Chinese patients treated with asparaginase-based regimens reported an overall response rate of 88.2%, with a median OS of 9.5 months and a 1-year OS rate of 45%. 31 Evidence suggests that the improvement in survival obtained from the asparaginase-based regimes is transversal to all ENKTL. In this group OS was 9.5 months and the 1-year OS rate was 45%. Our case was treated with a DDGP protocol and showed similar results. The study also found that an older age (>60 years) and an ECOG >2 was associated with worse OS and PFS. Another 20-patient case series found lower survival rates in female patients. 32 Our case, treated with the DDGP regimen, demonstrated similar outcomes. Poor prognostic factors include age >60 years and an Eastern Cooperative Oncology Group (ECOG) performance status >2. Additionally, a separate case series found worse survival in female patients, potentially due to estrogen-mediated modulation of the tumor microenvironment and enhanced EBV cytolytic activity. 33
The coexistence of multiple lymphomas is well-documented. The term “synchronous lymphoma” refers to two histologically distinct lymphomas occurring simultaneously. Chemotherapy for classical Hodgkin lymphoma (cHL) has been linked to the subsequent development of both Hodgkin and B-cell non-Hodgkin lymphomas, likely due to intrinsic immunodeficiency and chemotherapy-induced genomic damage. 33 However, the emergence of ENTKL following cHL treatment is exceptionally rare, with only eight cases reported in the literature. 34 Among these, patient age ranged from 34 to 67 years, with a balanced sex distribution. The mean time from cHL treatment to ENTKL diagnosis was approximately 2 years (15–108 months). This report describes the first known case of primary pulmonary ENTKL following cHL chemotherapy.
Several mechanisms have been proposed to explain the development of ENTKL after cHL treatment. First, cHL-associated T-cells (CD4+, CD25+, and CCR4+) may acquire genomic rearrangements that promote a tumorigenic microenvironment. Second, EBV infection plays a pivotal role in early ENTKL pathogenesis by infecting T-cells via the CD21 receptor. 35 Third, persistent EBV exposure in the context of chemotherapy-induced DNA damage may drive the oligoclonal proliferation of malignant T-cells. Some authors suggest a common origin for both malignancies based on T-cell receptor (TCR) rearrangements in patients with both malignancies. However, this hypothesis is based on a single case report, and subsequent studies have not confirmed a shared clonal lineage. In contrast, another case report found CD3 expression in only 3–10% of cHL cells and identified aberrant immunoglobulin gene rearrangements, arguing against a common origin. 36 Furthermore, cytogenetic analyses reveal distinct chromosomal abnormalities in cHL and ENTKL, supporting independent clonal origins. 37 Among the patients with metachronous cHL and ENTKL, four underwent TCR rearrangement testing, and none exhibited positive results. 34
There is a need for robust studies to establish the epidemiology of synchronous cHL and ENKTL, the exact pathophysiology underlying the development of ENKTL following cHL treatment, and the most effective treatment regimen for these scenarios. This need is particularly urgent in pulmonary ENKTL, where specific information remains lacking. We present the first reported case of primary pulmonary ENKTL related to cHL treatment, a diagnostically and therapeutically challenging overlap of distinct lymphoid neoplasms. Its strengths include precise clinicopathologic definition, comprehensive immunophenotypic validation, and documented complete metabolic response of both entities to asparaginase-based chemotherapy with allogeneic transplantation. Limitations stem from its single-case nature, the absence of molecular or clonal analyses to clarify potential lineage relationships, and the relatively short follow-up, which restrict broader inference. Nonetheless, it contributes to the refinement of diagnostic and therapeutic frameworks for concurrent lymphoid malignancies, emphasizing the need for integrated molecular characterization in future reports. This case underscores the importance of histopathological evaluations in patients with late relapse of lymphoma and considering primary pulmonary lymphoma as a differential diagnosis in patients presenting with lung nodules.
Footnotes
Ethical considerations
Ethics committee approval was not required for a single case report according to institutional guidelines.
Consent to participate
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author contributions
D.C.O, L.B., and M.A.Z. contributed to the conceptualization, data collection, supervision, and writing of the article. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data is available upon request.
