Abstract
Mural nodules are rarely identified in cystic ovarian neoplasms, and have been categorized into sarcoma-like, sarcomatous, and anaplastic carcinomatous types. Most reports of these mural nodules have been described in mucinous ovarian tumors. In this case report, we describe an ovarian serous borderline tumor with mural nodules composed of high-grade carcinoma with anaplastic features and necrosis, including the morphologic features, immunoprofile, and results of tumor DNA sequencing. Omental involvement was also identified. Recognition of this phenomenon in serous tumors is important, so that thickened areas of cyst wall in ovarian serous tumors will be thoroughly examined.
Case
Informed consent was sought, but unable to be obtained, as the patient was deceased at the time of preparation of the manuscript, and the patient’s legally authorized representative was unable to be contacted. This case report is not classified as research by the Human Research Protection Office at the University of Pittsburgh and therefore approval is not required. No Health Insurance Portability and Accountability Act (HIPAA) identifiers or other identifying characteristics are included in this manuscript. The patient, a mid-70s female, presented with longstanding gastrointestinal symptoms and underwent computed tomography (CT) of the abdomen and pelvis, showing a 14 cm cyst probably arising from the right ovary. Physical examination revealed a large cystic smooth pelvic mass on bimanual examination. Pre-operative CA-125 was elevated at 380.6 U/mL, and carcinoembryonic antigen testing was within normal limits. The patient underwent laparotomy, revealing a large cystic mass arising from the right ovary with some adhesions. Intraoperative rupture was noted during dissection of adhesions. The right adnexal mass was described on gross examination as a previously opened, 15 cm in greatest dimension, uniloculated and cystic mass with minimal papillary excrescences, although abundant thickened areas with tan-white focally hemorrhagic and focally necrotic cut surfaces were identified.
Frozen section diagnosis of the mass was requested, and definitive diagnosis was deferred to permanent histopathologic examination, with a diagnosis of “at least borderline tumor with extensive inflammation and necrosis”. A 3 cm firm white omental nodule was also identified intraoperatively. Staging procedure was ultimately performed, to include total hysterectomy, bilateral salpingo-oophorectomy, omentectomy, pelvic and peritoneal biopsies, and pelvic and para-aortic lymphadenectomy.
