Abstract
Tumors originating from soft tissues are uncommon, among these tumors, liposarcomas are the most frequent. These tumors remain asymptomatic for a long time, and only revealing themselves when they reach an important size. In such cases, treatment is difficult, requiring extensive surgery procedures that can excise several adjacent structures, potentially completed by adjuvant radiotherapy. Despite successful treatment, the recurrence rate remains very high. We report the case of a giant liposarcoma requiring a monobloc extensive resection involving the removal of the tumor, left kidney, left adrenal gland, and a portion of the posterior abdominal wall.
Introduction
Retroperitoneal liposarcomas (LPS) are rare primitive malignant mesenchymal tumors originating from fat cells in the retroperitoneal space. 1 These tumors remain challenging to diagnose and to treat. 2 The standard treatment is complete surgical excision, but this is often difficult, given the absence of anatomical boundaries outside the renal cavity in the retroperitoneum, and the large size of these sarcomas. Therefore, successful resection with uninvaded R0 margins is difficult to achieve 3 and the prognosis of these tumors remains poor, with a high rate of locoregional recurrence. 3 We present a case of giant sarcoma to highlight the anatomical particularities and surgical challenges of this localization within the retroperitoneum.
Case report
A 56-year-old patient, with no previous history, presenting a painless abdominal mass of gradually increasing volume for 2 years, accompanied by vomiting, with significant weight loss and general asthenia.
Physical examination revealed an enormous mass occupying all abdominal quadrants (Figure 1). Biological analyses revealed anemia with hemoglobin at 9 g/dl, lymphocytes at 12,000/m3 and a CRP at 137 mg/L, with normal tumoral markers values. Computed tomography (CT) revealed a 45 cm wild retroperitoneal mass, incorporating the left kidney and adrenal gland and displacing the aortocaval, iliac and mesenteric vascular axes alongside the small intestine, colon, spleen and liver, without infiltration (Figure 2(A)–(C)). A complementary magnetic resonance images (MRI) showed a giant retro and intra-peritoneal, multi-loculated mass, with mixed T2 hypersignal areas and iso-signal zones heterogeneously enhanced measuring 38 cm, englobing the left kidney and displacing the adjacent structures and compressing the left ureter with ureterohydronephrosis (Figure 2(D)). Based on this finding, the tumor was judged unresectable by a first medical team, which decided to perform a biopsy to confirm the diagnosis before initiating neoadjuvant chemotherapy with Gemcitabine in aim to reduce the tumor size. After 1 year of treatment, the patient’s condition showed no improvement a new discussion during a multidisciplinary meeting led to the decision to perform surgical excision of the tumor, along with resection of the biopsy tract. Giant abdominal masse englobing the entire abdomen. Computed tomography (CT) and magnetic resonance imaging, axial view. (A) Contrast-enhanced abdominal CT scan showing a 45 cm diameter giant intra and retroperitoneal heterogeneous tumor (blue arrows) occupying the pelvic and abdominal cavity. (B) The tumor (blue arrows) is incorporating the left kidney, ureter and adrenal gland (yellow arrows) and displacing the transverse and left colon (green arrows). (C) The mass is displacing the spleen, its pedicle and the left pancreas (with arrows). (D) Abdominal MRI showing a giant retro and intra-peritoneal, multi-loculated mass, with mixed T2 hypersignal areas and iso-signal zones heterogeneously enhanced measuring 38 cm, englobing the left kidney and displacing the adjacent structures.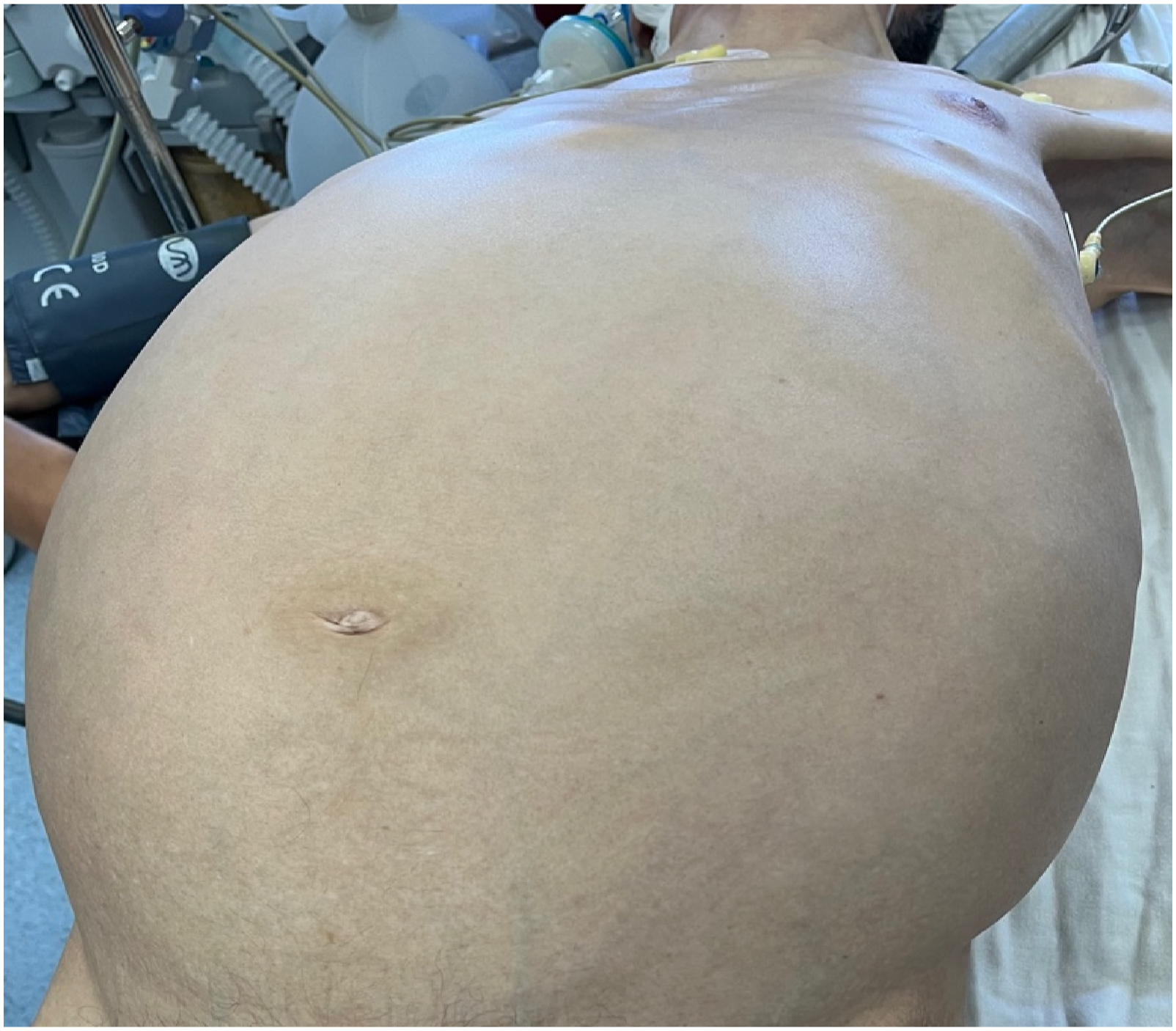

A xypho-pubic laparotomy revealed a giant tumor reaching the pelvis, incorporating the left kidney, ureter and adrenal gland, and displacing without invading the transverse and left colon and englobing the splenic vein and the left pancreas (Figures 3 and 4). The considerable size of the tumor facilitated its separation from the colon, which was displaced forward. However, the mesocolon, weakened by the tumor’s mass effect, coupled with the close contact with the tail of the pancreas and the splenic pedicle, significantly increased the risk of bleeding. This required meticulous and progressive dissection, starting with a left colo-parietal detachment allowing the liberation of the left colon and access to the retroperitoneal mass. Following a careful dissection of the spleen, its pedicle and the left pancreas from the tumor, an extended single-block resection passing by macroscopically non-invaded areas was performed, removing the tumor, the left kidney and left adrenal gland. The surgical procedure lasted 3 h with a 150 cc blood loss. Xypho pubic laparotomy revealing a giant tumor pushing forward the left colon. (blue arrows). Fully exposed liposarcoma.
