Abstract
Signet-ring cell carcinomas are an aggressive, poorly differentiated, and highly invasive adenocarcinoma carrying a poor prognosis. Most of these tumors originate in gastrointestinal organs; however, primary lung signet-ring cell adenocarcinomas can rarely occur. Tumoral lymphatic infiltration is a complication of these tumors and can cause phenomena such as lymphangitic carcinomatosis, characterized by a nodular thickening of the pleura, pleural effusions, and mediastinal lymphadenopathies. We report a case of a 63-year-old ex-smoker with a 2-week clinical course of dyspnea and pleuritic chest pain in which a nodular thickening of the pleura and pleural effusion were documented and led to the diagnosis of a primary signet-ring cell adenocarcinoma of the lung with lymphangitic carcinomatosis. This complication has never been described in the context of a primary lung tumor of this subtype. Both entities carry a high mortality and have no therapeutical options. This report adds to the information available about them.
Introduction
Signet-ring cell carcinomas (SRCCs) are a subtype of mucinous adenocarcinoma characterized by the presence of cells with high intracellular mucin content that pushes the nucleus peripherally, giving it a “signet ring” morphology. 1 SRCCs originate from undifferentiated cells of the lamina propria and have the ability to penetrate the submucosa and generate distant metastases. Because of their lack of differentiation, rapid growth, and invasive nature, SRCCs are usually tumors with a poor prognosis, with an average survival time of about 6 months and a 5-years survival rate of less than 10%.2,3 According to the SEER (Surveillance, Epidemiology, and End Results) registry, which collected data from 2001 to 2015, the vast majority of SRCCs were of gastrointestinal origin; 64% were gastric, 18% colon, 5% esophageal, 3.5% rectal, 1.8% pancreatic, and 1.1% gallbladder, while in contrast, SRCCs originating in other organs such as lung, breast, and bladder were much less frequent. The estimated incidence of primary lung SRCC is rare and ranges from 0.14 to 1.9% in the previously reported cohorts.1,4 Patients with this type of tumor tend to be younger, have a more advanced stage at diagnosis, and have higher requirements for chemotherapy compared to patients with other lung