Abstract
Introduction
Granulosa cell tumor of the ovary is a rare subtype of ovarian cancer originating from the sex-cord stromal component of the ovary. GCTs present with two clinically and molecularly distinct subtypes: the juvenile and the adult type. 1 GCT is a clinically distinct ovarian cancer due to its prominent hormonal activity and production of estrogen and inhibins. 1 GCTs have a remarkably indolent behavior compared to other ovarian malignancies, however, they are characterized by an unpredictable disease course with reported recurrence rates between 6% to 50%.2–6 The outcomes of patients with GCTs were good, with nearly 95% of patients surviving 5 and 10 years. 7 Because the majority of patients are diagnosed with stage I disease, the major clinical challenge in the management of GCT remains the identification of prognostic factors and/or tumor markers that are able to predict disease recurrence. 1 GCT requires the active follow-up for 10–15 years after primary diagnosis, since recurrences may develop late, asymptomatically and in multiple anatomical locations. 8 Furthermore, when the tumor recurs, 80% of patients will succumb to their disease.9,10 The prognosis was related to initial stage, presence of residual tumor after initial surgery, and tumor size. Different surgical methods and/or adjuvant therapy appear not to affect the outcome. 7 Here, the current study was investigated to access the clinical and paraclinical features, treatment findings, survival outcomes, and explored the prognostic factors in the granulosa cell tumor of Vietnamese women.
Methods
Patients and sample selection
This is a retrospective analysis of histopathologically diagnosed cases of ovarian granulosa cell tumors, conducted at the National Cancer Hospital, Vietnam from 2016 to 2020. Only patients with treatment-naïve tumors were selected. Patients who presented with second malignant tumors orrecurrent disease were excluded. During the study period, we identified 28 patients with ovarian GCT who met the inclusion criteria, including both ovarian adult and juvenile granulosa cell tumors. All patients were reviewed to extract information about demography, clinicopathology, tumor marker as CA12.5, treatment, and survival.
Ethical clearance for the conduction of the study was obtained from the institutional ethic committee. All private information was deleted or disguised in order to ensure patient anonymity.
Imaging features
All 28 patients underwent ultrasonography (US). Ultrasound findings were described in this study including tumor size, structure of mass, and echogenicity. The maximum diameter of tumors was calculated. Computed tomography (CT) or magnetic resonance imaging (MRI) was performed in 26/28 cases. Four malignant features included solid or papillary structure, multilocular tumors, ascites, and abdominal lymph nodes extracted from medical records when combining three above radiologic methods.
Histopathology
All specimens were received in the operating room and then transferred to the pathology department. Samples were fixed in 10% neutral formalin for 24 hours. Tumor samples were obtained by routine pathological techniques, such as hematoxylin and eosin staining. Experienced pathologists evaluated all histopathological features, such as tumor size and histopathological type. Histopathological types were classified according to 2020 WHO classification. 11
Variables of interest
Variables used for analyzing include age (mean, < 50, and ≥ 50 years), parity (≤ 2 and >2), clinical presentation, radiological features, serum CA-125 levels (mean, < 35 and ≥ 35 IU/mL), tumor size (mean, <10 and ≥10 cm), histopathological types (adult and juvenile), FIGO (International Federation of Gynecology and Obstetrics) stage (stage I-II and stage III), residual lesion during surgery (present or absent), adjuvant chemotherapy (yes or no), recurrence (yes or no), and follow-up. Variables were categorized for comparison based on ovarian GCTs features and previously published data. Stage was defined according to the criteria of the International Federation of Gynecology and Obstetrics (FIGO, 2013). Residual lesion was evaluated on macroscopy by surgeons and was extracted from surgery protocol. Survival of all 28 patients were contacted via phone or messages to collect information.
