Abstract
Carcinosarcoma (CS) is a rare, aggressive malignancy of the Mullerian system often termed mixed malignant Mullerian tumor (MMMT). It is biphasic in nature, differentiating into epithelial and sarcomatous components. Tumor-node-metastasis (TNM) staging and mismatch repair (MMR) status is the basis for both prognostication and therapeutic decision making. However, stromal differentiation (SD) is a new frontier in the field of histopathology and many studies have demonstrated its prognostic significance. The present study is the first study to evaluate the role of SD in carcinosarcoma. Here we found immature SD to be a significant prognostic signature (p = 0.04). It outperformed age, nodal metastasis, and lymphovascular invasion for predicting cancer-free survival. Immature SD also corelated with both myometrial invasion (p = 0.01) and tumor stage (p = 0.02). Carcinosarcoma has been previously thought to have universally poor outcomes; however, mature SD was found to be protective in this cancer subtype. Our findings support the integration of SD into the synoptic reporting for carcinosarcoma; however, this will require pathologists to shoulder the adoption of SD into clinical practice.
Introduction
More than 35 years ago, Dvorak brilliantly termed cancer as “a wound that does not heal.” 1 In doing so, Dvorak implied that biological processes controlling wound response also occurred in developing tumoral stroma. 2
Since then, much research has focused on the extracellular matrix (ECM), deciphering cellular components of the tumoral microenvironment, and how they are influenced by each other. In colorectal cancer, Ueno et al. 3 pioneered the field of (SD) and showed that different stromal phenotypes existed with distinct prognostic significance. Stroma has been classified as immature in the presence of amorphous myxoid material (>40× focus), intermediate with keloid like collagen, and mature with fine mature collagen fibers.
The prognostic significance of stroma is not specific to colon; today, stromal differentiation is being investigated in many other cancer subtypes.4–6 Notable cancers include breast, cervix, esophagus, and stomach.4–7 In the breast, stromal differentiation has been found to predict clinical outcomes, immune profiles, and molecular phenotypes. 7 Overall, immature stroma is a bad player, associated with reduced survival and higher pathologic stage.
As of now, the classification of stromal differentiation has primarily been based on the three-tier grading system originally introduced by Ueno et al.3,8 However, Hacking et al. 9 recently proposed a semi-quantitative approach for grading stromal differentiation in colorectal carcinoma (CRC). This allows stromata to be subdivided into low grade and high grade based on the degree of myxoid degeneration, variability in absolute difference in intensity between stromal matrix regions, and continuity of myxoid stroma. More practically, in high grade myxoid stroma is the predominate stromal pattern (>50%) at the extramural tumoral front. 10
The purpose of this study is to evaluate SD in carcinosarcoma (CS) or mixed malignant Mullerian tumor (MMMT), a rare cancer subtype which contains the malignant combination of tumor epithelia and mesenchyme. 11 These cancers are now understood to arise from a monoclonal origin, having variable degrees of mesenchymal differentiation. 11 This tumor subtype has very poor prognostic outcomes and therapeutic strategies have resulted in minimal gains for improved overall survival. 11 It is broken down into a homologous type with sarcomatous parts recapitulating tissues native to the uterus (smooth muscle); whereas in the heterologous-type, non-native tissues are present: cartilage, skeletal muscle and bone. 11 Similar to other carcinomas, carcinosarcomas also display prominent desmoplastic reaction (DR); although not extensively studied as other cancer subtypes.
Apart from squamous cell carcinoma of the cervix, there is very little data pertaining to stromal differentiation in gynecological pathology. 4 In this publication, we collected a large cohort of CS patients, a tumor seen to have uniformly poor clinical outcomes. Our primary aim is to evaluate the role of SD as a prognostic signature, assessing its overall relationship to the clinicopathologic profile and cancer-free survival (CFS). Innovations here could lead to improved prognostication, further optimized therapeutics, and improved clinical outcomes for women diagnosed with carcinosarcoma.
Materials and methods
Institutional review board
Institutional Review Board approval from the Office of the Human Research was obtained for all experiments. All methods were carried out in accordance with relevant guidelines and regulations. Patient consent was not required by the institutional review board (IRB) committee due to the retrospective nature of the study (Northwell Health IRB number: 18-0890).
Design
This was a retrospective study of Carcinosarcoma patients diagnosed at the Northshore University and Long Island Jewish Hospitals, Northwell Health. Routine hematoxylin and eosin (H&E) stained slides of these cases were collected between 2010 and 2020. Only resection specimens were selected and one representative slide for stromal grading was selected per case from a single slide containing the largest portion of tumor. Clinicopathological data was collected for each case and analysed in relation to stromal differentiation grading. Cancer-free survival data was provided by the Northwell Health Cancer Registry.
