Abstract
Gastric adenocarcinoma of the fundic gland is a rare, well-differentiated gastric cancer entity, and very few patients transition to poorly differentiated tubular adenocarcinoma during progression. Gastric adenocarcinoma of the fundic gland originates from the mucosa of the gastric fundic gland, usually without chronic gastritis or intestinal metaplasia. Histologically, the tumor cells are closely arranged to form anastomosing tubular glands, and more than 95% of tumor cells differentiate towards chief cells. Most gastric adenocarcinoma of the fundic gland cases are characterized by submucosal involvement, but the tumor volume is usually small, with lymphatic and vascular invasion rarely observed. Therefore, endoscopic submucosal dissection can be an ideal treatment, leading to a favorable prognosis, and recurrence and metastasis of the disease are uncommon.
Introduction
Morphologically, gastric cancer features a wide range of morphological heterogeneity, resulting in a variety of different histological subtypes. 1 According to the Lauren classification, gastric cancer is classified into intestinal and diffuse types 2 ; according to the Nakamura classification, gastric cancer is grouped into differentiated and undifferentiated types. 3 Based on molecular and genetic profiling, gastric cancer is divided into four genetic subtypes: EpsteinBarr virus (EBV)infected tumors, microsatellite instability (MSI) tumors, genomically stable tumors, and chromosomally unstable tumors. 4 With the development of immunohistochemistry (IHC), it has been confirmed that intestinal gastric cancer contains a gastric phenotype. The gastric phenotypes include foveolar type, pyloric gland type, and fundic gland type.
In 2007, gastric adenocarcinoma of the fundic gland (GA-FG) was first reported as a novel tumor entity by Tsukamoto et al. 5 According to tumor cell differentiation, GA-FG can be divided into three subtypes: chief cell-predominant type, parietal cell-predominant type, and mixed type. The GA-FG chief cell-predominant (GA-FG-CCP) type accounts for approximately 99% of all GA-FG cases. 6 With the increasing number of reported GA-FG cases, the purpose of this review is to summarize the clinicopathological characteristics, endoscopic appearance, pathogenesis, molecular phenotype, and prognosis of GA-FG to increase understanding of clinicians and pathologists regarding this type of disease.
Clinicopathological characteristics
Characteristics of reported gastric adenocarcinoma of the fundic gland.

––: Relevant data is not available; M: male; F: female; U: upper third of stomach; M: middle third of stomach; L: lower third of stomach; H. pylori: Helicobacter pylori; Type 0-I: protruded type; 0-IIa: superficial and elevated type; 0-IIb: superficial and flat type; 0-IIc: superficial and depressed type; T2: ulcerative; M: tumor confined to the mucosa; SM1: tumor confined to the submucosa, but depth is within 0.5 mm of the muscularis mucosae; SM2: tumor invasion is 0.5 mm or more deep into the muscularis mucosae; SS: tumor invades the subserosa; +: positive; -: negative; *: partial data is unknown.
Endoscopic imaging features
Endoscopically, most GA-FG lesions are located in the middle/upper third of the stomach. According to a previous review, the proportion of GA-FG lesions located in the middle/upper third of the stomach is up to 98% among the total GA-FG cases. 6 In our current analysis, the proportion was approximately 97.4%, which is similar to the previous results. The number of cases located in the upper, middle, and lower third of the stomach was 156, 34, and 3, respectively. Macroscopically, according to the available data, 3 tumor lesions showed a protruded type (T0-I); 1 tumor lesion presented an ulcerative type (T2); and the number of cases of superficial and elevated (0-IIa), superficial and flat (0-IIb), superficial and depressed (0-IIc), and mixed type was 94, 14, 39, and 20, respectively. Narrow band imaging can enhance visualization of mucosal vessels and show irregular patterns, which indicate heterogeneity of microvessels in GA-FG.7,14 The use of narrow band imaging and endoscopic ultrasound can improve the chance of complete resection through endoscopic mucosal resection (EMR) technology.