Treatment, outcomes, and follow-up
Surgery was performed initially for all patients. Fertility sparing surgery was defined as unilateral salpingo-oophorectomy. Radical surgery consisted of total abdominal hysterectomy with bilateral salpingo-oophorectomy. Residual lesion was extracted from surgery protocol reviewed by surgeons during the operation. Postoperative chemotherapy was indicated for all patients with stage IC-IV. The chemotherapy regimens included BEP (bleomycin, etoposide, and cisplatin), EP (etoposide and cisplatin), or a combination of paclitaxel and carboplatin. One received EP and two received paclitaxel/carboplatin due to older age or pre-existing pulmonary disease.
Our 28 patients were observed to evaluate recurrence or death up to 6 years. Overall survival (OS) was defined as the time from the date of diagnosis to the date of death due to ovarian cancer or the last available date before loss of follow-up. Death dates of the patients were acquired from death certificates issued by the commune government in Vietnam. Radiological and/or histopathological data were used to determine the recurrence. Disease-free survival (DFS) was calculated as the interval from diagnosis until evidence of disease recurrence or the date of the final follow-up.
Statistical analysis
Statistical analysis was performed using SPSS ver. 20.0. Pearson’s Chi-squared or Fisher’s exact test was used to compare the differences in proportions. The Mann-Whitney test was used to analyze the continuous variables. Survival curves and rates were calculated using the Kaplan-Meier method and the Log-Rank test was used to compare survival between groups. Univariate and multivariate analyses were performed using Cox’s regression model. A two-sided p-value < 0.05 was considered to be significant.