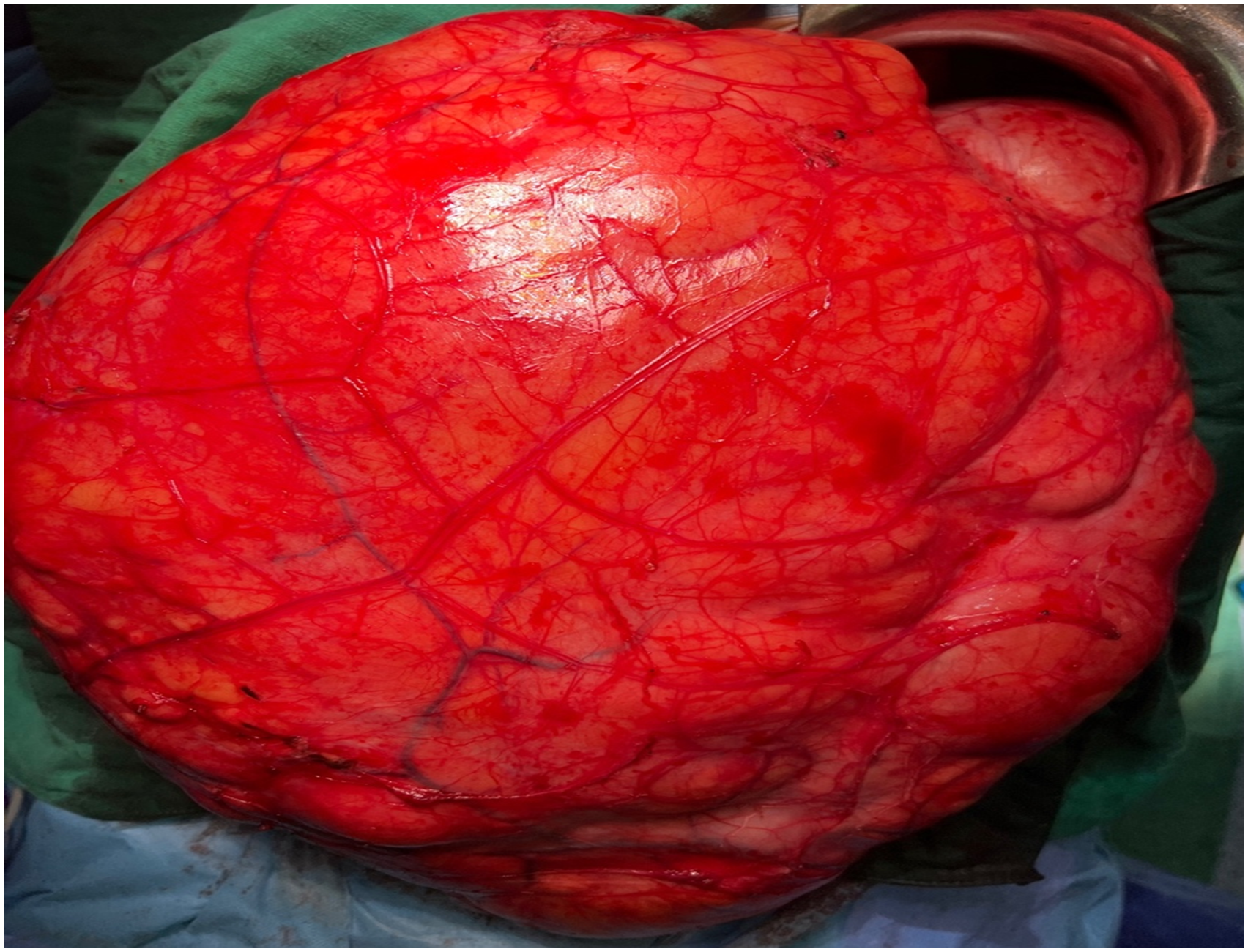
The post-operative follow-up showed no complication, and the patient was discharged 4 days after surgery.