Histopathological features
The histopathological findings for GA-FG are shown in Figure 1. The tumor surface area is typically still covered with normal foveolar epithelium. Chronic inflammation, atrophic glands, and intestinal metaplasia are rarely observed in the surrounding mucosal background. Tumor lesions are composed of closely arranged and anastomosing glands, forming an “endless gland.” Tumor cells are similar to fundic gland cells, appearing as well-differentiated, mildly heterogeneous columnar cells with moderately enlarged nuclei. In approximately 99% of cases, tumor cells differentiate into chief cells.
6
In our study, according to the relevant data available, the number of GA-FG-CCP cases was 163, accounting for 96%. In addition, 2 cases appeared to exhibit the foveolar epithelium type, 1 case showed mixed type differentiation, and 3 cases differentiated towards the mucous neck cell type. GA-FG originates from the deep layer of the lamina propria. Although most tumor lesions show submucosal invasion, lymphatic and vascular invasion is very rare.7,8,15 According to our analysis, among 163 cases of GA-FG, 119 cases had submucosal infiltration (73%), and only two cases had subserosal infiltration. Although tumor submucosal involvement has been identified in most reports, thin wisps of muscularis mucosae with the absence of desmoplasia were identified throughout the lesions. Therefore, Singhi et al.16,17 suggested that this change represents prolapse-type growth to the submucosa rather than the more typical submucosal infiltration seen with malignant tumors. Some researchers believe that it is excessive and exaggerated to name this disease “GA-FG,” and this type of disease should be considered benign. They prefer to use “oxyntic gland polyps/adenomas” rather than “GA-FG.”
16