Digital slides
We utilized digital slides for the purpose of analyzing histology for this study. Whole slide images (WSI) were accessed through the Aperio vendor agnostic whole slide image viewer and slides were scanned on a Leica Aperio AT2 (Leica Biosystems, Buffalo Grove, Illinois, USA) whole slide scanner at 20×.
Stromal differentiation
In the assessment, a detailed search was done to assess the stromal, with particular attention to the tumoral front. Scoring was based on the 3-tier grading system proposed by Hacking et al. 9 (Table 1). Mature stroma (SD1) contained fine and mature collagen fibers, often stratified into multiple fibrous layers at the invasive tumoral front. Immature stroma was assessed with a semiquantitative approach, based on the degree of myxoid amorphous amphophilic material. This myxoid stroma contained a basophilic to grey extracellular matrix, occasionally intermingled with haphazardly oriented and hyalinized collagen. Low grade stroma contained a high variability in absolute difference in intensity between stromal matrix regions (mosaic pattern) with less contiguous areas of myxoid stroma (Figure 1). Practically, low grade stroma contained a minimum amount of myxoid stroma (×40 field) and intermediate stroma containing “keloid like” collagen was designated low grade. 12 High grade stroma contained low variability in absolute difference in intensity between stromal matrix regions and stromal cells surrounded by contiguous regions of myxoid stroma. Practically, myxoid stroma is the predominate stromal pattern (>50%) at the extramural tumor front.
Classification of stromal differentiation.


A Semi-Quantitative Approach to Stromal Differentiation. Low grade should contain a minimum amount of myxoid stroma (×40 field), while high-grade is the predominate stromal pattern (>50%) at the extramural tumor front.
Clinicopathological variables
Clinicopathological data was collected for each case and analysed in relation to stromal differentiation grading. In exploratory analyses, consensus stromal differentiation scores were compared to multiple variables including cancer-free survival, age, lympho-vascular invasion (LVI), tumor stage, lymph node metastasis, tumor size, heterologous differentiation, myometrial invasion (MI), cervical involvement, and mismatch repair (MMR) status.
Mismatch repair
Immunohistochemistry was performed on formalin-fixed and paraffin-embedded tumor sections cut at 4-micron thickness and stained on a Ventana Bench Mark Autostainer (Ventana Medical System, Tucson, Arizona). The following rabbit monoclonal primary antibodies were used: MLH-1 (M1Ventana), PMS2 (EPR 3947, Ventana), MSH2 (G219-1129, Ventana), and MSH6 (44, Ventana). Antibodies were pre-diluted by the manufacturer and staining was performed following the manufacturer’s protocols in collaboration with platform vendors. Technical methodologies and quality assurance were performed at the Immunopathology Laboratory of Long Island Jewish Medical Center (Northwell Health System, New Hyde Park, NY). Complete loss of any of the foregoing markers lead to the designation of MMR deficient.
Statistical analysis
The primary objective of the statistical analysis was to assess the relative prognostic importance of stromal differentiation in CS. Descriptive statistics such as frequencies and percentages were calculated for the categorical variables. The Kaplan-Meier method was used to evaluate the disease-free survival rate as a function of time. The two tailed Fisher exact test was used to assess relationships between mature (SD1), immature, low grade (SD2) and immature, high grade (SD3) stromal differentiation. Statistical Analysis was performed using Prism graphpad 8.4.2. A p value of <0.05 was used to indicate statistical significance.
Results
Patient characteristics
50 cases of carcinosarcoma were retrieved from our health system for this study. Stromal differentiation was assessed in the manner previously mentioned. Out of the 50 cases, 21 (42%) were classified as mature, 16 (32%) as immature, low-grade, and 13 (26%) as immature, high-grade (Figure 2). Out of these patients, 24 were under the age of 70, while 26 were age 70 or older at diagnosis. 22 patients had lymphovascular invasion, while 28 did not. Regarding tumor stage, 26 patients presented with low stage (I–II) and 24 presented with high stage (III-IV). A total of 9 patients presented with nodal metastasis and 41 did not. The majority of patients had tumors larger than 4 cm: 41 versus 9. Heterologous differentiation was seen in 17 cases and homologous in 33. >50% myometrial invasion was seen in 20 cases, while 30 cases had <50% myometrial invasion. Cervical involvement was seen in 15 cases, while 35 cases were not involved. Finally, 4 patients had MMR deficiency, while 46 did not.

