Abstract
Soft tissue sarcomas are a rare group of mesenchymal malignancies which can range from low to high grade. These tumors have different clinical, radiographic, and histopathological characteristics. Beta human chorionic gonadotropin is a naturally secreted hormone by placental syncytiotrophoblast cells during pregnancy. On very rare occasions, sarcomas can develop the ability to ectopically produce human chorionic gonadotropin. Very few cases exist in the literature of soft tissue sarcomas expressing this hormone. We report the case of a 55-year-old female who presented with a posterior thigh soft tissue sarcoma who on the day of surgical resection was found to have an unusually elevated serum human chorionic gonadotropin. Positive immunohistochemical staining of the resected mass confirmed the sarcoma as the source of the beta human chorionic gonadotropin.
Introduction
Soft tissue sarcomas are a rare group of malignancies of mesenchymal tissue origin. There are an estimated 12,000 cases of soft tissue sarcomas diagnosed annually in the United States. 1 These cancers are most often diagnosed in patients over the age of 50. However, there are specific subtypes, such as synovial sarcoma which can be diagnosed in young children. Soft tissue sarcomas can exist from low to high grade and may differ in their level of aggressiveness. Approximately 16% of patients diagnosed with soft tissue sarcomas will have metastatic disease at initial diagnosis and annually nearly 5000 patients die from related issues annually in the United States. 1 There are approximately 70 different types of soft tissue sarcoma and each is diagnosed based on a combination of clinical imaging, histopathologic morphology, special stains, and evaluation for translocations.
With such a diversity of tumor subtypes, secretion of ectopic beta human chorionic gonadotropin (β-hCG) by the tumor is so uncommon that it is not routinely tested for in cases of sarcoma. A few cases have been published in the literature in which β-hCG was produced by sarcoma, and most often these cases involve osteosarcoma instead of soft tissue sarcoma. This case report documents an unusually rare circumstance of β-hCG production by a soft tissue sarcoma in a 55-year-old patient. We provide a detailed discussion of the case as well as review of the literature.
Case report
A 55-year-old female presented to our musculoskeletal oncology clinic with a chief complaint of a growing, painless posterior thigh mass for approximately 6 months. We obtained radiographs of the femur, which demonstrated a soft tissue shadow in the posterior thigh with some apparent calcifications within the mass (Figure 1). We then ordered a magnetic resonance imaging (MRI), which showed a large, heterogeneous soft tissue tumor abutting the neurovascular structures of the posterior thigh (Figures 2 and 3). Based on the concerning imaging, we moved forward by obtaining a core needle biopsy of the mass. The biopsy findings suggested the mass to be a hyalinizing tenosynovial giant cell tumor. Because the imaging and clinical history did not match well with our suspicion that his was a soft tissue sarcoma, we decided to move forward with a wide excision of the tumor, prepared for the possibility that final pathology would prove this mass to be a sarcoma. On the day of the procedure, a preoperative pregnancy test was ordered as is routine and the serum β-hCG was positive at 1122 IU/L. The patient reported that she was in menopause and had not had sexual activity in nearly a year. She denied any symptoms of pregnancy. The decision was made to proceed with surgery, understanding that there was a possibility that the tumor was secreting this hormone. The tumor was resected with a wide margin and sent for histopathologic evaluation (Figure 4). Sections from the resection specimen showed a proliferation of spindle cells and pleomorphic nuclei. By immunohistochemistry, there was strong staining of tumor cells for β-hCG. The morphologic and immunophenotypic features were consistent with an unclassified pleomorphic sarcoma with ectopic β-hCG production (Figures 5 and 6). Unfortunately, no molecular analysis is available for the specimen. Postoperatively, the patient’s wound healed without complication, she received postoperative radiotherapy, and at 2 weeks postoperatively her β-hCG test was negative. At 1 year postoperatively, she was without evidence of local or metastatic disease and her β-hCG continued to be negative.

Lateral radiograph of the femur demonstrating a posterior thigh soft tissue shadow with internal calcifications.