Although Yasuhiro Okumura reported a rare case of GA-FG that gradually transitioned into aggressive tubular adenocarcinoma with poorly differentiated components in the muscularis propria and subserosal layers, there were only two cases of subserosal infiltration in our analysis.13,18 Histopathological findings of representative GA-FG case. (A) In low-power view (×40), the tumor originated from the deep layer of the lamina propria and invaded submucosal layer. The surface was covered with non-atypical foveolar epithelium; (B) and (C): In high-power view (×100 and 200), the tumor comprised well-differentiated columnar cells mimicking the fundic gland cells with mild nuclear atypia.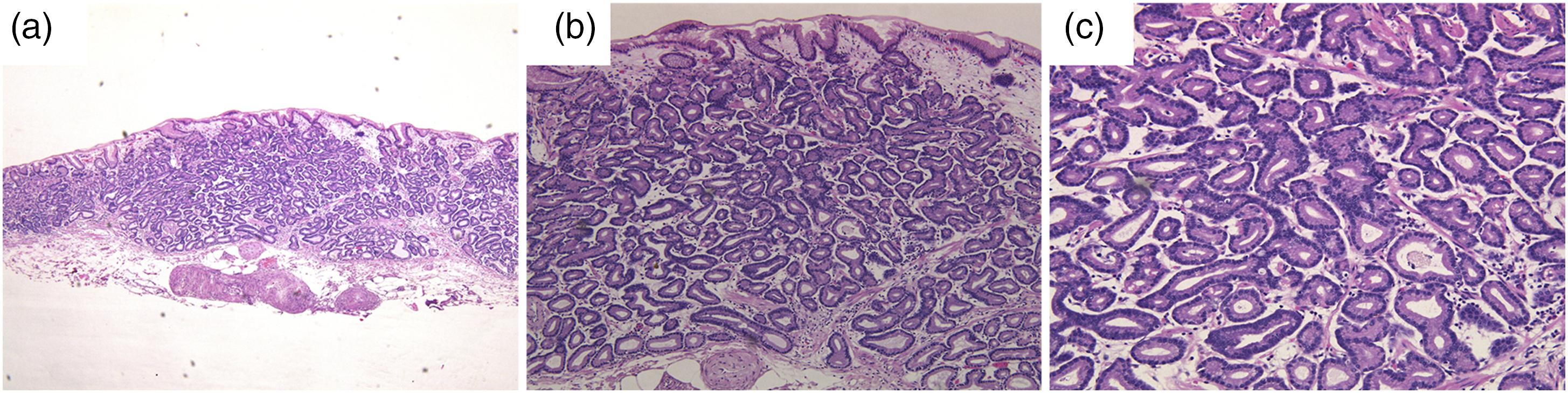
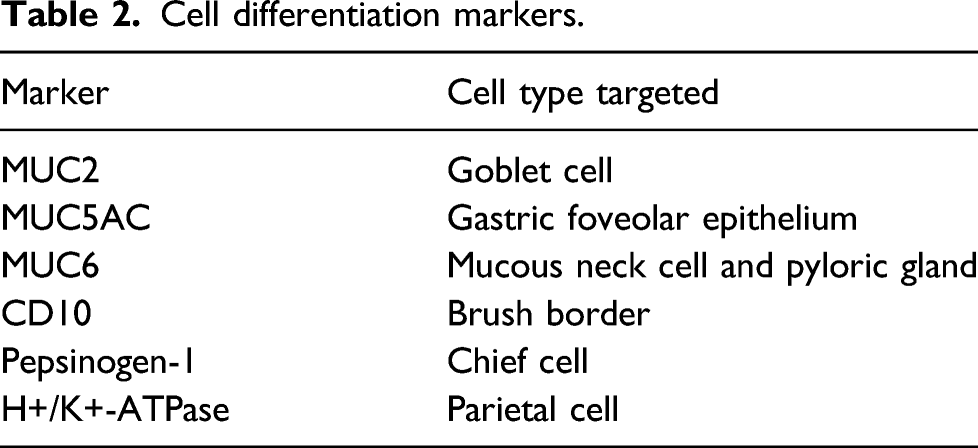
Cell differentiation markers.
Regarding expression of p53 protein and the Ki67 labeling index, none of the reported cases showed an overexpression of p53, and the Ki67 labeling index was very low (less than 5%).21,22
Pathogenesis and molecular analysis
Although many studies have been performed to elucidate the pathogenesis of GA-FG, it is still unclear. H. pylori is a gram-negative, flagellated, acid-resistant bacterium that infects more than 50% of the world’s population. 23 In etiology, H. pylori infection is associated with nonatrophic or atrophic gastritis, intestinal metaplasia,15,24 and peptic ulcers and is an increased risk factor for developing gastric adenocarcinoma and mucosa-associated lymphoid tissue lymphoma. 23
A recent study reported the effect of H. pylori infection in GA-FG. Compared with the H. pylori-negative group, a decreasing number of fundic glands and thinned foveolar epithelium covering tumor ducts were observed in the H. pylori eradication group. 15 Takashi Chiba et al. 25 reported 20 cases of GA-FG, of which 15 were H. pylori positive or experienced H. pylori eradication, but almost all lesions occurred in nonatrophic gastric mucosa. Therefore, the association between H. pylori infection and GA-FG still needs to be further studied.
Some GA-FG cases showed distended fundic glands mixed with foveolar cells, similar to fundic gland polyps. Therefore, some hypotheses have been raised regarding whether the occurrence of GA-FG is related to the use of proton pump inhibitors. However, the associated research is limited, and the relationship between the occurrence of GA-FG and the use of proton pump inhibitors is not clear.