Histopathologic examination of the mass demonstrated characteristic features of serous borderline tumor lining the cystic spaces, composed of mostly bland columnar epithelial cells with apical snouts and inconspicuous nucleoli. In some foci, comprising greater than 10% of the serous component of the neoplasm, epithelial proliferation and hierarchical branching were noted (Figure 1(a)). No evidence of mucinous differentiation was identified, and no high-grade nuclear features were noted in the cyst lining cells. Within the wall of the cystic mass, multiple nodules were identified, composed of a spindled and epithelioid proliferation which abutted the serous lining in some areas but did not exhibit a biphasic growth pattern (Figure 1(b)). The epithelioid areas demonstrated cells with enlarged vesicular nuclei, moderate nuclear pleomorphism, and abundant eosinophilic cytoplasm with admixed inflammatory cells predominantly composed of lymphocytes and neutrophils. Focally, the epithelioid cells showed a papillary growth pattern with more marked nuclear pleomorphism (Figure 1(c)). Other nodules were composed of cells with similar nuclear features; however, the cells showed spindled morphology with some fascicular growth pattern with more conspicuous mitotic figures and multiple foci of necrosis (Figure 1(d)). Tumor involving the omentum showed high-grade features and a predominantly spindled morphology, similar to the tumor seen in Figure 1(d). Representative hematoxylin and eosin (h and e) images of the tumor. (a): Focus of epithelial proliferation with stratification, hierarchical branching, and mild nuclear atypia; (b): Nodule composed of spindle cells, abutting serous cyst lining; (c): Focal papillary epithelioid growth pattern within nodule; (d): Nodule with spindle morphology.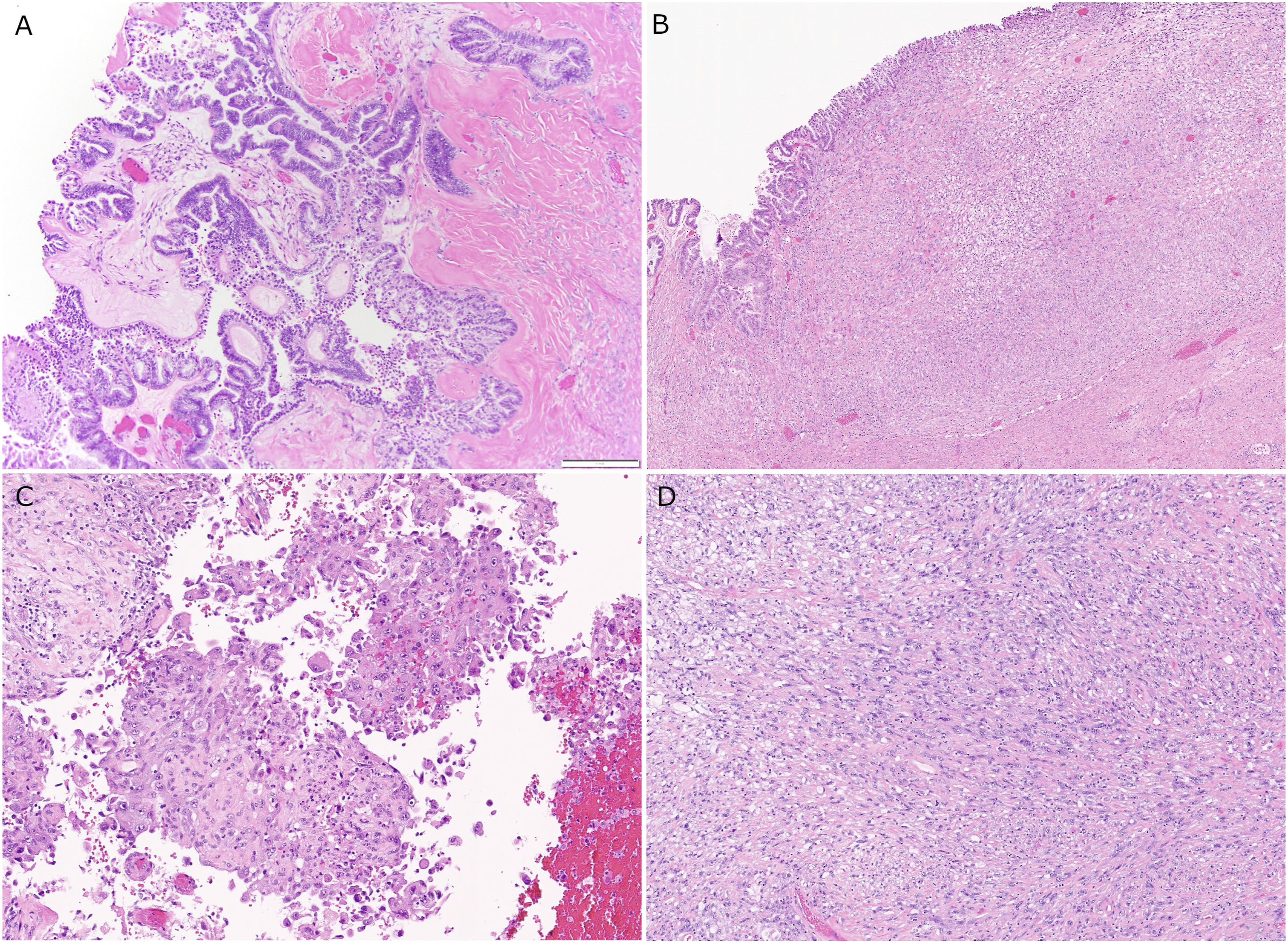
Immunohistochemistry results with antibody clone and vendor information.


Immunoprofile of serous cyst lining and anaplastic nodule. (a): Cytokeratin eight shows diffuse reactivity in cyst lining and nodule; (b): p53 shows wild-type reactivity in serous component, but diffuse strong nuclear reactivity in anaplastic nodule; (c): PAX-8 shows diffuse reactivity in cyst lining and nodule; (d): WT-1 shows strong nuclear reactivity in cyst lining but only weak expression in anaplastic nodule.
Mismatch repair protein immunohistochemistry showed preserved expression of MLH-1, PMS-2, MSH-2, and MSH-6. PD-L1 testing using the 22C3 antibody was reported as positive with a CPS of 40 (Caris Life Sciences). Tumor DNA sequencing (Caris Life Sciences) was reported to show pathogenic variants in KRAS (Exon 2, p.G12D) and TP53 (Exon 4, p.W91) genes. Variants of uncertain significance were also noted in the ATM (Exon 8, p.R337H) and BRAF (Exon 9, p.R389H) genes.
Final pathologic stage was International Federation of Gynecology and Obstetrics (FIGO) Stage IIIC due to omental involvement. The patient received adjuvant combination chemotherapy with carboplatin and paclitaxel. Unfortunately, intraabdominal recurrence in the liver and pelvic peritoneum was identified on imaging approximately 5 months following the surgical resection of the mass, treated with bevacizumab and doxorubicin. No significant response to treatment was identified on follow-up CT imaging 2 months later. The patient expired soon thereafter.