Results
Clinical and paraclinical features of ovarian GCT
Correlation of recurrence to clinical and paraclinical characteristics.
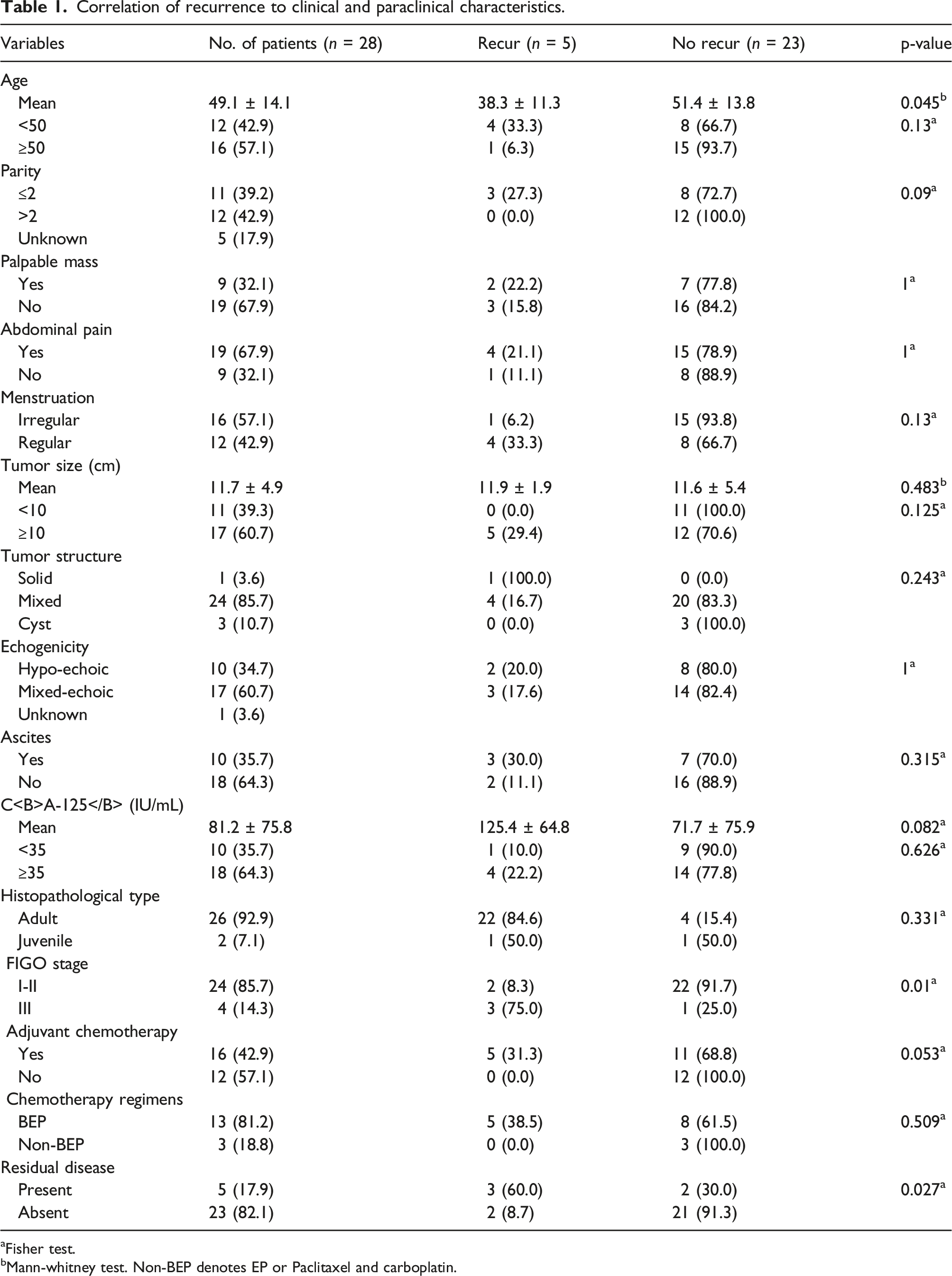
aFisher test.
bMann-whitney test. Non-BEP denotes EP or Paclitaxel and carboplatin.
Association between initial presentation and disease stage.

a: Fisher test; b: Mann-whitney test.
Treatment, outcome and follow up
Treatment details.

BEP denotes Bleomycin, etoposide, cisplatin; EP, Etoposide, cisplatin.
Disease free survival and overall survival based on clinical characteristics.

Non-BEP denotes EP or Paclitaxel and carboplatin.

OS and DFS of GCT based on age. The Log-rank test indicates that there was a significant difference between the two DFS survival curves according to two age groups (b). The Log-rank test displayed that there was not a significant difference between these five-year OS curves of two age groups (a).

OS and DFS of GCT based on menstruation status. The Log-rank test displays that there was a significant difference between the two survival curves of DFS according to regular or irregular menstruation (b). However, Log-rank test displayed that there was not a significant difference yet between these five-year OS curves of menstruation status (a).

OS and DFS of GCT based on stage I-II versus stage III. The Log-rank test demonstrates that there was a strongly significant difference between the two survival curves according to the GCT stages, both OS (a) and DFS (b).