The specimen (Figure 5) was weighted at 23 kg. Anatomopathological analysis (Figure 6) revealed a 59 cm diameter well-differentiated lipoma-like liposarcoma that included the renal and adrenal parenchyma without invading them, and with invaded R1 excision limits, however despite that the specimen was well-marked and properly oriented the significant volume of the tumor and its multiple contacts with adjacent structures made it impossible for the pathologist to precisely determine the area of tumor residue making it difficult to determine the area for potential radiotherapy. Therefore, during a multidisciplinary meeting and considering the slow progression of the tumor, surveillance was recommended. Resection specimen weighted at 23 kg. Histopathological findings: (A) adipocytic tumor proliferation encompassing renal parenchyma without invasion. (B): Well-differentiated tumor proliferation showing a few atypical adipose cells mixed with non-atypical adipose cells within an inflammatory stroma. (C): Atypical adipocytic cells exhibit irregular and hyperchromatic enlarged nuclei.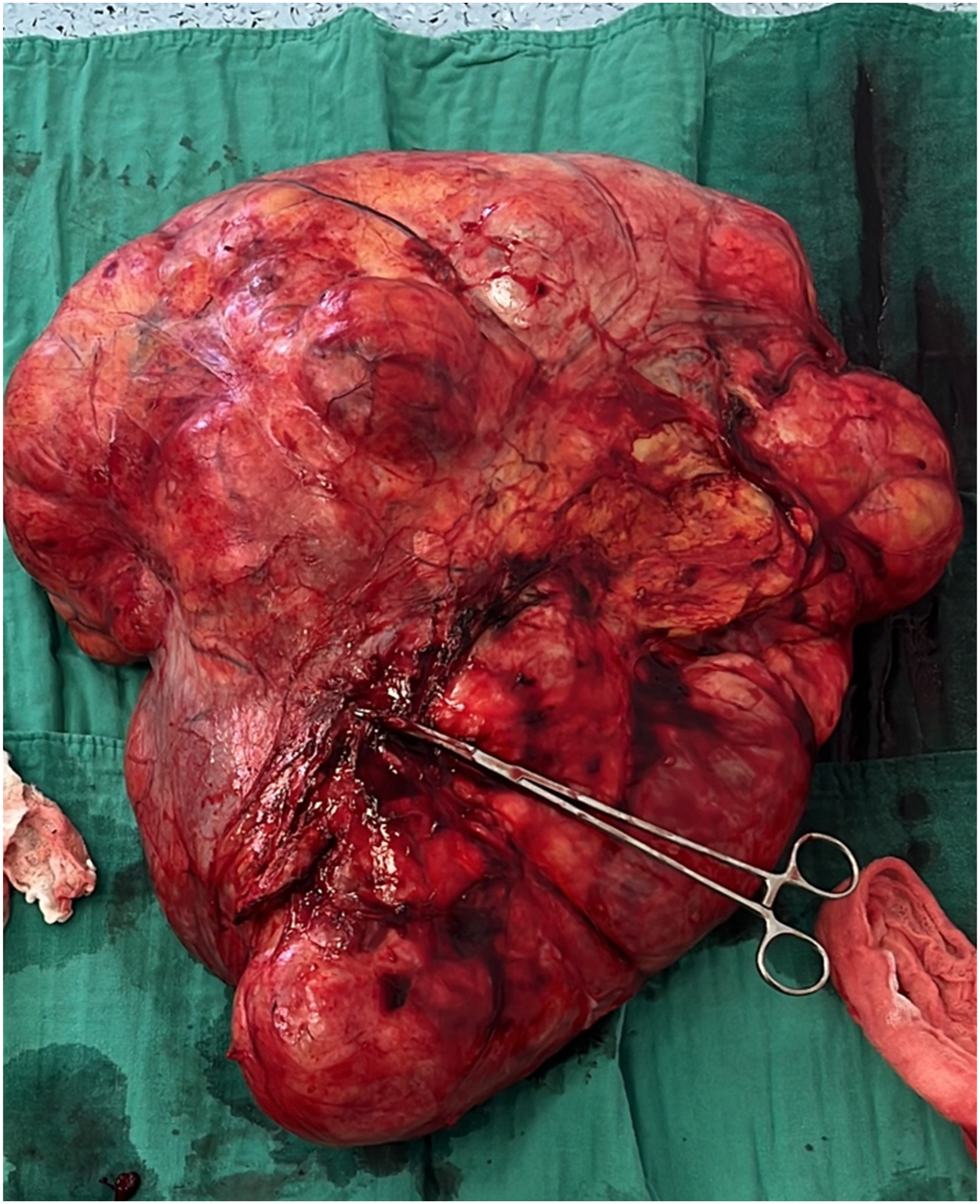

Discussion
Liposarcomas are the most common adult soft-tissue tumors (12.8% all localizations combined). 4 LPS is considered rare, but several recent studies have reported an overall increase in its incidence. The retroperitoneal location accounts for only 10% to 20% of sarcomas, with an overall incidence of 0.3% to 0.4% per 100,000 of the population, 5 with male predominance (60% of cases) and a preference for caucasians over 60 years old. 6
Their etiological factors have not yet been clearly determined, but environmental genetic factors, history of irradiation, viral infection or immunodeficiency, have been incriminated. 7 Finally, rare cases of post-traumatic liposarcoma have been reported. 8
The 2020 WHO classification divides LPS into five major subtypes, 9 all of which develop in the retroperitoneum and present specific histological aspects and genetic mutations 10 :
Well-differentiated liposarcomas (WDLPS) are locally aggressive tumors with low metastatic potential, accounting for 40–45% of all liposarcomas. 11 From a histological perspective, it can be categorized into three distinct subgroups that can co-exist simultaneously within the same tumor: well-differentiated “Lipoma-like,” sclerosing and inflammatory LPS. 11 Dedifferentiated liposarcoma (DLPS) is an atypical lipomatous tumor arising de novo 90% of the time and 10% develop in recurrences. 12 The myxoid form (MLPS) which is more aggressive, with greater recurrence potential and a poorer prognosis and represents 20 to 30% of all LPS. 12 Pleomorphic liposarcomas (PLPS) that have the poorest prognosis. 11 Lastly, myxoid pleomorphic liposarcoma which is also an exceedingly rare adipocytic malignancy, most commonly occurring in the mediastinum, the limbs and neck region. 12
Retroperitoneal LPS are generally asymptomatic for a long time, 9 when diagnosed, they may have reached significant sizes, with over 50% of tumors exceeding 20 cm, manifesting compressing signs of the nearby structures. 9 This results in the diagnosis frequently being established radiologically. CT and MRI imaging are the two most effective modalities in retroperitoneal LPS. Enabling differentiation between the different subtypes. 13 WDLPS typically contains more than 75% adipose tissue with septations thicker than 2 mm and small internal nodular areas discernible on CT images. On T2 MRI these septations and nodular areas exhibit hyperintense signals, distinguishing it from other subtypes. 13