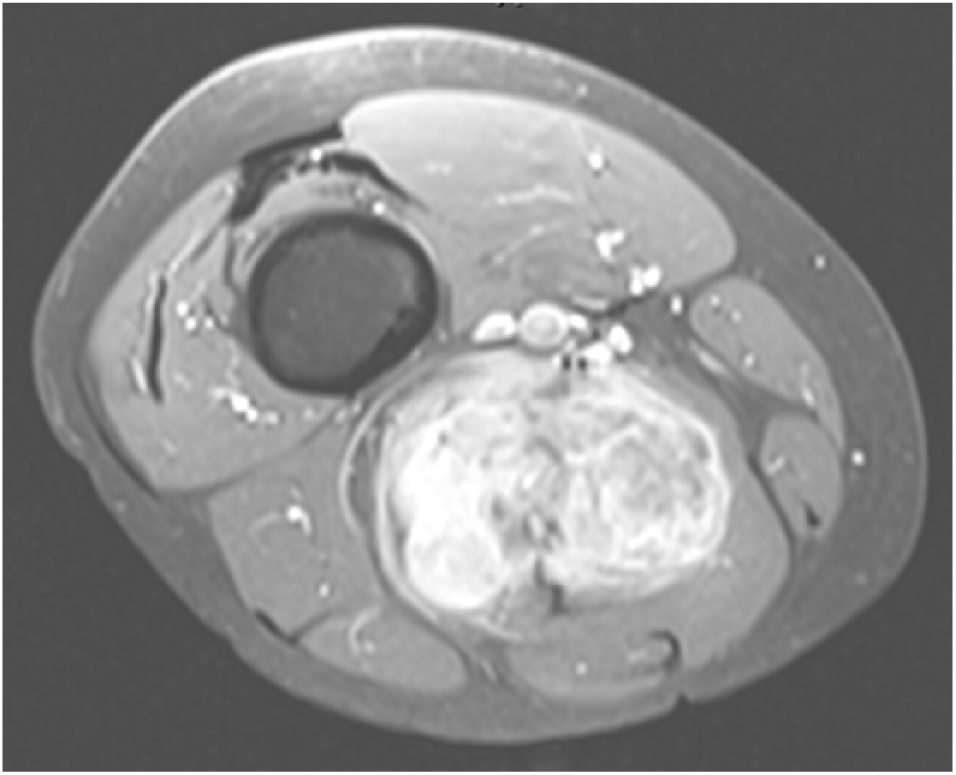
T2-weighted axial MRI of the thigh demonstrating a large, posterior tumor which is enhancing, heterogeneous, and appears to abut the neurovascular structures.

T2-weighted sagittal MRI of the thigh demonstrating a large, posterior thigh tumor which is enhancing and heterogeneous. Also note some proximally and distally tracking edema within the muscle.
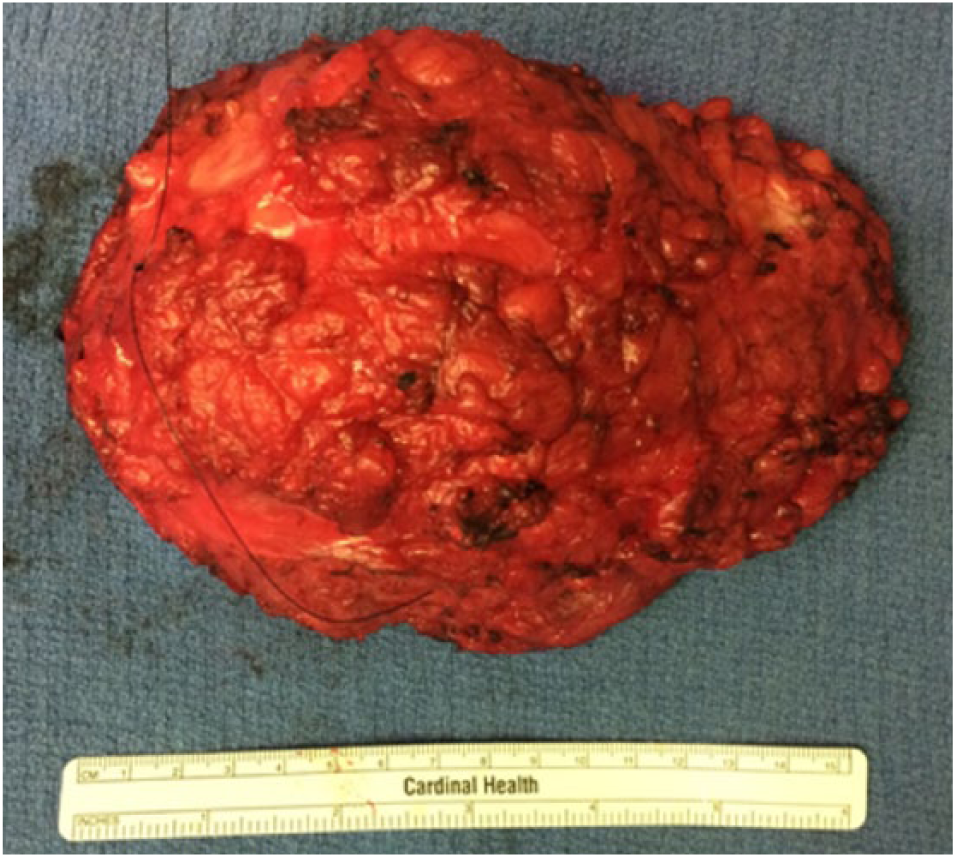
Clinical photograph of the tumor which was resected with a wide margin.

Sections from the resection specimen show a proliferation of spindle cells with admixed large polygonal cells with abundant pale eosinophilic cytoplasm and pleomorphic nuclei. There was no obvious tumor necrosis and up to 10 mitotic figures were identified in 10 high-power fields.

By immunohistochemistry, there was strong staining of tumor cells for β-hCG without staining for desmin, smooth muscle actin, S100 protein, SALL4, GATA3, cytokeratin, or CD34. The morphologic and immunophenotypic features were consistent with an unclassified pleomorphic sarcoma with ectopic β-hCG production.
Discussion
Soft tissue sarcoma is a rare malignancy of mesenchymal tissue origin which can exist in one of nearly 70 variants. Each of these subtypes is diagnosed based on a combination of clinical imaging, histopathologic morphology, special stains, and evaluation of translocations.
Although there is a great deal of diversity in these soft tissue tumor subtypes, secretion of ectopic β-hCG by the tumor is so uncommon that it is not routinely tested in cases of sarcoma. Very few cases have been published in which β-hCG was associated with a sarcoma, and most of these were cases involving osteosarcoma instead of