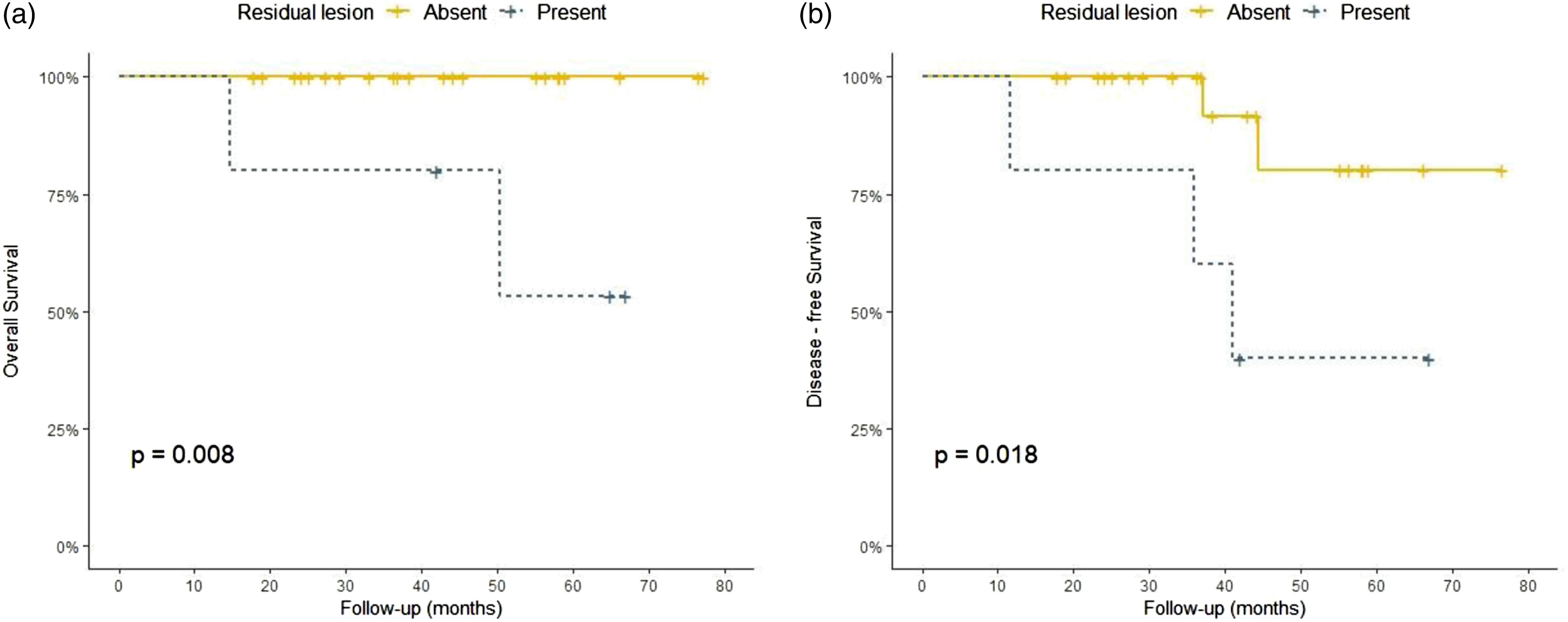
OS and DFS of GCT based on the residual lesion status. The Log-rank test demonstrates that there was a strongly significant difference between the these survival curves according to the residual tumor status for OS (a) and DFS (b), as well.

Univariate and Multivate Cox regression models of GCTs to assess the association between variables and DFS.
Discussion
GCTs can occur over a wide age range, but most GCTs are usually diagnosed in perimenopausal women with a median age of diagnosis between 50 and 54 years, although it can occur throughout the adult woman’s lifespan. 12 The mean age at diagnosis (52.9 years) and the mean tumor size (10.8 cm) did not change significantly over time. 13 Previous data showed the majority of patients present with one or a combination of the following clinical symptoms: abdominal pain, distention, mass, and menstrual disturbances.14–17 Abdominal distension and pain might be related to the fact that GCTs tend to be large and may rupture spontaneously.18–20 The most common presenting symptoms are abnormal vaginal bleeding (45%), but 14% were asymptomatic , 13 and abdominal pain or bloating (10–20%). In premenopausal patients, adult GCT typically causes irregular bleeding, amenorrhea, and more infrequently infertility. 13 In postmenopausal patients, abnormal vaginal bleeding associated with a unilateral ovarian mass is the most common clinical presentation. 12 Due to the slow growth and distinguishable hormonal symptoms, GCTs are generally diagnosed at an early stage. The majority (50–80%) are diagnosed at stage IA.7,13,21,22 In Bryk et al.‘s study, the stage I disease was present in 89.2%; stage II, in 7.0%; stage III, in 3.8%; and no one is staged in IV. 13 In Sun et al.‘s research, the stage distribution at diagnosis was stage I in 77.8% of patients, stage II in 5.1%, stages III-V in 6.1%, and unknown in 11% of patients. 7 Ovarian GCTs are divided into adult and juvenile types based on different clinical and histopathologic features.11,23 According to literature, adult type accounts for more than 90% of the GCTs and this figure in our study was 92.9%. The most present findings were similar to the above studies.