adenocarcinomas.5,6 Because of the low prevalence of primary lung SRCCs, information regarding the clinicopathological characteristics, therapeutical options, and the treatment recommendations for this type of tumor remain unknown. Among the complications of the thoracic metastatic disease, lymphangitic carcinomatosis (LC) is one of the rarest, characterized by an irregular nodular thickening of the pleura, pleural effusions, and mediastinal lymphadenopathies secondary to lymphatic tumoral invasion. 7 SRCCs of gastrointestinal origin have been reported to cause LC but never in the context of primary lung SRCCs.
We report a case of a primary lung SRCC whose initial finding was an unspecific nodular thickening of the pleura in the imaging studies and our diagnostic approach to LC as a secondary complication. This case adds to the missing information regarding SRCCs and rare subsequent complications like lymphangitic carcinomatosis.
Case report
We present the case of a 63-year-old male patient who consulted the emergency department of our institution with a 2-week clinical course consisting of progressive dyspnea, deterioration of his functional class, and pleuritic chest pain in the right hemithorax. His previous medical history included end-stage chronic kidney disease secondary to IgA nephropathy on renal replacement therapy with peritoneal dialysis, atrial fibrillation, and arterial hypertension. The patient reported regular tobacco use for 20 years that had stopped 15 years before the onset of symptoms. Daily medication included losartan (50 mg every 24 h) and metoprolol succinate (50 mg every 12 h). On physical examination, the patient was in acceptable general conditions, vital signs were in normal ranges, and the only pathological finding was the decrease of respiratory sounds in the base of the right lung. Paraclinical findings were normal except for a markedly positive D-dimer (4.4 μg/dL, reference range adjusted for age: <0.6 μg/dL) and a chest X-ray with right pleural effusion (Figure 1). According to the YEARS protocol, a chest CT angiography was performed on the patient and pulmonary embolism was ruled out. Nonetheless, the study revealed an irregular nodular thickening of the visceral and mediastinal pleura with late contrast enhancement. Lung parenchyma showed several nodular hyperdense