soft tissue sarcoma. Certainly, elevated β-hCG in a 55-year-old woman can be caused by a number of etiologies aside from a viable pregnancy including pituitary human chorionic gonadotropin (hCG) production, trophoblastic disease, phantom hCG, ectopic pregnancy, or miscarriage. Considering that this patient reported no sexual activity in over a year and she had a concerning appearing soft tissue mass, our clinical suspicion of a β-hCG secreting tumor was high.
Sarcoma and β-hCG have demonstrated some association in the literature in regards to use as a prognostic tool as well as a treatment. Some authors have published that β-hCG may function as a marker providing prognostic value in soft tissue tumor response. 2 Other authors have investigated its role in treatment. Early reports documented using β-hCG to treat Kaposi sarcomas with mixed outcomes but significant side effects.3,4 Although Kaposi sarcoma is not a true sarcoma, it does share many morphologic characteristics with soft tissue sarcoma.
Among the first reports of β-hCG production from a sarcoma was a case report published by Mack et al. 5 in 1977. In this case, a 26-year-old male was found to have a large acetabular lesion. The lesion was biopsied and the results were inconclusive; however, a germ cell line tumor was suspected. Because of concern for a germ cell tumor, a urine pregnancy was also performed and found to be positive. An open biopsy was then performed to obtain more tissue and a diagnosis of chondrosarcoma was made. At the time of this publication, it was established that malignancies could produce hormones, including β-hCG; however, this was the first report of a markedly elevated clinical level in the setting of sarcoma.