Regarding to the treatment of GCTs, surgery remains the cornerstone of treatment for patients with ovarian GCTs.14,17,24 Surgery was initially performed on all patients in our study. Of them, there were two patients given a fertility-sparing procedure (unilateral salpingo-oophorectomy). For postmenopausal women, a total abdominal hysterectomy and bilateral salpingo-oophorectomy are typically performed. Because, ovarian GCTs tend to present at a younger age with early stage disease, therefore making conservative operation an important issue. According to current guidelines, fertility-preserving approach is also reasonable in younger patients at stage I GCT who have a desire to preserve fertility.15,24 Postoperative chemotherapy was given for 16 cases in this study including all patients at advanced stage or stage IC. Several studies have shown that adjuvant chemotherapy is not associated with improved survival of GCT patients.25–27 Oseledchyk et al. found that regardless of stage, there were no significant differences in 5-year DFS between patients who received chemotherapy and those who did not. 26 But in fact, patients who receive adjuvant chemotherapy often have the greatest number of risk factors for recurrence. Current guidelines recommend adjuvant chemotherapy for advanced stage disease. For patients with high-risk stage I tumors including tumor rupture, stage IC, poorly differentiated tumor, and tumor size of more 10–15 cm, postoperative recommendations include observation or consideration of platinum-based chemotherapy. 24 Chemotherapy regimens for GCTs generally include bleomycin, etoposide, and cisplatin (BEP) and etoposide and cisplatin (EP) or carboplatin with paclitaxel. BEP was the most used regimen in this study (81.4%). Other alternative options included EP and paclitaxel/carboplatin, given for those who were at an older age or had pre-existing pulmonary disease. The Gynecologic Oncology Group is conducting a randomized phase II trial of BEP versus the combination of paclitaxel and carboplatin for patients with newly diagnosed and recurrent chemo-naïve sex cord-stromal tumors of the ovary (GOG 264). Hormonal therapy is a promising therapeutic approach in the management of granulosa cell tumors because these tumors generally express steroid hormone receptors. 28 This therapy was added in NCCN guideline as a treatment option in recurrent disease. 24
GCT is considered as a malignant tumor with an in dolent course and a tendency toward late recurrence.17,23,28 However, every third patient will eventually develop a tumor relapse, and this will lead to death in 50–80% of the relapsed patients,5,13,29 warranting life-long surveillance, especially in the high-risk patients. In Khosla et al.‘s study, the 5-year OS and event-free survival were 84.6% and 76.5% after 71.4 months of follow-up. 15 Mangili et al. reported that 5-year and 10-year OS were 97% and 95%, and 5-year and 10-year DFS were 91.5% and 71.6% accordingly. 21 Similarly, McConechy et al. demonstrated five-, ten-, and 15-year OS were 93.3, 84.4, and 72.1%, respectively. 5 A unique feature of GCTs is that they recur at extended time intervals from primary therapy, the median time to recurrence is 6 years.30,31 In Sun et al.‘s research, the recurrence rate was 21% with the median follow-up period of 60.7 months. 7 In the other study, Bryk et al. showed the median time to the first relapse was 7.4 years, and 75% of relapses occurred within ten years after primary diagnosis. 13 Wilson et al. demonstrated that 32.0% of patients were relapsed with a median time to relapse (TTR) of 12.0 years. Higher relapse rates (43% vs. 24% p = 0.02) and shorter TTR (10.2 vs. 16.2 years, p = 0.007) were seen with stage IC versus stage IA disease. 22 After the medium follow-up of 43.17 months (14.73 to 77.36 months), we detected five patients with relapsed disease and two of them died due to ovarian cancer. The mean time from diagnosis to recurrence was 40.2 months. In concordance with previous studies, our series showed a favorable prognosis with the 5-year OS and DFS were 87.7% and 69.7% respectively. Survival rates in our report were slightly lower since there were more advanced stage cases than that in previous studies. In addition, shorter follow-up time and small numbers of patients might be significant limitations in this study. Premenopausal status at initial diagnosis, FIGO stage IC versus IA, and tumor rupture associated with relapse. 8 Tumor rupture (stage IC) has a significantly increased risk of disease relapse.8,13,22 In the current study, factors such as mean young age, advanced FIGO stage, and residual lesion during surgery were the significant differences in recurrent rate. Prior use of oral contraceptives and history of infertility improved survival rates. 13