Discussion
The differential diagnosis of ovarian cystic lesions is broad, and can include physiologic entities, simple epithelial cysts with a variety of lining cell types, borderline tumors, and malignant tumors. Borderline tumors show epithelial proliferation that falls short of a malignant diagnosis, and a diagnosis of carcinoma is generally reserved for epithelial neoplasms that demonstrate frank stromal invasion, confluent growth pattern, or unequivocal high-grade nuclear features. The most common types show a serous or mucinous epithelial lining. Mural nodules, since first described by Prat and Scully, 1 have been well-documented in the literature in mucinous tumors, despite their rarity. To be considered a mural nodule microscopically, these lesions should demonstrate little, if any, focal admixture with the associated tumor. 2 These mural nodules have been divided into reactive sarcoma-like, anaplastic carcinoma, and sarcomatous nodules. 3 Reactive sarcoma-like nodules are difficult to diagnose, but may be well-circumscribed, may show a prominent inflammatory reaction, do not have associated necrosis or vascular invasion, and may have a better prognosis. 4 Anaplastic carcinoma nodules usually show reactivity for some cytokeratins by immunohistochemistry, while sarcomatous nodules typically do not.
The pathogenesis of mural nodules in cystic ovarian epithelial neoplasms is poorly understood. In a prior case report of ovarian mucinous carcinoma with anaplastic carcinoma mural nodules in a 20-year-old, continuity between the epithelial element and sarcoma-appearing element was described, suggesting dedifferentiation and epithelial to mesenchymal transformation of the mucinous carcinoma. 5 In a recent study of 13 ovarian mucinous tumors with associated mural nodules, 12 of the cases showed genetic evidence of clonality between the mural nodule(s) and associated mucinous tumor, including in sarcoma-like mural nodules, although no single recurrent genetic alterations were identified to suggest a “trigger” for mural nodule development. 6 Desouki, et al. reported a case of a mucinous ovarian adenocarcinoma with mural nodule of anaplastic carcinoma showing identical K-RAS mutations in each component, suggesting a form of dedifferentiation of the mucinous carcinoma. 7 A K-RAS mutation (c.35G > A p.G12A) was also previously reported in both the mucinous and anaplastic carcinoma nodule components of an ovarian mucinous tumor with a mural nodule. 8
In contrast to mural nodules associated with mucinous ovarian tumors, only very rare examples have been described in cystic ovarian tumors with a serous phenotype. Baergen, et al., described a case with bilateral serous cystadenocarcinoma with peritoneal involvement, in which one ovary showed sarcomatous mural nodules, with positive vimentin, but negative cytokeratin, EMA, and desmin by immunohistochemistry. 2 Gungor, et al., published a report of a 54-year-old with ovarian serous borderline tumor with both sarcomatous and sarcoma-like mural nodules. 9 Parker, et al. described a high-grade abdominal recurrence of a serous borderline tumor (SBT) with noninvasive implants that showed sarcomatoid-type carcinoma with microscopic foci of better differentiated tumor that resembled the primary SBT; however, the spindled and high-grade areas were not identified in the primary tumor. 10
In our case, mucinous differentiation was not identified in the epithelial component. The epithelial cyst lining showed cuboidal to columnar cells with focal cytoplasmic clearing and hierarchical branching characteristic of an SBT. No high-grade nuclear features were identified in the cyst lining cells. Immunohistochemical stains showed a characteristic profile of serous phenotype, including expression of CK7, ER, PR, and WT-1, with negative CK20 and wild-type p53 expression pattern. Based on the morphology and unusual nature of the histologic findings, carcinosarcoma (malignant mixed Mullerian tumor) was considered in the differential diagnosis, but a true biphasic growth pattern was not present and the cyst lining component was not frankly malignant. Adenosarcoma could also be considered in the differential diagnosis, but again, this lesion did not show a biphasic growth pattern, the epithelial component showed significant proliferation not usually present in adenosarcoma, and the tumor was predominantly cystic. The presence of KRAS and TP53 mutations is also of interest, as serous borderline tumors and low-grade serous carcinomas of the ovary can show KRAS mutations, while TP53 mutations are more often identified in high-grade serous carcinoma and carcinosarcoma, although the significance of this finding is unclear because the testing was performed at an outside laboratory and it is unclear if tumor microdissection was performed.
Based on the morphologic features and immunoprofile of both the cyst lining component and the cells comprising the mural nodules, this case appears to represent an exceedingly rare example of anaplastic carcinomatous mural nodules involving a serous borderline tumor of the ovary.
Conclusion
Although very rare, mural nodules can be seen in serous ovary cystic tumors. The anaplastic carcinomatous component was identified in the omentum and likely contributed to the unfavorable outcome reported in this patient. It is important to recognize this phenomenon in order to perform adequate tissue sampling of thickened areas of the wall of the ovarian tumor, and in histologic examination to arrive at the correct diagnosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