Stromal phenotypes in carcinosarcoma.
Cancer free survival
Stromal differentiation was found to be a significant prognostic signature (p = 0.04), as well as LVI (p = 0.04), tumor stage (0.04) and myometrial invasion (p < 0.03). Age was not a significant prognostic indicator in our patient cohort (p = 0.42); however, patients over the age of 70 tended to have lower survival. Lymph node metastasis (p = 0.08) also trended toward immature SD but did not reach statistical significance (Figure 3).

Kaplan Meyer survival analysis.
Stromal differentiation and the clinicopathologic profile
Firstly, there was a tendency for older age and grade 2 and 3 stroma; however, this was not statistically significant. For lymphovascular invasion, there was no association with stromal differentiation (p = 0.56). There was a tendency for higher tumor stage and immature stroma, although this did not reach statistical significance (p = 0.12). For tumor staging, immature stroma was found to be associated with high stage tumors (p = 0.02). Lymphovascular invasion was more common in patients with immature stroma but not significant (p = 0.71). Tumors also tended to be larger in the immature cohort, which did not reach statistical significance (p = 0.14). Heterologous differentiation was not associated with stroma in this study (0.76). Myometrial invasion of more than 50% was found to be statistically associated with immature stroma (p = 0.01). For cervical involvement, tumors with immature stroma tended to be more involved; however, this did not reach statistical significance (p = 0.21). Results are available from Fisher exact analysis (Table 2) can and stromal percentages for significant variables (Figure 4).
Fisher exact analysis.
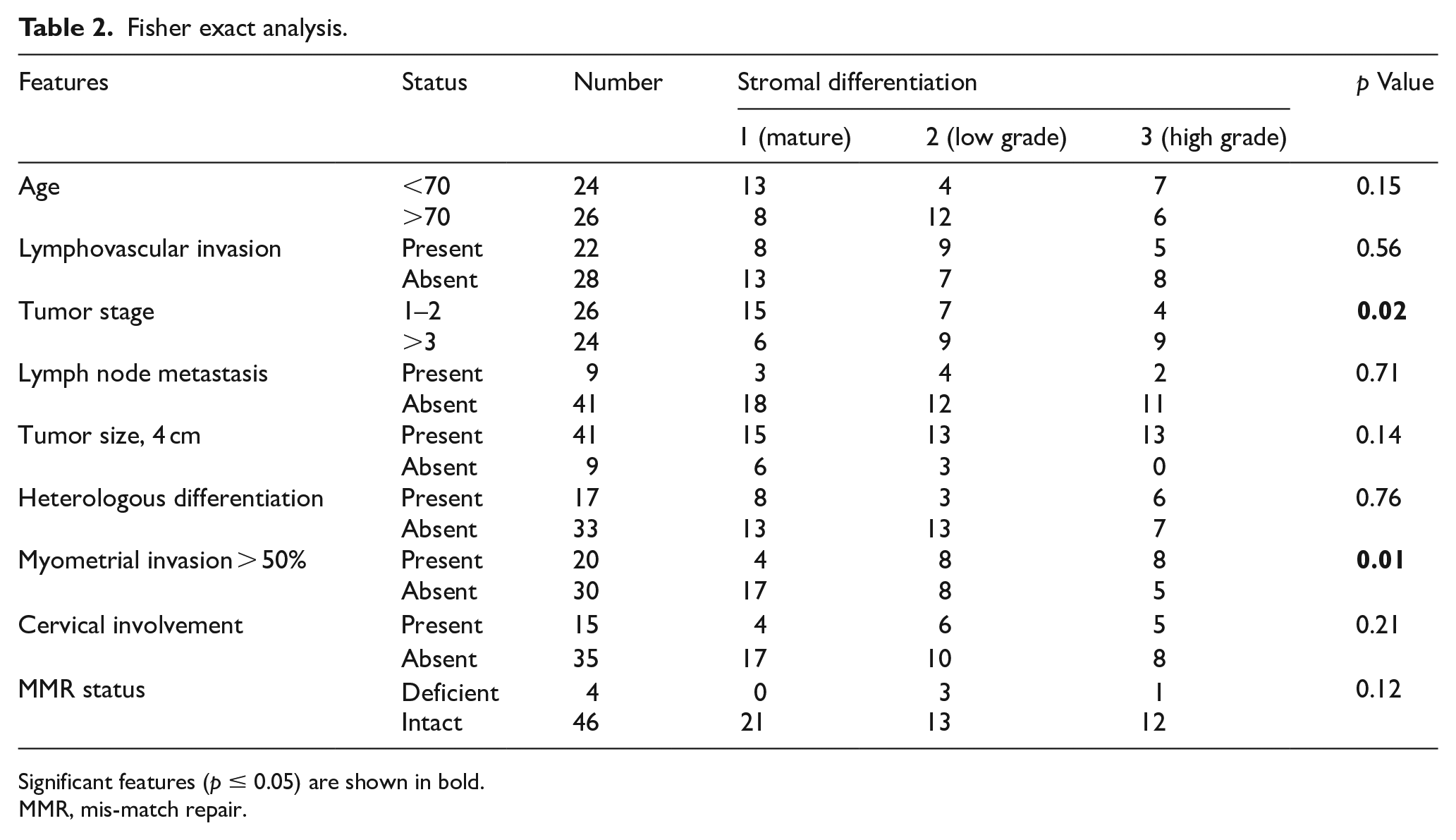
Significant features (p ≤ 0.05) are shown in bold.
