Abstract
Objective
This study aimed to systematically assess the effect of the metastasis associated lung adenocarcinoma transcript 1 (MALAT1) long noncoding RNA rs619586 polymorphism on cancer risk.
Methods
We conducted a literature search of the PubMed, Embase, and China National Knowledge Internet databases to identify relevant studies, and calculated the pooled odds ratios (ORs) and 95% confidence intervals (CIs) for the retrieved studies using RevMan software.
Results
Nine eligible studies including 5968 cases and 7439 controls were included in the meta-analysis. The pooled results showed that MALAT1 rs619586 polymorphism was significantly associated with cancer risk [(AG + GG) vs. AA: OR = 0.88; GG vs. (AG + AA): OR = 0.64; GG vs. AA: OR = 0.63; AG vs. AA: OR = 0.91; G vs. A: OR = 0.87]. However, subgroup analyses based on ethnicity and cancer type showed significant associations between MALAT1 rs619586 polymorphism and cancer risk in Asians and for cancers other than hepatocellular carcinoma, but not for Caucasians and hepatocellular carcinoma.
Conclusions
MALAT1 rs619586 polymorphism may play a role in cancer risk. However, further studies are needed to confirm these findings.
Keywords
Introduction
Single nucleotide polymorphisms are genetic variations that have been associated with the risk of complex human diseases, including cancer.1–3 The association between the rs619586 A>G polymorphism in metastasis associated lung adenocarcinoma transcript 1 (MALAT1) long noncoding RNA (lncRNA) and cancer risk has been widely reported. Liu et al. 4 found that rs619586 AG + GG genotypes were associated with a decreased risk of hepatocellular carcinoma (HCC), with borderline significance, while Peng et al. 5 found that individuals with the rs619586 AG genotype had a reduced risk of breast cancer. Zhao et al. 6 also showed that the rs619586 G allele was significantly associated with a decreased risk of colorectal cancer (CRC), and individuals with AG and GG genotypes had a lower risk of CRC. Qu et al. 7 found that the rs619586 GG genotype was associated with a lower risk of esophageal squamous cell carcinoma (ESCC) among individuals an ‘ever drinking’ subgroup, Wen et al. 8 showed that the rs619586 G allele could serve as a protective factor reducing susceptibility to papillary thyroid cancer (PTC), and in a study by Yuan et al., 9 younger individuals (<55 years) with the rs619586 G allele had a decreased risk of HCC. However, other studies have found no association between the MALAT1 rs619586 polymorphism and cancer risk.10–12
We therefore conducted a systematic review and meta-analysis of published studies to clarify the relationship between the MALAT1 rs619586 polymorphism and cancer risk, and to shed some light on biomarkers predicting cancer risk. 13
Materials and methods
Literature search
Clinical questions were proposed and translated for easy retrieval according to the patient, intervention, comparison, outcome (PICO) strategy. We performed a literature search of the PubMed, Embase, and China National Knowledge Internet databases up to 5 March 2020, using the following search terms: “MALAT1” and (“polymorphism” or “variation” or “SNP”) and (“cancer” or “carcinoma” or “tumor”). We also retrieved relevant references from the identified studies. The search was limited to studies published in English or Chinese.
The study was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
Inclusion criteria
Eligible studies were required to meet the following criteria: (a) case-control study assessing the association between rs619586 polymorphism and cancer risk; (b) available genotype and allele data for estimating odds ratios (ORs) and 95% confidence interval (95% CIs); and (c) genotype frequencies in the control group compliant with Hardy–Weinberg equilibrium (HWE).
Data extraction
Two authors independently selected eligible studies and extracted the following data: first author’s name, publication year, ethnicity, genotyping methods, cancer type, numbers of cases and controls, genotype and allele frequency, and HWE evidence. Any disagreement was resolved by discussion with a third author.
Statistical analysis
HWE was examined by goodness-of-fit χ2 tests and PHWE > 0.05 was considered to indicate compliance with the HWE. The association between the rs619586 polymorphism and cancer risk was evaluated by allelic (G vs. A), codominant (AG vs. AA, GG vs. AA), recessive [GG vs. (AG + AA)], and dominant [(AG + GG) vs. AA] models. The significance of pooled ORs was analyzed by Z tests, with PZ < 0.05 considered significant. Heterogeneity among studies was assessed by χ2 tests and I2 index, with PH < 0.05 or I2 > 50% indicating the existence of heterogeneity. In the event of heterogeneity, pooled ORs were calculated using a random-effect model; otherwise, a fixed-effect model was applied. Sensitivity analysis was performed by sequentially omitting each study to assess the stability of the results. Publication bias was determined based on Begg’s and Egger’s tests, and P < 0.05 was considered significant. Trial sequential analysis (TSA) was performed to quantify the statistical reliability of the meta-analysis. The required information size was calculated using a control event proportion of 20%, relative risk reduction of 8%, power of 80%, and type I error of 5%. All statistical tests were performed using RevMan 5.0 (Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark), Stata 12.0 (Stata Corporation, College Station, TX, USA), and TSA 0.9.5.10 (Copenhagen Clinical Trial Center, Denmark) software.
Results
A total of 428 studies were initially retrieved (Figure 1), of which 419 were excluded after reading the title, abstract and full text. Nine studies that included 5968 cases and 7439 controls were finally included in the study (Table 1), including eight studies conducted in Asians and one in Caucasians. Four studies focused on HCC, one on breast cancer, one on CRC, one on ESCC, one on PTC, and one on melanoma.