images as well. Diagnostic thoracentesis was consistent with a lymphocytic exudate: glucose 97 mg/dL (reference: <60 mg/dL), lactate dehydrogenase 335 U/L (reference: <133 U/L), total protein 3.8 g/dL (reference: <3.0 g/dL), 30 leukocytes/mm3 (75% lymphocytes and 25% neutrophils), 27200 red blood cells/mm3 (reference <5000 cells/mm3), and a negative culture. Adenosine deaminase and cell block studies were negative as well. The patient underwent parietal pleurectomy and chemical pleurodesis with pleural biopsy. The histopathological results showed atypical epithelial cells with the presence of signet-ring cells. Immunohistochemistry was consistent with primary lung adenocarcinoma: CK-7 (+), TTF-1(+), Napsin A (+), CK-20 (−), calretinin (−), WT-1 (−) and D240 (−). The complete pathology report and immunohistochemistry are available upon request. Endoscopic studies were performed to rule out alternative tumoral origins, with no evidence of malignancy-related findings and a normal gastric distensibility. Contrarily, a CT scan of the abdomen showed subdiaphragmatic peritoneal carcinomatosis and pericardial and phrenic lymphadenopathies. Notably, the walls of the stomach were regular, had normal thickness (<8 mm) and there were no signs of linitis plastica that could indicate a gastric origin of the tumor (Figure 2). Computed tomography of the chest (left) on axial view and of the abdomen (right) on coronal view showing irregular nodular thickening of the pleura, pleural effusion and peritoneal carcinomatosis.The gastric wall had normal thickness with no evidence of tumoral compromise on imaging.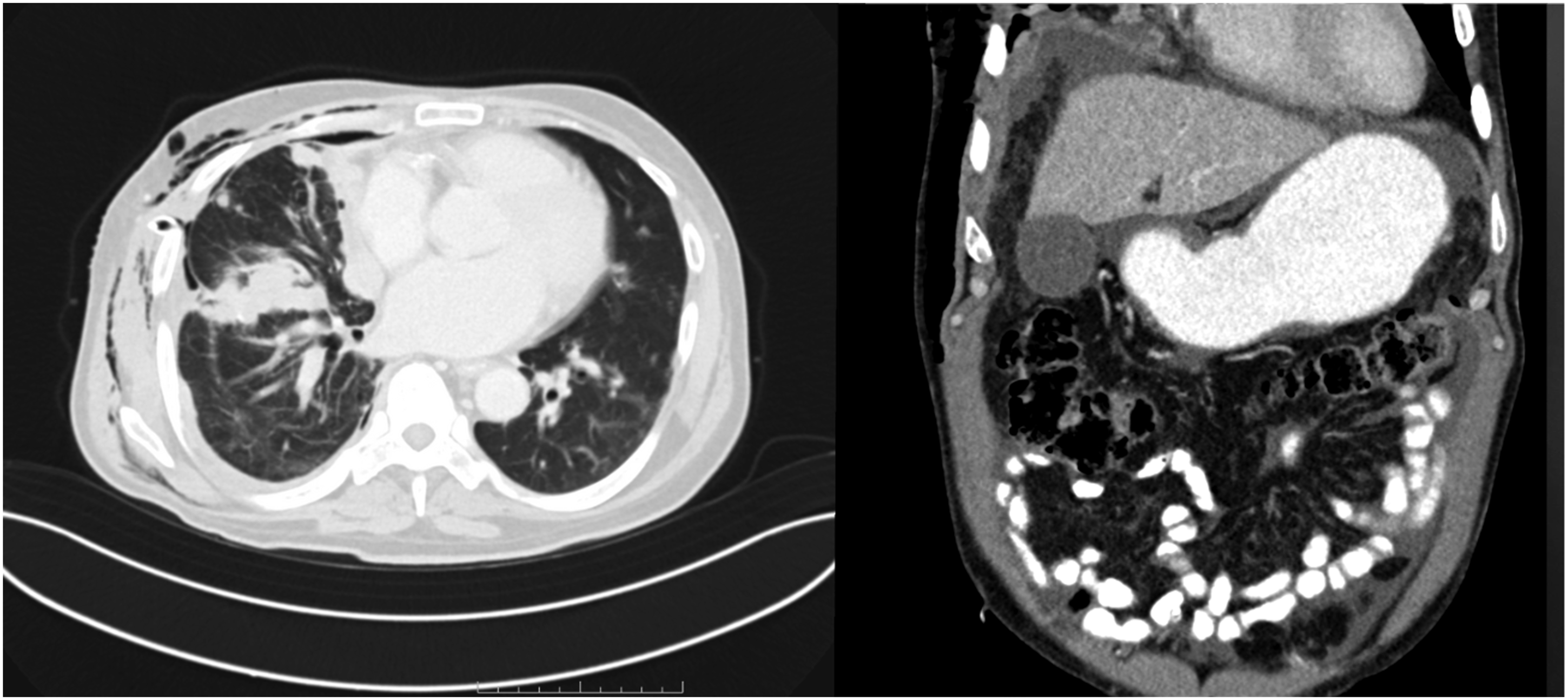
Metastatic primary lung SRCC with pleuro-peritoneal compromise was confirmed. Furthermore, the lymphatic invasion, the pleural effusion, the nodular appearance of the pleura, and radiologic pulmonary abnormalities corroborated the diagnosis of lymphangitic carcinomatosis. Finally, the patient was discharged for further ambulatory follow-up and palliative treatment oriented to comfort-seeking interventions exclusively. Posteroanterior and lateral views of chest x-rays at admission (above) and discharge (below).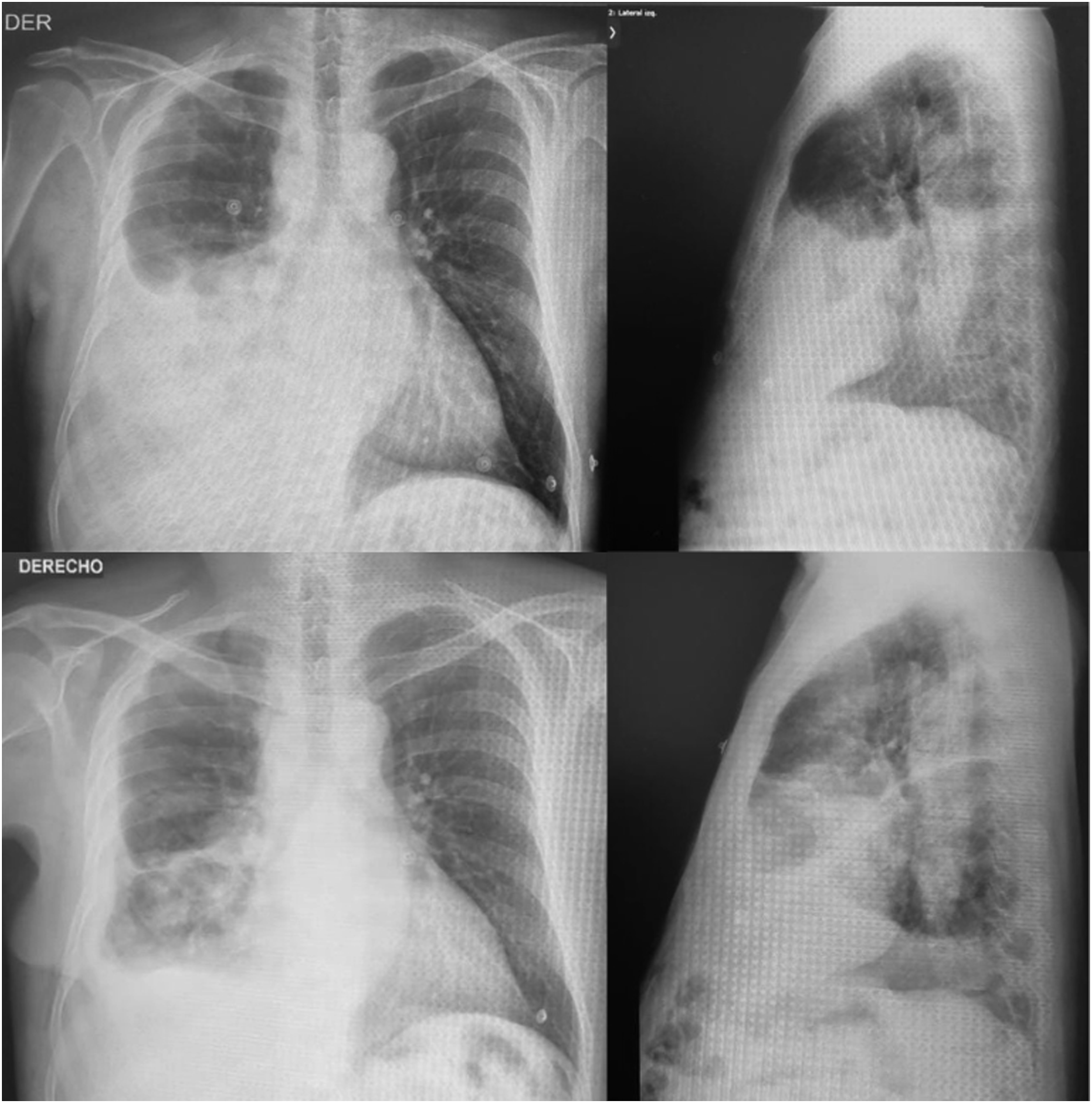
The patient authorized the publication of this case and related images through a signed informed consent available upon request.