MMR, mis-match repair.
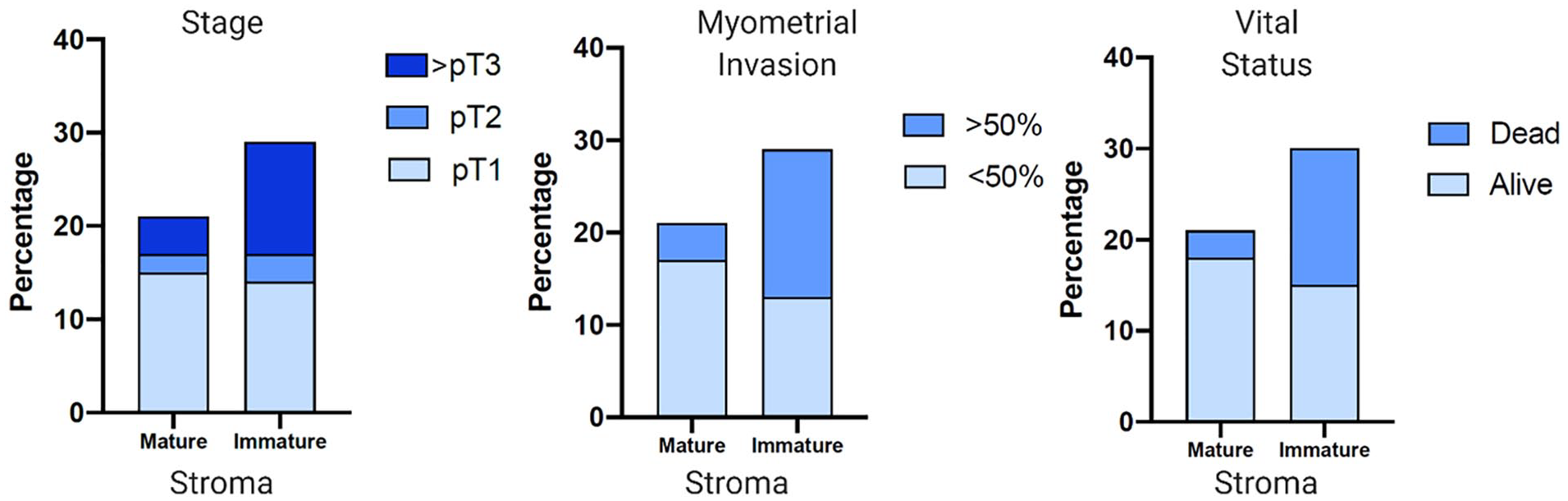
Stromal percentages for significant variables.
Stromal differentiation and mis-match repair
There were 4 cases of carcinosarcoma in out study which had total loss of any of the 4 mis-match repair proteins: MLH-1, PMS2, MSH2, MSH6. 0 of these cases had mature stromal maturation, 3 had low grade stroma and 1 had high grade stromal differentiation. Despite the tendency for MMR loss in patients with immature stroma, our results were not statistically significant (p = 0.12).
Discussion
Here we shed light into the role of stromal differentiation in Mullerian carcinosarcoma. The findings in our study were quite intriguing and support that stromal differentiation is both significant and biologically diverse in CS. Fineck et al. 13 demonstrated simular findings in breast cancer, showing that stromal gene expression predicted clinical outcomes. In ovarian cancer, genomic studies have identified alterations in tumoral stroma to be partially identical to its epithelial counterpart. 14 Suggesting that the stroma actively contributes to tumor development and patient mortality.