Flowchart of study selection procedure. HWE, Hardy–Weinberg equilibrium.
Main characteristics of studies included in the meta-analysis.

HCC, hepatocellular carcinoma; CRS-RFLP, created restriction site polymerase chain reaction-restriction fragment length polymorphism; CRC, colorectal cancer; ESCC, esophageal squamous cell carcinoma; PTC, papillary thyroid cancer.
The overall pooled analysis showed a significant association between MALAT1 rs619586 polymorphism and cancer risk (Table 2). Subgroup analysis based on ethnicity showed a significant association in Asians but not in Caucasians, and subgroup analysis based on cancer type showed that the MALAT1 rs619586 polymorphism was associated with the risk of cancers other than HCC, but not with the risk of HCC.
Association between MALAT1 rs619586 polymorphism and cancer risk.

HCC, hepatocellular carcinoma; OC, cancers other than hepatocellular carcinoma; EM, effect model.
Sensitivity analysis showed that omission of any single study did not significantly alter the overall pooled ORs under the (AG + GG) versus AA, GG versus (AG + AA), GG versus AA, and G versus A comparison models, but omission of some studies did significantly alter the overall pooled OR for AG versus AA, suggesting that these results need to be interpreted with caution (Figure 2). Begg’s and Egger’s tests found no evidence of publication bias (Table 3). TSA showed that the cumulative Z-curve did not cross the trial sequential monitoring boundary or reach the required information size, suggesting that the results of the meta-analysis remained inconclusive (Figure 3).

Sensitivity analyses in the current meta-analysis. CI, confidence interval.
Publication bias in the meta-analysis.