There are few studies on the molecular changes associated with GA-FG. Activation of the Wnt/β-catenin signaling pathway is considered to be related to GA-FG occurrence, but the detailed mechanism underlying the activation of this signaling pathway has not been elucidated. To elucidate the potential role of gene methylation in relation to the Wnt/β-catenin signaling pathway, Takashi Murakami et al. 26 performed β-catenin immunostaining and methylation-specific PCR detection of related genes, including SFRPs, APC, AXIN2, and MCC, in GA-FG-CCP. They found that the nuclear β-catenin labeling index was higher in GA-FG-CCP. SFRPs, APC, and AXIN2 are also more frequently methylated in GA-FG-CCP. A significant correlation was observed between nuclear β-catenin expression and SFRP1 methylation. In conclusion, the activation of this signaling pathway mediated by gene methylation may be related to the occurrence and development of GA-FG-CCP. In 2014, Ryosuke Nomura et al. 10 detected the mutation status of GNAS and the Wnt/β-catenin signaling pathwayrelated genes CTNNB1/AXINS/APC in 26 cases of GA-FG. They found that nearly half of the cases harbored at least one mutation in CTNNB1/AXINS/APC, leading to persistent activation of the Wnt/β-catenin signaling pathway. Among 26 cases of GA-FG, only 5 cases contained GNAS mutations (R201 C). GNAS mutation may occur in a small proportion of GA-FG as an alternative mechanism to activate the Wnt/β-catenin signaling pathway. Similarly, in 2013, Yasuhiro Hidaka et al. 17 detected the mutation status of the CTNNB1/AXIN1/AXIN2/APC genes. The results showed that 14 (51.9%) of 27 GA-FG-CCP type lesions contained at least one gene mutation in CTNNB1/AXIN1/AXIN2/APC. Among 27 cases of GA-FG, 17 cases presented low nuclear β-catenin expression, and 7 cases had high nuclear β-catenin expression. Therefore, as a unique nonaggressive tumor entity, GA-FG-CCP is characterized by accumulation of nuclear β-catenin and mutation of CTNNB1 or AXIN genes, indicating activation of the Wnt/β-catenin pathway. However, in a recent paper on 8 cases of GA-FG, 8 tumor nuclear β-catenin expression was negative in all cases, which may be attributed to the limited number of cases.
Treatment and prognosis
GA-FG is a well-differentiated, well-circumscribed, and low malignant potential gastric cancer entity that rarely exhibits lymphatic or vascular invasion. However, it is commonly observed in the submucosa. Endoscopic submucosal dissection (ESD) is considered the most effective regimen for restricted GA-FG. The present evidence shows that GA-FG has a favorable prognosis, and metastasis and recurrence rarely occur during follow-up. Li et al. 8 performed a follow-up of 8 cases of GA-FG from 5 to 33 months. None of the patients had recurrence or metastasis. According to these characteristics, GA-FG confined to the submucosa can be treated by ESD. However, extended gastrectomy is needed for GA-FG that has transitioned into a more malignant tumor, disease with suspected lymph node metastasis, or multiple gastric cancer.
Conclusion
GA-FG is a well-differentiated, low malignant potential, gastric cancer entity originating from the gastric fundic gland. In the surrounding mucosal background, chronic gastritis, atrophic glands, and intestinal metaplasia cannot be observed. In more than 95% of GA-FG cases, the tumor cells differentiate towards chief cells. Tumor cells with mild atypia and enlarged nuclei are closely arranged to form anastomosing glands, the so-called “endless gland.” Tumor lesions are very prone to involve the submucosa, despite their low malignancy and invasiveness. This phenomenon may not be malignant invasion of the tumor but instead may be a prolapse-type growth pattern into the submucosa. Although abnormal activation of the Wnt/β-catenin signaling pathway is related to GA-FG, there are few studies on the molecular changes in GA-FG and its pathogenesis, and further studies on GA-FG pathogenesis are necessary.
Footnotes
Author contributions
Guang Yang, Ru-yi Zheng, Xiang-yu Meng, and Cheng-ji Dong developed the main content of this manuscript. Guang Yang and Ru-yi Zheng were involved in writing the manuscript. Cheng-ji Dong and Xiang-yu Meng edited the language of revised manuscript. Guang Yang supervised the project and contributed to the revision of the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
An approval of our Institutional Review Board or Ethics Committee was not needed to conduct this analysis.
Informed consent
Not applicable.