Clinically, mature stroma correlated with lower pathological tumor stage, negative nodal status, and better overall clinincal outcomes. This also supported by evidence from studies in numerous cancer subtypes.4,5,12 The present study used a low and high grade approach for immature stromal classification, this was preferred over the three-tiered classification proposed by (Ueno, Shinto and Shimazaki, 2015). 12 We feel that this method facilitated a more detailed assessment of the stroma. Moving forward, studies comparing reproducibility and prognostication will be important.
Pertaining to the clinicopathologic profile: myometrial invasion and FIGO stage were found to corelate with immature stromal differentiation in the current study. However, many variables trended toward immature stromal differentiation, although they did not reach significance. Due to the rarity of carcinosarcoma cases in clinical practice, retrieving more cases to reach statistical significance was a challenge. Kaplan Meyer survival analysis did find immature stromal differentiation to be prognostically significant.
The tumoral microenvironment is highly diverse and the present study illustrates the crucial role of stroma in determining oncological outcome in CS. Stromal assessment is not challenging and can be analyzed by a pathologist under the microscope, or on digital slides. Digital slides are becoming ubiquitous to the practice of surgical pathology, and were also used in our study. This allowed for ease of access by multiple reviewers simultaneously. Fortunately, hematoxylin and eosin (H&E) stained slides are readily available, and stromal differentiation does not require the use of additional biomarker testing. This is unlike the assessment of programmed death-ligand 1 (PD-L1) or MMR, which requires testing with immunohistochemistry or preferably molecular testing for MMR.15,16
On a biological level, the presence of immature stroma is much more than desmoplastic reaction. It is a snap shot into the epithelial to mesenchymal transition (EMT). The presence of immature stroma illustrates the degeneration of extracellular matrix components. This occurs secondary to ADAM9s being released by cancer associated fibroblasts (CAFs), facilitated by Tenascin-c, which bind to integrins on CAFs.17,18 In this setting, tumor cells can breach through basement membrane material leading to invasion. This is supported by findings in the present study: increased myometrial invasion, tumor stage, and worse survival outcomes.
SD has also been shown to effect immune cells and immune escape in breast cancer. 7 Currently, MMR status is seen to be the sole indication for PD-L1 based therapeutics, as supported by many trials showing improved outcomes in MMR deficient cancer subtypes. 16 In future, SD could be incorporated into immunotherapeutic decision making, which could ultimately improve outcomes in CS.
Applications in computer vision are also quite exiting in this space, here computation could predict outcomes and therapeutic response from different stromal phenotypes. In fact, Beck et al. 17 was the first to demonstrate by computer vision that the best predictor of patient survival was not found in tumor cells, but in adjacent stromal tissues. The use of advanced machine learning, particularly deep learning, will be important for harnessing stromal features from whole slide images.
Stromal genomics is also a space for innovation. In current practice, laser capture microdissection is used to obtain pure tumoral cell populations. However, this results in a mixture of different cell types being processed and such topologies may not capture the true complexity of the tumoral microenvironment. An environment which includes not only tumoral cells, but also immune cells and CAFs. Most interestingly, studies have discovered that genomic prognostic signatures in colorectal cancer seem to arise from stromal rather than epithelial tumor cells. 20 This is reminiscent to that seen with computer vision. However, stromal genomics is not routinely performed in clinical practice.
There are several pitfalls to our study and it is important to consider that this was a retrospective study with the potential for bias. Secondly, due to the rarity of this cancer subtype, obtaining a larger study cohort was difficult. Multi-institution collaboration will be important moving forward.
Due to its simplicity and ease of use, SD should be incorporated into pathology practice. In future, the stroma could optimize prognostic stratification and determine therapeutics, shifting currenting paradigms in precision oncology. Well-designed, robust clinical trials will be needed to prove this hypothesis.
Footnotes
Acknowledgements
We thank Claudine Alexis and Kathy Quinn for their role as research coordinators.
Author contributions
MN and SH developed the theoretical formalism. SH, DW, and HC contributed to the acquisition of data. RN and DW performed the analytic calculations and performed the numerical simulations. SH, DW, RN, HC, and MN contributed to the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Institutional Review Board approval from the Office of the Human Research was obtained for all experiments. All methods were carried out in accordance with relevant guidelines and regulations. Patient consent was not required by the institutional review board (IRB) committee due to the retrospective nature of the study (Northwell Health IRB number: 18-0890). Not applicable.
Informed consent
Patient consent was not required by the institutional review board (IRB) committee due to the retrospective nature of the study (Northwell Health IRB number: 18-0890).
Data availability
Pathology data and the statistical analyses for the current study are available from the corresponding author upon reasonable request.