Since that first documented case, a number of other reports have been published. Kalra et al. 6 published in 1984 a case of an osteogenic sarcoma producing hCG in a 22-year-old woman. In this case, pregnancy was excluded with a normal ultrasound as well as an endometrial curettage. Tuy et al. 7 in 2008 published a case report of a 37-year-old female who presented with an aggressive appearing lesion of the distal femur. The patient was admitted with symptoms of abdominal cramping and vaginal bleeding and a preoperative pregnancy test was positive. Workup by gynecology including pelvic exam, ultrasound, and laparoscopy, and dilation and curettage were negative for pregnancy. Workup and treatment of the femoral lesion revealed a high-grade osteosarcoma which stained positive for β-hCG. Oshrine et al. 8 in 2014 also reported a case of osteosarcoma in an adolescent female which was secreting β-hCG. Serum β-hCG levels became undetectable by the end of her sarcoma treatment.
Due to an increasing interest in an association between β-hCG and sarcoma, a number of groups in 2010 began larger investigations. Masrouha et al. 2 published in 2012 a study in which they retrospectively stained 37 histopathology samples of osteosarcoma and found that five tumors stained positive for β-hCG. They also found that all samples with positive staining showed poor histological response to neoadjuvant chemotherapy. Lee et al. 9 in 2014 performed a larger study and found that, in 49 pretreated osteosarcoma samples, 57% showed positive β-hCG expression. The group found no clinical difference between the tumors that stained positive and negative for the hormone.
In regards to soft tissue sarcomas, very few reports exist in the literature of tumors expressing β-hCG. The first report by Meredith et al. 10 was published on a case of leiomyosarcoma of the small intestine producing β-hCG in a 22-year-old female in 1986. This patient presented with symptoms of nausea and vomiting, which can be commonplace during pregnancy. A mass was eventually appreciated and elevated β-hCG levels were localized to the sarcoma cells upon resection. Certainly, in a 22-year-old female, the other causes of elevated β-hCG previously mentioned should be at the top of the initial differential. Another report of a leiomyosarcoma secreting β-hCG was published by Mansi et al. 11 in 2002. In this report, the patient was a 57-year-old male with a 30-cm retroperitoneal mass which tested positive for the hormone after resection. A case report by Steffensen et al. 12 in 2008 documented an epithelioid sarcoma which was metastatic to the placenta during pregnancy. They noted that only 8% of malignancies which metastasize to products of conception prove to be sarcoma. It is even more rare for these metastases to express β-hCG, as this case did. Inoue et al. 13 reported in 2011 a case of a nonrhabdomyosarcoma soft tissue sarcoma in a 3-month-old with a large back mass. Unfortunately, due to subtotal resection and chemotherapy resistance, the tumor progressed and this patient died of disease at 22 months old. Finally, Stevens et al. 14 reported in 2013 a case of synovial sarcoma in the hip of a 45-year-old female. The author noted a significant delay in diagnosis due to consideration of more common etiologies of the elevated hormone including ectopic pregnancy and a trophoblastic neoplasm. At the time of final diagnosis, the pelvic sarcoma was deemed unresectable and she was treated with chemotherapy and radiation.
β-hCG expression by sarcomas is incredibly rare as has been evident by the scarcity of literature and case reports of the subject. It does seem that osteosarcoma may more commonly express this hormone than its soft tissue counterpart. Although some authors have looked into the prognostic and therapeutic aspects of the hormone in sarcoma, no definitive outcome has been established. The most significant point to be taken from the few case reports published on the occurrence would be that it is a possibility. Especially, in cases of very low likelihood of pregnancy such as our patient, the surgeon should always consider the possibility that the malignancy has developed the ability to secrete the hormone β-hCG. We recommend in cases of a positive preoperative pregnancy test, in the setting of known or likely malignancy, a candid discussion with the patient as well as consult to gynecology and pelvic ultrasound if necessary.
Footnotes
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent
The patient agreed to have her de-identified information included in this case report for academic/educational purposes.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