Considering to the prognostic factors, generally, GCTs have a remarkably indolent behavior compared to other ovarian malignancies. Multiple clinical and histological prognostic factors have been investigated for their role in GCT prognosis. Disease stage, tumor size, tumor rupture, age at diagnosis, nuclear atypia, mitotic index, and presence or absence of residual lesion after initial surgery had been shown to be the prognostic significance in various studies.7,16–19,32,33 However, the results have been inconclusive and varied significantly between published reports. Stage of the disease is the most widely accepted GCT prognostic factor that has been linked to tumor relapse and survival.7,13,23,28,34 Five-year survival is reported to be 80–95% for patients with early stage disease and survival declined to 25–40% in patients with advanced stage disease. 7 In this study, the 5-year DFS and the 5-year OS of early stage (stage I or II) was significantly higher than that rate for advanced stage (stage III) (80.8% and 25.0%; 100% vs 50% respectively, p < 0.05). None of patients with stage I had recurrent disease during the period following. In respect to residual disease, all cases with stage II or III in our study underwent cytoreduction. During primary operation, five patients had residual lesion. Three of them had recurrent disease during follow-up. Our findings showed a significant difference in the 5-year DFS and OS between patients with and without residual tumors (p < 0.05). Presence of residual disease was demonstrated to be a prognostic factor for inferior survival in several series32,35,36 which was in concordant with our finding (HR: 6.7, 95%CI, 1.1–41.0, p = 0.04). Residual disease was associated with poor survival and outcome in various studies. Ayhan et al. showed that there was a decrease in 5-year survival rate from 90% to 25% in those with or without the gross residual lesion after surgery, respectively. 35 Chan et al. also indicated that absence of residual disease was an independent prognostic factor to the improved survival. 32 It is hard to clearly confirm no residual tumor after operation, careful examination during surgery and image examination such as computer tomography can help. 7 There has been some controversy about the effect of age on survival. Some studies indicated that younger age at diagnosis was a prognostic factor to improved survival.32,37,38 In contrast to previous studies, patients with less 50 years old were significantly associated with inferior DFS in our study (47.1% vs 83.3%, p = 0.039). The OS rate in the age group of less than 50 was lower, but not statistically significant than that proportion in the older group (p = 0.112). Our finding on age at diagnosis was similar to Lee et al’s and Ayhan et al’s cohorts.17,35 There were some reasons that might be contributing factors to the worse outcome of the younger group. Firstly, mean age of advanced stage group was significantly lower than that of early stage group (36.0 vs 51.3%, respectively, p = 0.041). In addition, all four cases of stage III were at younger group (<50 years). However, age at diagnosis was not a significant prognostic variable in multivariate analysis after accounting for stage of disease. Additionally, regarding the menstruation, in this study, survival of patients with irregular menstruation was significantly better than those with normal menstruation (p = 0.033). We also found that patients with stage I presented abnormal menstruation more than those with stage II-III which was statistically significant (50.0% vs 7.1%, respectively, p = 0.017). Menstrual problems included abnormal vaginal bleeding, oligomenorrhea, or secondary amenorrhea accounted for 60.7% in our study. These clinical symptoms were signs of hyperestrogenism due to hormone production secreted by theca cells in granulosa cell tumors. Approximately 70% of patients with GCT have elevated circulating levels of estradiol. 39 In many series, menstrual symptoms are the most common reasons for seeking medical assistance. Considering to the tumor size, GCTs with more 10–15 cm are associated with inferior survival in some series.23,32,33,38 Chan et al. showed that the enlarged tumor with ≥ 10 cm was a prognostic factor to decrease the survival in both univariate and multivariate analysis. 32 We were not able to validate the prognostic significance of tumor size in ovarian GCTs (with a cutoff of 10 cm). Nevertheless, all five cases of recurrence showed that tumors were larger than 10 cm, in our study. The enlarged tumors often have a long indolent growth phase and already existed for a long time before discovery and diagnosis. In addition, large size might associate with the risk of tumor rupture before and during operation.