While similar, WDLPS and DLPS can be differentiated by larger non-lipomatous components containing nodular areas. 13 MLPS usually exhibits low signal intensity in T1 and intermediate signal intensity in T2. 13 PLPS shows minimal fat attenuation on CT. However, hemorrhage and necrosis commonly occur in PLPS, causing imaging heterogeneity and complicating diagnosis. 13
Although theoretically distinguishable, no specific subtype was diagnosed before surgery in our case.
Surgical excision is the treatment of choice. The trans-peritoneal laparotomy approach is the most commonly used, but depending on the tumor location, a thoracoabdominal, median or transverse approach is also possible. 5 A few cases of laparoscopic resection have been reported, 14 but these appear to be reserved for tumors measuring less than 10 cm without signs of local invasion. One case of recurrence at trocar insertion has been reported. 14
The challenge of sarcoma surgery is to achieve adequate local surgical control, which is often difficult due to the presence of large vessels, and retroperitoneal organs, requiring extensive resections. A unanimous definition of a safety margin for retroperitoneal LPS remains elusive. 15 However, there is a consensus that resection margins displaying macroscopic invasion elevate the likelihood of recurrence, emphasizing the importance of preserving the tumor integrity. Therefore, piecemeal resection should be avoided since it also increases tumor cell dissemination and the procedure duration which is also an important predictor for 1-year recurrences. 16
Some studies 17 emphasize the importance of nephrectomy in LPS surgery, as it increases R0 resection rates and reduces local recurrence. 17 Mortality rate is 10% 18 and the R0 resection rate varies from 38% to 74% depending on the series. 11
Adjuvant and neoadjuvant therapy is controversial. 16 Neoadjuvant chemotherapy (doxorubicin, alkylating agents) is indicated only in locally advanced or metastatic tumors, where it can reduce tumor volume therefore improving surgical performances. 11 Radiotherapy has been shown to significantly reduce the risk of local recurrence 19 at the cost of increased gastric and enteric morbidity (radiation-induced gastroenteritis, gastro-enteric fistulas, intestinal occlusions), which may require additional surgery. 11 Currently, high-dose neoadjuvant radiotherapy (45 to 50 Gy) is preferred. 11 Pre-, intra- or post-operative radiotherapy has, however, been of benefit in some patients, but, in most instances, does not improve patient prognosis. 5
LPS are characterized by their high recurrence rate, 15 which can be up to 65% at 5 years. 11 These recurrences can occur between 6 and 24 months after the initial procedure. 20 Numerous predictive factors have been identified, including age, tumor grade and histological type. 21 Size has long been considered an independent risk factor for recurrence, 20 but several studies have demonstrated that this is highly unlikely. 22
The management of LPS recurrences is also a surgical matter, these surgeries can be difficult and even impossible sometimes. 15 Few retrospective series have shown that a longer time to recurrence is associated with better oncologic outcomes. 18 So a surveillance approach can spare the patient the morbidity of such a procedure and since the probability of long-term disease-free survival is low, a period of observation may be helpful in selecting appropriate patients for resection. 15
Conclusion
Retroperitoneal liposarcomas are rare tumors, primarily treated surgically. This surgery is often difficult because of the tumor’s large size and location, which increases the probability of incomplete resection and local recurrence.