Discussion
Primary SRCC of the lung accounts for less than 0.5% of lung adenocarcinomas and carries a poor short-term prognosis. 3 About half of primary lung SRCCs are diagnosed in stage IV, have a lower histopathological degree of differentiation, and tend to affect younger patients. 6 Independent factors that have been associated with a worse prognosis include advanced age, a T4 stage, and the presence of distant metastases, which are frequent at the time of diagnosis. The pleural, peribronchial, and perivascular lymphatic infiltration by tumoral cells has been described in gastrointestinal SRCCs and is a contributing factor to distant disease development. 8 In these cases, the edema generated by neoplastic proliferation and tumoral secretions can cause a desmoplastic reaction and thus facilitate thickening of the interstitial pleura, which acquires a nodular appearance. This phenomenon is known as lymphangitic carcinomatosis and was firstly described by Troiser in 1873. Approximately 6–8% of all intrathoracic metastatic processes can induce LC, and adenocarcinomas stand for 80% of the causative tumors. 9 Breast, stomach, pancreas, lung, and prostate adenocarcinomas are among the most common origins, even though any metastatic compromise could generate this complication. The presence of signet-ring cells in carcinomas causing LC has been documented in several case reports, all of which were secondary to SRCCs of gastrointestinal origin.9–12 To our knowledge, there is no report of LC secondary to primary lung SRCCs to this date. Nonetheless, other reported cases of primary lung SRCCs share similar characteristics, and thus sub-diagnosis of LC among reports remains feasible.13,14 The relationship between the presence of signet-ring cells and the development of LC may exist, but studies with larger cohorts are needed to prove this affirmation.
As most SRCCs arise from the gastrointestinal tract, gastric linitis plastica should be excluded. These tumors infiltrate the submucosa and muscularis propria, and there may be no superficial findings in the endoscopic examination besides a poor distensibility of the stomach. CT scan images should rule out the presence of thickened stomach walls that could point to linitis plastica as the true origin of SRCC, and secondary lymphadenopathies or peritoneal carcinomatosis. 15 In our report, gastric flexibility was adequate, and the stomach walls had normal thickness on imaging, lowering the possibility of a gastric origin of the tumor.
As in our case, the reported symptoms of LC are nonspecific and correspond mainly to progressive dyspnea, pleuritic pain, and cough. 16 Radiographic changes tend to occur predominantly on the right side and include pleural effusions, diffuse nodular opacities, and hilar lymphadenopathy. However, there are no pathognomonic radiological findings, and the X-ray can be normal in up to 50% of cases. 17 The diagnostic method of choice is the chest CT in which irregular thickening of the pleura, subpleural nodules, ground-glass opacities, pleural effusions, and perihilar and mediastinal lymphadenopathies can be evidenced. There are no curative treatment options for this disease, although corticosteroids have historically been used as symptomatic relief. The mortality attributed to LC remains very high, with an average survival of only 2 months. 18 The evidence suggests that a signet-cell component in the biopsy is an independent marker of poor prognosis in patients with gastric adenocarcinoma regardless of conflicting results.19–21 To this date there are no prognostic studies including patients with primary lung SRCC and secondary LC.
Conclusions
The SRCCs are aggressive tumors with a poor prognosis. The evidence for this type of adenocarcinoma is still scarce, particularly in SRCCs of extra-gastrointestinal origin. The relationship with the development of LC may occur. Therefore, this entity should be considered among the differential diagnoses in patients with progressive dyspnea, without previous lung disease, and with imaging findings of nodular and irregular thickening of the pleura.
Footnotes
Authors’ contributions
DCO and LPV are equally responsible for conception, drafting and design of the case study. Both authors are responsible for the literature search and intellectual content. All authors read and approved the final manuscript.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Data availability statement
The authors indicate that data regarding this publications is available and shared.