Bryk et al indicated that the GCT-related deaths were associated with old era, patient age of more 60 years, tumor sizze greater than 10 cm, advanced stage, presence of residual tumor, and use of hormonal adjuvant therapy in the univariated analysis, however, in the multivariate analysis, higher stage was only the independent prognostic factor. 13 In another study, Sun et al.‘s showed that the presence of residual tumor after initial surgery and tumor size were significantly associated with recurrence, in the multivariate analysis. 7 Similarly, Auranen et al. also demonstrated that other parameters inconsistently reported and providing overall less convincing evidence as being important for prognosis include patient age, primary tumor size and rupture of tumor, mitotic activity, and nuclear atypia. 40 In the current findings, in univariate impact were factors such as stage and residual tumor. Nevertheless, these parameters were not demonstrated as an independent prognostic indicator in the multivariate analysis.
Limitations of the study
At present, some limitations still remained in the present study. It is a retrospective analysis that has inherent biases. This weak point, however, is common in studies on GCT due to the rarity and characteristics of these tumors. The rarity of GCTs makes it hard to conduct prospective and randomized clinical trials. Another drawback of this study is the small number of patients and the short time of observation. The indolent course of this tumor leads to the need for a long-term follow-up period. Our findings should be confirmed in more other cancer centers. Therefore, continued follow-up and new patient recruitment are planned to confirm the valuable prognostic factors of GCTs.
Conclusions
Old age, irregular menstruation, early stage, and absence of residual lesion were significantly the predictors for improving survival. In univariate analyses, FIGO stage and residual lesion during surgery had significant differences in recurrent rate.
Footnotes
Acknowledgements
The authors would like to thank the leading board of the National Cancer Hospital and National Cancer Institute, Vietnam, and unlucky patients, as well for their assistance and support with this study.
Author Contributions
Duc Thanh Le should be considered the major author. He participated directly in diagnosis, treatment, and follow up of the patients, patient recruitment, performed literature review, and assisted in drafting of the components of the article, and assisted in formatting the presented material
Tu Anh Do and Kien Hung Do took part in the diagnostic and treatment consultant and, assisted in literature review
Linh Ly Thi Nguyen performed the patient recruitment and follow up of the patient, review of patient’s chart, literature review, data analysis, assisted in drafting of the components of the article
Chu Van Nguyen performed the diagnostic consultant of the HE stains and, literature review, took illustrated figures, and assisted in analyzing data, drafting of the components of the article, and assisted in formatting the paper material.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s note
This manuscript has not been previously published on any journals.
Ethical approval
All objects of the protocol’s this study was approved by the Science and Ethical Committee of Hanoi Medical University, Vietnam as number: 6811/QD-DHYHN. The present study was performed in accordance with the Helsinki Declaration of 1964, and its later amendments.
Informed consent
Written informed consent was applied to all patients before enrolling them to the study. Patients could withdraw from the study at any time without any threats or disadvantages and for no stated reasons.
Patient involvement
All individual information was deleted or disguised, in order to make sure anonymity for the patient.
Data availability
All data analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