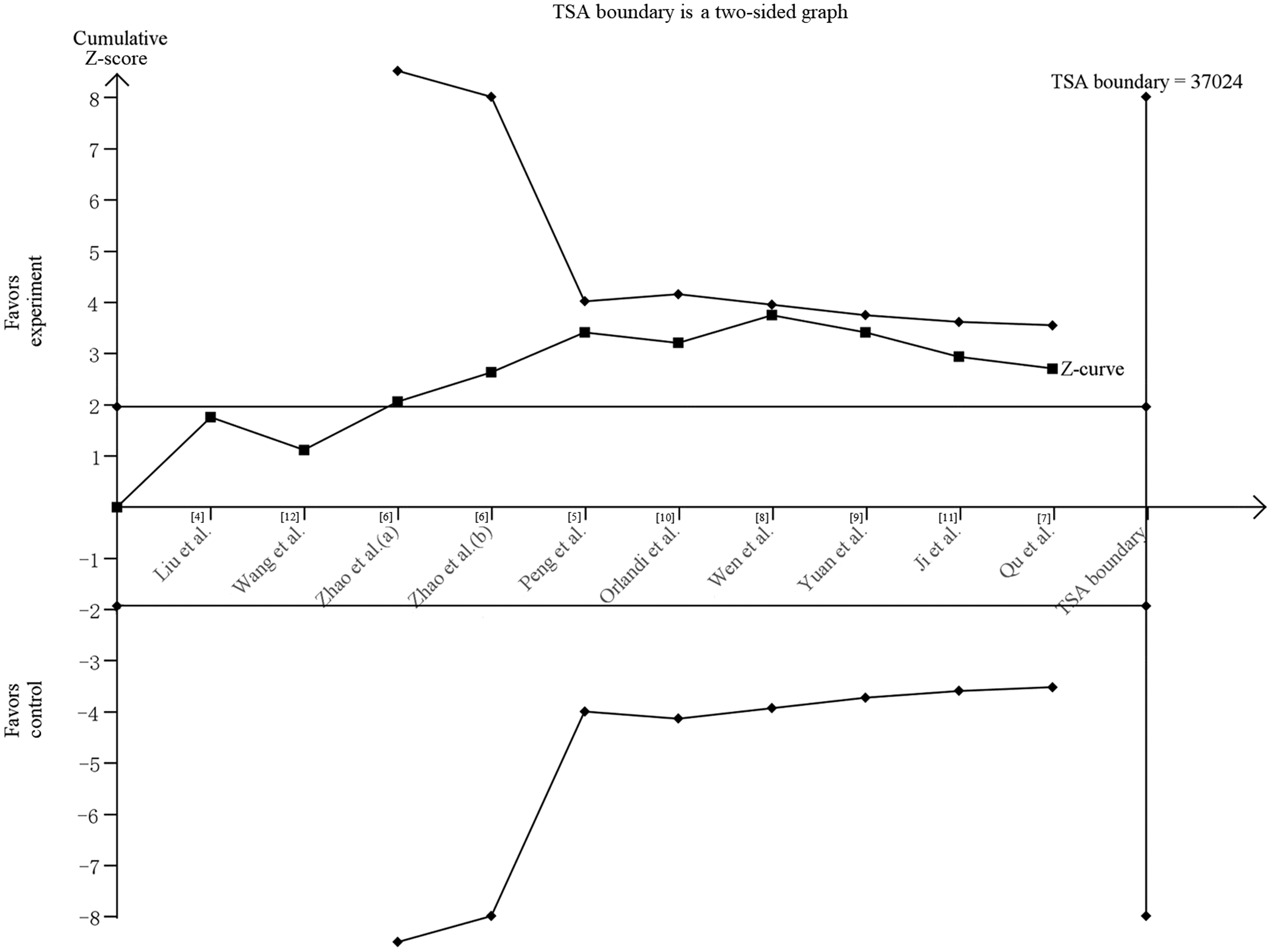
Trial sequential analysis of association between MALAT1 rs619586 polymorphism and overall cancer risk for (AG+GG) versus AA. Cumulative Z-curve constructed using a fixed effect model. The required information size was calculated using a control event proportion of 20%, relative risk reduction of 8%, power of 80%, and type I error of 5%. TSA, trial sequential analysis.
Discussion
LncRNAs are transcripts longer than 200 nucleotides that lack protein-coding ability, but which are involved in cancer initiation and progression. 14 MALAT1 is an 8.5 kb lncRNA located on chromosome 11q13.1, which could serve as an oncogene and to modulate cancer-related signaling pathways.15,16 Dong et al. 15 found that MALAT1 promoted the proliferation and metastasis of osteosarcoma cells by activating the phosphoinositide 3-kinase/Akt pathway, while osteosarcoma patients with high expression of MALAT1 were more likely to develop pulmonary metastasis and to have poor nodal status. Guo et al. 16 showed that down-regulation of MALAT1 inhibited the proliferation, invasion, and migration of ovarian cancer cells, arrested cell cycle progression in S phase, and induced cell apoptosis by inhibiting activation of the Wnt/β-catenin signaling pathway. The MALAT1 rs619586 polymorphism has attracted recent attention. Plasma expression levels of MALAT1 rs619586 AG, GG, and AG + GG genotypes were significantly lower than that of the AA genotype in cancer-free controls, indicating that the rs619586 polymorphism could affect MALAT1 expression. 5 Furthermore, the rs619586 G allele was associated with significantly decreased MALAT1 expression, reduced PTC proliferation, and increased PTC apoptosis. 8 The association between MALAT1 rs619586 polymorphism and cancer risk has also been widely investigated; however, the results have been inconsistent and there is thus a need to clarify this relationship. A meta-analysis, as a powerful tool for analyzing cumulative data, was applied to explore the effect of the MALAT1 rs619586 polymorphism on cancer risk, but it only included two studies. 17 However, several new studies have since been published and should thus be included in the pooled analysis. The current pooled analysis including these new studies identified a significant association between the MALAT1 rs619586 polymorphism and cancer risk, especially in Asians and cancers other than HCC.
This meta-analysis had some limitations. First, the limited number of relevant studies might have led to an overestimate of the effect of the rs619586 polymorphism on cancer risk. Second, the study was conducted under an unadjusted assessment, and a more precise analysis should be carried adjusting for individual data, such as lifestyle and environmental factors, if these become available. Third, the study was not registered with the PROSPERO database, which should be done in future studies.
In conclusion, the meta-analysis indicated that the MALAT1 rs619586 polymorphism was associated with cancer risk. However, further well-designed studies should be performed to confirm these findings.
